Abstract
Background:
Implementation fidelity is critical in evaluating effectiveness of interventions.
Aim:
Identifying and summarising strategies to improve and assess the level of reporting of implementation fidelity in randomised controlled trials of palliative care complex interventions.
Design:
Systematic review.
Data sources:
Published and completed randomised controlled trials from 2000 to current evaluating effectiveness of specialised palliative care services on patient-centred outcomes in adult patients were examined. MEDLINE was searched from 2008 to 29 September 2015 and supplemented by randomised controlled trials identified in a 2008 systematic review.
Results:
Altogether, 20 randomised controlled trials involving 8426 patients were reviewed using 40 subcomponents of five elements of implementation fidelity (resulting in 20 × 40 = 800 items). Over 88 strategies were identified, classified under the following elements: ‘treatment design’, ‘training providers’, ‘delivery of treatment’, ‘receipt of treatment’ and ‘enactment of treatment skills’. No single overarching strategy was discovered. Strategies under ‘treatment design’ aimed to ensure equivalent treatment dose between and within intervention and control groups, and delivery of necessary ingredients. Ongoing ‘training (of) providers’ included supervision and ensuring skill acquisition. Use of treatment manuals and implementation checklists aimed to aid ‘delivery of treatment’. Research teams aimed to improve ‘receipt of treatment’ by transmitting clear information and verifying understanding, while improving ‘enactment of treatment skills’ by reviewing and reinforcing prior content. Only 26% of the items received sufficient reporting; 34% were either not used or reported on.
Conclusion:
Implementation fidelity in palliative care is under-recognised. A table to collate these strategies to improve implementation fidelity in palliative care research and clinical practice is proposed.
Palliative care is a complex intervention.
The effectiveness of an intervention can only be determined if there is implementation fidelity (meaning the extent an intervention is implemented as intended).
Current understanding of implementation fidelity and strategies to improve this have been largely derived from behaviour change interventions, but there has been no analysis of this for palliative care.
Implementation fidelity in palliative care is under-recognised.
Strategies used to improve implementation fidelity in randomised controlled trials of palliative care can be categorised under the following elements: ‘treatment design’, ‘training providers’, ‘delivery of treatment’, ‘receipt of treatment’ and ‘enactment of treatment skills’.
Over 88 strategies have been identified to improve implementation fidelity.
A table, modified from the data extraction form used in this review, representing the elements of fidelity, their subcomponents, and showcasing the strategies identified, has been produced.
The strategies identified could be used, not just in research but in clinical practice, to guide all phases of the development and evaluation of palliative care interventions.
Substantial administrative burden in the application of the identified strategies suggests that further investigation is required to identify which strategies are more effective in improving, as well as assessing, the level of reporting of implementation fidelity in palliative care interventions.
Introduction
The two conditions for experimental interventions to be adopted as standard care are as follows: they are effective and were implemented with a minimum implementation fidelity standard. Implementation fidelity is important because without it, conclusive statements about the effectiveness of an intervention cannot be made.1–8 However, implementation fidelity is challenging in palliative care. Current understanding of implementation fidelity is largely focused on interventions aimed at changing health behaviours,1,2,4,5,9 that is, behavioural change interventions, which often only makes up one component of palliative care. Therefore, behavioural change implementation fidelity strategies and findings may not be easily transferable to palliative care, a complex intervention. Palliative care, with a heterogeneous patient population both in terms of primary diagnosis and setting for service delivery, 6 requires individualised care 10 and becomes ineffective if any of its vital interacting components are left out.5,11 Therefore, understanding how to maintain and improve implementation fidelity in palliative care is especially important in order to avoid errors of poor implementation of complex interventions. In this systematic review, we aimed to identify and summarise strategies used in recently published randomised controlled trials (RCTs) of palliative care complex interventions to improve implementation fidelity.
