Abstract
Background:
Little research has explored the detail of practice when using sedative medications at the end of life. One work package of the I-CAN-CARE research programme investigates this in UK palliative care.
Aims:
To investigate current practices when using sedative medication at the end of life in London, UK, by (1) qualitatively exploring the understandings of palliative care clinicians, (2) examining documented sedative use in patient records and (3) comparing findings from both investigations.
Design:
We conducted focus groups with experienced palliative care physicians and nurses, and simultaneously reviewed deceased patient records.
Setting/participants:
In total, 10 physicians and 17 senior nurses in London hospice or hospital/community palliative care took part in eight focus groups. Simultaneously, 50 patient records for people who received continuous sedation at end of life in the hospice and hospital were retrieved and reviewed.
Results:
Focus group participants all said that they used sedative medication chiefly for managing agitation or distress; selecting drugs and dosages as appropriate for patients’ individual needs; and aiming to use the lowest possible dosages for patients to be ‘comfortable’, ‘calm’ or ‘relaxed’. None used structured observational tools to assess sedative effects, strongly preferring clinical observation and judgement. The patient records’ review corroborated these qualitative findings, with the median continuous dose of midazolam administered being 10 mg/24 h (range: 0.4–69.5 mg/24 h).
Conclusion:
Clinical practice in these London settings broadly aligns with the European Association for Palliative Care framework for using sedation at the end of life, but lacks any objective monitoring of depth of sedation. Our follow-on study explores the utility and feasibility of objectively monitoring sedation in practice.
Keywords
Sedative medication may be used to manage intractable symptoms at the end of patients’ lives.
No UK guidelines specifically address the detail of how sedatives should be used, but international guidelines endorse monitoring the depth of sedation, and the European Association for Palliative Care (EAPC) framework recommends that monitoring should relate to the aim of using sedatives.
Despite internationally agreed guidelines and recommendations, use varies widely between countries and settings, including the depth of sedation sought, and the dosages administered.
This study shows that usual practice when using sedative medication in two palliative care settings in London, UK, is predominantly to use low dosages of midazolam to achieve patient comfort, rather than to sedate patients.
Practice in these London settings broadly aligns with EAPC recommendations for proportionate use of sedatives at the end of life.
Nevertheless, although the EAPC framework also recommends systematic objective monitoring to monitor the effects of sedatives, clinicians in these settings use only clinical observation, never structured objective tools, even when using high doses of sedatives.
The term ‘palliative sedation’ does not usefully describe all uses of sedative medication in palliative care, since this implies sedation is the aim, which is not always the case. Proportionate sedation might be a preferable term for the type of practice we found in our study.
Palliative care guidelines and definitions should clearly distinguish between deep sedation and other uses of sedatives in palliative care.
When higher doses of sedative medication are used and/or when the specific intention is to sedate a patient, clinicians may need to employ more structured monitoring of sedative effects.
Background
Two 2015 reports on palliative and end-of-life care indicated that further research and/or improvement of care in relation to symptom management would be of value. ‘Dying without Dignity’ 1 identified ‘poor symptom control (pain and agitation)’ as one area where care of dying patients needed improvement. The research priorities for palliative care identified by the Palliative and End-of-life care Priority Setting Partnership 2 in the same year included managing symptoms and medication.
The Marie-Curie-funded I-CAN-CARE research programme began in 2016. One of its two work packages addresses the use of sedative medication at the end of life in the United Kingdom. Sedative medications are used internationally3,4 and may be used for patients with refractory symptoms, primarily aiming to relieve distress by reducing patient consciousness.5–7 A recent Cochrane review of pharmacological sedation for terminally ill adults concluded that studies should address the effect of sedatives on ‘a person’s quality of life, or peacefulness and comfort during the dying phase’, and how well sedatives control distressing symptoms. 8
Concerns have been expressed that palliative care patients may be over- or under-sedated, with potentially adverse consequences for their care and their relatives’ experiences.9–11 Guidelines generally recommend that sedative use at the end of life should be proportionate, with doses being ‘individually tailored’, 12 although Schildmann et al. 13 reviewed national and international guidelines for using sedation at the end of life and concluded that guidelines are inconsistent and of limited quality. Abarshi et al. 14 suggest in their later systematic review, however, that the European Association of Palliative Care (EAPC) Framework for using sedation in palliative care 15 is an acceptable standard. This Framework recommends that patients receive proportionate sedation, that is, minimum doses of sedatives to palliate their suffering, and be regularly monitored for the level of sedation, 15 although stating further that, if the aim of using sedatives is to ensure comfort for an imminently dying patient, the only critical parameters to be monitored are those pertaining to that person’s comfort.13–15
However, terminology also lacks clarity and consistency,6,14,16,17 and the frequently used term ‘palliative sedation’ does not always accurately describe practices, 6 which vary by country18–21 and setting (community, hospice or hospital),22–25 and in depth and type of sedation used (light, deep, continuous or intermittent).26,27 Morita et al.5,6 discuss this variability, and, following Quill et al., 7 recommend more precision in the terminology used, including for approaches such as the EAPC’s proportionate sedation. 6
Previous qualitative research exploring UK clinicians’ perceptions suggested that in the United Kingdom sedative drugs may be prescribed at the end of patients’ lives to ‘settle’ agitation or ensure comfort, rather than unconsciousness. 28 A 1999 retrospective review of notes for deceased UK hospice patients, which defined sedation as receiving daily doses of ⩾10 mg midazolam, found that (by this definition) 48% of patients had been sedated. 29 A subsequent study of sedative use in another UK hospice, using the same definition, found decreasing use between 1996 and 2006; 30 another UK study found a slight decrease between 2009 (80% patients sedated), 2011 (62%) and 2014 (73%). 31
As a first step in the I-CAN-CARE sedation project, we conducted a study to explore the detail of current practice when using sedatives at the end of life in two settings in London, UK, employing qualitative description32,33 plus descriptive statistics. This article presents findings on sedative drugs and dosages used, reasons for using sedative medications and how their effects were monitored or assessed, obtained from two sources: qualitative focus group (FG) discussions and a retrospective review of patient records, and compares findings from both.
