Abstract
Background:
Little is known about place of death in chronic neurological diseases. Mortality statistics are ideal for examining trends in place of death, but analyses are limited by coding rule changes.
Aim:
To examine the relationship between place of death and underlying cause of death in Parkinson’s disease, multiple sclerosis and motor neurone disease and the impact of coding rule changes on analysis of place of death.
Design:
Population-based study. Proportion ratios for death in hospice, home, care home and hospital were calculated according to underlying cause of death, using multivariable Poisson regression.
Participants:
Deaths in England (1993–2010) with any mention of Parkinson’s disease, multiple sclerosis or motor neurone disease as a cause of death, identified from national mortality data.
Results:
In this study, 125,242 patients with Parkinson’s disease, 23,501 with multiple sclerosis, and 27,030 with motor neurone disease were included. Home deaths ranged from 9.7% (Parkinson’s disease) to 27.1% (motor neurone disease), hospice deaths ranged from 0.6% (Parkinson’s disease) to 11.2% (motor neurone disease) and hospital deaths ranged from 43.4% (Parkinson’s disease) to 55.8% (multiple sclerosis). In Parkinson’s disease and multiple sclerosis, cancer as underlying cause of death increased likelihood of hospice death (proportion ratio (PR): 18.8, 95% confidence interval (CI) = 16.1–22.0; 8.88, 95% CI = 7.49–10.5) and home death (PR: 1.91, 95% CI = 1.80–2.04; 1.71, 95% CI = 1.56–1.88). Dementia as underlying cause of death increased likelihood of care home death in Parkinson’s disease (PR: 1.25, 95% CI = 1.19–1.32), multiple sclerosis (PR: 1.73, 95% CI = 1.22–2.45) and motor neurone disease (PR: 2.36, 95% CI = 1.31–4.27).
Conclusions:
Underlying cause of death has a marked effect on place of death. The effects of coding rule changes are an essential consideration for all research using underlying cause of death to study place of death.
Introduction
Chronic neurological conditions such as Parkinson’s disease (PD), multiple sclerosis (MS) and motor neurone disease (MND) are characterised by progressive deterioration in neurological function and increasing dependence over time. In the absence of a cure, quality of care and advance care planning in these conditions are key governmental strategies. 1 –3
Place of death is used as an indicator of the quality of end-of-life care; this is because the majority of people would prefer to die at home, 4 but in reality, most deaths occur in institutional settings, 5 –8 and there is some evidence that the quality of end-of-life care in institutions is less favourable than at home. 9 Of the chronic neurological conditions, place of death in dementia has been best studied, 10,11 and less is known about place of death in PD, MS and MND.
Death certificates are the most useful source of national data on cause and place of death. The mortality statistics generated from death certificates inform clinical practice and policy. 5,12 A strength of using mortality data is the potential to include the whole population and the use of internationally agreed methods of death classification and coding, thereby allowing national and international comparisons. 12 Furthermore, use of historical mortality data allows analysis of the changing pattern of place of death over time, which is essential for future projections, to assess historical policy initiatives and to guide appropriate health policy decisions. 5,13
Since 1993, mortality statistics have included all certified causes of death (termed as ‘mentions’), in addition to an underlying cause of death (UCD) that is selected automatically using software. 14 Analyses of place of death from mortality statistics most often consider UCD, rather than any mention of disease. 10,11,13 However, a potential limitation of using UCD to analyse mortality trends is that the procedures for selecting UCD have changed over time. 15 Specifically, internationally implemented changes to coding rules (in particular Rule 3) with the introduction of the International Classification of Diseases, 10th Revision (ICD-10) in 2001 led to a reduction in the number of deaths assigned to bronchopneumonia as UCD and a reciprocal increase in the number of deaths assigned to chronic conditions such as neurological diseases. 16,17 Therefore, the aims of this study were to use national mortality statistics to explore the relationship between UCD and place of death in PD, MS and MND and the impact of coding rule changes on analysis of place of death.
Methods
Design
Population-based study including all deaths in England with a mention of PD, MS or MND.