Methods
Theoretical framework and definitions of implementation fidelity
There is no consistent definition for implementation fidelity. For the purpose of this review, implementation fidelity is defined as ‘the extent an intervention is implemented as intended’1,12 and interpreted to mean the same as ‘intervention fidelity’ 9 and ‘treatment fidelity’. 2 In order to be inclusive, implementation ‘fidelity’ has also been interpreted to mean the same as ‘integrity’. 2 Without a universally agreed definition for implementation fidelity, some authors have taken an alternative approach to defining implementation fidelity by elucidating its essential elements. The current understanding of implementation fidelity by its elements is summarised, as in Figure 1, building on existing conceptual frameworks.2–4,9,12,13
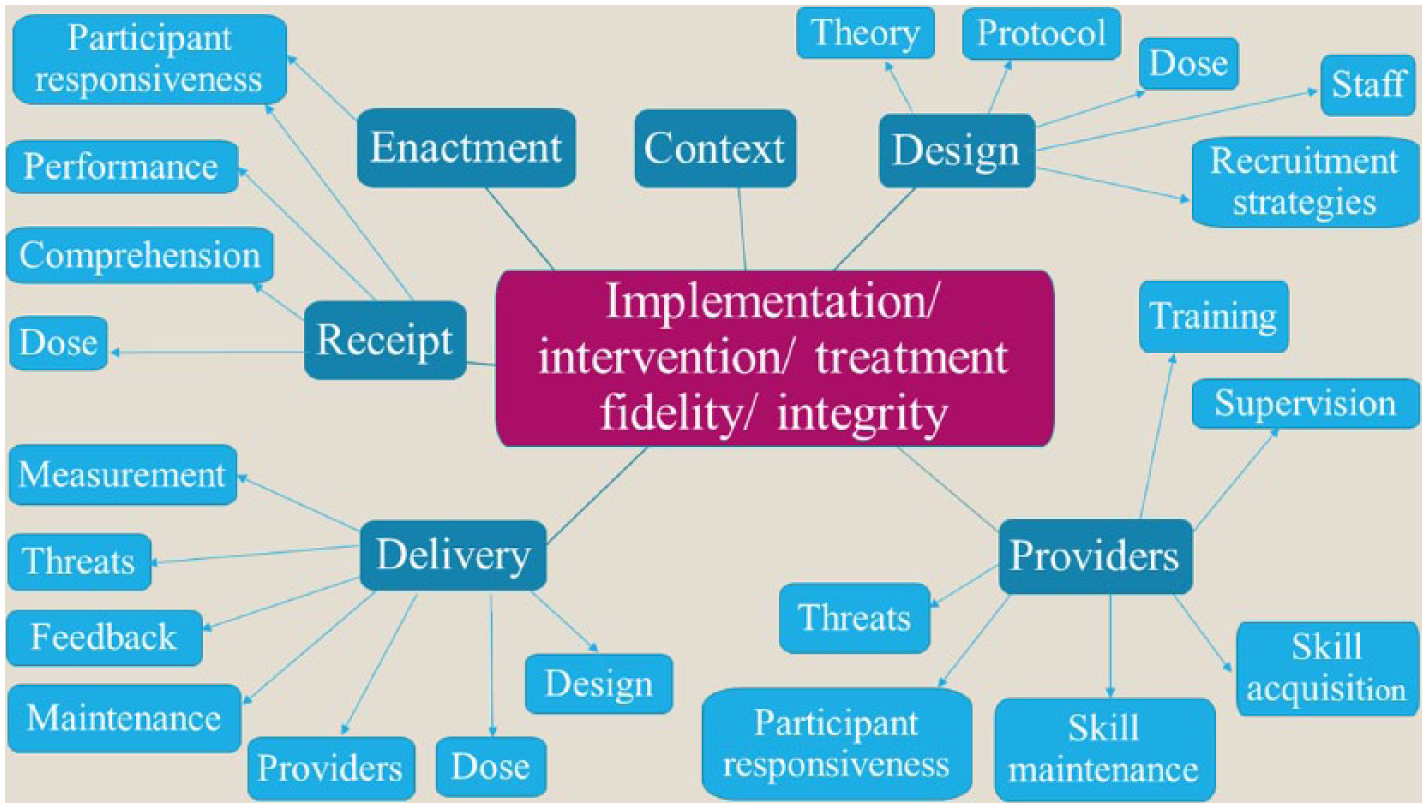
Modified conceptual framework of implementation fidelity.
As represented in Figure 1, implementation fidelity is thought to have six core elements (in dark blue) each with different components (in light blue). These are ‘context’, ‘design’, ‘providers’, ‘delivery’, ‘receipt’ and ‘enactment’. Each corresponds to the various stages of an intervention and is examined sequentially below.
The first element is the ‘context’, or in other words, the ‘surrounding social systems, such as structures and cultures of organizations and groups, inter-organizational linkages, and historical as well as concurrent events’ 12 of the intervention. While relevant to recognise, ‘context’ is usually an uncontrollable variable so not many strategies are expected to result therefrom.
The second element is the ‘design’ which refers to the study’s ability to adequately test its hypotheses underlining its theory and clinical processes. 2 As seen in Figure 1, ‘design’ has five components.
The first component is the ‘theory’ 2 underlining the intervention. The second component is a comprehensive ‘protocol’ or manual 9 for implementation of the intervention to ensure ease of replication and evaluation of the intervention, such as detailed guidelines, troubleshooting assistance and setting of standards. The third component is determination of the ‘dose’ and its ideal and minimally acceptable standards. ‘Dose’ refers to the frequency and duration of the intervention, and also the content of the ‘dose’ (inclusion of right active ingredients(s) necessary for desired outcomes to be achieved). 3 To preserve flexibility, guidance on adaptation can set parameters for dose variations. 9 The fourth component is ‘staff’ (intervention providers) standards. 9 The fifth and last component of ‘design’ is ‘recruitment strategies’: procedures used to attract potential intervention participants, 12 as this impacts the extent to which those eligible for the intervention participate in it. 3
The third element is the ‘providers’ who should be adequately trained to deliver the intervention, and subjected to assessment and ongoing evaluation. 2 This influences the quality of delivery, defined as the ‘extent to which the provider approaches a theoretical ideal’. 3 This element consists of six components: standardised ‘training’ and ‘supervision’,3,9 measures to ensure ‘skill acquisition’ and ‘maintenance’,3,4 and ‘participant responsiveness’ (the extent the providers are engaged in an intervention 3 ), as well as countering ‘threats’ to fidelity.
The fourth element is ‘delivery’, which directly impacts quality of delivery. 3 This entails monitoring that the intervention is delivered as intended:2,3 following the ‘design’ (second element), delivering the intended ‘dose’, adherence to ‘providers’ (third element) standards, 2 ‘maintenance’ of delivery standards over time and ‘feedback’ on collected fidelity data at each stage of the intervention resulting in corrective adjustments, to address further ‘threats’.3,9 Finally, valid and reliable ‘measurement’ of exposures and outcomes 9 are essential to maintaining delivery.
