Abstract
Background:
Increasing number of people are dying with advanced dementia. Comfort and quality of life are key goals of care.
Aims:
To describe (1) physical and psychological symptoms, (2) health and social care service utilisation and (3) care at end of life in people with advanced dementia.
Design:
9-month prospective cohort study.
Setting and participants:
Greater London, England, people with advanced dementia (Functional Assessment Staging Scale 6e and above) from 14 nursing homes or their own homes.
Main outcome measures:
At study entry and monthly: prescriptions, Charlson Comorbidity Index, pressure sore risk/severity (Waterlow Scale/Stirling Scale, respectively), acute medical events, pain (Pain Assessment in Advanced Dementia), neuropsychiatric symptoms (Neuropsychiatric Inventory), quality of life (Quality of Life in Late-Stage Dementia Scale), resource use (Resource Utilization in Dementia Questionnaire and Client Services Receipt Inventory), presence/type of advance care plans, interventions, mortality, place of death and comfort (Symptom Management at End of Life in Dementia Scale).
Results:
Of 159 potential participants, 85 were recruited (62% alive at end of follow-up). Pain (11% at rest, 61% on movement) and significant agitation (54%) were common and persistent. Aspiration, dyspnoea, septicaemia and pneumonia were more frequent in those who died. In total, 76% had ‘do not resuscitate’ statements, less than 40% advance care plans. Most received primary care visits, there was little input from geriatrics or mental health but contact with emergency paramedics was common.
Conclusion:
People with advanced dementia lived with distressing symptoms. Service provision was not tailored to their needs. Longitudinal multidisciplinary input could optimise symptom control and quality of life.
Keywords
The World Health Organization has named dementia as the seventh leading cause of death worldwide, and deaths due to dementias more than doubled between 2000 and 2015.
Detailed longitudinal data on symptom burden and healthcare utilisation in advanced dementia are sparse.
Intervention development for people with advanced dementia has lacked a strong clinical evidence base.
People with advanced dementia live with chronic and persistent symptoms, including pain and agitation.
There are few acute clinical events that predict impending death and thus prognosis is uncertain.
Despite complex physical and psychiatric needs, most care is provided by general practitioners or emergency services with little input from specialists such as geriatricians or palliative care.
Nursing home residents with advanced dementia require multidisciplinary, multi-speciality care, and nursing home staff need more support from external healthcare services.
The potentially long, slow decline in advanced dementia as death approaches and uncertainty about the duration need to be taken into account in planning and delivery of services.
People with advanced dementia would benefit from care focussing on management of physical and psychological symptoms. Further study is required to establish the best models for providing this care.
Introduction
Approximately 850,000 people in the United Kingdom have dementia. 1 One-third of people aged over 65 years in the United Kingdom will die with some form of dementia, 2 the majority will die in care homes3,4 and advanced dementia is becoming a leading cause of death in the United Kingdom and across the world.5–7
It has been suggested that the key goals of end-of-life care for people with dementia should be to maximise comfort and quality of life,8,9 but concerns have been raised about the quality of care provided.10,11 Barriers to providing good end-of-life care include structures of service provision, difficulties in estimating prognosis and uncertainty around when the person is entering the terminal phase. 12 Studies from the United States, The Netherlands, Belgium and Italy13–16 have demonstrated a high symptom burden in people with advanced dementia, but there has not been a detailed, prospective longitudinal study of physical and psychological symptoms and service use in this population in the United Kingdom. Data on the UK context would inform service provision and may facilitate international comparisons in treatment and outcomes, enhancing our understanding of how context influences the provision of end-of-life care.
The 3-year Compassion Programme (National Institute for Health Research (NIHR) reference number CRN-PCRN 12621; 12623) used realist methods 17 and combined data from multiple sources (including this cohort study)18,19 to develop and implement a complex intervention to improve end-of-life care in advanced dementia.20,21 In this article, we report on a 9-month longitudinal cohort study of people with advanced dementia. Our aims were to
Describe the course of physical and psychological symptoms;
Examine health and social care service utilisation;
Describe the care received at the end of life.
