Abstract
Background:
Carers of people with advanced cancer play a significant role in managing pain medication, yet they report insufficient information and support to do so confidently and competently. There is limited research evidence on the best ways for clinicians to help carers with medication management.
Aims:
To develop a pain medicines management intervention (Cancer Carers Medicines Management) for cancer patients’ carers near the end of life and evaluate feasibility and acceptability to nurses and carers. To test the feasibility of trial research procedures and to inform decisions concerning a full-scale randomised controlled trial.
Design:
Phase I-II clinical trial. A systematic, evidence-informed participatory method was used to develop CCMM: a nurse-delivered structured conversational process. A two-arm, cluster randomised controlled feasibility trial of Cancer Carers Medicines Management was conducted, with an embedded qualitative study to evaluate participants’ experiences of Cancer Carers Medicines Management and trial procedures.
Setting:
Community settings in two study sites.
Participants:
Phase I comprises 57 carers, patients and healthcare professionals and Phase II comprises 12 nurses and 15 carers.
Results:
A novel intervention was developed. Nurses were recruited and randomised. Carer recruitment to the trial was problematic with fewer than predicted eligible participants, and nurses judged a high proportion unsuitable to recruit into the study. Attrition rates following recruitment were typical for the study population. Cancer Carers Medicines Management was acceptable to carers and nurses who took part, and some benefits were identified.
Conclusion:
Cancer Carers Medicines Management is a robustly developed medicines management intervention which merits further research to test its effectiveness to improve carers’ management of pain medicines with patients at the end of life. The study highlighted aspects of trial design that need to be considered in future research.
Keywords
Carers play an important role in managing patients’ pain medicines at the end of life.
Evidence suggests carers have concerns about pain medicines and lack information and support.
Interventions for carers have not been adequately developed or tested for effectiveness.
A description of a complex intervention to support carers in managing medications at the end of life;
Preliminary evidence of the intervention is acceptable, feasible and beneficial for patient–carer dyads;
Identification of features trial design that could be successfully replicated on a larger scale as well as those that require modification.
The feasibility trial has helped illuminate important design issues to be considered in future research.
The intervention should be evaluated further to examine its effect on outcomes in carers and patients.
Introduction
Many people with advanced cancer experience persistent pain 1 and are typically prescribed analgesics, including opioids. Carers – defined as ‘anyone who cares, unpaid, for a friend or family member who due to illness, disability, a mental health problem or an addiction cannot cope without their support’ (https://carers.org) – often help patients to manage medicines, especially near the end of life (EoL), and their contribution can be crucial to enabling patients to remain at home.2,3 Medication management requires knowledge and practical skill and involves carers in monitoring and interpreting symptoms, as well as selecting, administering and evaluating the effectiveness of medicines.4,5 Internationally, research has repeatedly shown that carers experience difficulties as a consequence of their beliefs about pain and analgesics, particularly opioids,6–8 knowledge deficits 9 and lack of access to information and support.10,11 Most studies conclude that healthcare professionals need to provide carers with more information, training and continuing support.4,8,10–12
While reports and small-scale evaluations of carer participation in medicines management can be found in the literature,13,14 these focus on practice development initiatives, and there is a lack of reliable research on effective methods of supporting carers with medicines management. 15 No study has attempted to integrate interventions with routine palliative care, the majority of interventions lack theoretical underpinning and none were designed with input from carers and clinicians. Evaluating complex interventions in palliative care settings inevitably raises ethical and methodological challenges, 16 but studies reviewed included limited discussion of these and none used qualitative methods to explore participants’ views on the feasibility and acceptability of interventions or study procedures.
Aims and objectives
The study reported here (author submission checklist available online) drew on authors’ previous experience of developing nurse-delivered interventions17,18 and followed Medical Research Council guidance on complex interventions. 19 Phase I aimed to develop a novel, theoretically based, intervention for carers of patients with advanced cancer designed to improve pain medicines management (Cancer Carers Medicines Management (CCMM)) to be delivered by nurses providing home-based palliative care. The aim of Phase II was to conduct a feasibility study, comprising a randomised controlled trial (RCT) and qualitative process evaluation, to test trial procedures and assess acceptability and feasibility of CCMM to carers and nurses to inform decisions concerning a full-scale RCT. Objectives were to determine rates of recruitment and retention, to test acceptability and feasibility of trial procedures, to investigate variability in outcome scores, to establish acceptability and feasibility of CCMM and to identify factors that promote or inhibit its utilisation.
