Abstract
Background:
Despite fast-growing ‘older old’ populations, ‘place of care’ trajectories for very old people approaching death with or without dementia are poorly described and understood.
Aim:
To explore end-of-life transitions of ‘older old’ people across the cognitive spectrum.
Design:
Population-based prospective cohort (United Kingdom) followed to death.
Setting/participants:
Mortality records linked to 283 Cambridge City over-75s Cohort participants’ cognitive assessments <1 year before dying aged ≥85 years.
Results:
Overall, 69% were community dwelling in the year before death; of those with severe cognitive impairment 39% were community dwelling. Only 6% subsequently changed their usual address. However, for 55% their usual address on death registration was not their place of death. Dying away from the ‘usual address’ was associated with cognition, overall fewer moving with increasing cognitive impairment – cognition intact 66%, mildly/moderately impaired 55% and severely impaired 42%, trend p = 0.003. This finding reflects transitions being far more common from the community than from institutions: 73% from the community and 28% from institutions did not die where last interviewed (p < 0.001). However, severely cognitively impaired people living in the community were the most likely group of all to move: 80% (68%−93%). Hospitals were the most common place of death except for the most cognitively impaired, who mostly died in care homes.
Conclusion:
Most very old community-dwelling individuals, especially the severely cognitively impaired, died away from home. Findings also suggest that long-term care may play a role in avoidance of end-of-life hospital admissions. These results provide important information for planning end-of-life services for older people across the cognitive spectrum, with implications for policies aimed at supporting home deaths.
MeSH Terms:
Cognitive impairment, Dementia, Aged, 80 and over, Aged, frail elderly, Patient Transfer, Residential characteristics, Homes for the aged, Nursing Homes, Delivery of Health Care, Terminal care
Other key phrases:
Older old, Oldest old, Place of death, Place of care, End-of-life care.
Keywords
More people will be dying in very old age and dying with dementia or cognitive impairment.
Half the very old move in the last year of life and die away from their usual residence, but it is poorly understood how cognitive impairment affects these transitions.
Prospective population-based research with very old people, including cognitive assessments, reveals patterns in individual trajectories through care settings that routine data cannot explore.
Although 39% of ≥85-year-olds with severe cognitive impairment live in the community in their last year of life, only 8% die there, compared with 20% home deaths among cognitively unimpaired ≥85-year-olds.
The proportions of very old people dying in hospitals or in care homes decrease and increase, respectively, with worsening cognitive impairment.
Community end-of-life care provision needs to increase if more very old people are to be supported to die at home.
Developing services and training in dementia and end-of-life care for frail older people in hospitals and care homes should be high priority, as these are currently where most very old people die.
As populations age, good dementia care is an increasingly crucial element of good end-of-life care in all settings.
Introduction
Dementia is an important cause of cognitive impairment in older people, affecting more than 35 million people worldwide in 20101,2 and is forecast to increase most steeply in low- and middle-income countries.3–5 It is associated with significant disability for patients, considerable caregiver burden and increased use of health and social services.6–10 The United Kingdom estimates forecast an increase from approximately 750,000 individuals currently affected to more than 1.7 million by 2051 as the population ages. 6 Dementia prevalence not only increases with advancing age but also with proximity to death across all older age-bands.3,6,11 As the fastest growing population sector in the developed world is people aged 85 years or older, the rising number of very old people, with or without dementia, needing care towards the end of their lives is a growing public health concern.2,12,13
National Health Service (NHS) end-of-life care policy seeks to facilitate ‘good deaths’ by enabling more people to die at home if they wish and reducing admissions to hospitals in the last weeks of life. 14 Place of death and changes in locus of care before death have been suggested as quality indicators for end-of-life care,14,15,16 and provide important information for planning and organising end-of-life services and monitoring improvements. To date, little has been reported about changes in place of care of older people before death, 17 as prospective studies of the population must follow sufficient numbers to death to collect such evidence.
We have previously reported transitions in care and place of death of ‘older old’ people aged at least 85 years in the last year of life, 18 52% dying away from their place of residence. Whether these transitions are different for individuals with cognitive impairment has not been investigated previously, despite the fact that their care needs and capacity to consent may differ compared with the cognitively unimpaired. The present analysis aims to identify cognitive impairment, including dementia, among individuals interviewed for the Cambridge City over-75s Cohort (CC75C) study less than 1 year before they died aged 85 years or more, and to compare place of death and end-of-life transitions in place of residence or care experienced by ‘older old’ people of different cognitive status.
Methods
Study cohort
The CC75C study is a longitudinal population-based cohort study of cognitive and functional changes in advanced old age (http://www.cc75c.group.cam.ac.uk). The study design and methods have been described elsewhere19,20 following Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines (http://www.strobe-statement.org/). Men and women aged 75 years or over (n = 2610) were recruited from seven socially and geographically representative general practices in Cambridge; six practices continued in the follow-up. Participants were interviewed in their own home at baseline (1985–1987, response rate 95%) and re-interviewed every few years. Mortality accounts for most of the study’s attrition between interview waves: 76% overall and even higher in older age-bands. Loss to follow-up was minimised by approaching relatives of participants for proxy informant interviews if participants were too frail or confused to participate themselves. Cognitive assessments at each survey included the Mini-Mental State Examination (MMSE), 21 and sub-samples had further diagnostic assessment, the Cambridge Mental Disorders of the Elderly (CAMDEX). 22 Retrospective informant interviews conducted after a participant’s death contributed to the clinical diagnosis of dementia at death 23 (see Box 1). Each CC75C study phase was approved by Cambridge Research Ethics Committee.
Definition of cognition and dementia status.
Assessments of cognitive function in every survey included the Mini-Mental State Examination (MMSE). 17 Participants with MMSE scores from assessments conducted in the last year of life were included in the study’s primary analysis (n = 283), categorised as
‘Severe cognitive impairment’ – MMSE scores 0–17/clinical diagnosis of moderate/severe dementia.
‘Mild/moderate cognitive impairment’ – MMSE scores 18–25/clinical diagnosis of minimal/mild dementia.