Methods
We wished to explore the detail of current practices in depth, so we conducted FGs with experienced hospice and hospital/community palliative care clinicians, while simultaneously retrospectively reviewing hospital and hospice records of recently deceased patients. We then compared FG participants’ perceptions with documented data.
Focus Groups
No ethical approvals are required for research with clinicians in the United Kingdom, but other required approval was given (HRA ref. 16/HRA/1670) for conducting FGs with experienced physicians and nurses in a London hospice and on palliative care teams at a London hospital and linked community services. Potential participants were purposively selected for their level of experience in palliative care and informed of the study by email and/or team presentations. We arranged group discussions, organised by profession and setting, in the research offices or participants’ places of work.
All participants used pseudonyms and gave written informed consent. The research lead, B.V., facilitated each group, following a topic guide (TG; Supplemental material online). Another researcher (J.H. or S.D.) observed and took notes. B.V. and J.H. are both social scientists, with lengthy experience in palliative care research, and S.D. an experienced nurse and researcher.
Our TG began with an open question, followed by more focused questions exploring details of practice. The first FG served partly to pilot-test the TG. Only minor adjustments were made subsequently (to the question order and to include some additional prompts), so those data were retained. All FGs were audio-recorded and transcribed. B.V. and J.H. reflected throughout to determine when topics had been saturated.
Transcripts were checked against recordings and notes, then the more focused TG questions used to construct a framework for framework analysis.34,35 B.V. and J.H. independently analysed the transcripts using constant comparison,36,37 then discussed and agreed the final analysis.
Patient records
Simultaneously, and independently, L.B.-Q., a palliative care specialist registrar (SpR) working in another setting, retrospectively reviewed deceased patient drug charts and medical/nursing notes, working backwards from 31 December 2015 to locate records for 25 people in both hospice and hospital who received continuous subcutaneous infusion (CSCI) of midazolam in the last 24 h of life. We were advised that this work should be classified as an audit, which requires no ethical approvals, but permission to access the records from hospice management and the hospital’s clinical audit lead was required and obtained.
Patients’ ages, genders, diagnoses (cancer or non-cancer) and length of the final period under palliative care were extracted from all records, plus, for people who had received CSCI midazolam, doses of sedative and analgesic medication prescribed and administered during their final 72 h, together with any notes regarding aims of using sedation, monitoring of symptoms and/or depth of sedation, and/or medication titration.
We used standard conversion ratios to convert opioid analgesic doses to oral morphine equivalents (OMEs), as follows: 2:1 morphine:oxycodone; 100 µg fentanyl = 10 mg morphine; 2 mg oral morphine = 1 mg oral methadone; 2 mg oral methadone = 1 mg subcutaneous methadone. 38 Patients’ responses to medications vary individually, but we were unable to determine these from the reviewed records, and therefore employed an approximate, broadbrush calculation. We summarised all quantitative data by descriptive statistics. Written patient notes were analysed qualitatively, following the framework determined for the FG data, initially by L.B.-Q, and then independently by B.V., then discussed and agreed.