Methods
Phase I: intervention development
CCMM was developed using a multi-method, iterative process, with expert input from a purposive sample of patients (n = 3), carers (n = 12) and health professionals (n = 42: 35 community and specialist palliative care nurses, 3 doctors and 4 pharmacists) from the study sites, who provided information on carers’ needs for support with managing medicines (see Online Appendix 1). The study also included carers from a wider geographical area, who took part in user involvement group consultations (n = 23). Interview guides were developed from critical insights from the initial literature review and were focused on key medicines management issues and possible intervention components. Data from all sources were synthesised and with reference to a theoretical framework (Lazarus’ theory of adaptation, 20 underpinned by Ward’s representational approach to patient education 21 and self-efficacy theory 22 ), a prototype intervention was developed. This was refined further using participatory methods (nominal group technique and ethnodrama) with two groups of nurses (n = 16) working in community palliative care. A panel of nurses (n = 16) and carers (n = 6) then reviewed the refined CCMM materials to confirm relevance to clinical context.
CCMM addresses carers’ beliefs, knowledge and skills and promotes self-evaluation of competence. It centres on a structured conversational process between nurse and carer, which has six components forming the acronym CARERS (Table 1).
Components of Cancer Carers Medicines Management (CCMM).

Phase II: CCMM feasibility study
Trial design
A two-arm, parallel group, randomised controlled feasibility trial of CCMM was conducted at two sites, in south Wales (SW) and southern England (SE). Nurses were randomly allocated to intervention or control (usual care) arms of the study at each site. Clusters were of patient–carer dyads on the caseload of each nurse. This cluster design was used to minimise risk of contamination. The nature of the intervention precluded concealment of treatment allocation. Further details can be found in the study protocol. 23
Setting
In SW, health board–employed district nurses had generic caseloads that included patients with palliative care needs and in SE, two independent hospice palliative care providers employed nurse specialists to work in the community. These contrasting models are typical of nurse-delivered EoL care in the United Kingdom and were chosen to assess feasibility issues associated with each. Nurse volunteers for the study were sought at each site: invitations were given directly to nurses who had recently participated in EoL care training (SW) and issued via nurse managers (SE).
Prior to randomisation, nurses completed the Knowledge and Attitudes Survey Regarding Pain 24 to compare pain management knowledge across the two groups of nurses. Each nurse was given a sealed envelope containing group allocation, determined independently (by P.S.) using simple randomisation and concealed until nurses opened the envelopes.
Participants
The study used a consecutive convenience sample of patients on the study nurses’ caseloads. Nurses screened patients for eligibility (see Table 2) and were asked to consider all dyads meeting eligibility criteria as potential participants, in combination with clinical judgment about appropriateness of inviting participation.
Carer eligibility criteria.

Capacity to consent was judged to be present when the patient and carer could understand the information relevant to participating in the study, retain that information, use or weigh that information as part of the process of making the decision and communicate his/her decision.
Intervention delivery
Intervention group nurses received one day’s training on CCMM, designed and facilitated by specialist palliative care educators (S.A., S.D.). Training included preparation for and rehearsal of the CCMM intervention, as well as an overview of the study design and processes. To support intervention, delivery nurses were provided with a conversational process script, access to an online video demonstration and a prompt card. Nurses were asked to document use of CCMM components by completing a structured reflective record soon after each CCMM consultation.
