Abstract
Background:
Maintaining quality of life including physical functioning is highly prioritized among older cancer patients. Geriatric assessment is a recommended approach to identify patients with increased vulnerability to stressors (frailty). How frailty affects quality of life and physical functioning in older cancer patients has scarcely been investigated.
Aim:
Focusing on physical functioning and global quality of life, we investigated whether frailty identified by a geriatric assessment was associated with higher risk of quality-of-life deterioration during cancer treatment and follow-up.
Design:
Prospective, observational study. Patients were classified as frail or non-frail by a modified geriatric assessment. Quality of life was measured using the European Organization for Research and Treatment of Cancer Core Quality-of-Life Questionnaire at inclusion, 2, 4, 6 and 12 months.
Setting:
Eight Norwegian outpatient cancer clinics.
Participants:
Patients ⩾70 years with solid tumours referred for palliative or curative systemic medical cancer treatment.
Results:
Among 288 patients included, 140 (49%) were frail and 148 (51%) non-frail. Frail patients consistently reported poorer scores on all functioning and symptom scales. Independent of age, gender and major cancer-related factors, frail patients had significantly poorer physical functioning and global quality of life during follow-up, and opposed to non-frail patients they had both a clinically and statistically significant decline in physical functioning from baseline until 12 months.
Conclusions:
Geriatric assessment identifies frail patients with increased risk of physical decline, poor functioning and high symptom burden during and following cancer treatment. Frail patients should therefore receive early supportive or palliative care.
Frailty identified by geriatric assessment is associated with increased risk of death and treatment complications in older cancer patients.
Although geriatric assessment is recommended for older cancer patients, it is rarely implemented into clinical practice.
Studies investigating the impact of frailty on highly relevant outcomes like physical functioning and quality of life are scarce.
Frailty identified by geriatric assessment independently predicts a clinically significant decline in physical functioning.
Frailty is associated with worse global quality of life, poorer functioning and a higher symptom burden throughout the cancer trajectory
Including routine geriatric assessment for older cancer patients undergoing systemic medical cancer treatment will aid oncologists in identifying frail patients who need early supportive and palliative care.
Introduction
Prolonging survival is usually considered the main goal of cancer care. However, maintaining or improving quality of life can be equally important. This applies especially to older patients, who have poorer survival in comparison with their younger counterparts and may be less willing to exchange current quality of life for smaller survival benefits.1,2 The quality-of-life concept embraces multiple dimensions: emotional, social, existential as well as physical, the latter including aspects such as patient-reported somatic symptoms and physical functioning. Physical functioning is strongly associated with independent living, which is highly prioritized among older patients,3,4 and is also a key driver for how they perceive their overall quality of life.5,6 Thus, making appropriate treatment decisions for older cancer patients requires knowledge on how quality of life may be affected and ability to identify patients at risk of deterioration. Particular attention to physical functioning seems essential.
Frailty is defined as increased vulnerability to adverse changes in health status 7 and is associated with increased mortality, postoperative complications and intolerance to cancer treatment.8,9 Frail patients have been found to have poorer quality of life than non-frail patients,10–12 but longitudinal studies investigating the impact of frailty on quality of life during and after cancer treatment are scarce. Results from those available are not consistent, having shown both similar changes in quality-of-life trajectories of frail and non-frail patients10,11 as well as accelerated decline of some dimensions among frail patients. 13
A challenge to all frailty research is the lack of universally accepted operational criteria. Over 70 different methods for measuring frailty have been developed, most of which are linked to the two dominating pathophysiological theories of frailty: the physical frailty phenotype and the cumulative deficit model.14,15 In the oncology literature, geriatric assessment is the recommended approach to identifying frailty 14 and to guide treatment decisions for older patients. 16 This approach includes a systematic assessment of areas such as functional status, mobility, cognitive function, comorbidity and geriatric syndromes.8,16 Still, geriatric assessment remains to be widely implemented into oncology practice, perhaps hampered by its comprehensiveness. Simpler frailty screening tools are more time-efficient and might be easier to implement into clinical practice, but their lower sensitivity and specificity is a challenge. 17 Thus, geriatric assessment is considered the gold standard, 14 although screening tools may be used to select patients for a complete geriatric assessment. 18 There is, however, no general agreement on how frailty should be defined based on a geriatric assessment. Varying domains and thresholds have been applied in different studies, 8 but the criteria as proposed by Balducci and Extermann 19 have commonly been used.20,21
We have formerly demonstrated that frailty identified by a modified geriatric assessment and a modification of the Balducci criteria22,23 was independently predictive of survival in cancer patients ≥70 years of age. 24 In this study, targeting the same population, we aimed at investigating whether frailty was associated with higher risk of quality-of-life deterioration during treatment and follow-up. Our main hypothesis was that patients classified as frail upon start of treatment would experience a steeper decline in both physical functioning and global quality of life than non-frail patients.
Materials and methods
Patients
Patients were consecutively recruited from January 2013 until April 2015 at eight Norwegian outpatient oncology clinics (two university hospitals and six local hospitals). Eligible patients were ⩾70 years and referred for systemic medical cancer treatment (chemotherapy, hormonal or targeted therapy) with a histologically confirmed solid tumour (newly diagnosed or first relapse after previous curative treatment). Patients provided written, informed consent.