Methods
Setting
We recruited people with advanced dementia from May 2012–December 2014, from six Clinical Commissioning Groups (CCG; National Health Service (NHS) organisations overseeing healthcare delivery in England) across Greater London. To reflect the estimated place of residence of people with advanced dementia in the United Kingdom, we aimed to recruit 100 people with advanced dementia, 70 from nursing homes and 30 residing in their own homes. 1 We purposively selected 14 nursing homes ensuring a representative range of
Care Quality Commission (CQC; public body of the Department of Health which regulates and inspects health and social care services in England) ratings;
Gold Standards Framework (GSF; national training programme enabling staff to provide generalist care at the end of life) implementation; 22
Number of beds;
Ownership (private company or state services);
Areas of socio-economic and ethnic diversity.
The majority (90%) of UK nursing homes are privately owned, but residents should have equitable access to all core NHS services, such as primary and secondary care (including mental health), and palliative care. However, access to chiropody, opticians and physiotherapy is less consistently provided by statutory services and nursing homes, and residents or their families may purchase this care privately.
To recruit people with advanced dementia residing in their own homes, we used general practices in five CCG areas of greater London; practice staff screened patient lists to identify potential participants. To ensure access to primary care records, we recruited participants (both those residing at home and those in nursing homes) who were registered with practices linked to the Primary Care Research Network which works with general practitioners (GPs) to support research.
Inclusion and exclusion criteria
Nursing home and primary care staff identified potential participants meeting the following criteria: clinical diagnosis of dementia aged over 65 years and at Functional Assessment Staging (FAST) grade 6e and above 23 (one of: doubly incontinent and speaks only a few words, unable to walk, loss of intelligible speech, unable to smile, unable to hold their head up). For recruitment and consent procedures, see Jones et al. 17
Data collection
At study entry, we documented demographics, Charlson Comorbidity Index (CCI) 24 (19 diseases weighted on their association with mortality); prescribed medications; 25 advance directives, advance statements and lasting power of attorney. Research staff assessed participants using validated scales, through direct observation or in consultation with carers and nursing home staff. Study measures (see Table 1) included physical symptoms: pain (Pain Assessment IN Advanced Dementia (PAINAD) Scale, 30 scores of 2 or greater indicate clinically significant pain), 31 pressure sore risk (Waterlow Scale) 28 and pressure sore severity (Stirling Scale); 29 neuropsychiatric symptoms: Neuropsychiatric Inventory (NPI) 33 and Cohen Mansfield Agitation Inventory (CMAI; scores of over 39 indicate clinically significant agitation), 32 comfort (Symptom Management at the End of Life in Dementia (SM-EOLD)), 34 quality of life (Quality of Life in Late-Stage Dementia (QUALID)) 35 and service use with the Client Services Receipt Inventory (CSRI) and the Resource Utilization in Dementia-Lite (RUD-LITE) Scales.36,37