The fifth element is ‘receipt’ of the intervention. This assesses whether the participant receiving the intervention (assessed by quantity of ‘dose’ received in terms of frequency, duration and content), ‘comprehends’2,9 the intervention, and is able to demonstrate the knowledge or ‘perform’ the intervention skills taught. 2 ‘Participant responsiveness’, measuring participants’ engagement in the intervention, applies to this and to the next and final element – ‘enactment’. 3
The sixth element ‘enactment’ refers to the intervention participant demonstrating knowledge or performing intervention skills in relevant real-life settings. 2
Design
We followed standard systematic review methods; 14 the protocol was registered on PROSPERO and can be accessed at http://www.crd.york.ac.uk/PROSPERO/display_record.asp?ID=CRD42015027950.
Inclusion and exclusion criteria
Recently published (from 2000 to current) and completed RCTs which had evaluated the effectiveness of specialised palliative care (SPC) complex intervention services on patient-centred outcomes in adult palliative care patients were included. We took the view that these recently published palliative care RCTs would be the most useful source of information on implementation fidelity and an efficient means for a systematic review, given the likelihood that more comprehensive reporting will be found in RCTs which are high-quality evidence, 15 and reporting of recent RCTs was seen to be of higher quality. Pilot and feasibility trials, and trials where only the protocol had been published, were excluded. Studies published in languages other than English were also excluded.
We defined an SPC service as a professional service that provides or coordinates comprehensive care. This was also the definition used in Zimmermann et al.’s 16 systematic review. Only multifaceted or multicomponent complex interventions were included. Drug or procedural trials, and trials evaluating singular variables, for example, communication, were excluded, in order to reflect the complexities of an SPC service. There were no exclusions based on the comparator interventions used as the aim of the review was to identify strategies for improving implementation fidelity, rather than assessing the effectiveness of interventions.
Trials were included if its participants were adult patients requiring palliative care. This meant adult patients with advanced, progressive, symptomatic and life-threatening disease, for whom the focus of care is maximising their quality of life, through expert symptom management, psychological, social and spiritual support. Trials were excluded if the participants targeted were informal caregivers.
Trials were included if they reported on patient-centred outcomes (e.g. symptom control, quality of life, survival or satisfaction). Trials that only reported on one aspect of a patient-centred outcome (e.g. effectiveness of pain medication on pain) were excluded.
Search methods for identification of studies
The search was conducted using MEDLINE from 2008 to 29 September 2015. This was supplemented by published RCTs from 2000 to January 2008 identified through Zimmermann et al.’s 16 systematic review on the effectiveness of SPC services. Key trials (Appendix 1) were identified by experts in the field of palliative care research (I.J.H. and W.G.). Keywords used in the final search were ‘palliat$ or terminal$ or hospice$’, ‘quality of life or well being’ and ‘randomised controlled trial$ or randomized controlled trial$ or RCT or random allocation or randomisation or randomization’. The detailed search strategy can be found in Appendix 2. Searching of other resources, such as grey literature, hand searching reference lists and cited reference searching was not done.
Data collection and analysis
Selection of studies
In considering inclusion/exclusion, a first decision was made by one researcher (K.A.) based on titles and abstracts available. A study was excluded if information clearly indicated that it did not meet the inclusion criteria. When a decision could not be made with certainty, the full paper was inspected. Any doubt in the selection of studies was discussed with a second researcher (I.J.H.) and resolved through consensus. A record of excluded studies and the reasons for exclusion were kept.
Data extraction and management
Data extraction was carried out on the included trials using the 40-item Treatment Fidelity Assessment and Implementation Plan checklist formulated by National Institutes of Health Behaviour Change Consortium (NIHBCC). 4 This checklist was the most comprehensive and acceptable in pilot-test coding when compared against four other checklists5,9,17,18 from the literature review that informed the above-modified conceptual framework (Figure 1). Data extracted from each trial included the following: the author and year of publication, setting (country), number of patients in the trial, brief patient inclusion and exclusion criteria, brief description of the intervention and control intervention, brief research outcomes, the 40 items in the Treatment Fidelity Assessment and Implementation Plan checklist themselves 4 and also whether or not other strategies outside of those coded in the checklist for use in behaviour change 4 could be identified. The last item in the checklist 4 was amended from ‘a strategy would be used to assess performance …’ (deemed a typo error) to ‘a strategy would be used to improve performance …’, as published in the original Treatment Fidelity checklist. 18
Where included trials referred readers to previously published studies with likely additional information on implementation fidelity, these referenced studies were retrieved. This was to allow our review to be as inclusive and comprehensive as possible in gathering strategies for implementation fidelity. Strategies were coded on the same checklist such that only one checklist was generated for each trial. A record of the additionally retrieved studies was kept.
Each of the 40 items was rated with ‘A’ for ‘absent but should be present’ and ‘NA’ for ‘not applicable’. 18 The ‘present’ rating 18 was differentiated 9 into ‘++’ for ‘present sufficiently’ (especially if there was detailed or extensive coverage/reporting) and ‘+’ for ‘present insufficiently’ (especially if only briefly mentioned, inferred or one was unsure if it fulfils criteria for ‘present sufficiently’). Empty space for free text was also provided such that even if insufficiently present, the strategy used for each of the items could be filled in. Strategies suggested in the ‘Discussion’ section of the included trials were also described in free text, even if the item was rated as ‘A’ for absent.
Standard rules for rating were set to maximise reliability in coding, enable differentiation of ratings and optimise the checklist such that it would be applicable to palliative care. Examples provided by Borrelli 4 were added to guide the coder on what to look out for. Two trials were also pilot-test coded independently by a second researcher (N.H.) and disagreement resolved with further refining of the standard rules. Where consensus was not achieved, the opinion of a third researcher (I.J.H.) was sought and the standard rules further refined to eliminate ambiguity.