Assessments
Oncologists reported cancer type (10th revision of the International Statistical Classification of Diseases and Related Health Problems (ICD-10)), stage of disease, planned treatment and the Eastern Cooperative Oncology Group (ECOG) performance status. Data on administered treatment were retrieved from the patients’ medical records. Physical functioning and global quality of life was assessed by the European Organisation for Research and Treatment of Cancer Quality-of-Life Core Questionnaire (QLQ-C30) 25 at inclusion and after 2, 4, 6 and 12 months. QLQ-C30 consists of 30 questions comprising five functioning scales, nine symptom scales/items and a global quality of life scale. The functioning scales include physical, role, social, cognitive and emotional functioning. Symptoms include fatigue, pain, nausea/vomiting, sleep disturbances (insomnia), appetite loss, diarrhoea, dyspnoea and constipation and financial impact. The raw scores are transformed into scales from 0 to 100 points. 26 Higher scores on the functioning and global quality-of-life scales represent better functioning, whereas higher scores on symptom scales/items indicate a higher symptom burden.
Frailty was identified by a geriatric assessment which we have referred to as modified since it was not performed by an interdisciplinary team, but by trained oncology nurses and patients’ self-report, 24 using well-known and validated instruments for each included domain27–31 (Table 1). Our frailty definition was predefined, and following the Balducci criteria, patients were categorized as frail if they fulfilled at least one of the following: dependencies on activities of daily living, significant comorbidity or one or more geriatric syndromes (cognitive function, depression, malnutrition and falls). Similar to Kristjansson et al., 22 we included polypharmacy as a criterion and added impairment according to Timed Up and Go, 27 a sensitive and specific measure of frailty. 32 Cut-off values for each domain were chosen in line with former reports and practice (Table 1).23,33–40 A detailed explanation is found in a previous paper. 24 To screen for deficits in activities of daily living, a question from the QLQ-C30 physical functioning scale (‘Do you need help with eating, dressing, washing yourself or using the toilet?’) was used.
The modified geriatric assessment a .
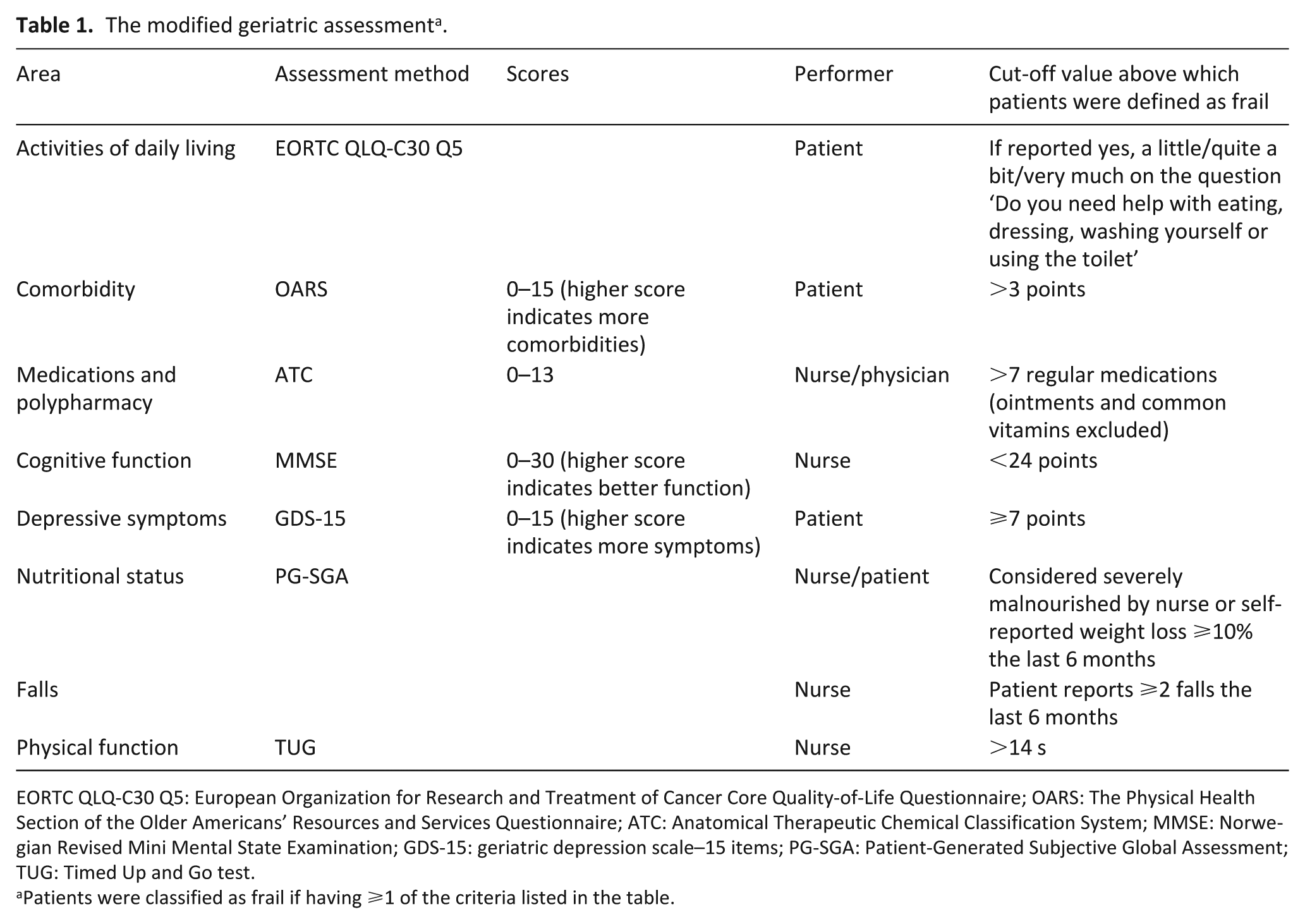
EORTC QLQ-C30 Q5: European Organization for Research and Treatment of Cancer Core Quality-of-Life Questionnaire; OARS: The Physical Health Section of the Older Americans’ Resources and Services Questionnaire; ATC: Anatomical Therapeutic Chemical Classification System; MMSE: Norwegian Revised Mini Mental State Examination; GDS-15: geriatric depression scale–15 items; PG-SGA: Patient-Generated Subjective Global Assessment; TUG: Timed Up and Go test.