‘No cognitive impairment’ – MMSE scores 26–30/dementia clinically absent.
Clinical diagnostic assessments, following the Cambridge Mental Disorders of the Elderly Examination (CAMDEX), 18 were conducted by old age psychiatrists in sub-samples of participants. Additionally, clinical diagnosis of dementia at death was made by at least two clinicians experienced in old age psychiatry according to the American Psychiatric Association’s criteria (Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) for participants in CC75C’s brain donation programme. This diagnosis was based on review of all available information, including survey data, proxy informant data, general practitioner (GP) report of dementia diagnosis, death certificates and retrospective data from informant interviews conducted after participant’s death. 19 Participants for whom it was possible to confirm the absence or presence of dementia at death (n = 142) were included in the study’s secondary analysis, categorised as
‘Moderate/severe dementia’ – CAMDEX assessment/consensus clinician review/death certificate recording any dementia/GP report
‘Minimal/mild dementia’ – CAMDEX assessment in the last year of life/consensus clinician review
‘No dementia’ – CAMDEX assessment in the last year of life/consensus clinician review
Study sample
A total of 321 participants died aged 85 years or over within a year of taking part in one of CC75C’s Surveys 1 to 7 (1985–2007), 80% of those alive at survey, see Figure 1. Participants who had insufficient evidence to confirm presence or absence of cognitive impairment close enough to death (n = 37), and who had no death registry residence information available (n = 1), were excluded from analysis. This article presents analyses of participants in terms of their cognitive status (n = 283): similar results from analyses of the smaller sub-sample with known clinical dementia status (n = 142) are available from the authors on request.
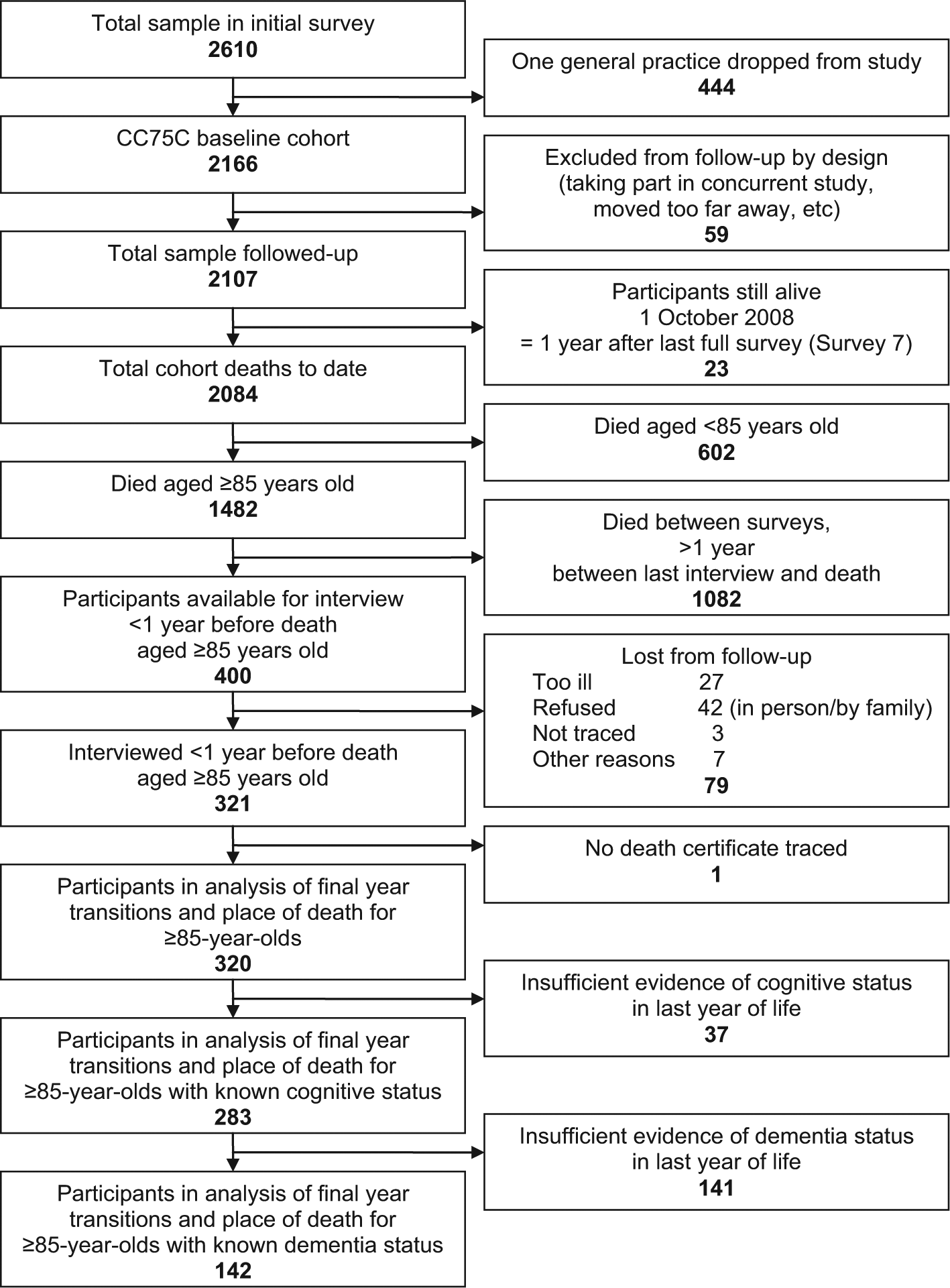
CC75C study sampling frame: participants who died aged ≥85 within a year of interview with cognitive assessment or confirmed dementia status. Flowchart illustrating how the sample for these analyses was derived from the full cohort: n = 283 people with a cognitive assessment in their last year of life and n = 142 people with confirmation of dementia diagnosis or absence of dementia.