Study measures.
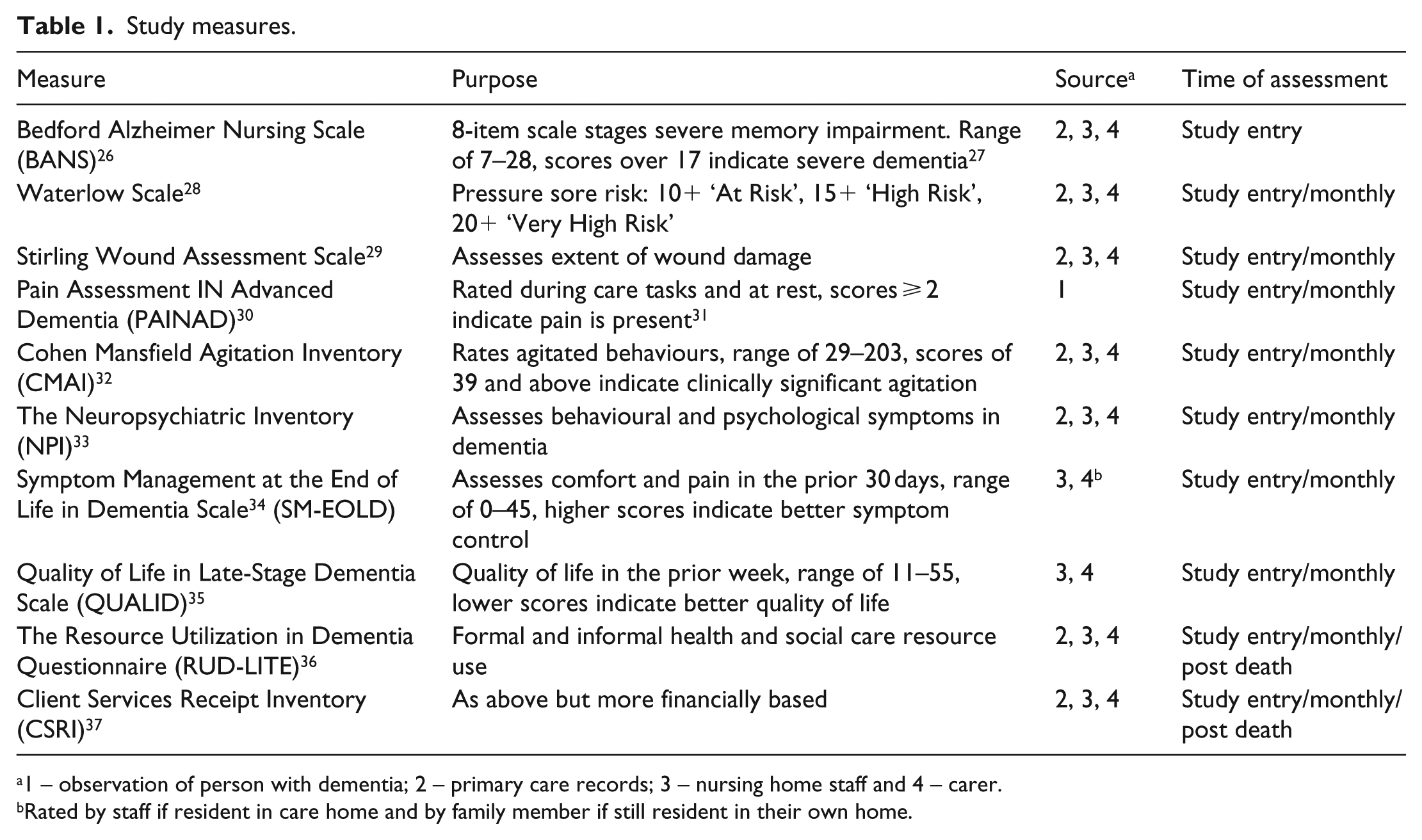
1 – observation of person with dementia; 2 – primary care records; 3 – nursing home staff and 4 – carer.
Rated by staff if resident in care home and by family member if still resident in their own home.
Participants were assessed every 4 weeks for a maximum of 9 months. In addition, we documented events from the prior month:
Painful or burdensome interventions, for example, enteral feeding tubes, venous or arterial blood tests and mechanical ventilation. 38
Symptoms documented as being common or problematic in advanced dementia: dehydration, constipation, difficulty swallowing, insufficient oral intake, weight loss, aspirating/choking on swallowing and difficulties breathing. 13
Acute clinical events: seizures, fever, urinary tract infection, pneumonia and septicaemia. 39
After death, we collected information from staff and/or family carers on whether the death had been ‘expected’, whether there was a ‘do not attempt resuscitation’ (DNAR) order at the time of death, place and primary cause of death.
Data were collected prospectively on paper case-report forms. A new form was used at each visit so raters would not be influenced by previous scores.
Analysis
Participant characteristics were described using mean values or medians for continuous variables (with standard deviation (SD) or inter-quartile range (IQR)) and frequencies (percentages) for categorical variables at four time points: (1) study entry, (2) for those who died at the final visit prior to death, (3) for those who were still alive at the end of the study at the final visit and (4) for the whole cohort at the final visit. If more than 20% of questionnaire items were missing, the total was set as missing for the individual as this may have implied non-completion of the whole questionnaire. If less than 20% of items were missing per individual, mean scores were imputed. 40 Analyses were performed using Stata 13.
Ethics
Our approach was based on the UK Mental Capacity Act 2005, and a personal or a nominated ‘professional’ consultee gave their informed written agreement for the person with advanced dementia to participate 17 (National Research Ethics Committee East of England approval 12/EE/0003).
Results
Of 159 potential participants with advanced dementia, 85 were recruited, 79 resided in nursing homes and 6 in their own homes (Online Figure 1). At study entry, all questionnaires were completed. In total, 21 (0.6%) individual questionnaires over follow-up had one or two missing items which were imputed. At the last visit, data collection varied from missing in two participants (2%) on the entire Confusion Assessment Method (CAM), QUALID and PAINAD at rest scales to missing in six participants (7%) on the PAINAD activity scale and symptoms proforma.