Patients were classified as frail if having ⩾1 of the criteria listed in the table.
Statistical analyses
Medical and sociodemographic factors were compared between frail and non-frail patients by independent samples t-tests or χ2-test. Our predefined main endpoints were changes in physical functioning during the first 2 months of follow-up (primary) and changes in physical functioning and global quality of life during 12 months (secondary). Changes during 12 months for the remaining QLQ-C30 scales and items were assessed by exploratory analyses using the same approach as for the main endpoints.
Differences between frail and non-frail patients in changes over time were assessed by linear mixed models. All models included random intercepts for cancer clinics and for patients nested within cancer clinics to account for intra-patient correlations due to repeated measurements and possible within-clinic cluster effect. The models also included fixed effects for frailty group, time (as second-order polynomial to account for non-linear trends in models assessing data on 12-month follow-up), and the interaction term between frailty group and time (frail × time). A significant interaction term would imply that there were differences in change between frail and non-frail patients. Models adjusting for age, sex, cancer type, performance status, stage and treatment were also estimated. Treatment was classified as (1) curative treatment, that is, patients referred for neoadjuvant chemotherapy treatment, adjuvant chemotherapy and/or endocrine treatment after curative surgery or curative radiotherapy; (2) palliative chemotherapy; (3) other palliative systemic cancer treatment and (4) non-systemic palliative treatment the first 2 months after inclusion (i.e. radiotherapy, surgery or palliative care). Performance status was classified as 0–1 or 2–4 and stage as local, locally advanced or metastatic. The results were tabulated as regression coefficients with standard errors (SE) and p values for the primary and secondary analyses of physical functioning and global quality of life. The results from unadjusted models were also presented graphically as estimated mean values with 95% confidence intervals (CI) for all QLQ-C30 scales/items. Within- and between-group differences with the corresponding 95% CI and p values were calculated from the models. Significance level was set at 5%. A difference of ⩾10 points on the functional and symptom scales/items was considered a clinically significant change. 41
Missing values in QLQ-C30 multi-item scales were imputed according to the official manual if at least half of the scale had been answered. 26 The study was approved by the Regional Committee for Medical and Health Research Ethics South East Norway 09.02.2012 (Reference number 2012/104) and registered at clinicaltrials.gov (NCT01742442).