Place of residence and place of death
Interview data (‘accommodation type when last interviewed’) and death registration data (‘usual address at time of death’ and ‘place of death’) were used to identify transitions in place of residence or care between the last interview and death. Community-dwelling categories were either ‘own home’ (‘house/flat/granny flat’ regardless of ownership) or ‘sheltered housing’ (individual apartments in housing scheme for older people with limited support). For analyses comparing community versus institutional residents, the category ‘long-term care’ included ‘residential care home’, ‘nursing home’ and ‘long-stay hospital ward’.
Analysis
Analyses were primarily descriptive, but analyses of association also explored significance of between-groups differences. Pearson’s χ2 or Fisher’s exact tests were used for differences in proportions of categorical variables, Kruskal–Wallis non-parametric analysis of variance (ANOVA) for differences in medians of non-normally distributed continuous variables and Cochran’s Q test for differences in proportions in different settings at different time-points. All analyses were performed using Stata Version 9.2.
Results
Table 1 shows the demographic characteristics of the 283 participants stratified by cognitive status. When interviewed in the last year before death, two-fifths had no cognitive impairment, one-quarter had mild/moderate cognitive impairment and one-third had severe cognitive impairment. The mean age at time of death of all the participants was 90.4 years (standard deviation (SD) = 4.5; range = 85–106), median 89 years (interquartile range (IQR): 86–93) and age at death was higher among those with greater cognitive impairment (p < 0.001). Most were women (67%), had left school before the age of 15 years (70%) and were widowed (66%), especially those with severe cognitive impairment. Approximately, half of the final interviews were conducted in the last 6 months of life; proximity to death had no relation to cognition. Among those whose dementia status was known, one-quarter had no dementia, one-quarter minimal to mild dementia and half moderate to severe dementia.
Demographic characteristics of the sample by cognitive status.
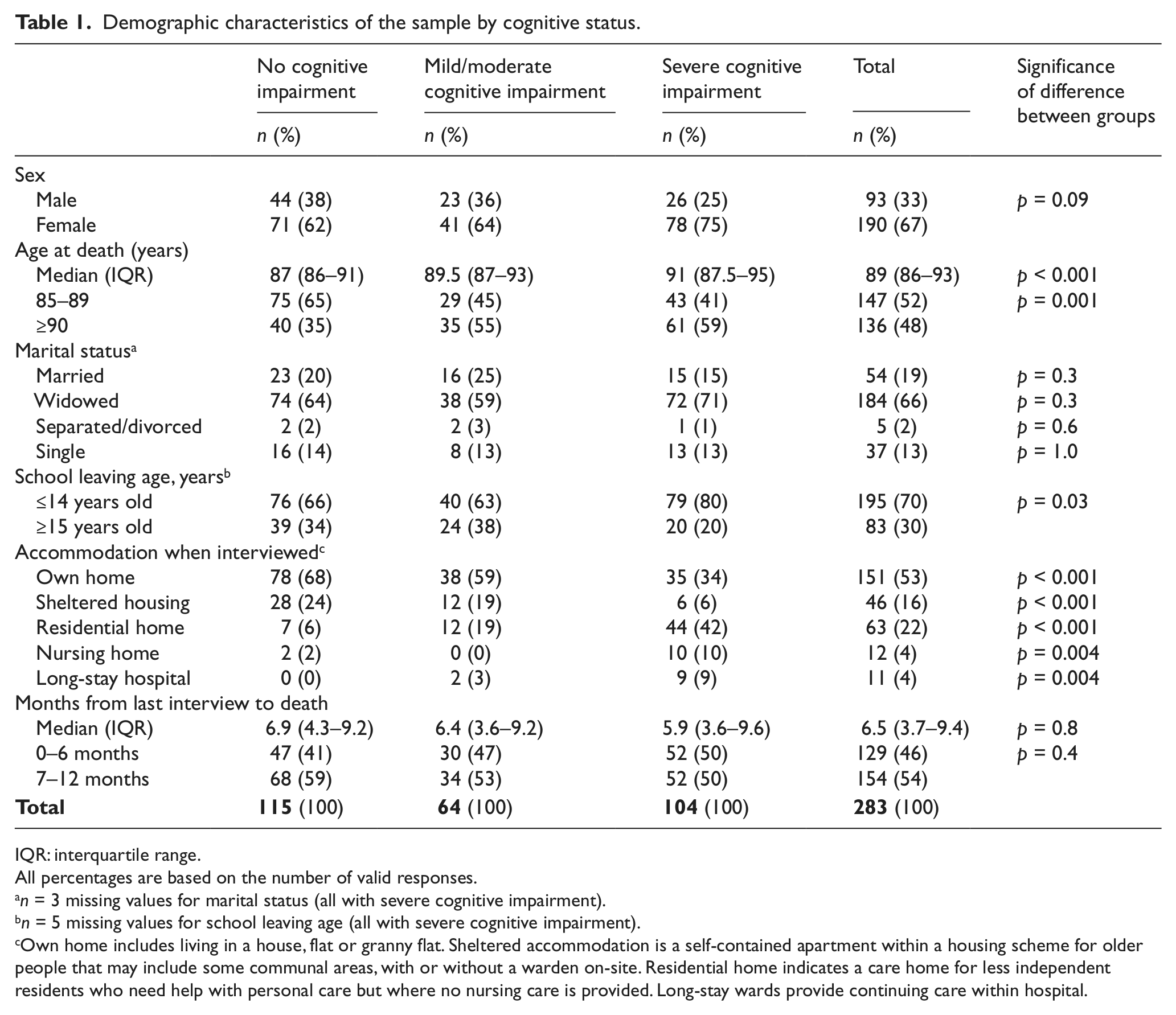
IQR: interquartile range.
All percentages are based on the number of valid responses.
n = 3 missing values for marital status (all with severe cognitive impairment).
n = 5 missing values for school leaving age (all with severe cognitive impairment).
Own home includes living in a house, flat or granny flat. Sheltered accommodation is a self-contained apartment within a housing scheme for older people that may include some communal areas, with or without a warden on-site. Residential home indicates a care home for less independent residents who need help with personal care but where no nursing care is provided. Long-stay wards provide continuing care within hospital.