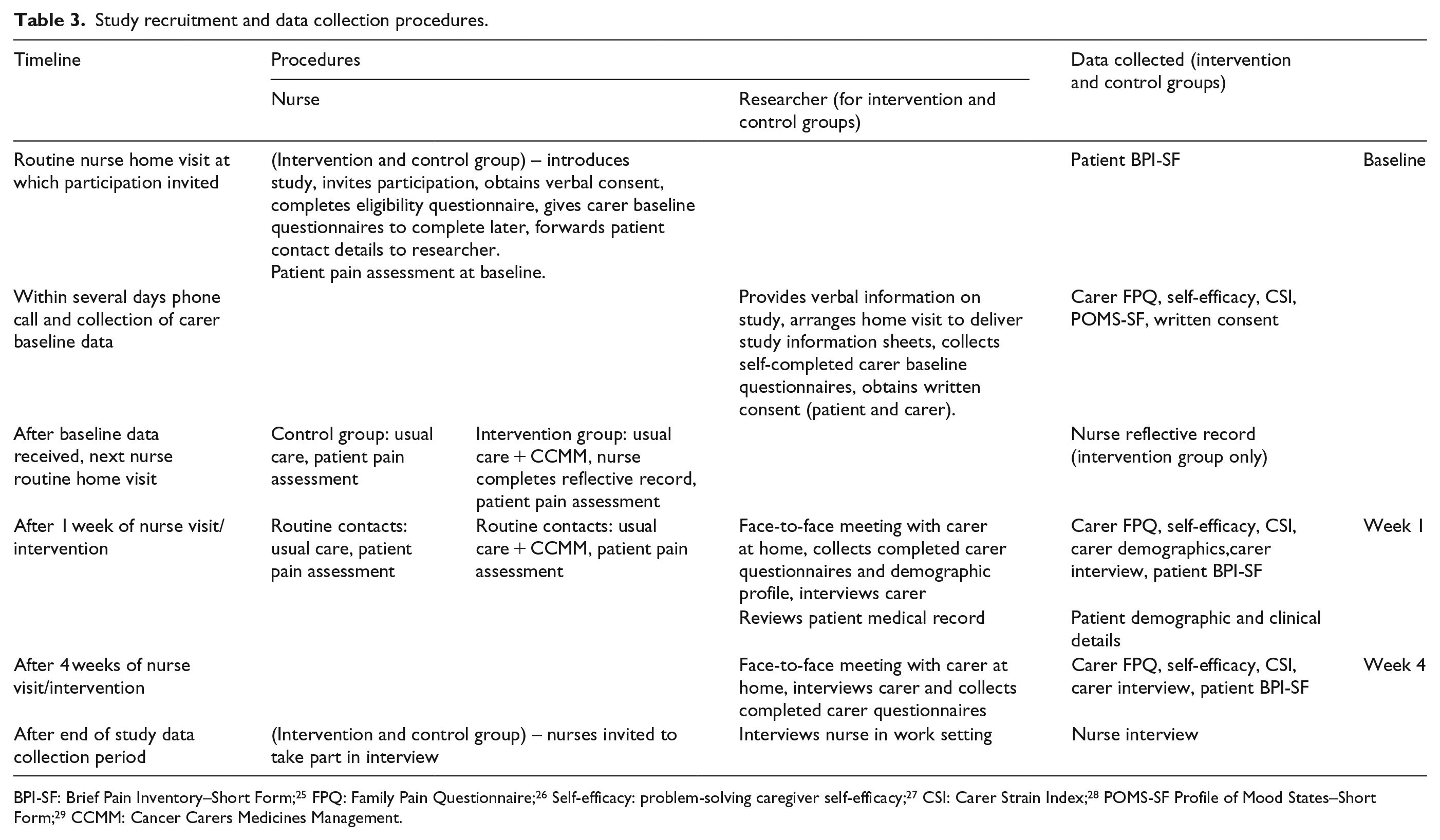
Trial procedures
Study recruitment and data collection procedures are summarised in Table 3. Following carer completion of baseline questionnaires, nurses in the intervention group delivered CCMM to carers at the next appropriate home visit. At subsequent routine contacts, nurses could address participants’ questions and reinforce the intervention (Table 1).
Study recruitment and data collection procedures.
Outcomes
Feasibility and acceptability
Data were collected on numbers of dyads screened, assessed as eligible, recruited and retained. Nurses’ and carers’ experiences of research procedures and CCMM (intervention group) or pain medication management practices (control group) were explored in face-to-face semi-structured qualitative interviews. Interview guides were informed by normalisation process theory (NPT), a framework for assessing compatibility of trial procedures with clinical practice and identifying factors that influence perception and utilisation of complex interventions. 30 Intervention group nurses’ reflective records were used to capture intervention delivery and utilised in conjunction with interview data to assess fidelity of intervention delivery.