Results
Patients
From January 2013 to April 2015, a total of 307 patients were included. One patient withdrew consent and 18 had missing baseline questionnaires and therefore incomplete geriatric assessments. Thus, 288 (94%) patients were eligible for the present frailty study. A total of 140 patients (49%) fulfilled one or more of the predefined criteria and were categorized as frail. The most frequent deficits were comorbidity (n = 82, 28%), malnutrition (n = 43, 15%), polypharmacy (n = 37, 13%) and depressive symptoms (n = 35, 12%). In all, 40 patients (14%) had deficits in physical functional aspects: activities of daily living (12 patients), Timed Up and Go (18 patients) and number of falls (10 patients). Nine patients (3%) had cognitive impairment. Of the 140 patients categorized as frail, 67 (48%) patients had two or more registered deficits. Only one patient was classified frail based on the activities of daily living criterion alone, which was screened for by using question 5 from the physical functioning scale of QLQ-C30.
The patients’ baseline characteristics are shown in Table 2. Mean age was 76.9 (5.1) years, 56% were male and the most common cancer types were colorectal (29%), lung (21%) and prostate cancer (19%). The majority of patients had distant metastases (56%), and overall, 68% received palliative treatment. A higher percentage of frail compared to non-frail patients had lung cancer, distant metastases, performance status 2–4 and received palliative chemotherapy.
Baseline patient characteristics according to frailty status.
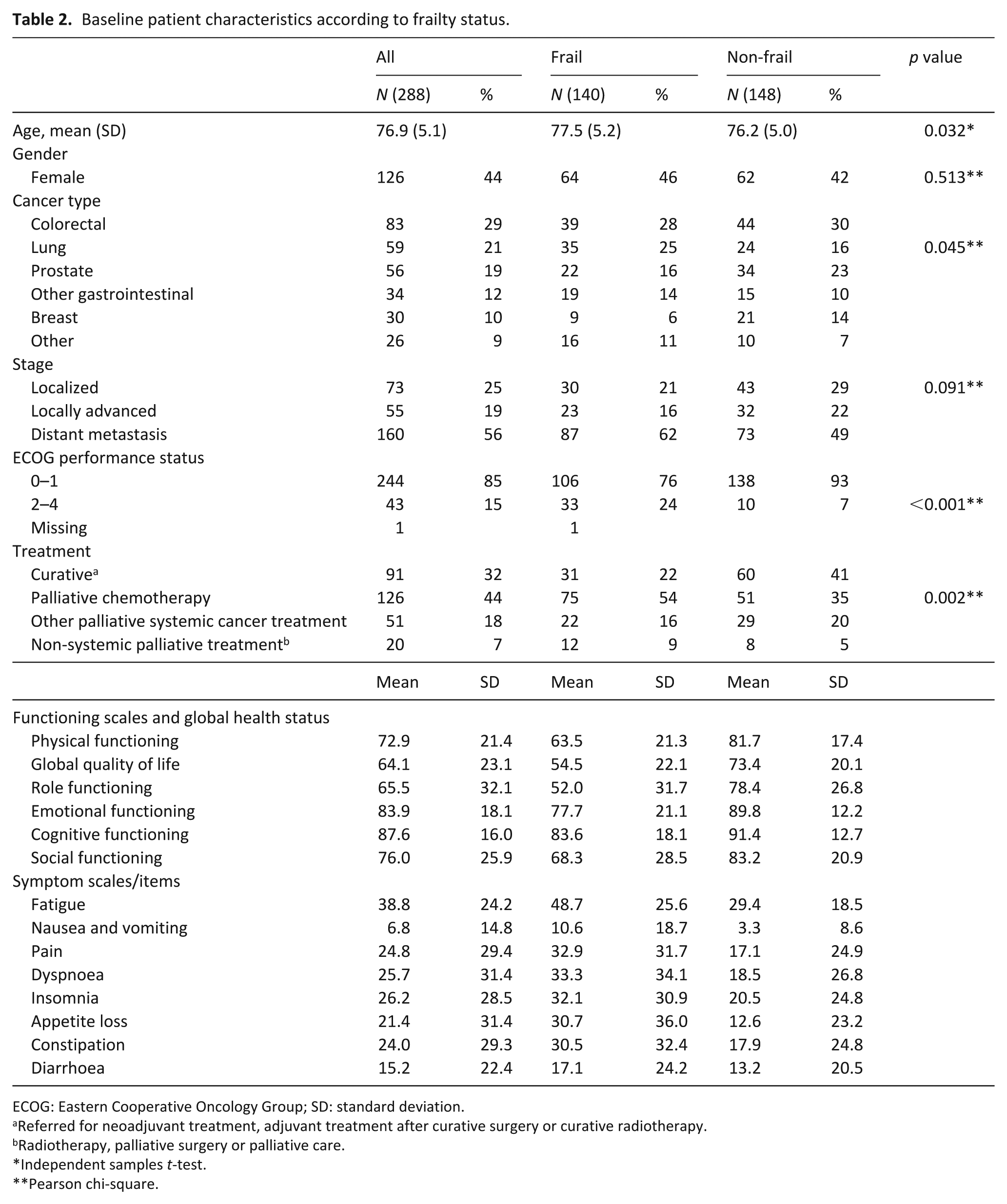
ECOG: Eastern Cooperative Oncology Group; SD: standard deviation.
Referred for neoadjuvant treatment, adjuvant treatment after curative surgery or curative radiotherapy.
Radiotherapy, palliative surgery or palliative care.
Independent samples t-test.
Pearson chi-square.
At 2, 4, 6 and 12 months of follow-up, 13 (5%), 27 (9%), 52 (18%) and 93 (32%) patients of the overall cohort had died. Median overall survival was shorter among frail than non-frail patients (15 vs 29 months). 24 The first 12 months, 83 (59%) of frail and 112 (76%) of non-frail patients were alive, resulting in relative risk of death of 1.7 (95% CI: 1.2–2.4) for frail compared to non-frail patients. The proportion of completed questionnaires ranged between 89% and 95% for those alive at the various assessment points (Figure 1). The mean proportion of missing items ranged from 0.51% to 0.96%.
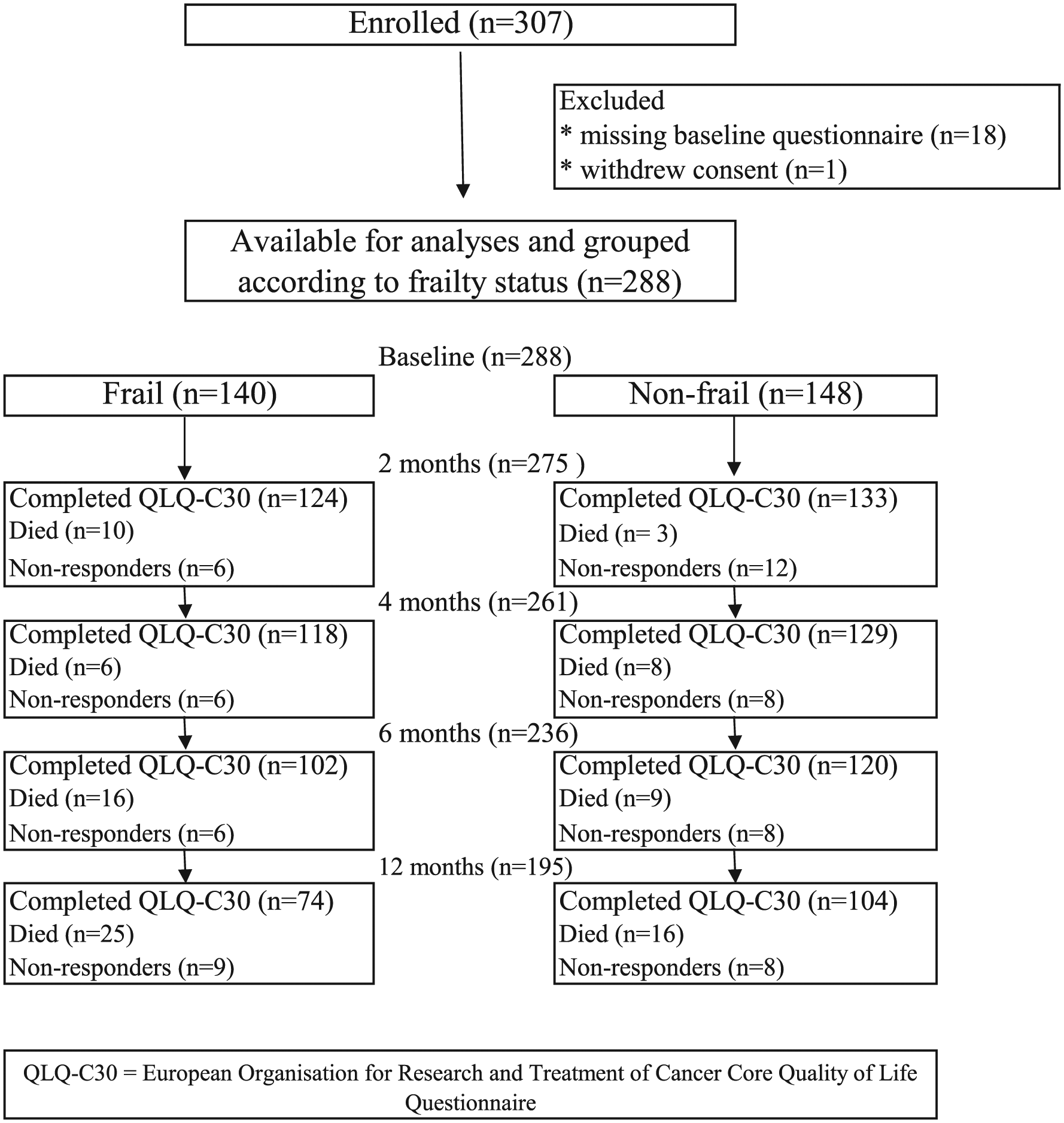
Flow diagram of patient enrolment and follow-up.