Address when interviewed less than a year before death
Two-thirds of the participants were community dwelling when last interviewed, more 85- to 89-year-olds than ≥90-year-olds (80% vs 59%, p < 0.001) and more men than women (84% vs 63%, p < 0.001). A third of those with severe cognitive impairment lived at home and 6% in sheltered housing. Individuals who lived in care homes at their final interview included relatively more ≥90-year-olds than 85- to 89-year-olds (35% vs 18%, p = 0.001) and more women than men (33% vs 14%, p = 0.001).
Usual address at death
According to the ‘usual address’ recorded at death registration, relatively few participants changed their address during the last year of life. Figure 2 displays, by cognitive status, the proportions of participants living in different settings at death and the proportions of participants dying at different places of death. Table 2 summarises the detail in Figure 2 into four residential categories for all three known time-points in the last year of life. By the time of death most of those with ‘no cognitive impairment’ (90%) or ‘mild/moderate cognitive impairment’ (78%) remained community dwelling. Participants with severe cognitive impairment were more likely to live in care homes than in the community, although more than a third were still living in the community at death.
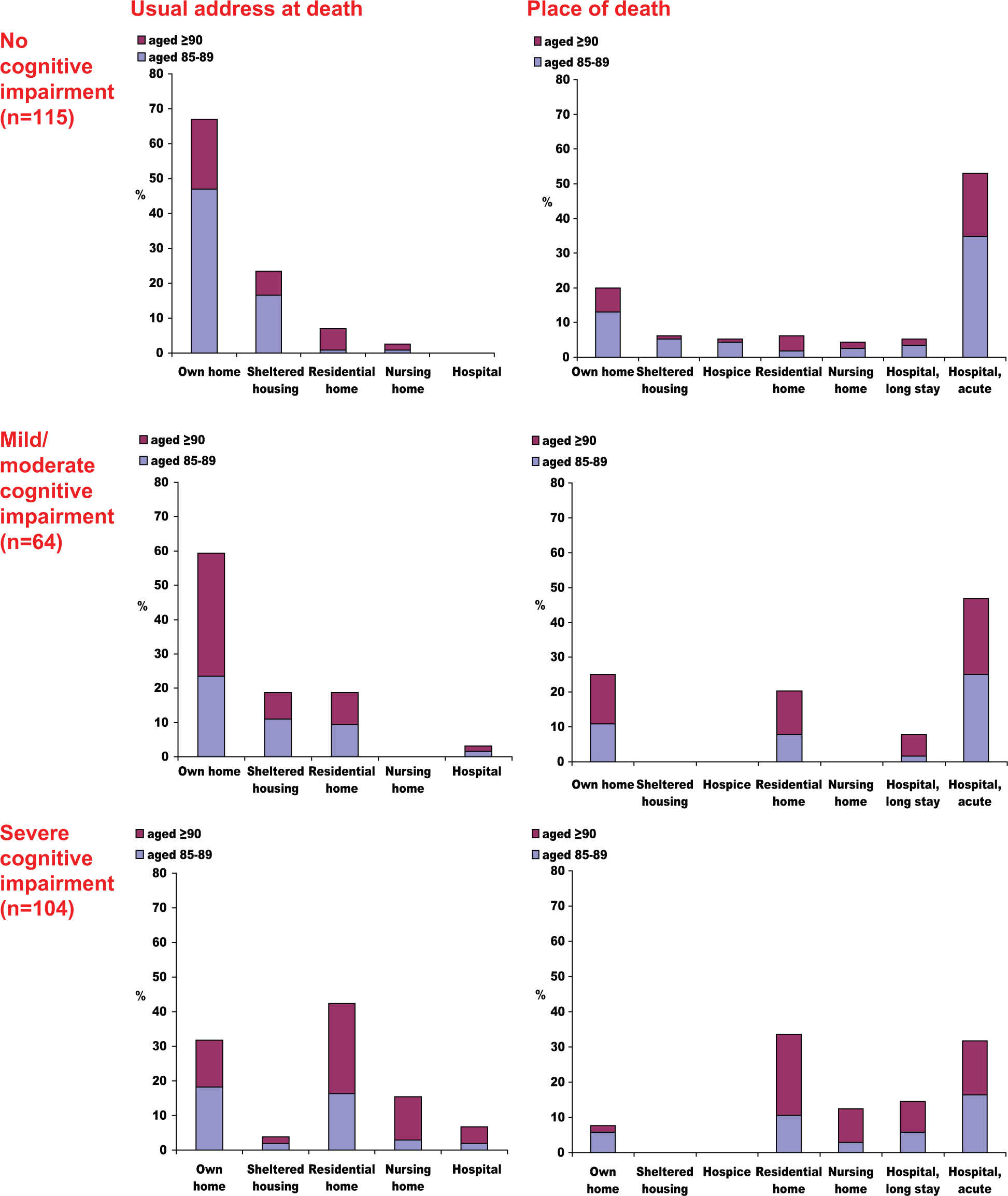
Usual address at death and place of death by cognitive status. Patterns of residence (‘usual address’ registered at death) and place of death were markedly different for people with severe cognitive impairment compared with people with no/mild/moderate cognitive impairment.