Data source
Mortality data were collated for all deaths with any mention of PD, MS or MND registered in England between 1993 and 2010. By law, all deaths in England must be registered within 5 days (unless a coroner’s inquest is necessary). Mortality data comprise information recorded on the death certificate such as the date of death, age and sex of the patient and the cause(s) of death. Since 1993, mortality statistics have included the UCD, the disease that initiated the train of events leading to death, as well as contributory causes, defined as part of the causal sequence leading to death or contributing to death. Where a condition is listed as either the UCD or a contributory cause, this is termed a mention. The data were obtained from the deaths database held by the Office for National Statistics (ONS), the national statistics institute of the United Kingdom, in a joint collaboration with South West Public Health Observatory. ICD codes used were as follows: PD ICD-9 332, ICD-10 G20; MS ICD-9 340, ICD-10 G35; MND ICD-9 335.2, ICD-10 G12.2.
Analysis
The UCD was grouped into the neurological condition (PD, MS or MND), bronchopneumonia (ICD-9 485-486, ICD-10 J18.0, J18.2-J18.9), cancer (ICD-9 140-239, ICD-10 C00-D48), cardiovascular disease (ICD-9 390-429, 440-459, ICD-10 I00-52, I70-99), cerebrovascular disease (ICD-9 430-438, ICD-10 I60-69), chronic lower respiratory disease (ICD-9 490-494, 496, ICD-10 J40-47), dementia (ICD-9 290, ICD-10 F01 F03) and ‘other disease’ (all remaining ICD codes).
Place of death was grouped into hospice, care home (includes nursing and residential homes), hospital and home based on routinely used ONS coding categories. For 473 (0.4%) of the PD mentions, 111 (0.5%) of the MS mentions and 236 (0.9%) of the MND mentions, place of death was unknown (e.g. occurring on the street). Place of death was classified as ‘elsewhere’ (e.g. ‘home for incurables’ and ‘psychiatric hospital’) for 1278 (1.0%) of the PD mentions, 717 (2.9%) of the MS mentions and 177 (0.6%) of the MND mentions. Subjects for whom place of death was either unknown or ‘elsewhere’ according to ONS coding categories were removed from the analysis.
Change in age at death by year was studied using linear regression. Place of death was determined for the total population (any mention of disease). Multivariable Poisson regression analyses were used to provide age- and sex-adjusted proportion ratios (PRs) for death in hospice, care home, home and hospital, compared with any other place of death, for deaths coded with neurological disease as UCD compared with each separate UCD. For the analysis of neurological condition as UCD with bronchopneumonia as UCD, the data were limited to 1993–2000. All analyses used Stata version 10 (StataCorp, College Station, TX, USA).
Ethics
This study was based on anonymised records, and no ethical approval was required according to national guidelines and those of the King’s College London Research Ethics Committees.
Results
In this study, 125,242 patients who died with a mention of PD, 23,501 with a mention of MS and 27,030 with a mention of MND were included. The neurological condition was the sole recorded cause of death in 6.2% (PD), 10.8% (MS) and 39.6% (MND) of patients. There was one additional contributory cause in 38.1% (PD), 47.6% (MS) and 40.3% (MND) of patients. There were two or more contributory causes of death in 55.7% (PD), 41.5% (MS) and 20.1% (MND) of patients.
The average ages at death were 81.4 years (standard deviation (SD) = 7.1 years) (PD), 63.3 years (SD = 13.1 years) (MS) and 70.3 years (SD = 11.6 years) (MND). The age at which patients died from all three conditions increased across the study period (1993–2010): in PD, from 81.1 to 81.9 years (annual increase = 0.06, 95% confidence interval (CI) = 0.05–0.07, p < 0.001); in MS, from 63.2 to 65.3 years (annual increase = 0.13, 95% CI = 0.09–0.16, p < 0.001); in MND, from 69.2 to 71.3 years (annual increase = 0.15, 95% CI = 0.12–0.18, p < 0.001). Most PD and MND mentions were men (57% and 55%, respectively). Most MS mentions were women (65%). For all three conditions, the number of patients dying per year increased over time: from 7107 to 8055 in PD, from 1227 to 1428 in MS and from 1198 to 1982 in MND for the period 1993–2010.
Place of death for any mention of PD, MS or MND, 1993–2010
Home deaths were most common in patients with a mention of MND (27.1%) and least common in patients with MS (18.4%) and PD (9.7%). Care home deaths were most common in PD (46.3%) and least common in MS (23.3%) and MND (16.6%). Of the MS, MND and PD mentions, 55.8%, 45.1% and 43.4%, respectively, died in hospital. Hospice deaths were extremely uncommon in PD and MS (0.6% and 2.5%) but more common in MND (11.2%).