Quality-of-life analyses
At baseline, frail patients reported poorer functioning and more symptoms than non-frail patients on all scales/items (Table 2). Both frail and non-frail patients reported a statistically, but not clinically significant decline in physical functioning from baseline to 2 months. The decline was not significantly different between frail and non-frail patients (unadjusted model: p = 0.181, adjusted model: p = 0.218). According to the unadjusted linear mixed model, there were, however, statistically significant differences in physical functioning scores between the two groups in disfavour of frail patients, mean 18.2 (95% CI: 13.3–23.1) points at baseline and 15.0 (CI 9.9; 20.0) points at 2 months (p < 0.001; Figure 2, Table 3). The differences remained statistically significant when adjusting for age, gender, cancer type, stage, performance status and treatment (12.2 (95% CI: 7.5–16.9) points at baseline and 9.2 (95% CI: 4.4–14.1) at 2 months; p < 0.001; Figure 2, Table 3).
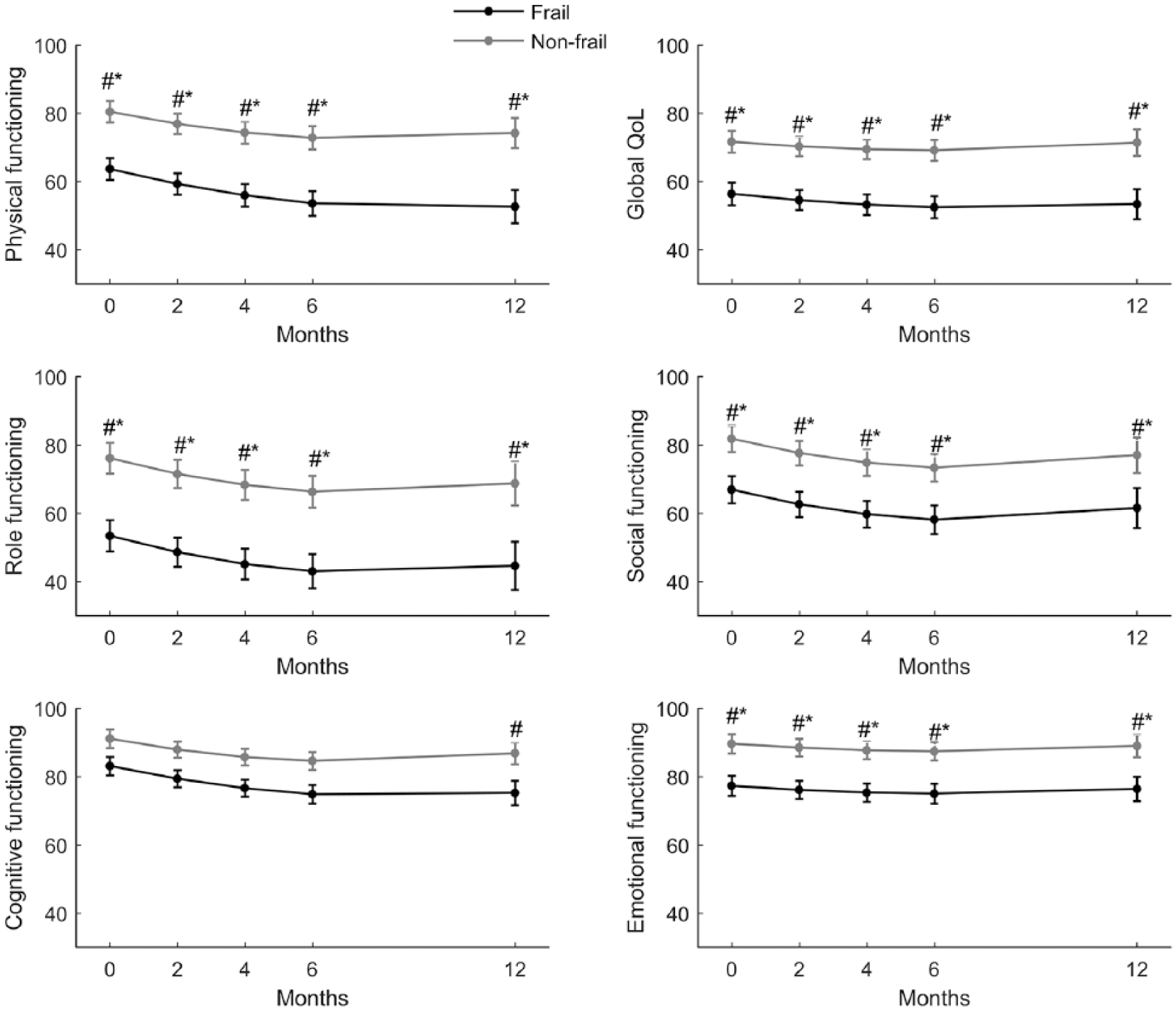
Global quality-of-life and function scores for frail and non-frail patients at baseline and at 2, 4, 6 and 12 months of follow-up, according to unadjusted mixed linear models.