Results
Focus Groups
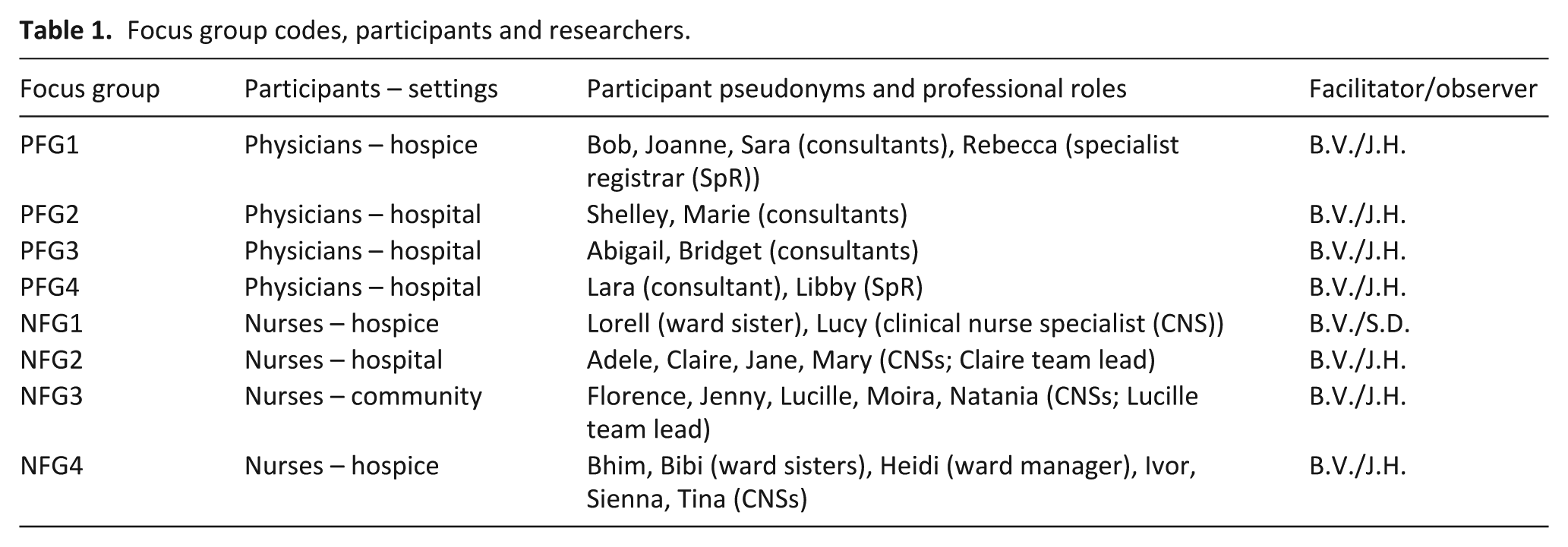
We conducted eight FGs with 27 clinicians: four groups with 10 physicians (8 consultants and 2 SpRs) and four groups with 17 senior nurses (11 clinical nurse specialists (CNSs), 2 team leads, 3 ward sisters and 1 ward manager) (Table 1).
Focus group codes, participants and researchers.
Following the open-ended question on what participants understood by sedation, the more focused TG questions (2–6) asked when and why sedative medications were used, which specific drugs and dosages were employed, and how effects, including depth of sedation, were monitored. FG participants’ responses to these more focused questions are presented below (section ‘Themes from responses to TG questions 2–6’ (a)–(c)), and findings from related patient record data follow (section ‘Patient records’ (a)–(c)). Findings from the more open, exploratory TG questions will be presented in later publications.
Themes from responses to TG questions 2–6
(a) Reasons for using sedative medications
All participants said sedative medication was primarily used for people experiencing intractable symptoms, particularly distress and/or agitation, at the end of life. All our participants, both physicians and nurses, also emphasised that sedatives were only used after exploring possible causes and other interventions for patient distress or agitation:
Participants said the aim when using sedatives was for patients to be ‘relaxed’, ‘calm’ or ‘comfortable’, not sedated per se, and stressed that the aim was to ‘settle’ the patient and/or their symptoms, and if a level of sedation occurred, this was a side effect, or a ‘by-product’. However, participants in most FGs discussed requests from family members that their relative be ‘made sleepy’, and the need for conversations with family and/or patients regarding how sleepiness, while not the aim, might be an outcome of the medication:
(b) Drugs and dosages used
Reinforcing comments on exploring other causes and treatment options before beginning sedative medication, participants all said they would reflect carefully before deciding to begin administering sedatives and always begin with low doses, usually of midazolam:
If patients’ experiences and/or past histories indicated that small dosages would be ineffective, participants said that they would adjust dosages and/or drugs accordingly: younger patients who are on, you know, huge amounts of opiates, and huge amounts of neuropathic agents, and you just know that when it comes to that terminal event, that 2.5 milligrams [of midazolam] … is not going to touch it, so I wouldn’t even start with something like that […] I’d easily start with 5 or 10 […] The very young or the very old […] who’ve never had anything more than a paracetamol … who’ve become a bit agitated, where I might just start at 1.25.
If midazolam proved ineffective, participants said they would then, after further consideration of causation, most commonly use levomepromazine, followed by phenobarbitone, and then, very rarely, propofol:
(c) Monitoring and assessing levels of sedation
Participants all said they used clinical observation rather than any objective monitoring tools when assessing the effects of sedative medication, and most said they did not only assess the patient personally, but also discussed with colleagues and/or patient relatives how they perceived patients’ situations. Some strongly preferred using clinical observation and judgement:
All our participants discussed looking for particular features of behaviour or patients’ movements and/or expressions when making their assessments:
Emergent related themes
Our TG did not include any questions on hastening death, but this theme emerged in all FGs.