Carer and patient outcomes
Measures assessed for acceptability and responsiveness to CCMM exposure in carers were knowledge, beliefs and skills in cancer pain medicines management (Family Pain Questionnaire 26 ); self-efficacy in pain management (Zeiss Caregiver Problem-Solving Self-Efficacy 27 ) and strain (Caregiver Strain Index 28 ). A secondary outcome was patients’ perceived pain (Brief Pain Inventory–Short Form 25 ). Data collection points were selected to test sensitivity of measures and to test feasibility of completing a measure at 4 weeks post-intervention in people approaching EoL. Carers also self-completed an assessment of mood (Profile of Mood State–Short Form 29 ) at baseline only. Carers in both intervention and control groups were asked to complete measures at baseline, 1 and 4 weeks and to take part in 1- and 4-week interviews.
Sample size
A target sample size of 30 patient–carer dyads per arm was selected to enable estimation of sample size for a possible follow-on RCT 31 based on the outcome measure showing greatest change in response to CCMM. Previous research with a similar group of carers suggested we should assume a 50% non-participation and attrition rate; 32 therefore, the plan was to approach up to 120 patient–carer dyads.
Analysis
Rates of eligibility, recruitment and attrition were calculated as percentages. Interviews were audio-recorded, transcribed verbatim and anonymised. Analysis, using the framework approach, 33 was led by one researcher (E.L. – carer interviews and J.A.H. – nurse interviews) with research team members (S.L., J.B.H., A.R. and C.M.) reading a sample of transcripts. For the nurse interviews, thematic frameworks, generated inductively and informed by NPT, were used to organise data. Due to the small sample size of carers, case reports were constructed; themes were synthesised from these into a composite, interpretive ‘case study’. 34
Ethics approval