Linear mixed models of the trajectories of physical functioning in frail versus non-frail patients during 2 months of follow-up and of physical functioning and global quality of life during the first 12 months of follow-up.
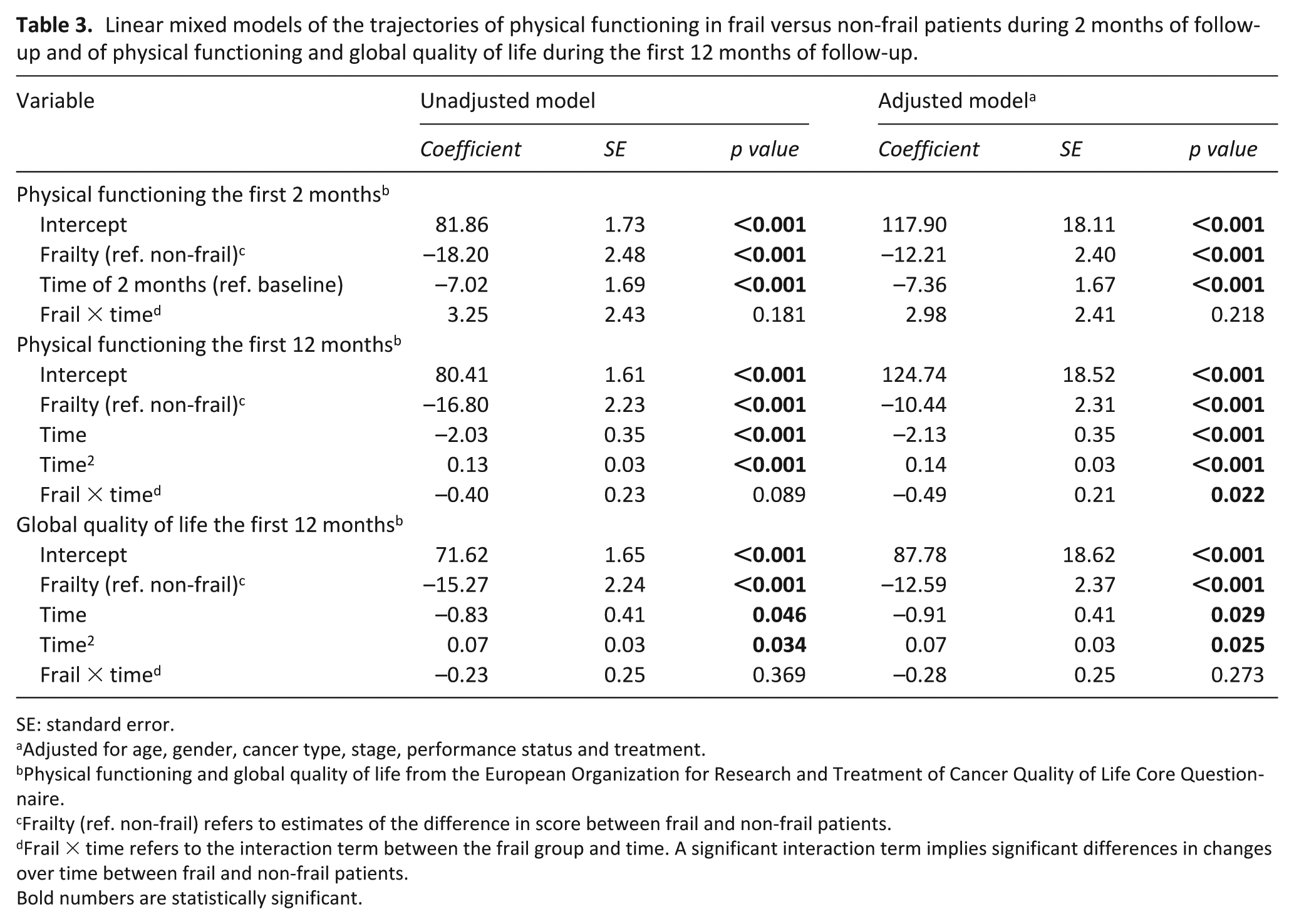
SE: standard error.
Adjusted for age, gender, cancer type, stage, performance status and treatment.
Physical functioning and global quality of life from the European Organization for Research and Treatment of Cancer Quality of Life Core Questionnaire.
Frailty (ref. non-frail) refers to estimates of the difference in score between frail and non-frail patients.
Frail × time refers to the interaction term between the frail group and time. A significant interaction term implies significant differences in changes over time between frail and non-frail patients.
Bold numbers are statistically significant.
For our secondary endpoint, physical functioning during 12 months of follow-up, a statistically significant decline was found for non-frail patients from baseline to 6 months and for frail patients from baseline to both 6 and 12 months. Only frail patients had a clinically significant (⩾10 points) decline. In unadjusted models, the decline in physical functioning for frail and non-frail patients was not significantly different (p = 0.089; Table 3, Figure 2). However, when adjusting for age, gender, cancer type, stage, performance status and treatment, the decline was found to be significantly steeper for frail patients (p = 0.022; Table 3). Thus, the observed difference in scores in disfavour of frail patients during the first 2 months increased throughout the follow-up period and remained statistically and clinically significant, both according to unadjusted (Figure 2, Table 3) and adjusted models (Table 3; p < 0.001).
For global quality of life during 12 months of follow-up, there was no significant difference between frail and non-frail patients in the course of changes (p = 0.369 in unadjusted models; p = 0.273 in adjusted models; Table 3). Both models demonstrated that frail patients had statistically and clinically significantly worse scores compared to non-frail patients at all assessment points (p < 0.001; Figure 2, Table 3).