Cohort characteristics
The majority were women (median age of 85 years, IQR of 81–90). Median length of nursing home residence was 3 years (IQR of 1–5). For the 53 (62%) participants alive, at end of follow-up, a median number of 9 (IQR of 8–9) assessments were completed. Mortality at 9 months was 37%, 32 participants died, with a median of four assessments (IQR of 2.5–6.5). At study entry, 76% of the cohort had a DNAR order, 30% had an advance care plan, 40% a documented preferred place of death and 40% a lasting power of attorney.
Diagnoses and comorbidities
Alzheimer’s disease followed by vascular dementia were most common. The mean Bedford Alzheimer Nursing Scale (BANS) score was 21 (SD = 3.4), indicating participants were severely impaired. Participants had an average Charlson comorbidity score of 6 (SD = 1.5), and 38% were taking 6–9 different classes of drugs (Table 2).
Characteristics of people with advanced dementia (n = 85).

IQR: inter-quartile range; FAST: Functional Assessment Staging Scale; SD: standard deviation.
Clinical symptoms
At study entry, 53% were at very high risk of pressure sores (Waterlow Scale). On the Stirling pressure sore severity scale, 22% had early signs of pressure sores and 16 participants (19%) had partial-to-full skin thickness loss. Pressure sore risk and prevalence of pressure sores remained constant throughout follow-up and there were no differences in pressure sore risk between those who died and those who were still alive at the final assessment (Table 3 and Figure 1).
Physical and psychological symptoms, quality of life and comfort in people with advanced dementia (n = 85).
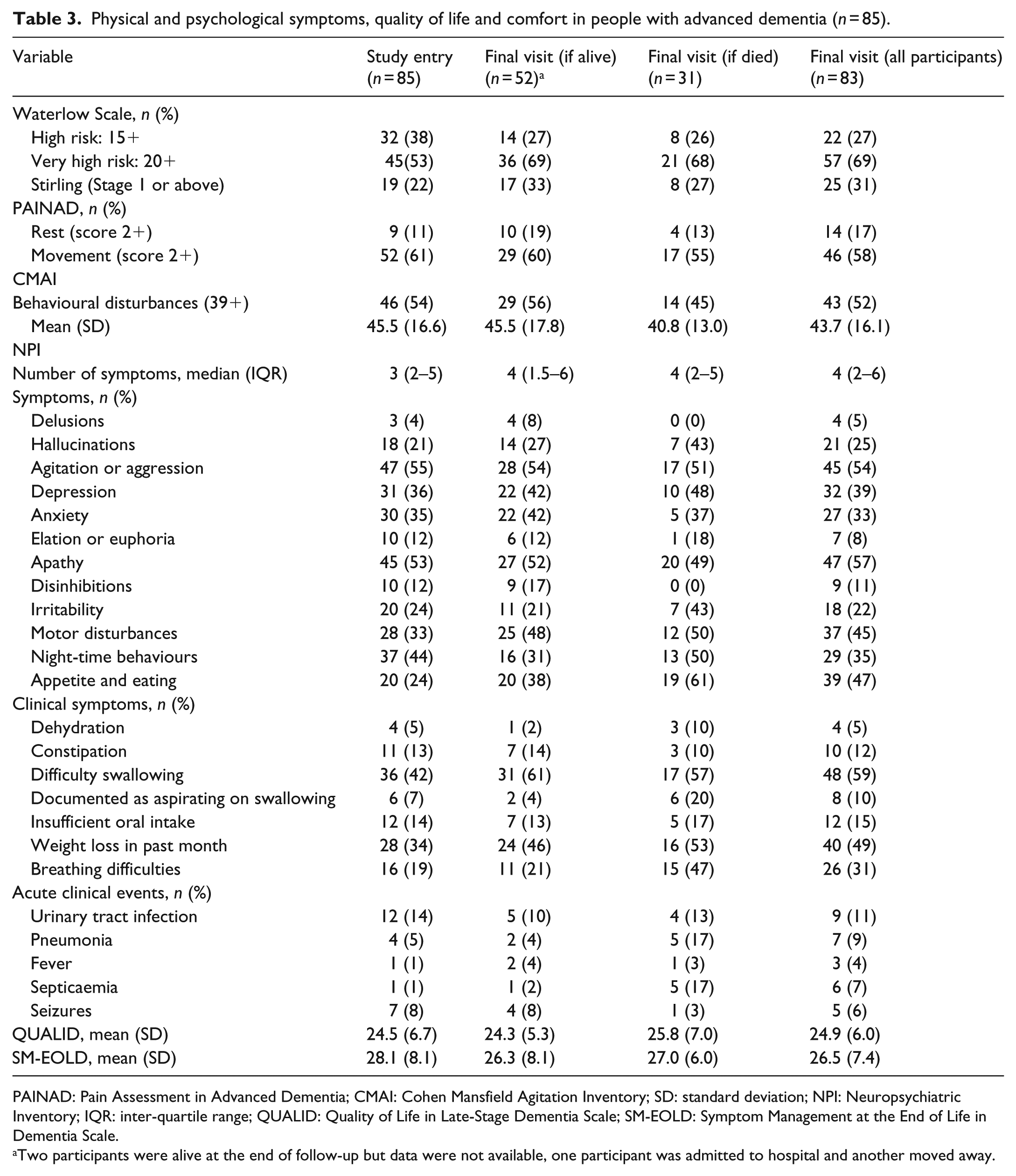