Effect of 2001 coding rule changes on UCD
The proportion of deaths in each condition where the neurological condition was the UCD was 48.1% (PD), 59.1% (MS) and 87.4% (MND). For all three conditions, this proportion was greater for the period 2001–2010 than 1993–2000, reflecting changes in coding rules (in particular Rule 3), which reduced the number of deaths assigned to bronchopneumonia and increased the number of deaths assigned to chronic neurological diseases as UCD. This effect was greatest for PD and least apparent for MND (Figure 1).
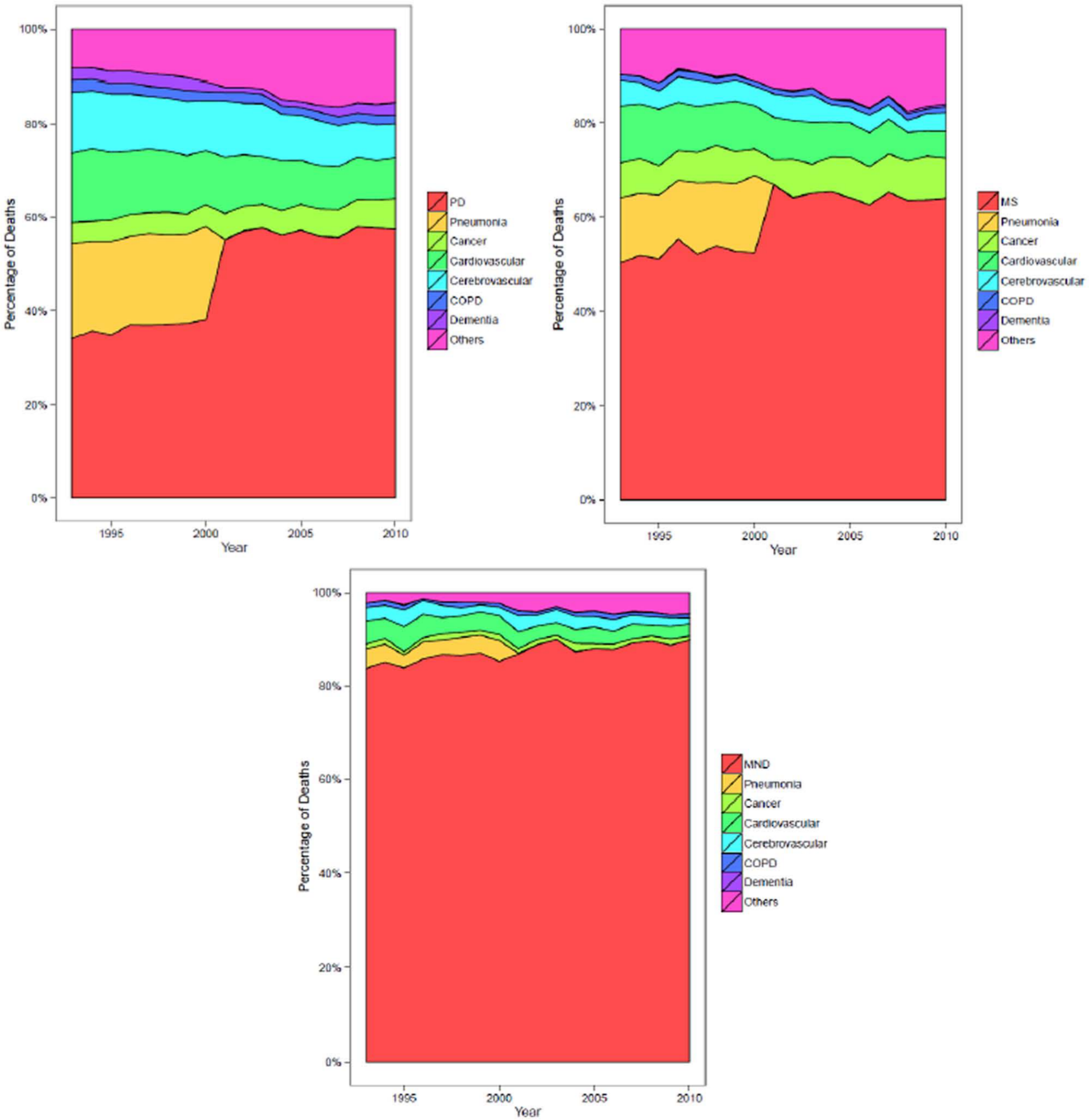
Underlying cause of death as a proportion of all mentions of PD, MS and MND, 1993–2010.