Unadjusted trajectories for frail and non-frail patients for the remaining functioning and symptom scales are shown in Figures 2 and 3. Differences that were both statistically and clinically significant according to unadjusted and adjusted analyses are indicated. In the adjusted model, frail patients had a clinically and statistically significant decline in role functioning from baseline to 6 months (p < 0.001). None of the other scales showed any clinically significant changes from baseline in the adjusted models, neither in frail nor non-frail groups. Except for diarrhoea (adjusted model, p = 0.023), with a statistically but not clinically significant increase in symptoms from baseline to 6 months for frail patients, the course of the trajectories was not significantly different between the groups. However, adjusted models showed that frail patients had statistically and clinically significant more constipation (p < 0.01) and worse role- (p < 0.001), social- (p < 0.01) and emotional functioning (p < 0.01) at all assessments. Accordingly, significant differences between the frailty groups were found at some but not all assessment points for dyspnoea, insomnia, appetite loss and fatigue (Figure 3).
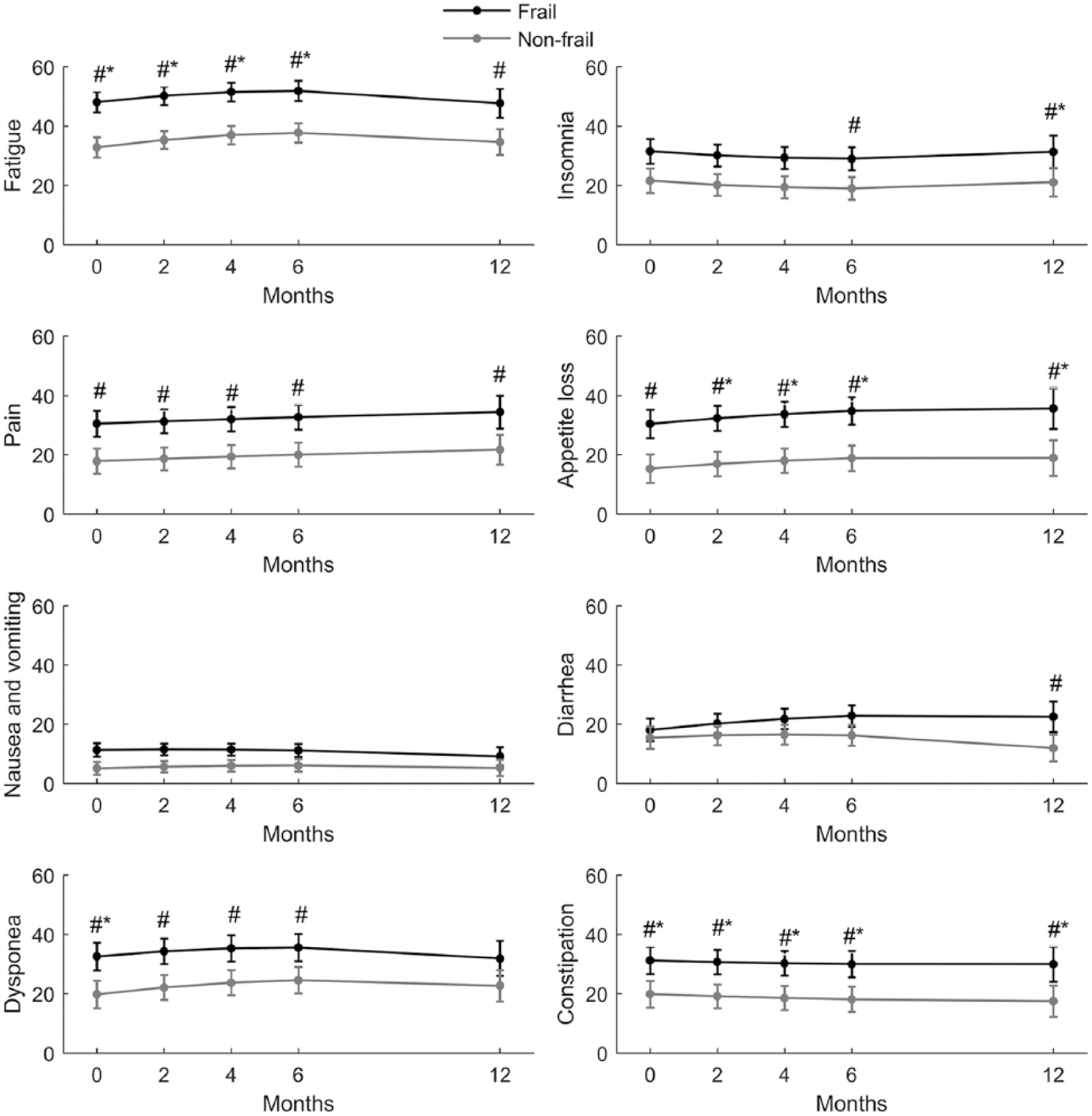
Symptom scores for frail and non-frail patients at baseline and at 2, 4, 6 and 12 months of follow-up, according to unadjusted mixed linear models.
Discussion
In this longitudinal study, older cancer patients were assessed by a modified geriatric assessment, and we identified a group of frail patients who in comparison to non-frail patients had substantially poorer functioning and more symptoms. Independent of age, gender and major cancer-related prognostic factors, they reported significantly worse global quality of life; physical-, role-, social,– and emotional functioning and more constipation during treatment and follow-up. They also reported a long-term decline in physical functioning that was clinically significant and significantly steeper than for non-frail patients.