PAINAD: Pain Assessment in Advanced Dementia; CMAI: Cohen Mansfield Agitation Inventory; SD: standard deviation; NPI: Neuropsychiatric Inventory; IQR: inter-quartile range; QUALID: Quality of Life in Late-Stage Dementia Scale; SM-EOLD: Symptom Management at the End of Life in Dementia Scale.
Two participants were alive at the end of follow-up but data were not available, one participant was admitted to hospital and another moved away.
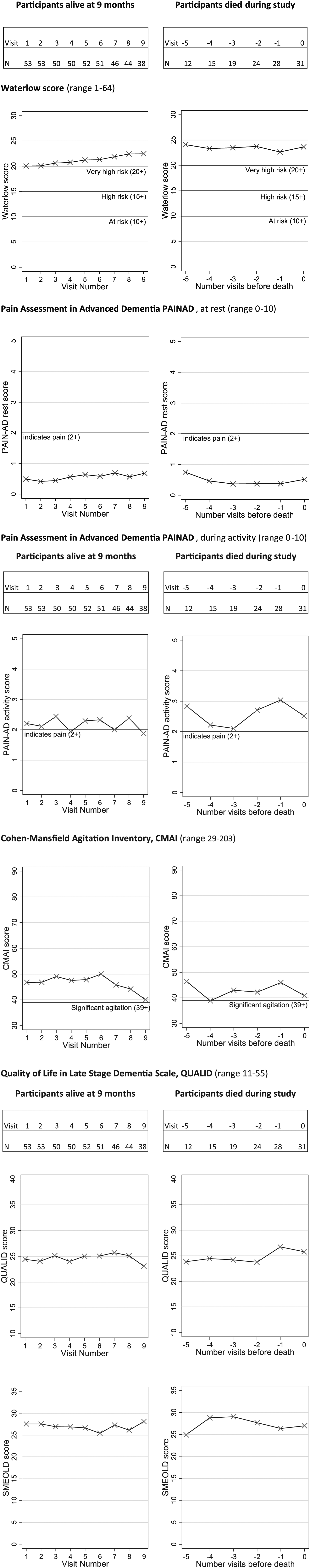
Changes in symptoms over time in people with advanced dementia (n = 85).
The proportion of participants observed to be in pain at study entry, both at rest (11%) and during movement (61%), changed little during follow-up (Figure 1). Over half of participants had clinically significant agitation on the CMAI at study entry (54%) with a slight reduction at the final assessment for those who died (45%) compared to those still alive (56%). Other common psychiatric symptoms at study entry were depression (36%), anxiety (35%), apathy (53%), motor disturbances (33%) and night-time behaviours (waking during the night or sleeping excessively during the day (44%)). The prevalence of these remained unchanged throughout follow-up for all participants, apart from depression which increased to 42% of those alive at the end of the study and 48% who died and motor disturbances which increased to 48% of those who remained alive and 50% of those who died (Table 3).
The commonest physical symptoms at study entry were difficulty swallowing (42%) and weight loss in the prior month (34%). At the final study visit, a higher proportion of those who died experienced aspiration on swallowing (20% compared to 4% those who remained alive) and breathing difficulties (47% compared to 21% of those who remained alive).