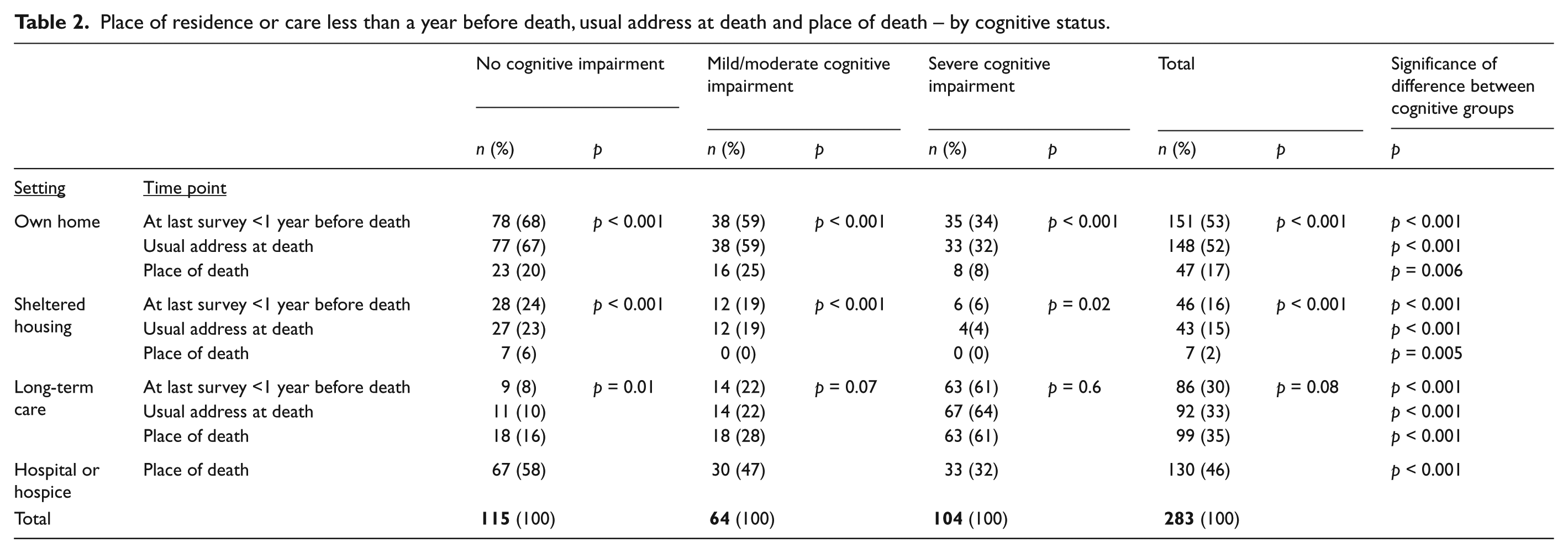
Place of residence or care less than a year before death, usual address at death and place of death – by cognitive status.
Place of death
No one with any cognitive impairment died in a hospice, the setting for only 2% (n = 6) of deaths overall. Acute hospital deaths were the most frequent for those who were cognitively intact (58%) or with mild/moderate cognitive impairment (47%). A third of those with severe cognitive impairment died in acute hospitals, while nearly two-thirds died in long-term care (34% residential homes, 13% nursing homes, 14% long-stay wards). Care homes were place of death for only one-tenth of those without cognitive impairment and one-fifth of those with mild/moderate cognitive impairment; a further 5% and 8% of those without cognitive impairment and those with mild/moderate cognitive impairment, respectively, died in long-stay hospital wards. Dying at home was more likely among individuals with mild/moderate cognitive impairment (25%) or none (20%) than among individuals with severe cognitive impairment (8%). Only seven individuals died in sheltered housing, all without cognitive impairment (Figure 2, Table 2).
Women and those aged 90 years or above were more likely to die in care homes (men 10%, women 34%, p < 0.001; 85- to 89-years-olds: 16%, ≥90-year-olds: 36%, p < 0.001), especially those with severe cognitive impairment. Of those with any cognitive impairment, men and those aged 85–89 years died more often in acute hospitals (men: 53%, women: 31%, p = 0.008; 85- to 89-years-olds: 46%, ≥90-year-olds: 31%, p = 0.05), but these age and sex differences in hospital deaths were not seen for those without cognitive impairment (data by age and sex available on request).
Transitions before death