The study was approved by NRES Committee South Central Hampshire B (ref. 12/SC/0365, 4 September 2012). Research governance approval and permission for researcher access were granted by NHS organisations, independent healthcare providers and other gatekeeper bodies. All patients, carers and healthcare professionals participating in Phase II provided written informed consent.
Results
Phase II
Feasibility and acceptability of trial procedures
Recruitment and retention of nurses
The target number of 12 nurses took part in the study. Two nurses at each site (2 from control group and 2 from intervention group) withdrew for reasons unrelated to the research. A total of 10 nurses took part in post-study interviews.
Nurse pain knowledge and attitudes
Nurses at the two study sites had similar baseline pain knowledge and attitudes scores: nurse specialists (SE): mean = 29/40, range = 18–35; district nurses (SW): mean = 27/40, range = 24–31. Post-study re-testing showed little change (SE: 30/40, 25–34 and SW: 29/40, 26–32).
Randomisation
No difficulties were encountered in randomising nurses. At interview, some nurses said they had entered the study with a preference for the intervention group, but all accepted their group allocation. Analysis of interviews indicated control group nurses had not become aware of intervention details or altered their pain management practice. Interviews with carers did not reveal any concerns about their nurse’s group allocation.
Eligibility, recruitment and retention of carer participants
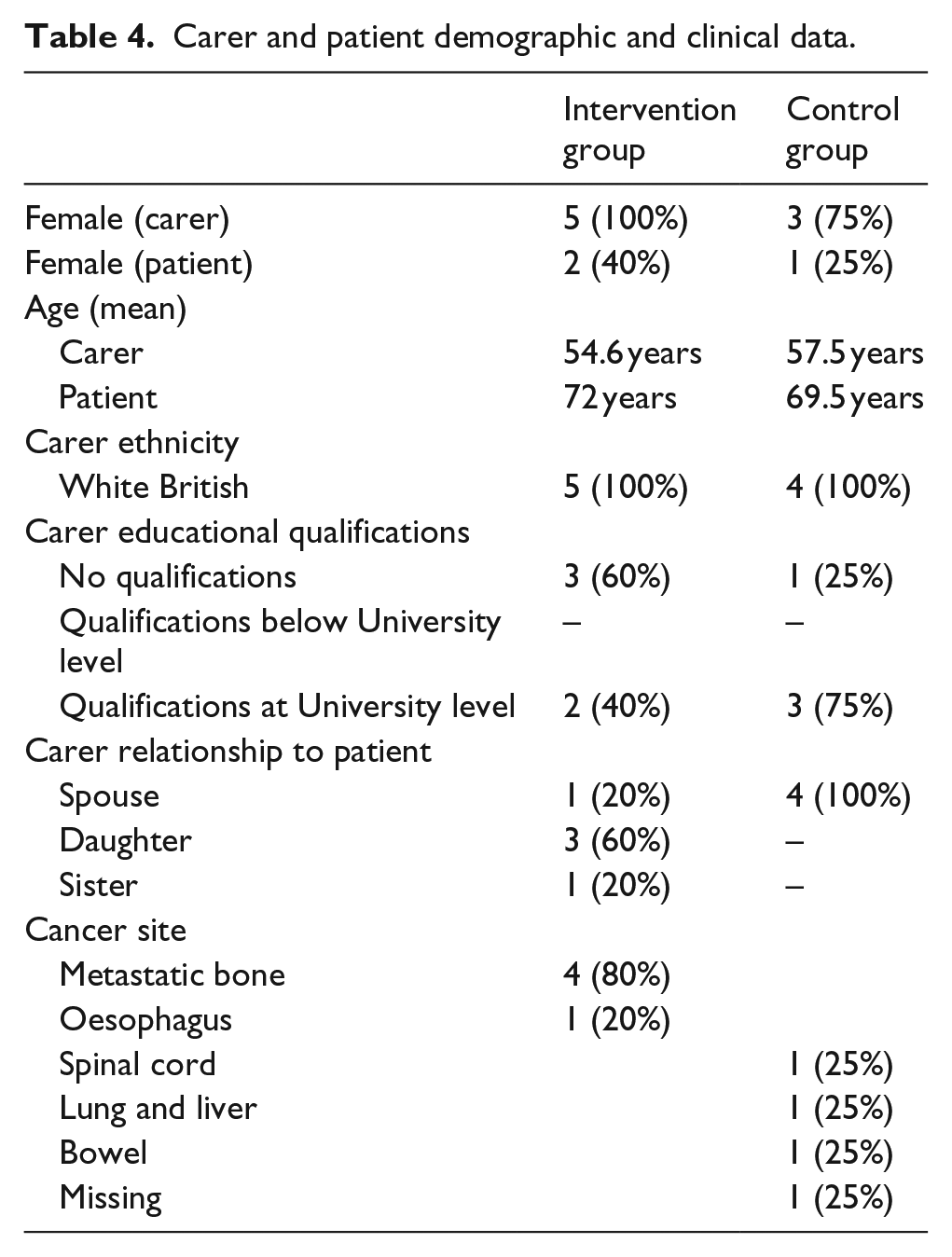
Data collection took place between March and September 2014. The flow of participants through the trial is shown in Figure 1. Full demographic and clinical data were collected on patients and carers (n = 9) who completed the trial (see Table 4).
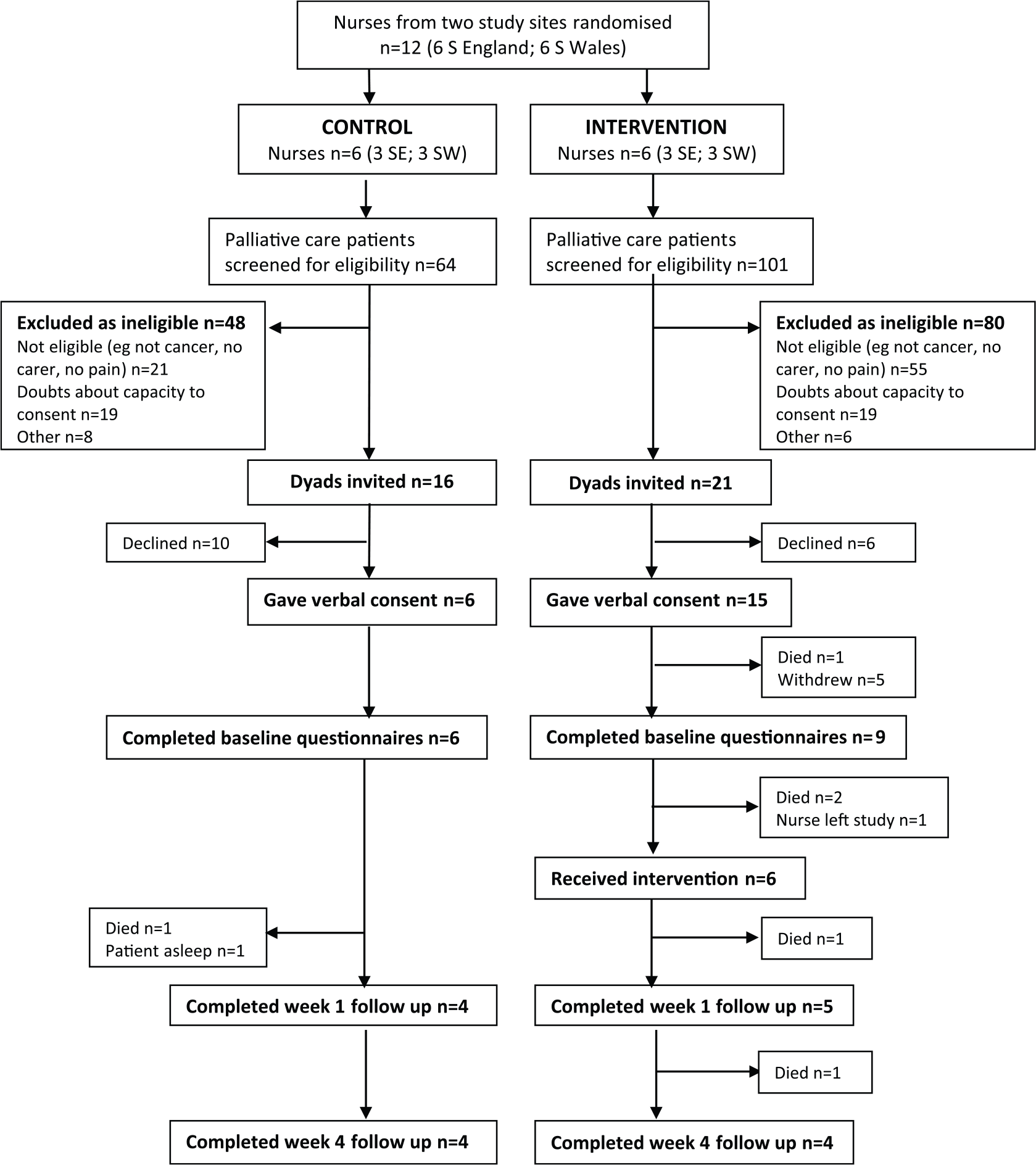
CONSORT flow diagram for the CCMM study.
Carer and patient demographic and clinical data.