Acute clinical events
Urinary tract infection was the commonest acute medical event at study entry (14% of participants in the prior month). Septicaemia and pneumonia occurred in 17% of participants who died compared to those who were alive at the final study assessment (septicaemia 2% and pneumonia 4%).
Quality of life and comfort
Quality of life (QUALID Scale) remained constant from study entry (24.4, SD = 6.8) until final study assessment (24.3, SD = 5.3) deteriorating slightly towards death (25.8, SD = 7.0) (higher scores indicate lower quality of life) as did scores for comfort on the SM-EOLD (Table 3 and Figure 1).
Service use
General (acute) hospital admission was infrequent in the month prior to study entry (0.18 admissions per participant) and remained low during follow-up (Table 4). Use of outpatient services was more frequent. In the month prior to study entry, there were 0.31 contacts per participant with paramedics or emergency ambulance services and 10.3% saw a paramedic. In the month prior to death, there were 0.75 paramedic contacts per participant and 19% were seen by a paramedic at the nursing home.
Service use data from Client Services Receipt Inventory and Resource Utilization in Dementia Questionnaire in people with advanced dementia (n = 85).
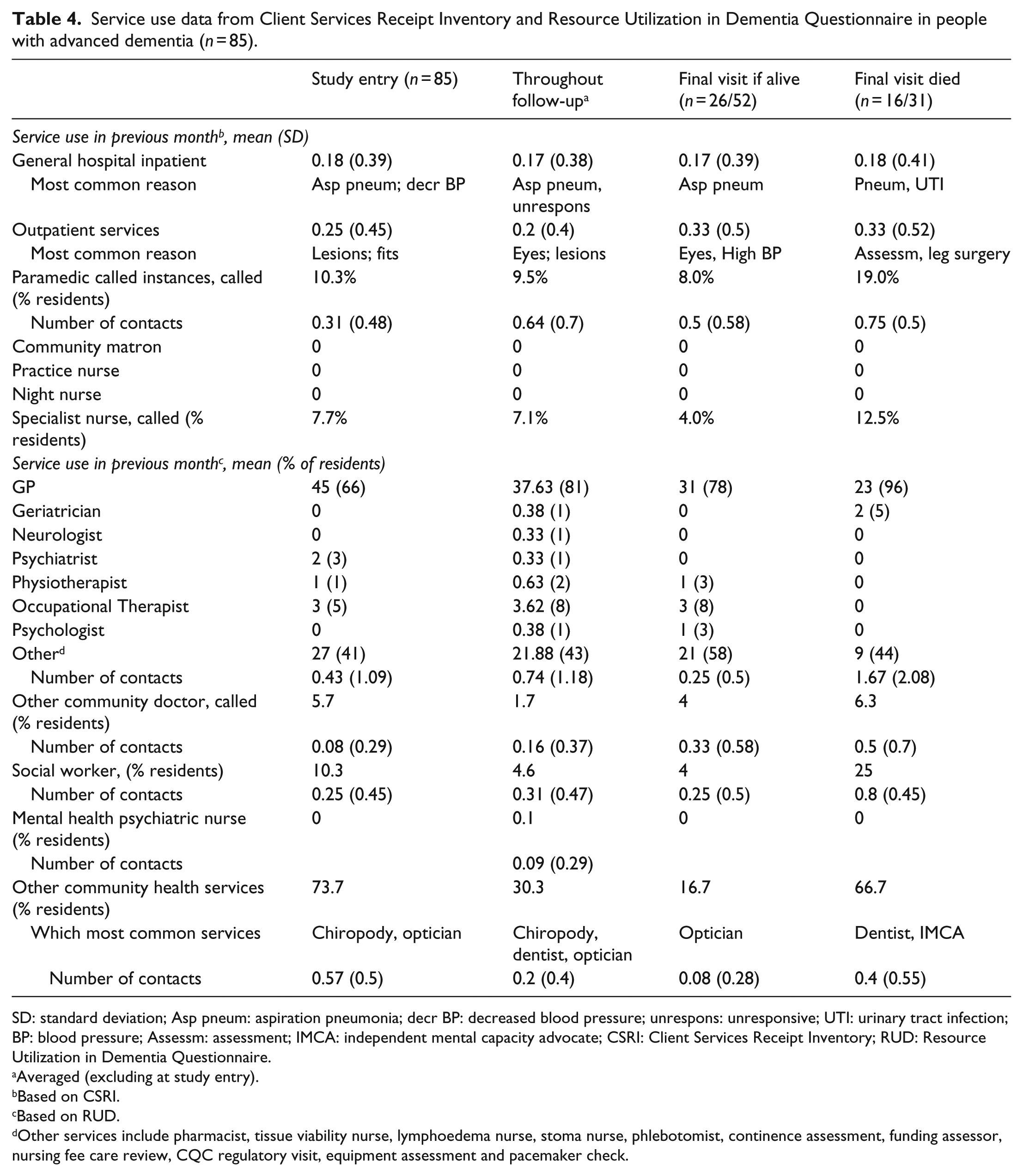
SD: standard deviation; Asp pneum: aspiration pneumonia; decr BP: decreased blood pressure; unrespons: unresponsive; UTI: urinary tract infection; BP: blood pressure; Assessm: assessment; IMCA: independent mental capacity advocate; CSRI: Client Services Receipt Inventory; RUD: Resource Utilization in Dementia Questionnaire.