Subcomponents of the modified conceptual framework (Figure 1) were also included, with ‘training’ for the providers interpreted to include supervision, and ‘participant responsiveness’ interpreted as a means to improve participant comprehension of the intervention. ‘Recruitment strategies’ was not included as it was thought to be more relevant to the fidelity of the trial, as opposed to the fidelity of the intervention which is the scope of this review.
K.A. is a physician, while NH holds a doctoral degree; both have a special interest in palliative care. I.J.H. is a consultant in specialist palliative care and also holds a doctoral degree. Inter-coder reliability was not calculated.
Data synthesis
Strategies identified through the data extraction form were coded on the 40-item checklist. Each item was rated ‘++’, ‘+’, ‘A’ or ‘NA’ according to the level of reporting of the strategy and analysed in table form against the five core elements (the first element ‘context’ was not considered as it is usually an uncontrollable parameter):
‘treatment design’, that is, whether the study is able to sufficiently test its hypotheses in relation to its underlying theory and clinical processes; 2
‘training providers’, that is, whether the providers are satisfactorily trained to deliver the intervention and are subjected to assessment and ongoing evaluation; 2
‘delivery of treatment’, that is, whether the intervention is delivered as intended;2,3
‘receipt of treatment’, that is, whether the intervention participant has received and ‘comprehends’2,9 the intervention, and is able to demonstrate the knowledge or ‘perform’ the intervention-related skills taught; 2
‘enactment of treatment skills’, that is, whether the intervention participant is able to demonstrate knowledge or perform intervention-related skills in relevant real-life settings. 2
Results
A study flow diagram as recommended by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement 14 is illustrated in Figure 2.
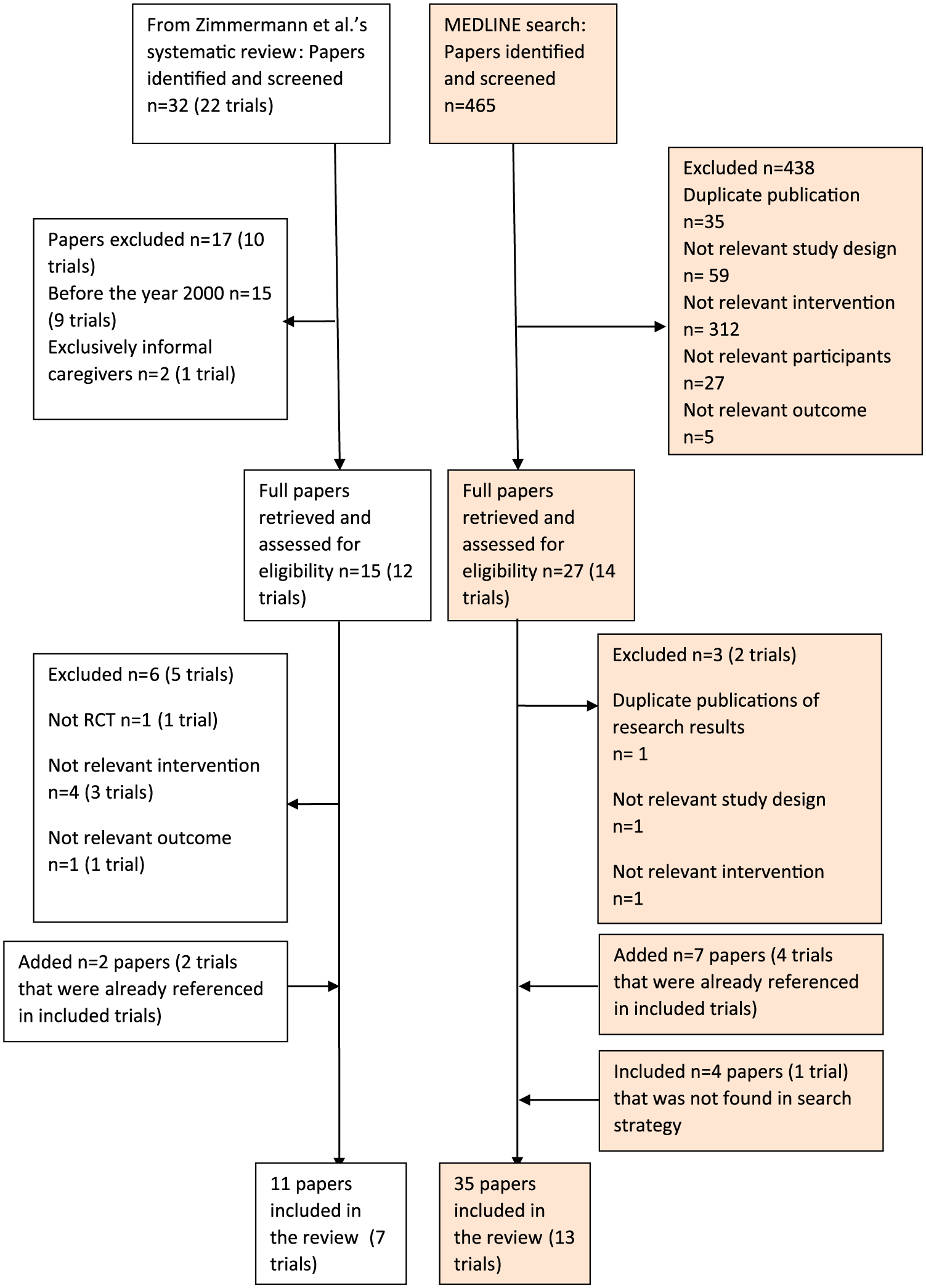
PRISMA flowchart.