Between 1993 and 2000, 10,535 (19.5%) patients with a mention of PD, 1358 (14.0%) patients with a mention of MS and 371 (3.7%) patients with a mention of MND were coded with bronchopneumonia as UCD. Compared to deaths coded with PD, MS or MND as UCD, bronchopneumonia deaths were more likely to occur in hospital (PR: 1.16, 95% CI = 1.12–1.21; 1.25, 95% CI = 1.15–1.36; 1.49, 95% CI = 1.31–1.71, respectively) and less likely to occur in hospice (PR: 0.25, 95% CI = 0.11–0.60; 0.34, 95% CI = 0.17–0.70; 0.12, 95% CI = 0.05–0.29, respectively) and at home (PR: 0.67, 95% CI = 0.61–0.73; 0.70, 95% CI = 0.60–0.82; 0.46, 95% CI = 0.35–0.60, respectively). To enable comparable analysis of data before and after the change to coding rules in 2001, for subsequent analyses, cases with bronchopneumonia as UCD were combined with cases with the neurological disease as UCD for each of the three datasets.
Place of death by underlying cause, 1993–2010
To explore further the relationship between UCD and place of death, PRs for each place of death were calculated according to UCD, with the neurological disease as UCD as the reference (Table 1). Cardiovascular disease as UCD increased the likelihood of home death in PD (PR: 1.75, 95% CI = 1.67–1.83), MS (PR: 1.49, 95% CI = 1.36–1.64) and MND (PR: 1.17, 95% CI = 1.04–1.32). Cancer as UCD increased the likelihood of home death in PD (PR: 1.91, 95% CI = 1.80–2.04) and MS (PR: 1.71, 95% CI = 1.56–1.88). Cancer as UCD also increased the likelihood of hospice death in PD (PR: 18.8, 95% CI = 16.1–22.0) and MS (PR: 8.88, 95% CI = 7.49–10.5). Cancer as UCD did not significantly affect the likelihood of hospice or home death in MND, and all other UCDs reduced the likelihood of hospice death in MND. For all three conditions, the likelihood of care home death was increased in cases with dementia as UCD (PR: 1.25, 95% CI = 1.19–1.32; 1.73, 95% CI = 1.22–2.45; 2.36, 95% CI = 1.31–4.27), and the likelihood of hospital death was increased by ‘other’ as UCD (PR: 1.53, 95% CI = 1.49–1.57; 1.49, 95% CI = 1.42–1.56; 1.56, 95% CI = 1.44–1.69).
Underlying cause of death in PD, MS and MND, and adjusted proportion ratios (95% CI) for death in hospice, care home, home and hospital by underlying cause of death, 1993–2010.