Table 3 shows the significance of differences in end-of-life transitions from the community and long-term care by cognitive status. Figure 3(a)–(c) illustrates the transitions in place of residence or care in the last year of life by cognitive status, from each individual’s address when interviewed less than 1 year before death, to their ‘usual address’ at death and to their place of death. For each group, the direction of transition was mainly up the ladder of care. The only exceptions were one cognitively intact individual who moved from a care home to her child’s home and three people with severe cognitive impairment who moved from long-stay hospital wards into care homes.
End-of-life transitions from different residential settings (community or long-term care) – by cognitive status.
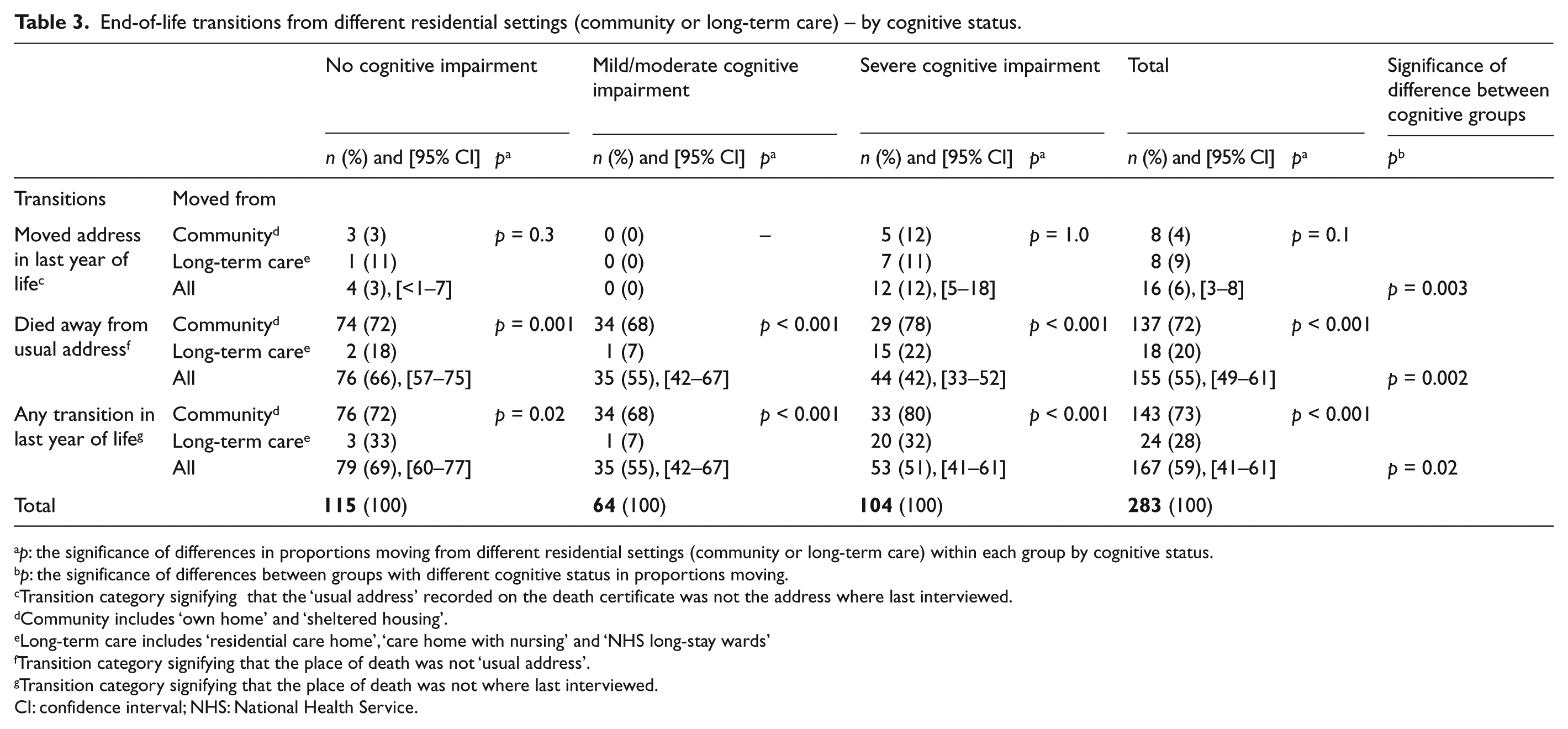
p: the significance of differences in proportions moving from different residential settings (community or long-term care) within each group by cognitive status.
p: the significance of differences between groups with different cognitive status in proportions moving.
Transition category signifying that the ‘usual address’ recorded on the death certificate was not the address where last interviewed.
Community includes ‘own home’ and ‘sheltered housing’.
Long-term care includes ‘residential care home’, ‘care home with nursing’ and ‘NHS long-stay wards’
Transition category signifying that the place of death was not ‘usual address’.
Transition category signifying that the place of death was not where last interviewed.
CI: confidence interval; NHS: National Health Service.
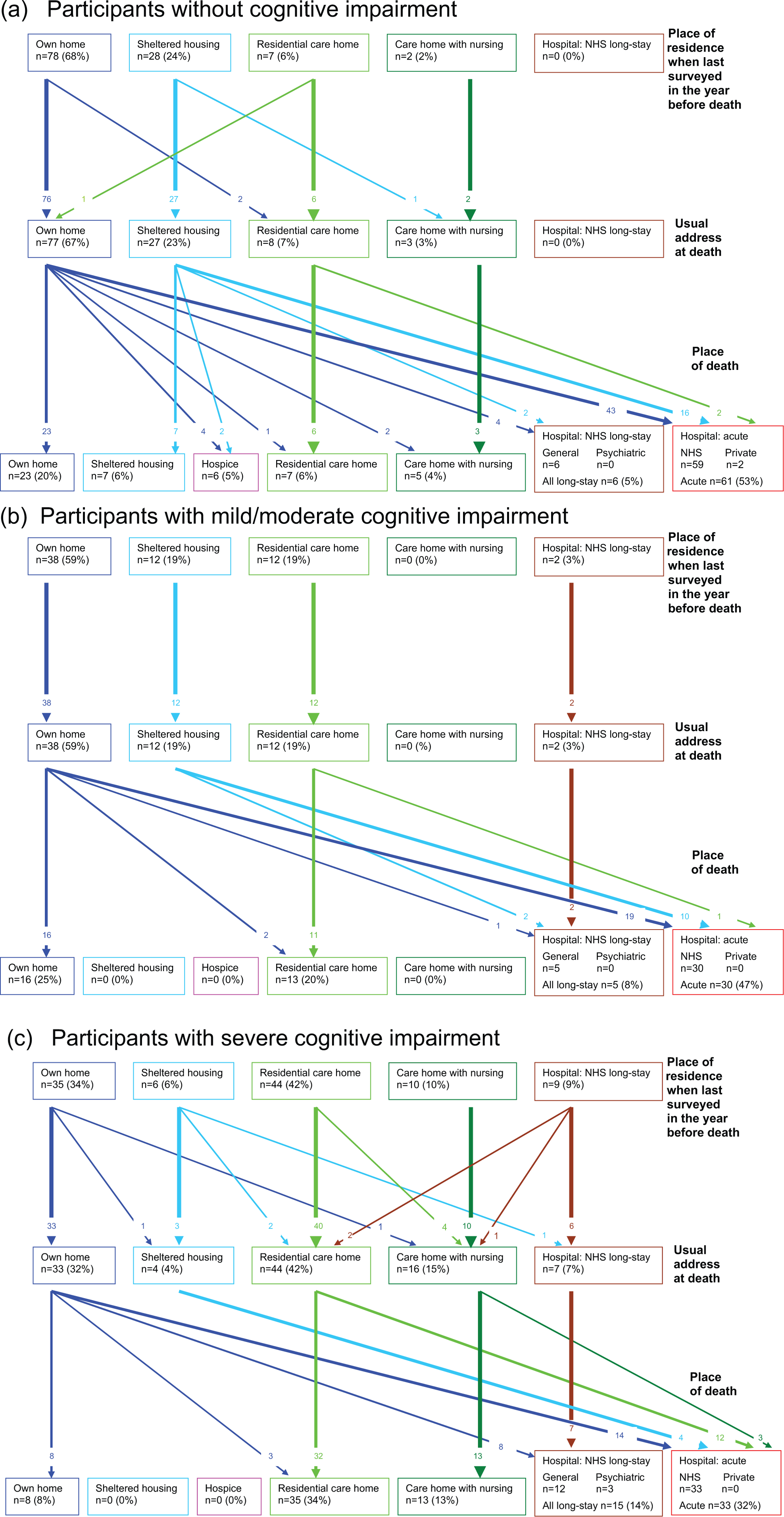
Transfers in place of residence or care at the end of life of 283 study participants, (a) without cognitive impairment (n = 115), (b) with mild/moderate cognitive impairment (n = 64) and (c) with severe cognitive impairment (n = 104). Figure 3 traces individual-level transitions between settings from where individuals lived at the time last interviewed less than a year before death to the location registered as their ‘usual address’ when they died and to their place of death, showing different cognitive groups separately.