Three papers were excluded upon full inspection. One of these was a quasi-experimental study 19 and two were duplicate publications of results from the ENABLE II trial.20,21 The ENABLE II trial was excluded as the intervention was a nurse-led psychoeducation intervention; however, the papers reporting the trial were included as they contain information relevant to implementation fidelity in ENABLE III. 22 Another five published RCTs from 2000 to January 2008 identified through Zimmermann et al.’s 16 systematic review were excluded upon full inspection. Further details of the excluded studies can be found in Appendix 3.
Of the key trials used to ensure sensitivity of the search strategy, one could not be recovered in the final search strategy; it was included in the data analysis.23–26 Nine papers20,21,27–33 which had been referenced in the included trials were later added to the review as they were likely to contain additional information on strategies used for implementation fidelity.
Description of studies
A total of 46 papers reporting on 20 RCTs of SPC complex intervention services involving 8426 patients were included. In total, 10 trials were conducted exclusively with cancer patients,20–22,27,32–51 one exclusively on multiple sclerosis,23–26 one exclusively on chronic heart failure,52,53 whereas the remaining eight trials were not confined to a single diagnostic group.28–31,54–65 All were carried out in developed countries with the majority being in the United States (nine of them)20–22,28,30,31,33,39–45,54–57,60–65 and in the United Kingdom (five).23–27,29,37,38,58,59
As the focus of this review was to extract implementation fidelity strategies rather than to evaluate the effectiveness of the interventions used, only brief notes on the study participants, sample size, setting (country), intervention and outcome measured have been tabulated. This table of characteristics of included studies is in Appendix 4.
Strategies for improving implementation fidelity in RCTs of palliative care complex interventions
Under treatment design, an important subcomponent is to ensure the dose is equivalent between and within the intervention and control groups, with similar access and attention to both groups 38 and within each group. The latter could be via specifying the number of contacts29,58 with a proviso that this could be increased when necessary,38,46 or limiting the number of contacts (referring on if more is needed);23–26 specifying a range of minutes to be spent;48–50 using a ‘structured-visit format’; 55 or promoting protocol adherence, such as early site visits, review of medical records 60 and conference calls.55,60 Other key strategies identified include (1) specification of provider credentials needed, 47 (2) specification and incorporation of active ingredients, demonstrating treatment differentiation,27,28,34–50,52–54,56,59 (3) use of expert/protocol review groups to determine implementation protocol fidelity to underlying theoretical model and (4) identification of potential confounders.
Under training providers, key strategies include provision of pre-implementation or ongoing training, such as observing experts, working with relevant clinicians, clarifying of job scope,27,37 regular supervision sessions,20–22,27,33–37 regular didactic sessions by experts,28,34–36,56 grand rounds, workshops and video presentations.30,31,61–65 Other strategies include role-play practice sessions with feedback to assess provider skill acquisition,20–22,33 record and review of intervention sessions to assess provider skill maintenance,20–22,33 assessment of providers’ suitability for particular interventions 54 and appropriate training for trainees from different disciplines.30,31,61–65
For treatment delivery, key strategies are the usage of a treatment manual,34–36 usage of an implementation checklist,39–44 site visits/surveys to ensure adherence to intervention plan,54,60 interviewing patients or caregivers on their experience,23–26,29,32,45,51–53,58 checking whether medications given were appropriate,32,51 limiting specific intervention to intervention group patients and preventing exposure by control group, 59 and using a cluster randomised trial design,30–32,34–36,51,61–65 preferably with only one unit of randomisation per cluster and restricting the intervention to trial sites.32,51
In relation to ‘delivery of treatment’, a summary table on the level of reporting of implementation fidelity is presented in Table 1, and the remaining summary tables for the remaining core elements (excluding ‘context’) are presented in Appendix 5. As can be seen from Table 1, only about one-third (62 items) of the strategies were sufficiently reported (rated ‘++’), while another third (60 items) were either not used or reported (rated ‘A’); none of the trials mentioned having set a standard for implementation fidelity that the trial would have to meet.
Level of reporting of implementation fidelity strategies in included studies: delivery of treatment.