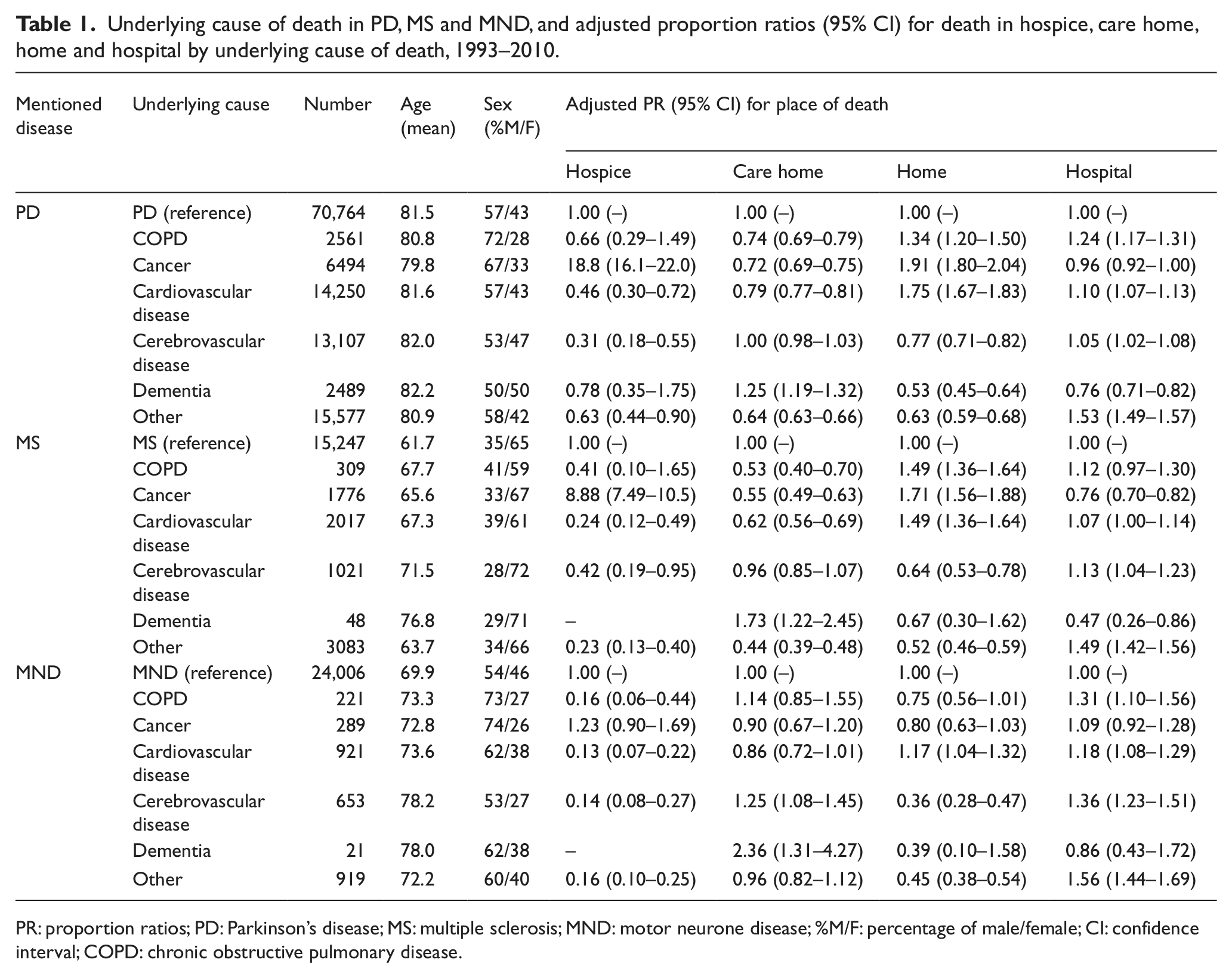
PR: proportion ratios; PD: Parkinson’s disease; MS: multiple sclerosis; MND: motor neurone disease; %M/F: percentage of male/female; CI: confidence interval; COPD: chronic obstructive pulmonary disease.
Discussion
This population-based study of deaths in PD, MS and MND in England from 1993 to 2010 demonstrates the striking relationship between UCD and place of death and the effect that changes to coding rules with the introduction of ICD-10 had on UCD. The proportion of deaths coded as being due to the neurological condition as underlying cause was greatest for MND. Similarly, MND deaths had fewest contributory causes of death. This indicates that patients with MND are most likely to die as a direct consequence of the disease, which is consistent with autopsy series, which have attributed 73%–80% of deaths in patients with MND to the disease. 18 In contrast, patients with PD are less likely to die as a direct consequence of their disease, especially as age at diagnosis increases. 19
Our analyses have shown that UCD is a key component of the complex network of personal, demographic and environmental factors that affect where people die. 20 The influence of UCD on place of death is likely to be mediated through health-care factors such as home care provision and disease-related factors such as trajectory of decline. For example, in PD and MS, the increased likelihood of hospice and home death in patients with cancer as UCD may reflect access to palliative care services. In contrast, in patients with MND, who are more likely to receive palliative care services than patients with other neurological conditions, 21 cancer as UCD was not associated with an increased likelihood of home or hospice death.