Very few of those without cognitive impairment (3%; 95% confidence interval (CI): <1%−7%), and nobody with mild/moderate cognitive impairment, had changed address. More with severe cognitive impairment (12%; 95% CI: 5%−18%) had changed address by the time of death. However, more than half (55%; 95% CI: 49%−61%) of all the participants had a ‘usual address’ that differed from their ‘place of death’, that is, their place of residence or care changed in the period prior to death. Most of these movements were into hospital, with just n = 8 (5%; 95% CI: 2%−9%) dying in a care home that was not their ‘usual address’. Two-thirds (66%; 95% CI: 57%−75%) of those without cognitive impairment, more than half (55%; 95% CI: 42%−67%) of those individuals with mild/moderate impairment and 42% (95% CI: 33%−52%) of those with severe impairment had a ‘usual address’ that differed from their place of death (p = 0.002; Figure 3(a)–(c); Table 3).
This trend reflects transitions being far more common overall from the community than from institutions: 73% (95% CI: 66%−79%) and 28% (95% CI: 18%−38%) from the community and institutions, respectively, did not die where last interviewed (p < 0.001). However, participants with severe cognitive impairment living in the community when last interviewed were the most likely group of all to move: 80% (95% CI: 68%−93%) of them died somewhere other than their residence at last interview. No long-stay-hospital residents moved. Everybody with cognitive impairment in sheltered housing moved to a hospital (Table 3).
Cognitive impairment and dementia
Repeat analyses using only the smaller sample (n = 142) with known dementia status – clinically confirmed diagnosis or absence – produced very similar results to those presented above for the larger sample (n = 283) with known cognitive status but not necessarily confirmed dementia status (data available from the authors on request).
Discussion
Summary of main findings
This is the first study, to our knowledge, comparing such very old people (≥85-year-olds) across the cognitive spectrum, mapping transitions in place of end-of-life care. Mortality records were linked with prospectively collected population-based data from cognitive assessments or clinical diagnosis, identifying high levels of cognitive impairment and dementia at the end of life. Less than 1 year before death, the majority of the total sample was community dwelling, as were 39% of those with severe cognitive impairment. By the time of death, only a small minority had changed their usual address, but 55% died away from their usual address. The direction of these changes in place of residence for all categories of cognitive impairment was towards increased care, mainly from the community to hospital, the most common place of death for all but the most cognitively impaired. In sheltered housing, all those with any cognitive impairment, and two-thirds without, moved to hospital before dying. By contrast, residents of care homes and long-stay hospitals were more likely to have cognitive impairment and less likely to move prior to death. There were thus fewer transitions among individuals with severe cognitive impairment or dementia overall, but those of them living in the community were most likely to die elsewhere.
Strengths and limitations of this study
A strength of this study is its prospectively collected data from a representative population-based cohort, allowing individual-level comparison of transitions in place of residence or care experienced by very old people less than 1 year before death. Study design enabled analysis not only by cognitive status in a larger group but also by dementia status from detailed assessments of half the sample, confirming similar patterns in both.
The CC75C study is the longest running cohort of older old people followed-up with interviews even into their last year of life. Our focus on deaths in advanced old age inevitably meant that our sample, although sizeable for this age range, indeed to our knowledge the largest in comparable published research, is nonetheless too small to fully examine the independent effects of age and sex as well as cognition. A limitation is that information on participants’ place of residence or care was gathered at only three time-points in the last year of life, potentially missing intervening transitions. Moreover, policy changes during the period of the study (1985–2008), such as closure of long-stay wards, may have influenced care options available; however, we have previously compared CC75C study findings to national statistics and found comparable trends in place of death. 18 The study had no access to medical records that might clarify reasons for transitions and their proximity to death.
Death certificates can be a valuable data source for examining place of death, albeit with known limitations.24,25 However, the authors recognise further validation is required of ‘usual address at death’ data based on report from the deceased’s key informant. In our sample, for instance, study investigators aware of the recently deceased’s circumstances were able to correct a few inconsistencies noticed.
Comparison with existing literature
The prevalence of cognitive impairment and dementia in our study sample by the time of death is in line with findings from the other large population-based study to examine prevalence of cognitive impairment and dementia before death, 11 confirming the enormous rising challenge of caring for those approaching death in very old age.
Research on where older people with cognitive impairment and dementia die is limited, and generally methodologically limited by reliance on death registration: dementia is under-reported on death certificates. 26 Australian and US studies27–29 and a cross-national European one 30 examined place of death of older people above 65 years old whose death certificated causes of death included dementia. In these studies, a slightly higher percentage of dementia-related deaths occurred in care homes than among our ≥85-year-olds. However, death certificate recording of dementia diagnosis is more likely for nursing home residents than for individuals dying in non-institutional care settings. 26 Population-based research provides more robust estimates of place of care, but cognitive data are rare in these research studies. 31 A Belgian nationwide mortality study through the Sentinel general practitioner (GP) network retrospectively reported place of death for ≥65-year-olds with GP-confirmed dementia. 32 In this study, 71% of those with severe dementia died in a care home, as did 41% with mild dementia. Although these percentages are again higher than we found, these findings are in line with our findings that dying in a care home is more likely with increasing cognitive impairment (mild/moderate: 28%, severe: 61%). All these studies shared our finding that hospital was the most frequent place of death for individuals without dementia but also the second commonest location for deaths with dementia.