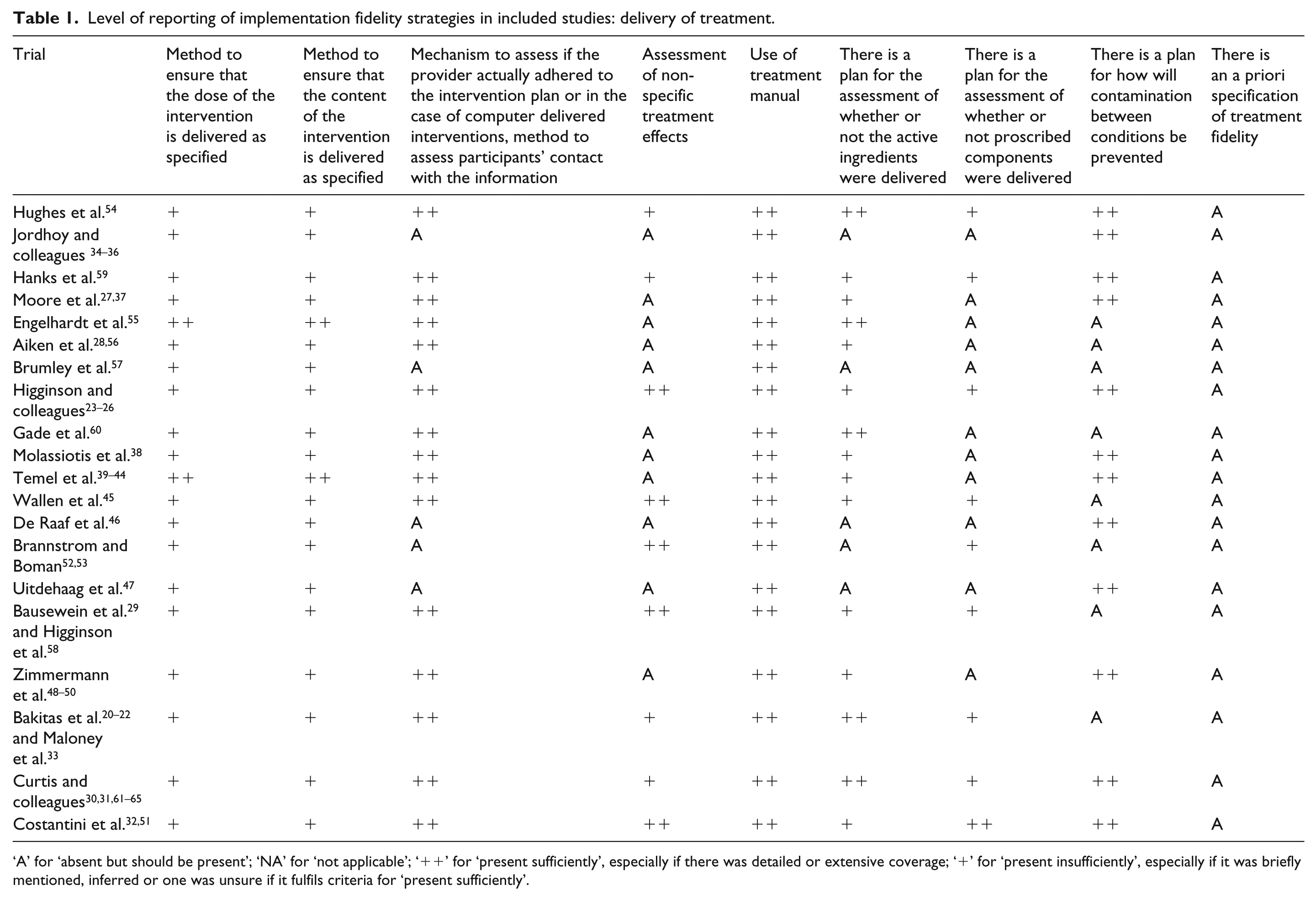
‘A’ for ‘absent but should be present’; ‘NA’ for ‘not applicable’; ‘++’ for ‘present sufficiently’, especially if there was detailed or extensive coverage; ‘+’ for ‘present insufficiently’, especially if it was briefly mentioned, inferred or one was unsure if it fulfils criteria for ‘present sufficiently’.
For receipt of treatment, key strategies are having a health literacy component, 55 having patients recall intervention suggestions, considering ‘participant responsiveness’ and recruitment of willing patients,34–36 assisting with understanding of medical terminology, 55 providing access to information adjusted for health literacy,28,55,56 answering questions,28,38,56 verifying understanding,28,39–44,56 summarising information29,58,60 and providing an information pack.29,58 Rehearsal of emergency situations and practising self-monitoring and reporting of patients’ results28,56 would aid in assessment of patients’ receipt. Consideration of cross-cultural communication in the educational component,30,31,61–65 amending feedback method for outcome data by removing ranking numbers, de-identifying comments, emphasising positive findings and positively presenting low score as opportunities for improvement30,31,61–65 were used to improve receipt.
Finally, key strategies for assessment of enactment include checking self-monitoring diaries, the status of earlier referrals and compliance with medical regime,28,29,56,58 while reinforcing prior content20–22,28,29,33,56,58 improves enactment.
These strategies are summarised in Table 2 (in black text). No other strategies for improving implementation fidelity outside of the elements used in behaviour change and coded outside the checklist 4 were identified. We also did not find examples of strategies that tried to alter the context of the implementation environment to facilitate implementation of the intervention.
Recommended data extraction form along with the standard rules and examples of strategies used.
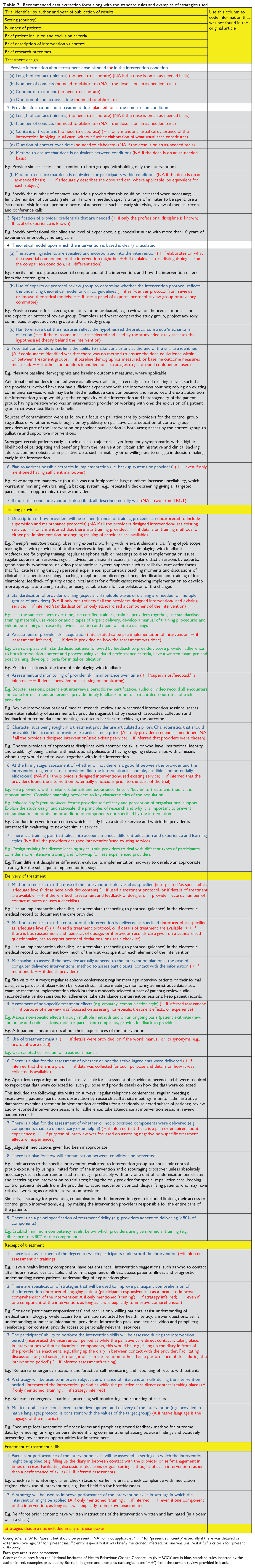
Coding scheme: ‘A’ for ‘absent but should be present’; ‘NA’ for ‘not applicable’; ‘++’ for ‘present sufficiently’ especially if there was detailed or extensive coverage; ‘+’ for ‘present insufficiently’ especially if it was briefly mentioned, inferred, or one was unsure if it fulfils criteria for ‘present sufficiently’.