To the best of our knowledge, this study is the first to report a longitudinal comparison of self-reported physical functioning between frail and non-frail older patients mainly receiving systemic cancer therapy and the first to suggest a more profound deterioration in this quality-of-life dimension among frail patients after adjusting for other relevant confounders. Our finding is supported by two former studies reporting frailty indicators to be predictive of observer-rated physical decline in older cancer patients receiving chemotherapy or neoadjuvant/adjuvant treatment.42,43 No such impact of frailty was found in studies of patients receiving surgery and radiochemotherapy, respectively.10,11 In the latter, however, specific assessments of physical functioning were reported only at 4 weeks after start of therapy, and as indicated by our results, a significant decline may take longer to develop. It is also likely that a protracted course of chemotherapy, which was the treatment received by most of our patients, may have a larger impact on frail patients’ physical functioning than surgery.
The results of the few previous studies that have investigated how frail older cancer patients perceive their quality of life are largely consistent with our remaining findings. Frail patients seem to be at a considerable disadvantage throughout the disease trajectory, reporting a substantial symptom burden and poor functioning compared to non-frail patients.10,11,44 In line with the findings for most quality-of-life aspects in our cohort, others have also found that although quality of life is poorer, changes mainly follow a similar course in frail and non-frail cancer patients. Increased risks of long-term deterioration has, however, been suggested.13,44 How an observed similarity of changes in quality-of-life trajectories of frail and non-frail patients should be interpreted is not obvious. One might argue that this indicates that frail patients tolerate cancer therapy equally to non-frail patients. However, as frail patients are worse off from the start, changes in the same magnitude may affect these patients more profoundly than those who are non-frail.
Our study has several strengths, that is, a fairly large patient cohort, 12 months follow-up, use of a well-validated quality-of-life questionnaire, high completion rate and statistics controlling for major factors that may affect quality of life. Still, the results should be interpreted with some caution. First, the population was heterogeneous, details of the chemotherapy regimens were not accounted for and we cannot rule out that frail patients received modified or less aggressive regimens than those who were non-frail. This is, however, unlikely as the physicians were blinded for the results of the modified geriatric assessment. Also, as formerly reported, there was only a fair agreement between the frailty classification based on this assessment and physician-rated frailty. 24 Second, we were not able to accurately register the number of potentially eligible patients who were not included at the various participating clinics. According to the project nurses, however, non-inclusion mainly occurred by random due to lack of time to identify and include patients among their routine clinical tasks. Still, there is some risk that the frailest patients with the poorest overall health more often declined participation or were less frequently invited to participate due to concerns of the additional burden the study tests represented. Third, due to a higher death rate among frail patients, attrition bias may have resulted in underestimation of differences between frail and non-frail patients. 45 Fourth, physical function, as assessed by Timed Up and Go, number of falls and one item from the physical functioning scale of the EORTC QLQ-C30, is a key component of a geriatric assessment and frailty definition and can probably explain some of the baseline difference we found in functioning between frail and non-frail patients. However, it is not inherent in our frailty definition that frail patients experience a steeper decline in physical functioning compared to non-frail. Moreover, only a minority of the patients fulfilled these criteria, and the main point to be noted is the overall burden of problems among these frail patients. An additional point of consideration is that we used one question from the QLQ-C30 physical functioning scale, which was also our main endpoint, to identify frailty. Only one patient was classified as frail based on this criterion alone; hence, we believe that this did not affect our results. Finally, as there is no consensus on how frailty should be identified, it may be discussed if our frailty definition captures the true concept. One may argue that it was too broad as only one criterion was needed to be classified as frail. A stricter definition might have resulted in larger discrepancies between frail and non-frail patients. However, our approach was adapted from the Balducci criteria, and a similar definition was found superior to the physical frailty phenotype in identifying postoperative complications in cancer patients. 46 There is a need for standardisations of cut-off-values for frailty; 8 nevertheless, the consistency of findings across studies indicates that geriatric assessment can identify patients who need particular attention.
Our study shows that frailty as identified by a modified geriatric assessment has a severe impact on the patients’ quality of life throughout the disease trajectory, independent of cancer-related factors. Thus, by introducing geriatric assessment into clinical work, a more correct individualization of treatment can be achieved. 47 Furthermore, targeted interventions to improve quality of life and maintain functioning may be initiated. Early introduction of palliative care has been shown to improve quality of life, reduce aggressiveness of treatment and improve survival. 48 Similar studies in frail old cancer patients are needed to examine whether improvement of quality of life can be obtained. Ideally, these studies should include interventions on geriatric deficits and measure their effect on quality of life. Particular attention should be paid on avoiding physical decline, which may considerably increase the risk of dependency, a predominant fear among older patients.3,4 As indicated by the findings in our study, frail patients report significantly poorer physical functioning than those who are non-frail, meaning that any decline is likely to have more serious consequences.
In conclusion, introducing geriatric assessment into routine clinical practice may help oncologists identify patients with significantly worse quality of life and enable better individualization of treatment. This may also facilitate early and correctly targeted interventions. Future research is, however, needed to explore whether intervening on frailty domains can improve functional status, global quality of life, symptom burden or tolerance to cancer therapy.
Footnotes
Acknowledgements
We want to thank all the patients for their dedicated participation in our project. We also want to thank the following nurses for their contribution in the assessment and follow-up of the patients: Signe Eldevik, Toril Nistad, Anne Mari Hanstad, Gunvor Hjelle, Bjørg Baklien, Gunhild Evenrud, Anne Glorvigen Hanstad, Astrid Rusten, Marit Opheim Auning, Eva Iren Haugen, Kathrine Engdal Horn and Unn-Cathrin Buvarp and an addition thanks to the local investigators Morten Brændengen, Oslo University Hospital and Olav Yri at Akershus University Hospital.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was publicly funded by Innlandet Hospital Trust.