(a) Relatives’ anxieties regarding hastening death
In most FGs, participants discussed the concerns and anxieties of patients’ relatives regarding syringe drivers, and their associations with hastening death. This theme arose particularly when speaking about starting with low dosages of medication:
(b) Non-specialist staff anxieties
Participants in many FGs also remarked that non-specialist staff were also anxious about hastening death:
Physician participants in two separate FGs used the phrase ‘dying at the end of a needle’ when discussing this issue in relation to the concerns of non-specialist staff, in both hospital and community settings, especially when injections were administered shortly prior to patients’ deaths:
Resonating with earlier discussions regarding how the decision to start sedative medication was not taken ‘lightly’ (NFG1, Lucy – section ‘Drugs and dosages used’ above), and although one hospital consultant commented that her team’s practice had always been cautious, many FG participants reflected on how staff concerns, and their consequent caution, possibly resulted from media representations of the Liverpool Care Pathway (LCP): 39
(c) Patients’ requests to be unconscious until death
In contrast to the discussion in all FGs of the concerns of relatives and non-specialist staff that patients’ deaths not be hastened, some FG participants also raised the opposite issue: wishes and/or explicit requests or expectations of some patients that they be made unconscious, so they could sleep until they died, and how difficult they and their colleagues found such requests:
One participant commented that occasionally patients themselves did not only ask to be made unconscious, but for their deaths to be hastened:
Patient records
To obtain 25 patients at each site who had received CSCI midazolam in their final 24 h necessitated retrieving 92 patient records (44 at the hospice, 48 at the hospital) prior to 31 December 2015 (Table 2).
Comparison of patients who received CSCI midazolam at the end of life with others.

CSCI: continuous subcutaneous infusion.
(a) Documented reasons for using sedative medications
For the 50 patients who received CSCI midazolam at the end of life, 44/50 records (88%) included both patient descriptions and plans/aims underlying introducing midazolam: C51 (M, age 80–84; starting dose 20 mg; 20–40 mg prescribed): Agitated++, distressed, trying to climb out of bed. No response to 2.5 mg midazolam therefore 5mg midazolam given with some response. Difficulty with oral medicines … Plan … Commence CSCI … Midazolam 20mg.
The other six records (12%) did not specifically state reasons or give a detailed plan: L22 (M, age 35–39; starting dose 5 mg; 5 mg prescribed): Patient looks like he is actively dying. Unable to rouse with voice /// Plan: End of life care.