Each grey area is one component.
Out of the 800 items rated (40 items for each of the 20 trials), 270 items were rated ‘A’ (34%), 159 ‘+’ (20%), 209 ‘++’ (26%) and 162 ‘NA’ (20%). When the individual trial was considered, less than 50% of the 40 items were rated ‘++’. The detailed extracted data are available from the corresponding author. Appendix 6, sorted into the five core elements (excluding ‘context’), presents a summary of strategies used and suggested, including the strategies which were insufficiently described, for improving implementation fidelity.
Discussion
This is the first systematic review of strategies used to improve implementation fidelity in RCTs of palliative care complex interventions. By reviewing and rating 20 recently published SPC service RCTs involving 8426 patients, we have shown that implementation fidelity in palliative care is under-recognised, with up to a third of assessed items rated ‘absent’ and another 20% insufficiently reported on. Moreover, it does not seem that the studies had intentionally reported on implementation fidelity. Nevertheless, over 88 strategies have been identified, sub-classified under the following elements: ‘treatment design’, ‘training providers’, ‘delivery of treatment’, ‘receipt of treatment’ and ‘enactment of treatment skills’. The list of strategies for each item should be viewed as a whole rather than individually as certain items rated absent may still have been accomplished partially in other items. Strategies were not mutually exclusive and can support several goals. The use of these strategies, albeit mostly in cancer patients in developed countries, suggests that the strategies are applicable to other palliative care RCTs.
Strategies in palliative care covered all subcomponents except for two: (1) having a plan to ensure that the measures reflect the hypothesised theoretical constructs/mechanisms of action and (2) an a priori specification of treatment fidelity. Although a potential source of administrative burden, theoretical underpinnings have been recognised as crucial for the success in complex interventions. 66 Similarly, high fidelity requires at least 80% adherence, with 50% considered low. 4 Although palliative care is a complex intervention, of which behaviour change may only constitute one component, no additional strategies outside of those coded in the checklist for use in behaviour change 4 could be identified. This suggests that strategies used in behaviour change and other fields, not yet identified as used in palliative care RCTs, may be used to improve implementation fidelity in palliative care RCTs and palliative care interventions in general, and that palliative care researchers could work more closely with behaviour change researchers.
As such, Table 2, modified from the data extraction form used in this review, incorporates examples of strategies used in behaviour change interventions 4 in green text. The strategies identified in this review have also been added on to the data extraction form in black text, as examples of strategies used in palliative care trials, for use in assessing the level of reporting of implementation fidelity. These examples provided would assist future coders on what to look for. The standard rules used in data extraction are in red text.
The strategies identified in this review could be used, not just in research but in clinical practice, to guide all phases of the development and evaluation of palliative care interventions, 66 to ensure that they are implemented more efficiently and produce more trustworthy results. However, there is also substantial administrative burden in the practical application of these strategies. Administrative burden is brought on by evaluating the ‘delivery of treatment’ and also in ‘training providers’ by having to assess skill acquisition in intervention providers trained to deliver the intervention, and to continually evaluate that they have maintained their skills.
An additional column for strategies identified from papers other than the main paper dealing with the trial has also been added to the data extraction form used in this review (see Table 2). The proportion of trials in which a strategy had been sufficiently present, or the mean proportion of adherence to strategies for each trial, could then be calculated. 18
With the use of the TIDieR checklist and guide, 67 which aims to improve the completeness of reporting of interventions by supplementing the CONSORT 68 and SPIRIT 69 statements, and which has 2 out of its 12 items dedicated to fidelity, the level of reporting of interventions is likely to improve. The TIDieR checklist and guide 67 suggest that both the planning and actual delivery of an intervention in terms of fidelity should be described, and that the description should include how intervention fidelity was ‘assessed and by whom, the strategies that were used to maintain or improve fidelity’, and the extent to which the intervention was delivered as planned (which also pertains to ‘delivery of treatment’). The use of Table 2 could also improve levels of reporting and reduce the proportion of items rated ‘A’. Table 2 could be updated if additional strategies are identified. The proportion of ‘A’ and ‘+’ ratings could also be used to track if the reporting of implementation fidelity has improved over time and provide an estimate of the acceptability and interpretability of the table when reporting and awareness of implementation fidelity are optimised.
Aside from Table 2, to optimise face and content validity, it would be best to consult an international implementation fidelity workgroup in palliative care to ensure that the modified checklist captures the views of all these stakeholders. Consensus on what would constitute a sufficient level of reporting, especially in the differentiation between ‘+ and ‘++, and ‘A’ and ‘NA’, and if each strategy identified ought to be scored individually, and whether certain aspects of implementation fidelity should be more heavily weighted than others, could be sought. The question of how much information on implementation fidelity is needed for an informed decision as to whether or not the intervention was implemented with a minimum implementation fidelity standard, and therefore adequately evaluated the intervention tested, can then be answered.