Nurses received fewer than predicted referrals during the study period and assessed a high proportion of them as ineligible (78%) (reasons included non-cancer, no pain, no carer and other perceived physical or psychological demands on them). Initial estimates of referrals may have been inaccurate, but during the trial, contextual factors (i.e. reorganisation of nursing teams, staff turnover and sickness absence) and changes in clinical practice (e.g. patients being referred very close to EoL to district nurses) impacted unpredictably on the number of eligible patients. On average, nurses approached three dyads each during the 6-month data collection period (median = 3 and range = 0–8), obtained verbal consent from 1.75 dyads (median = 1.5 and range = 0–6) and recruited 1.25 dyads (median = 1 and range = 0–3).
Analysis of nurse interviews distinguished various obstacles to recruitment (Table 5). These included reports of ‘protecting’ patients and carers from perceived additional burden and distress that might be raised by demands of the research and nurses’ not approaching some patients/carers to avoid their own disappointment caused by dyads declining participation.
Factors influencing recruitment of study participants.

[…]: text of transcript has been edited for clarity and brevity.
Quotes are tagged with a composite code that provides unique nurse study ID (first letter followed by two digits), group allocation (i or c, intervention or control) and study site (south England (SE) or south Wales (SW)).
The demographic and clinical profiles of participating dyads suggest that nurses were selective: carers were relatively young (mean age = 56 and range = 37–68) and the majority had university level education. Average scores for mood disturbance (POMS-SF; median = 25 and interquartile range (IQR) = 13–36) and carer strain (Carer Strain Index (CSI); median = 4 (out of 12)) were low. Most dyads had medicines management routines in place, and carers reported experience of medicines management in their personal or occupational background.