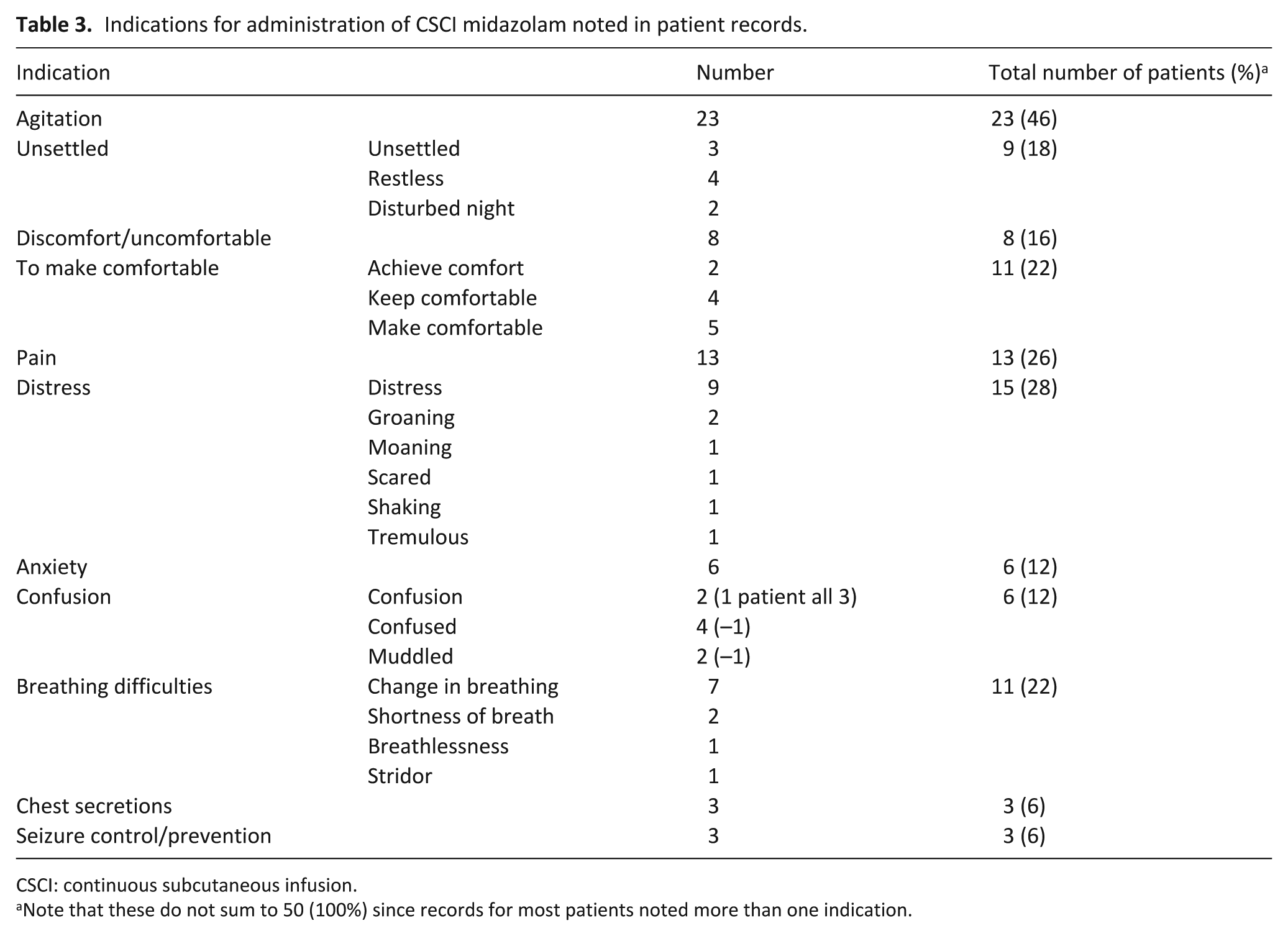
Reasons for starting CSCI midazolam were explicitly stated in 28/50 records (56%), with the most frequent being agitation (23/50; 46%). Other frequent indications were being unsettled (including restlessness or having a disturbed night), discomfort, pain, distress, anxiety, confusion and breathing difficulties (Table 3):
Indications for administration of CSCI midazolam noted in patient records.
CSCI: continuous subcutaneous infusion.
Note that these do not sum to 50 (100%) since records for most patients noted more than one indication.
C49 (F, age 50–54; starting dose 10 mg; 10–30 mg prescribed): Difficult day … Anxious+++, finding it a lot to cope with at times, finds lorazepam helpful … Anxiety a big problem for [her] at the moment … Add 10mg/24h Midazolam to CSCI to help with anxiety.
In total, 10 patient records noted just one indication; the other 35 noted more than one, most frequently (19/50) two (e.g. both agitation and distress). The largest number of indications was seven (for one patient). We found no pattern in the combination of indications recorded: L43 (F, age 30–34; starting dose 5 mg; 5 mg prescribed): Patient reviewed due to deterioration in condition. Has required a number of PRNs in last 24h … Currently breathing fast, has pain, feels sick, obviously dying … [She] asked what will happen next. I have explained we will review SC pump regularly and increase as needed to keep her comfort paramount. Plan: Suggest increase S/C pump to 80mg oxycodone. Use midazolam S/C to help breathing.
Some records noted patients’ behaviours and/or expressions, and some also included comments on the thoughts and impressions of patients’ relatives: L47 (M, age 80–84; starting dose 5 mg; 5 mg prescribed): Unresponsive. Small twitches/jerking noted. Frowning and screwing up face. Daughter present and notes that [he] often makes this face & she thinks it is a sign of pain – I agree … Plan … Start CSCI 10 mg oxycodone, 5 mg Midazolam, & 1.6mg hyoscine … PRN medications as needed.
One of the 50 records noted that the patient had expressed the wish to die more quickly: C54 (F, age 70–74; starting dose 15 mg; 10–20 mg prescribed): Had a very distressing night, lots of pain. 4× oramorph overnight … Had bowel motion through her vagina. She is very tearful this morning and ‘wants it all over’ and does not want to be here. Wants to die quicker. Explained that we cannot make it happen quicker but we can make her more comfortable and that maybe a SD is appropriate. Slept every two hours – diazepam did not help as well as before … Plan: Trial of syringe driver – nervous about needles but will try.