Limitations
This review is subjected to biases. There was bias in sample selection from searching only MEDLINE for completed RCTs on adult patients that were published in English, although experts in palliative care research were consulted to identify key trials (Appendix 1) to ensure that the final sample of trials retrieved would be as representative as possible. We also did not search trial registers for other papers reporting our included trials, for example, process evaluations. This could be a limitation of the study if papers detailing implementation fidelity were published at a later date. Measurement bias likely prejudices the level of implementation fidelity found in each trial, given the wide variation in level of reporting and limited word count in single publications, although data from multiple reports of the same trials were extracted onto a single checklist. To improve reporting in single publications, authors could be encouraged to publish fidelity information in an online resource and in their appended study protocol.
Conclusion
Implementation fidelity in palliative care is under-recognised. Nevertheless, over 88 strategies have been identified in palliative care RCTs. The use of these strategies and those identified in behavioural change interventions, along with greater awareness on the subcomponents of the elements of implementation fidelity (see Table 2), will be valuable in improving, as well as evaluating, implementation fidelity. Needless to say, more funds are needed in palliative care for education to maintain the minimum fidelity standard, facilitate the implementation of essential components of a complex intervention and to support the development and evaluation of research and clinical services. 70
Footnotes
Acknowledgements
The authors would like to thank Lim Ming Yi for his advice and assistance on earlier drafts; OPTCARE Neuro is a multicentre clinical trial funded by the National Institute for Health Research, Health Services and Delivery Research Programme (NIHR HS&DR, 12/130/47), led by King’s College London, Cicely Saunders Institute, Department of Palliative Care, Policy & Rehabilitation, UK. The views expressed in this publication are those of the authors and not necessarily those of the NHS, the National Institute for Health Research or the Department of Health; Co-Chief Investigators: Higginson, Gao; Co-Investigators: Al-Chalabi, Benz, Burman, Byrne, Chaudhuri, Crosby, Evans, Hotopf, Jackson, Leigh, McCrone, Murtagh, Pickles, Silber, Wilcock, Young; Project Manager: Hepgul, van Vliet (1 April 2014 to 1 April 2015); Site PIs: Ashford – Smith; Brighton – Lindsay; Cardiff – Byrne; Liverpool – Young; London – Bajwah; Nottingham – Crosby; Sheffield – Smith; Trial Statisticians: Gao, Pickles; Health Economists: McCrone, Yi; Collaborators: Turner-Stokes, Murphy; OPTCARE Neuro Team Members: Prof Ammar Al-Chalabi, Dr Sarah Awan, Dr Sabrina Bajwah, Dr Cynthia Benz, Ms Virginia Bray, Dr Rachel Burman, Dr Anthony Byrne, Dr Margred Capel, Ms Gillian Carey, Prof K Ray Chaudhuri, Ms Rebecca Cloudsdale, Dr Vincent Crosby, Ms Joanna Davies, Ms Marsha Dawkins, Dr Catherine Evans, Ms Mim Evans, Ms Sarah Farnan, Dr Wei Gao, Ms Terri Gazi, Dr Karen Groves, Dr Jo Hayes, Dr Nilay Hepgul, Prof Irene Higginson, Prof Matthew Hotopf, Dr Diana Jackson, Ms Kate Jones, Mrs Paramjote Kaler, Ms Loretta Kerr, Dr Nigel Leigh, Dr Fiona Lindsay, Ms Cathann Manderson, Prof Paul McCrone, Ms Caroline Murphy, Dr Fliss E M Murtagh, Mrs Jenifer Newton, Ms Caty Pannell, Ms Louise Pate, Miss Zaynah Sheikh, Prof Andrew Pickles, Dr Eli Silber, Dr Clare Smith, Dr Ellie Smith, Ms Caroline Sunderland, Miss Debbie Tonkin, Prof Lynne Turner-Stokes, Dr Liesbeth van Vliet, Dr Andrew Wilcock, Ms Rebecca Wilson, Dr Deokhee Yi and Prof Carolyn Young; Study Steering Committee Members: Prof Marie Fallon (Chair), Dr Cynthia Benz, Prof Mogens Groenvold, Prof William Hollingworth, Ms Denise Howel, Prof Huw Morris, Mr Foster Murphy, Dr Diane Playford, Prof Julia Riley, Prof Jane Seymour and Dr Andrew Wilcock; Data Monitoring and Ethics Committee Members: Prof Mike Bennett (Chair), Prof Gunn Grande, Dr David Oliver, Prof Raymond Voltz, Prof Stephen Walters; Patient and Public Involvement Committee Members: Dr Cynthia Benz, Mr Sanjay Chadha, Mr David Charlton, Mr Colin Fellows, Ms Helen Findlay and Mrs Savita Jain; The UKCRC-registered King’s Clinical Trials Unit at King’s Health Partners is part funded by the National Institute for Health Research (NIHR) Biomedical Research Centre for Mental Health at South London and Maudsley NHS Foundation Trust and King’s College London and the NIHR Evaluation, Trials and Studies Coordinating Centre (NETSCC). The authors would like to thank Ms Joanna Kelly for the management of the eCRF database and Mrs Beverley White-Alao for providing advice on the Trial Management process; The Collaboration for Leadership in Applied Health Research and Care (CLAHRC) South London is part of the National Institute for Health Research (NIHR) and is a partnership between King’s Health Partners, St. George’s, University London, and St. George’s Healthcare NHS Trust. This publication is independent research supported by the National Institute for Health Research Collaboration for Leadership in Applied Health Research & Care Funding scheme; Prof Irene J Higginson is an NIHR Senior Investigator; systematic review registration no. CRD42015027950.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplementary materials
Underlying research materials related to the paper are available from the corresponding author.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