Longitudinal epidemiological information on place of death is required to understand historical trends, to study the effects of policy initiatives and to plan appropriate end-of-life health-care policies. 5,12 However, longitudinal analyses of mortality statistics are limited by changes over time in classification, certification and coding practices. In this study, the annual number of deaths with a mention of MND increased from 1241 in 1993 to 2005 in 2010. This increase is partly caused by the misclassification in ICD-10 of progressive supranuclear palsy to MND, leading to an increase in the number of deaths classified as MND after 2001 by about 10%. 22 The World Health Organization is currently addressing this problem prospectively.
Changes to coding rules governing selection of the UCD have led researchers to recommend the use of any mention rather than UCD when analysing longitudinal mortality statistics in neurological diseases, in particular for mortality rates. 16,17 However, given the effect of UCD on place of death, using any mention may mask inequalities in place of death, particularly in PD and MS where a large proportion of patients die with UCD other than their neurological condition. In contrast, using any mention may be particularly appropriate for studies investigating the population burden of these diseases or the effects of co-morbidity on place of death.
A limitation of using UCD to analyse place of death longitudinally is the exclusion of patients coded with bronchopneumonia as UCD between 1993 and 2000. This has the greatest effect in PD and MS where 19.5% and 14.0%, respectively, of deaths were coded with bronchopneumonia as UCD, compared with just 3.7% in MND. We found a tendency of hospitals to record bronchopneumonia as UCD more than hospices and home death certifiers. This may reflect true clinical variation (e.g. bronchopneumonia could be a risk factor for dying in hospital) or be an artefact of certification practices in different settings, 23 for example, reflecting greater access to radiological and microbiological diagnostic facilities in hospital. The influence of place of death on death certification practices warrants further investigation. To overcome the limitation of changes to coding Rule 3 on longitudinal analyses using UCD, we combined cases of bronchopneumonia and neurological condition as UCDs. Published data on comparability ratios for recoding UCD in PD, MS and MND as UCD from International Classification of Diseases, 9th Revision (ICD-9) to ICD-10 suggested that this was valid. 24
Place of death has both societal and economic implications, and improving end-of-life care is a public health priority. 25 Development of policies aiming to reduce inequality in place of death requires an a priori understanding of where people die and the clinical and organisational factors that influence this. Mortality statistics have well-documented limitations, but they are the best tools available to study national trends in place of death, to improve understanding of the factors influencing where people die, and to allow international comparisons. 12,26
The effects of coding rule changes are an essential consideration for all research using mortality statistics to study place of death, particularly for those that use UCD to define their population of interest. For such studies, adjustment for coding changes over time can reduce potential artefact. The availability within mortality data of information on contributory causes of death presents opportunities to investigate the influence of co-morbidity on place of death. Further work will extend the findings of this study to examine the disease-related, demographic, temporal and regional factors that influence place of death in chronic neurological disease.
Footnotes
Acknowledgements
We thank Service User members Tony Bonser, Robert Lovick, Carolyn Morris and Stafford Scholes. We thank Andy Pring (Senior Analyst) and Tariq Malik (Deputy Director) of the South West Public Health Observatory and Katie Lindsey (Project Manager) of National End of Life Care Programme. We thank the ONS for supplying data. We thank Claudia Wells, Vanessa Fearn and Julie Messer of ONS.
I. J.H., W.G., J.V., M.G. and Barbara Gomes applied for funding for the GUIDE_Care Project. I.J.H. and W.G. are responsible for the day-to-day management of the project, and they both acquired data. Y.K.H. is responsible for data management. K.E.S., W.G., E.S. and I.J.H. contributed to the design and conceptualisation of the study and interpretation of data. K.E.S. performed the analysis, with input from Y.H.K. and supervision from W.G. and I.J.H. K.S. drafted the manuscript. All the authors revised the manuscript critically for intellectual content and approved the final manuscript for submission. I.J.H. is an NIHR senior investigator.
Declaration of conflicting interests
The views and opinions expressed therein are those of the authors and do not necessarily reflect those of the HS&DR programme, NIHR, NHS or the Department of Health.
Funding
Project GUIDE_Care is funded by the National Institute for Health Research Health Services and Delivery Research (NIHR HS&DR) programme (project number 09/2000/58). Visit the HS&DR website for more information. K.E.S. is funded by an NIHR Clinical Lectureship in Palliative Medicine.