(b) Documented drugs and dosages used
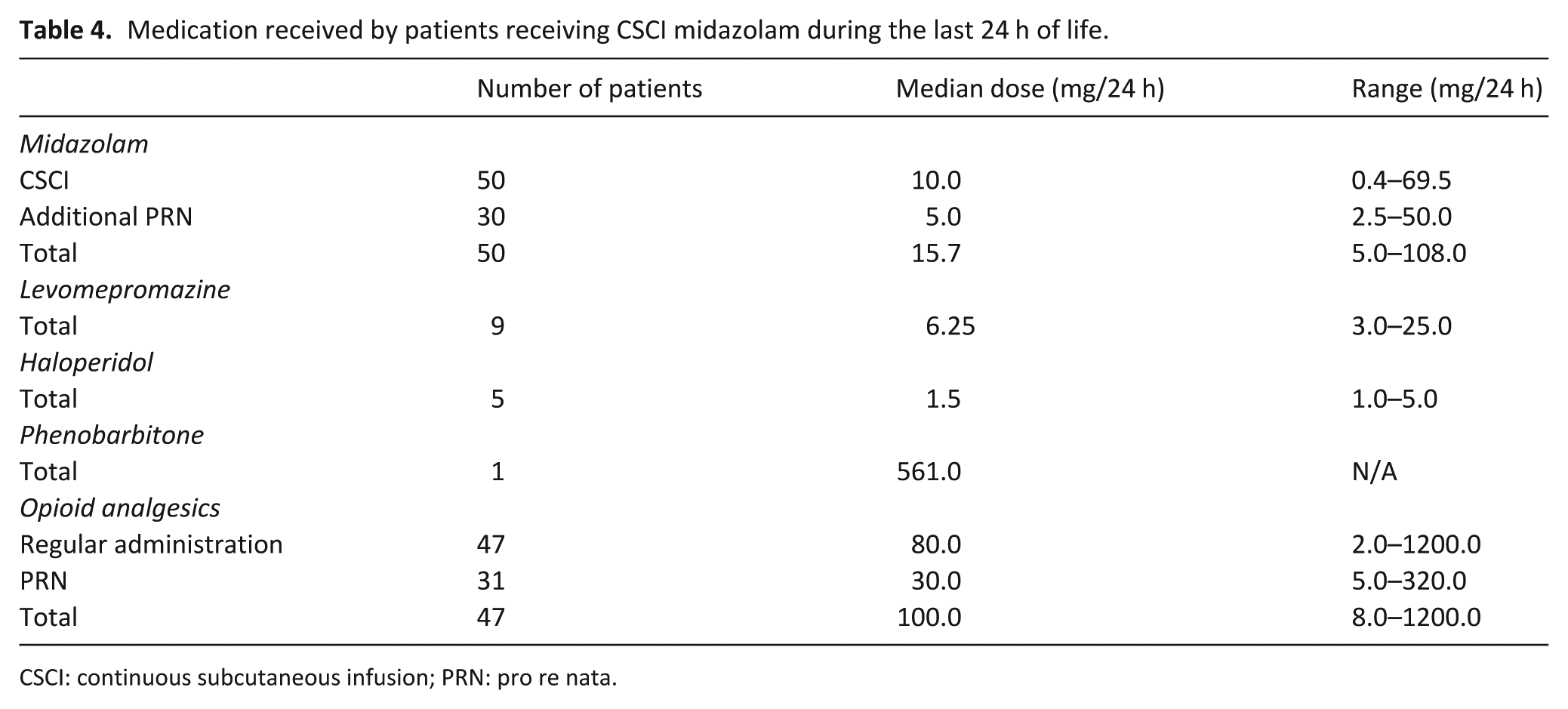
Patients began CSCI midazolam a median of 1.5 days (range: 0–19) before death. The median dose of CSCI midazolam received in the last 24 h of life was 10 mg (range: 0.4–69.5) (Table 4 and Figure 1), with 30/50 patients (60%) also receiving pro re nata (PRN, or ‘as required’) midazolam – median PRN dose 5 mg/24 h (range: 2.5–50). The median total dose of midazolam received in the last 24 h of life was 15.7 mg/24 h (range: 5–108 mg).
Medication received by patients receiving CSCI midazolam during the last 24 h of life.
CSCI: continuous subcutaneous infusion; PRN: pro re nata.
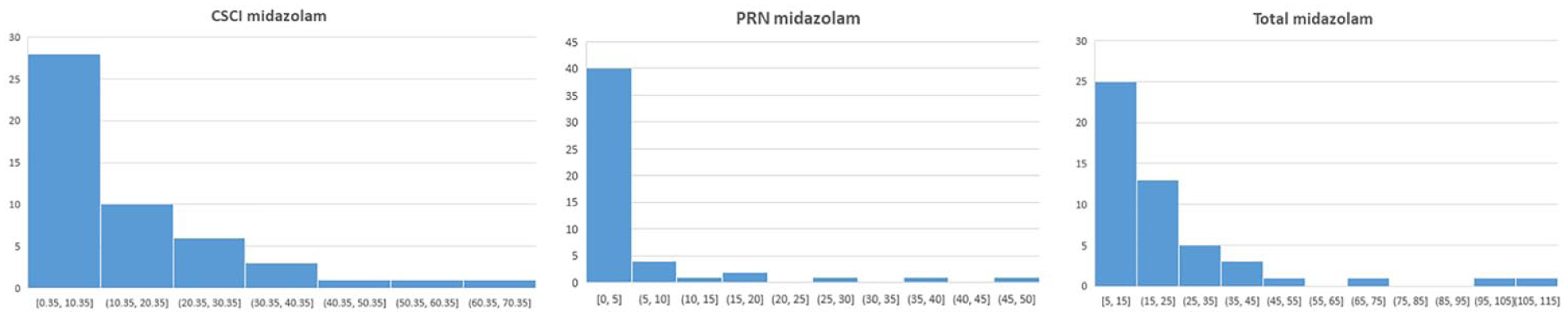
CSCI, PRN, and total midazolam for all 50 ‘CSCI patients’ in the last 24 h of life.
The majority of patients (47/50; 94%) who received CSCI midazolam also received CSCI opioid analgesia (median dose OME: 80 mg/24 h), with 31/50 (62%) also receiving PRN analgesia in the last 24 h of life (median total dose OME: 100 mg/24 h). In total, 15 of these patients (30%) also received additional sedatives: nine (18%) levomepromazine, five (10%) haloperidol and one (2%) phenobarbitone (Table 3). Three patients (6%) received hyoscine for chest secretions.