Data collection
Feasibility of self-completed questionnaires for measuring carer outcomes was confirmed: 100% of POMS questionnaires were returned at baseline and approximately 90% of all other questionnaires were returned at each time point, with relatively few missed items or obviously incorrect responses.
Nurses completed 85% of patient pain assessments with the patient using Brief Pain Inventory–Short Form (BPI-SF). At interview, some highlighted difficulties, particularly for patients who were very ill or reluctant to talk about their illness. Several nurses described the assessment as ‘cumbersome’ and ‘quite onerous’ and reported adapting the questionnaire. These findings raise questions about the feasibility of nurse-administered pain assessment using BPI-SF.
Three nurses completed reflective records for four of the six visits at which CCMM was initially delivered. The contemporaneous documents supplemented accounts of intervention delivery given in interviews. However, self-report provided insufficient data to reliably assess intervention fidelity; it would have been preferable to observe or audio-record a sample of nurse visits at which CCMM was used.
Acceptability and utilisation of CCMM
Nurses’ use of CCMM
Findings from intervention group nurse interviews (n = 6) were mapped against NPT categories of implementation work (sense-making, participation, action and monitoring) to assess CCMM’s implementation potential and identify how it might be optimized. 23 Full details are provided in Online Appendix 2 and summarised below.
CCMM made clinical sense to nurses, who recognised the challenges faced by carers managing analgesia at the EoL and saw potential benefits in improving education and support. However, nurses did not find CCMM sufficiently new or distinctive, and said they were already using some components of the intervention, although this claim was not supported by control group nurses’ descriptions of routine practice. Nurses tended to equate CCMM with providing written information and resources, rather than seeing it as a conversational and educational process.
Positive experiences of CCMM training helped nurse engagement. However, due to low recruitment, each nurse used CCMM with only one or two dyads and not all nurses became confident delivering it. Nurses gave examples of CCMM benefiting carers, although some nurses were concerned that focusing specifically on pain could potentially increase patient/carer anxiety about this issue. At the end of the trial, CCMM was compared favourably with current practice because it offered a more systematic and comprehensive approach to supporting carer management of pain medicines. Nurses particularly valued the toolkit resources, that is, information about opioids and simple charts for documenting pain and medication, because they were of immediate practical value to carers.
Nurses found CCMM generally compatible with existing work practices: it was easy and quick to deliver and adaptable to family circumstances, concerns raised by carers and the time available during a visit. However, CCMM resources were not always used selectively as had been intended: nurses tended to give the complete toolkit to carers. Nurses found CCMM’s specific focus on pain medicines difficult to reconcile with their holistic approach to managing a range of EoL symptoms and associated medicines. They also suggested that expanding the intervention to include life-limiting conditions other than cancer would increase its applicability and enhance its compatibility with palliative care nursing practice. Nurses also favoured introducing CCMM earlier in the course of a patient’s illness, which was felt more appropriate and of greater benefit to carers.
Carer experience of CCMM
Nine family carers were interviewed (five intervention and four control; 17 interviews). Although carers had some difficulty in isolating CCMM from their overall experience of caring for a patient at the EoL, most commented on the value of CCMM resources in the toolkit, particularly for information, reassurance and supporting problem-solving. Within the data, we identified some positive changes in medicines management, such as increased acceptance of the need for opiates, knowledge being reinforced or enhanced and behavioural change, for example, responding more readily to patients’ requests for pain relief and improved systems in place for giving and recording medicines. There were no reports from carers of CCMM increasing the burden of caring or otherwise causing harm or distress (Online Appendix 3).
Discussion
Phase I enabled the development of a novel, theory-based intervention to support carer management of pain medicines, grounded in current EoL care context, as well as research and expert evidence. Although caution in interpretation is required due to the small sample, data indicate that the intervention was acceptable to nurses and carers, was feasible to deliver in practice, and showed some benefits.
Qualitative data from carers were suggestive of potential benefits for their knowledge, beliefs and behaviours related to management of pain medicines. However, some nurses reported that CCMM could potentially raise patient and carer anxiety through its focus on pain. Due to problems with recruiting our target sample size, we were unable to conduct a reliable analysis of variability in outcome scores and therefore select a primary outcome or determine sample size for future research. All carer outcome measures proved feasible to complete, with little missing data, suggesting suitability for future research, but nurses’ reported difficulties measuring patient pain using the Brief Pain Inventory (BPI) and alternative methods may be required.