Averaged (excluding at study entry).
Based on CSRI.
Based on RUD.
Other services include pharmacist, tissue viability nurse, lymphoedema nurse, stoma nurse, phlebotomist, continence assessment, funding assessor, nursing fee care review, CQC regulatory visit, equipment assessment and pacemaker check.
Participants had no contacts with community nursing services, although contact with ‘specialist’ nurses increased to 12.5% of those who died (from 7.7% at baseline) likely due to palliative care nurses being called in. Contact with other professionals such as chiropodist, optician and dentist was more frequent. The majority of participants were seen by a GP during the study and 96% of those who died saw a GP in their last month. There was little contact with geriatricians, neurologists, psychiatrists or community psychiatric nurses.
Care received at the end of life
The most frequent interventions in the month prior to death were blood tests (14%) and urinary catheters (14%). Death was ‘expected’ in 90% of cases; 81% died in the nursing home and one person died in their own home. Of those who died, 34% were referred to and 28% were seen by the palliative care team. A DNAR was in place for 93% of those who died, and the commonest cause of death was aspiration pneumonia (Table 5).
Symptoms and interventions at dying, place of death in people with advanced dementia (n = 85).

I-V: intravenous; NG: nasogastric; PEG: percutaneous entero-gastrostomy; CPAP: continuous positive airway pressure; BIPAP: bi-level positive airway pressure; DNAR: do not attempt resuscitation order.
Determined by consensus discussion with family carers (where available), care home staff and primary care team.
Discussion
Course of physical and psychological symptoms
Mortality at 9 months (37%) was comparable to 6-month mortality rates in people with advanced dementia from the United States (18%), Italy (20%) and The Netherlands (37%).41–43 The commonest physical symptoms at study entry were difficulty swallowing and weight loss. Pain on movement, agitation and apathy were also frequent and persisted at clinically significant levels throughout the study. Aspiration and breathing difficulties increased in the month prior to death, and the commonest acute events prior to death were septicaemia and pneumonia.
Our participants were similar in disease severity and demographics to cohorts from The Netherlands, 44 Belgium 45 (for whom data were collected retrospectively after death) and the United States, 39 which also found pain, agitation and dyspnoea to be the commonest symptoms.39,46 As the end of life approaches, difficulties in swallowing, problems with eating and risk of pneumonia also increase. 13 We report a comparable symptom profile in the United Kingdom and, in addition, demonstrate how these symptoms were chronic and unchanging. This is important as our participants were unable to express their needs and were, therefore, vulnerable to long-term suffering. However, despite concerns regarding suboptimal care in the United Kingdom, 10 mean symptom management scores (SM-EOLD) were similar to those found in The Netherlands 44 and Belgium, 15 but lower than found in the United States. 47
Service use
General Practitioners were the main providers of medical care with most participants having contact during the study. Despite the prevalence of chronic pain and psychiatric symptoms, little specialist healthcare was provided. Of 85 participants, only 5 saw a geriatrician and a psychiatrist with negligible contact with community psychiatric nurses. Paramedics were major providers of assessment and healthcare towards the end of life (although few contacts led to acute hospital admissions) suggesting a reactive response to needs. In the United Kingdom, studies have found that nursing home residents are often poorly served by existing healthcare arrangements 48 and receive less planned and more emergency care than those living in the community. 49 Use of social care services also increased in the month prior to death; 25% of participants who died having contact compared to an average of 4.6% of participants during follow-up. There was more contact with chiropodists, dentists and opticians, reflecting how these services are often purchased-in by care homes or by relatives.