(c) Documented monitoring of level of sedation
No patient records contained any indication of structured monitoring. Three sets (all for patients who died 1–3 h after commencing CSCI) understandably did not document additional sedative doses nor clinical observations/patient descriptions. All other records (47/50; 94%) documented either or both; most (31/50; 62%) included both, as illustrated below (our emphases).
Low final doses of midazolam
L35 (F, age 75–79; total midazolam in the last 24 h of life 5.2 mg): Asked to review [patient] after difficult night of distress and coffee ground vomiting. Ryles tube passed since and patient sleeping. D[iscussion]/w[ith] nursing staff, patient appears to have massively deteriorated from last wk … Currently comfortable, inappropriate to move, needs EoL paperwork … Midazolam S/C to keep comfortable. Not reactive to verbal and pain stimuli … comfortable in bed … Plan … Midazolam PRN. Already on SD – seems comfortable so no changes for now Pain poorly controlled overnight and today in the morning … Patient with eyes closed, responds to verbal and pain stimuli with groaning. Doesn’t look comfortable. Cheyne-Stokes breathing … Plan: Increase SD midazolam (from 2.5 to 5) and oxycodone (from 10 to 15). At the moment SD with levomepromazine 12.5 and metoclopramide 30 + midazolam 5 + oxycodone 5. Palliative care to kindly review patient and analgesia, please. Seems comfortable now but was restless last night. Team has increased midazolam to 5mg and oxycodone to 15mg today. Continue with same.
High final doses of midazolam
Notes for patients receiving large final doses were similar to those for patients receiving low doses, that is, recording also solely clinical assessments of their level of comfort. The following extract indicates the monitoring of a hospice patient who received 107.9 mg in her last 24 h of life (the highest final total dose in all patient records reviewed): C53 (F, age 45–49; total midazolam in the final 24 h 107.9 mg (57.9 mg CSCI + 50.0 mg PRN): Patient in bed, drowsy. Was agitated earlier this morning … SD: Midazolam 60mg. ×3 Midazolam 10mg overnight. Discussed increasing sedation to keep comfortable with visiting friend. Prognosis short. Plan: … Add phenobarbitone 1600mg via 2nd SD [She] has been agitated at times and needed medication to help settle – We have added in 2nd syringe pump with additional medication (Phenobarbital) to help settle her and keep calm.
Patient is comfortable
Midazolam administered twice during the night for distress and agitation Appeared agitated at time of handover, 10mg of midazolam given with short effect.
We found no noteworthy differences between records such as those above for patients who received large doses of sedative medication at the ends of their lives, and those for the majority, who received low doses.
Discussion
Main findings
Our FG participants’ statements that when administering sedative medications they generally started with low dosages of midazolam, using other sedatives, infrequently, as second-line treatment, were corroborated by data from patient records. Just over half (50/92; 54%) of retrieved records indicated use of CSCI midazolam at the end of life, just under half of those (24/92; 26% of all records reviewed) received 10 mg or more. The median CSCI dose was 10 mg/24 h; the median total dose was 15.7 mg/24 h, lower levels of sedative use than found in the two recent retrospective reviews in the United Kingdom.30,31 Patient records showed infrequent use of other sedatives, of which the most frequent was levomepromazine (9/50 patients; 18%).
Our FG participants said they used sedatives primarily for exceptional distress or agitation, occasionally for other intractable symptoms, aiming for patient comfort, not unconsciousness. Again, data from patient records supported these statements; the most frequent indications were agitation, discomfort, distress, and pain. No FG participants mentioned following any guidelines, and all said that they never used structured tools to determine the effects of medications on patients, but rather clinical observations plus consultation with colleagues and relatives. The reviewed patient records also corroborated this. Records for patients receiving high or low total doses did not differ in their content, and we found no indication of any systematic structured monitoring, regardless of the sedative dose received (although high levels of sedative medication do not necessarily mean loss of consciousness).
In all our FGs, participants extended the discussion beyond the TG to the perceptions of some patients, relatives and non-specialist staff that using medication via syringe driver at the end of life implied hastening death. All spoke of the consequent need to manage related concerns, with some linking these negative perceptions with critical representations of the LCP. From the opposite perspective, some participants also discussed needing to address requests from some patients that they be made unconscious until they died, or even actively assisted in dying. One set of patient records noted a discussion with a patient who had indicated a desire for her death to be hastened.
Strengths and limitations
A strength of our study is its multi-method approach, triangulating qualitative findings with data from pre-existing patient records, which corroborated FG participants’ comments. However, as a small-scale exploratory study of current practice in two London settings, our study is limited in its generalisability, although our qualitative findings support those from recent research interviews in the United Kingdom. 28
Another limitation is that we assessed patient records, rather than directly observing patient care, and the recorded data cannot be taken to exhaustively represent patients’ conditions. The information recorded was what clinicians chose to document, and factors such as time pressure might affect their choices. Furthermore, because systematic objective monitoring tools were not used, the notes reflect clinicians’ subjective decisions regarding what to document, and in what terms. Although some terminology was common, occasionally clinicians used their own personal vocabularies and language which was not necessarily standard or consistent between individuals, for example, ‘muddled’ and ‘confused’ (Table 3). Our analysis therefore sometimes required interpretation regarding equivalent meanings.