The need for CCMM made sense to nurses and was largely compatible with their clinical practice and helped to systematise it. However, nurses highlighted challenges caused by focusing solely on pain, and in future research, consideration should be given to expanding CCMM to include medicines used to manage other symptoms at EoL, making it more compatible with nurses’ holistic focus. Nurses had some difficulty distinguishing CCMM from routine practice and tended to distribute the written resources indiscriminately to carers rather than engaging them in a conversational process. Nurses needed more opportunity to reflect on current practice and practise CCMM before the trial proper to be confident about and competent in using CCMM.
Some aspects of study design proved feasible and could be replicated in a larger scale trial: both specialist palliative care nurses and community nurses were recruited, and randomisation was acceptable; in keeping with findings from other studies, 35 using a cluster design with nurses randomised within-site did not prove problematic with respect to control group contamination. The study also reinforced the usefulness of qualitative methods of evaluation in studying experiences of acceptability and feasibility.
The main obstacle to feasibility was carer recruitment, partly attributable to fewer than expected eligible participants. Rather than simply accepting managers’ estimates of palliative care referrals, we could have carried out a rapid audit to check viability. Recruitment problems were also attributable to low proportions of eligible carers invited by nurses. Our findings are suggestive of a high degree of gate-keeping by nurses, similar to recruitment problems described in other recent trials.36,37
Organisational issues and fundamental conflicts between clinical and research roles appeared to exist. Schildmann and Higginson 16 recommend screening for eligibility is done by research staff, while Stone et al. 38 argue for careful monitoring of the recruitment process, so that reasons for low recruitment by clinical staff can be identified and managed. Research staff access to patients and carers may also be facilitated by recruiting at hospital discharge or through out-patient clinics.
Gatekeeping may also result in selection bias and thus a risk of inequity in patient and carer access to an intervention. The small sample of dyads recruited to this study did not appear to be typical – they were younger and functioning well – a theme in other studies. 39 In a pragmatic study, it is perhaps impossible to completely eliminate risks of selection bias, but checks can be introduced, so that biases can be identified. We did not collect data on all eligible participants, so could not systematically compare the characteristics of those nurses invited and those they did not. This should be part of the protocol of future studies in which healthcare professionals recruit participants, if ethics committees are prepared to allow this. 40
Finally, it may be important to reconsider the recruitment of dyads and focus on carer outcomes alone, as CCMM is a complex psychoeducational intervention for carers. Parallel consent of both patient and carer made the recruitment process complex with exclusion of some carers because the patient did not have capacity to consent. Others have also concluded that recruiting both patients and carers extends the recruitment phase. 41
Conclusion
CCMM is a robustly developed, novel medicines management intervention which merits further evaluation to test effectiveness at improving carers’ management of pain medicines with patients at EoL. This feasibility trial has helped illuminate important design issues that would need to be incorporated into a definitive trial.
Footnotes
Acknowledgements
The authors like to thank all of the nurses, carers and patients who participated in this study. Trial registration number: NCRI Palliative & Supportive Care Clinical Studies Group CSP: 105045. For access to research data from this study, please contact the corresponding author. S.L., J.B.H. and A.R. proposed the idea for and developed the study; S.L. and J.B.H. managed the study; E.L. and J.H. collected data; S.D. and S.A. facilitated the intervention development and training workshops and provided on-going education and clinical advice; M.B. provided on-going clinical and research advice; E.L., J.H., J.A.H. and J.B.H. conducted data analysis; C.M. advised on Normalisation Process Theory and contributed to qualitative data analysis; P.S. advised on statistical aspects of research design and data collection; J.A.H. and S.L. wrote the first draft of the paper; all authors contributed to further redrafting of the final manuscript; and S.L. is guarantor.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Professor Hopkinson reports personal fees paid to Cardiff University from Helsinn Healthcare, Chugai Pharma, Healthware and other from Chugai Pharma, outside the submitted work and independent member (non-executive director), Velindre NHS Trust, Cardiff, Wales. Ms Jane Hughes reports personal fees from University of Southampton during the conduct of the study. Professor Richardson received personal fees from Cancer Partners UK until May 2015, outside the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Dimbleby Marie Curie Research Fund DCMC-RF-12-05.