Care received at the end of life
Apart from septicaemia and pneumonia, indicators of impending death were not clearly identifiable. Palliative care outreach teams saw 28% of those who died (34% were referred), predominantly in the month prior to death. 40 This proportion is higher than in the United States (25%) 50 and The Netherlands (2.5%); 51 however, there are differences in healthcare provision between these countries. In The Netherlands, nursing homes have a dedicated physician who provides the majority of medical and palliative care ‘in-house’. Nursing homes in our study were poorly serviced by community medical services and did not have dedicated physicians, thus palliative care teams were called in at the end of life to assist with symptom management. Although 76% of our participants had DNAR orders, only 5% had an advance decision to refuse treatment, the same proportion as that found in The Netherlands, 52 40% had a recorded preferred place of death and 16% died in hospitals. Care planning focussed on resuscitation decisions and place of death, rather than the type of care that the person would like to receive. Numerous initiatives have tried to address a lack of advance care planning in nursing homes, 28 but there are systemic challenges to effective implementation. 44
Strengths and weaknesses
We did not reach our recruitment target despite extending our recruitment period and receiving support from the Primary Care Research Network; many GPs did not hold records of patients with advanced dementia, and it was difficult to locate people living in their own homes. We recruited from a range of nursing homes, ensuring these were representative by purposively sampling by size, CQC rating, ownership and local socio-economic factors to reflect the current UK nursing home market. In the United Kingdom, 25% of nursing homes are rated ‘inadequate’ or ‘requires improvement’; in London, 20% receive a similar rating and 28% of nursing homes in our study were in this category. Thus, by these measures, our study nursing homes are more representative of the United Kingdom in general, rather than of London. We cannot be sure that our participants are representative of people in the United Kingdom with advanced dementia as no comparative data are available. However, the demographic composition of our cohort is similar to that of UK 48 and international studies of advanced dementia.13,46 Nursing home documentation was not standardised, particularly for comorbidity, service use and interventions. Previous studies have been mainly retrospective, relying on symptom recall. We used detailed prospective methods44,45 and reliable validated tools to allow international comparisons.
Implications for clinical practice
Many current policies and tools focus on making an advance care plan, particularly regarding the preferred place of death. These can be beneficial but rely on identifying those who are approaching the end of life and, as our data suggest, this is challenging. Complex symptoms require active specialist intervention, multidisciplinary working and effective care coordination, but GPs feel they do not have the time or knowledge to do this. 53 Increased collaboration between geriatric medicine, palliative care and psychiatry may be helpful but should be coordinated and supported by adequate resourcing and service commissioning. 54 Although the evidence base for interventions in dementia end-of-life care remains limited, 55 considering data used in our development of a complex intervention to improve care in advanced dementia, 20 we suggest a coordinated needs-based longitudinal approach should be developed and tested. 12 A recent survey found ‘hospice enabled care’ is an emerging model for palliative care for people with dementia in the United Kingdom. 56 This involves palliative care specialists, predominantly nurses, providing care coordination and symptom management expertise to nursing home residents, 12 and this may warrant further formal evaluation.
Policy and end-of-life care interventions promote the importance of end-of-life care in dementia 8 and have focused on the place of death; but to improve comfort and quality of life, better management of chronic symptoms is required. 53 This would enable people to live and die well with dementia.
Supplemental Material
726443_Supp_mat – Supplemental material for Living and dying with advanced dementia: A prospective cohort study of symptoms, service use and care at the end of life
Supplemental material, 726443_Supp_mat for Living and dying with advanced dementia: A prospective cohort study of symptoms, service use and care at the end of life by Elizabeth L Sampson, Bridget Candy, Sarah Davis, Anna Buylova Gola, Jane Harrington, Michael King, Nuriye Kupeli, Gerry Leavey, Kirsten Moore, Irwin Nazareth, Rumana Z Omar, Victoria Vickerstaff and Louise Jones in Palliative Medicine
Footnotes
Acknowledgements
The authors thank their co-applicant Professor Steve Morris from University College London for providing expert advice throughout the programme and researchers Sharon Scott, Dr Margaret Elliott and Dr Kathryn Lord.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The Compassion Programme was funded by Marie Curie (grant reference: MCCC-FPR-11).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