What this study adds
There is no standard practice for use of sedation at the end of life; it is known that practice varies internationally, and locally by setting, and that guidelines are inconsistent.13,14 Our findings indicate that clinicians’ practices in these London settings, in relation to drugs and dosages used and indications for usage, align with the EAPC framework’s 15 recommendations for proportionate use of sedatives and echo previous qualitative findings with UK palliative care professionals. 28 We found lower levels of sedative use in both London settings than found in previous patient record reviews in other UK settings.29–31
The EAPC also recommends that sedative use for imminently dying patients should be regularly monitored, with patient comfort being the primary parameter, if this is the aim. 15 All records examined for patients receiving CSCI midazolam demonstrated monitoring of patient comfort, with no evident differences in approach between dosage levels.
Implications for practice
Our clinician participants did not mention following any guidelines, whether EAPC or otherwise, but practice when using sedatives at the end of life in these two London settings meets EAPC recommendations for proportionate use. However, the EAPC also recommends systematic, objective monitoring of sedative effects, which we did not find. Such monitoring might be beneficial by facilitating consistency in approach, thereby ensuring that patients are neither over- nor under-sedated, and also in clinicians’ terminology. Among other things, when handing over or comparing notes between clinicians, systematic language would help clarify whether clinicians’ perceptions are equivalent when using terms such as ‘muddled’ or ‘confused’, ‘agitated’ or ‘restless’, ‘settled’ or ‘comfortable’, and so ensure continuity of care.
Conclusion
Our FG findings, corroborated by patient record data, demonstrate that, when using sedatives at the end of life, current practice in the London settings studied is cautious and proportionate, as per EAPC recommendations. Clinicians first consider using sedatives if patients are agitated and/or experiencing considerable anxiety or distress, and, if used, begin with low doses if possible, aiming primarily to increase patient comfort. Sedation, if it occurs, is a by-product, so this approach is best described as proportionate sedation. The only variation we found from EAPC recommendations for drugs, dosages and monitoring was that clinicians did not objectively monitor sedative effects, even when using moderate–high doses of sedatives. The next step in this research programme therefore explores the feasibility of introducing such objective monitoring in clinical care.
Supplemental Material
826007_Topic_guide_for_usual_practice_Clinicans_v7_(1) – Supplemental material for Palliative care specialists in hospice and hospital/community teams predominantly use low doses of sedative medication at the end of life for patient comfort rather than sedation: Findings from focus groups and patient records for I-CAN-CARE
Supplemental material, 826007_Topic_guide_for_usual_practice_Clinicans_v7_(1) for Palliative care specialists in hospice and hospital/community teams predominantly use low doses of sedative medication at the end of life for patient comfort rather than sedation: Findings from focus groups and patient records for I-CAN-CARE by Bella Vivat, Lucy Bemand-Qureshi, Jane Harrington, Sarah Davis and Patrick Stone in Palliative Medicine
Footnotes
Acknowledgements
We thank the peer reviewers and editors for their helpful comments and feedback. We also gratefully thank all the FG participants for their time and thoughtful contributions, and colleagues at both sites for help with accessing patient records. Thanks also to all the current and former members of the study advisory and working groups: Bridget Candy, Alice Colum, Anna Gola, Tariq Husain, Yana Kitova, Anna-Maria Krooupa, Nuriye Kupeli, Philip Lodge, Rebecca Lodwick, Jon Martin, Stephen McKeever, Liz Sampson, Liz Thomas, Adrian Tookman, Vinnie Nambisan and Tim Wehner. Particular thanks to Hilary Bird and Kathy Seddon (Marie Curie Expert Voices PPI representatives on the Advisory Group). We thank Matthew Bemand-Qureshi for his help with statistical analysis, and Mia Davis, Anna Floyer and Nuriye Kupeli for transcribing. Parts of the data reported on in this paper were presented at the 15th World Conference of the EAPC, Madrid, 18–20 May 201740, 41, and at the Marie Curie/Royal Society of Medicine (Palliative Care Section) Palliative Care Research Conference, 6 October 2017 42 . P.S. conceived the overall study design and funding application. B.V. and P.S. conceived the detailed design and prepared the initial protocol and study documents. B.V., J.H., S.D. and L.B.-Q. performed the data collection. B.V., J.H., P.S. and L.B.-Q. performed the analysis. B.V. prepared the initial draft of the paper and performed initial revisions and response to editors/reviewers following peer review. All authors performed further drafting and revisions, approval of the final version, revisions and approval of resubmission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by Marie Curie (grant no. MCCC-FPO-16-U). BV, JH, SD, and PS are also supported by the UCLH NIHR Biomedical Research Centre.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