Transitions in place of residence or care experienced by older people with cognitive impairment and dementia are poorly investigated, most research having focussed on moves into long-term care. 33 The Longitudinal Ageing Study Amsterdam (LASA) retrospectively investigated transitions 3 months before death in younger old-age ranges. Despite differences in methodology, age range and time frame, LASA also found that 50% of individuals changed setting shortly before death. 34 However, these Dutch findings are not stratified by cognitive or dementia status, so no direct comparison can be drawn. The Belgian Sentinel study investigated transitions during the last 3 months and the last week of life, stratified by mild/severe dementia. Patients with severe dementia were half as likely to be transferred in the last 3 months and the last week of life compared with patients with mild dementia, in line with our finding that severity of cognitive impairment was overall associated with lower proportions dying away from previous addresses. The Belgian results do not include sufficient detail to compare whether they also found the contrasting situation for the sub-set of individuals with severe cognitive impairment living in the community whom we found were the most likely group of all to move. 32 Other Sentinel programme reports highlighted admissions to hospital as highest closest to death for all older people.17,35 Our data suggested most transitions occurred soon before death, but we lack data to compare time frames in such detail. They are nonetheless consistent with data from Western Australia showing only 5% of transitions shortly before dying were into long-term care. 28 The relative stability of our study’s care home residence in the final year reflects the fact that moves into long-term care in very old age often happen before this stage. 36
Implications
As the population ages, end-of-life care is increasingly about providing good dementia care. 11 The care setting is an important factor and moving in the latter stages of life, can be very distressing, especially for individuals with severe cognitive impairment.37,38 Although quality end-of-life care indicators are complex to define and measure,39–44 dying at home is often described as an important end-of-life aim. 14 Clarifying such preferences is challenging even for individuals without cognitive impairment,13,45–48 but particularly so with dementia. 49 Our finding that the majority of ‘older old’ people died away from home, particularly those living in the community and, of those, especially the most cognitively impaired, may suggest that community care could not or did not provide the support that community-dwelling individuals (or perhaps their informal carers 50 ) needed prior to death. We had insufficient data on who lived alone to analyse the impact of this crucial factor, 51 but our results suggest sheltered housing settings rarely provide sufficient support for their singly dwelling high-needs residents to die ‘in place’.
Care home residents and long-stay hospital patients had fewer moves close to death, but it was beyond the scope of this analysis to ascertain whether avoiding transfers up the ladder of care achieved better deaths. Current UK policy, in line with other countries,52,53 seeks to avoid ‘inappropriate admissions’, 13 but what proportion of our study participants’ transitions might have been ‘avoidable’ was unquantifiable. Hospital admission may have been appropriate for many; our findings could be interpreted as showing that a majority of cognitively intact very old people manage to remain at home until an acute illness close to the end of their lives.
Very old people with and without cognitive impairment have complex care needs towards the end of their lives, and prognostication can be difficult.40,54–58 Meeting these needs in different care settings is an increasingly high priority, and lack of understanding of the course of dementia is a barrier to improving care. 59 There is growing evidence of inequitable care and poor outcomes associated with hospitalisation of older people with dementia,60–62 prompting calls to improve their access to palliative care.63–66 We found long-term-care settings provide end-of-life care for a high proportion of the oldest old with some of the highest needs in their final ‘home’. Research has highlighted challenges,54,55,67 but a UK general practice audit showed care homes can reduce hospital admissions and increase proportions of residents dying in the home if adequate resources allow more frequent GP and district nursing visits. 68 Community care for cognitively impaired older people puts enormous demands on families,10,16,69 increasing nearer to the end of life; so, service providers should note the high proportions of the most severely impaired we found still living in the community less than a year before death. Community palliative care programmes can enable more people to die in their ‘preferred place of death’,70–72 but demographic projections and trends in place of death point to an urgent need for service expansion.25,51,73 Previous CC75C study findings showed markedly higher disability levels among people dying at age 90 years or older compared even with people dying at age 85–89 years, 74 indicating enormous implications for end-of-life care as life expectancy rises. However, alongside increased community provision for end-of-life care for the frail elderly, equally crucial is the need to develop training and service provision to improve end-of-life care in hospitals and care homes – the settings where the majority of very old people die.1,59,75,76
Footnotes
Acknowledgements
The authors would like to acknowledge particularly the CC75C study participants, their families, friends and the staff in many care homes and collaborating general practitioners without whose help none of this research would be possible. Furthermore, the authors gratefully acknowledge the contributions of previous investigators and past research team members and the helpful comments on earlier drafts of this article from current CC75C study collaborators (see full list at ![]() ).
).
Author contributions
J.F., S.B., M.F. and C.B. are principal investigators who devised the current programme of research into end-of-life care and proposed this research idea. A.J.P. and J.Z. prepared the data. A.J.P. analysed the data, produced tables and figures and wrote the first draft of the manuscript with J.F.. J.F. advised on the analysis, ran further analyses and was responsible for subsequent drafts. J.F., J.Z. and H.B. supervised this project. All co-authors interpreted the data and contributed to editing and revising the manuscript. A.J.P., J.F. and S.B. are guarantors for the article. C.B. is principal investigator for the CC75C study.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethical approval
Each CC75C study phase was approved by Cambridge Research Ethics Committee (current reference number: 05_Q0108_308).
Funding
The CC75C study has been financially supported by many sponsors over two decades (see http://www.cc75c.group.cam.ac.uk/pages/grant/default.htm for full list of project grants), particularly the BUPA Foundation (http://www.bupafoundation.com/) under their Health and Care of Older People grant. S.B. and M.F. are supported by Macmillan Cancer Support (http://www.macmillan.org.uk) as part of their Research Capacity Development Programme. J.F. is currently funded by the NIHR CLAHRC for Cambridgeshire and Peterborough (Collaborations for Leadership in Applied Health Research & Care; ![]() ). The funders had no role in the study design, data collection and analysis, decision to publish or preparation of the article.
). The funders had no role in the study design, data collection and analysis, decision to publish or preparation of the article.
