Abstract
Objective
To determine the effect of falls-prevention exercise compared with nothing, sham or another exercise intervention on reducing falls after stroke. To also determine the effect of falls-prevention exercise on balance, mobility and quality of life.
Data sources
Searches were conducted from inception to December 2025 on MEDLINE, EMBASE, Scopus and PEDro databases according to predefined search terms with details provided in supplementary materials.
Review methods
Randomised trials were included if the intervention targeted falls and the primary outcome was the rate of falls or the number of people experiencing one or more falls. Other outcomes of interest were balance, mobility and quality of life. Methodological quality was rated with the PEDro scale. Two researchers independently extracted data, which was synthesised by meta-analysis.
Results
Three trials (n = 677) were included in the review. Exercise trended towards a lower rate of falls compared to no/sham intervention (IRR 0.84, 95% CI 0.62 to 1.15, p = 0.29) with uncertainty in the estimate (confidence interval includes the possibility of no effect). Exercise did not reduce the number of individuals experiencing one or more falls (RR 0.98, 95% CI 0.81 to 1.18, p = 0.84). There was a trend towards improved balance (MD 0.49 steps in the Step Test, 95% CI −0.11 to 1.08, p = 0.11) and exercise did improve mobility a small amount (MD 0.04 m/s preferred walking speed, 95% CI 0.01 to 0.07, p < 0.01). No trials compared two exercise interventions.
Conclusion
There is a suggestion that exercise can reduce the rate of falls after stroke but does not affect the number of individuals falling.
Registration
PROSPERO CRD42024520272.
Introduction
Community-dwelling adults with stroke fall at up to twice the rate of the general population, 1 elevating the risk of fall-related injuries. 2 Moreover, people after stroke often fall repeatedly, impacting long-term health and well-being. 3 Given that 101 million people worldwide are living with the effects of stroke, and many of them are expected to experience a fall, falls after stroke are an urgent issue for the health system globally. 4 Due to the heterogeneity of post-stroke impairments falls risk factors can be varied, however reduced balance, cognitive changes and previous falls are some of the known risk factors. 3
In community-dwelling older people in general, there is strong evidence that exercise reduces the number who experience falls. 5 The Cochrane review synthesised 81 randomised trials (∼19,500 participants) and reported a lower rate of falls (IRR 0.77, 95% CI 0.71 to 0.83) and fewer participants experiencing one or more falls (RR 0.85, 95% CI 0.81 to 0.89) with high certainty in those who received exercise compared to control interventions. However, this review did not include trials of people with stroke. There is less evidence that exercise reduces the number falls in people living in the community after stroke. The Cochrane review investigating people after stroke synthesised 10 trials (969 participants) and reported a lower rate of falls (IRR 0.72, 95% CI 0.54 to 0.94) but no fewer people experiencing one or more falls (RR 1.03, 95% CI 0.90 to 1.19) with very low certainty in those who received exercise. 6 However, this review included trials where the intervention was not targeting falls prevention,7,8 trials that were not powered to detect falls reduction and trials with comparisons where both interventions targeted balance and/or falls prevention.9,10 Therefore, our review aims to provide specific evidence of the effect of exercise by including trials where the exercise is targeting falls prevention (e.g., exercise contains a challenge to balance via reducing the base of support or adding movement in standing 11 ) and the trial is powered to detect an effect in falls reduction.
The research questions were:
Is falls-prevention exercise effective at reducing falls after stroke compared with no/sham intervention? Does falls-prevention exercise improve balance, mobility and quality of life? Is one type of falls-prevention exercise superior to another?
Method
This systematic review is reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 12 The protocol was registered at PROSPERO (CRD42024520272) prior to commencement (22/03/2024).
Searches for relevant trials were conducted from inception to December 2025 on MEDLINE, Embase, Scopus and PEDro databases without date restrictions. The search strategy was devised by generating pilot searches on MEDLINE with up-to-date search strings from the Cochrane Library. Search terms were related to stroke, falls and clinical trials (see Supplementary Material for the full search strategy). Each title and abstract were screened by two reviewers (from AH, SD, IL and KS) to identify relevant trials. Full text copies of relevant papers were retrieved and independently screened by two reviewers (from AH, SD, KS and LA). Reference lists of these papers were screened to identify further relevant trials. The method section of the retrieved papers was extracted and reviewed independently by two reviewers (KS and LA) using predetermined criteria (Box 1). Disagreements or ambiguities were resolved by consensus after discussion with a third reviewer.
Inclusion criteria

The methodological quality of the included trials was rated using the PEDro score (internal validity and statistical information; score range: 0 to 10 points). PEDro scores were extracted from the Physiotherapy Evidence Database (www.pedro.org.au). Where a trial was not included in the database, it was scored independently by two reviewers (LA and KS). Methodological quality was considered to be poor (0 to 3 points), fair (4 to 5 points), good (6 to 8 points) or excellent (9-10 points). 13
Trials were included if they involved participants who were community-dwelling adults (>18 years) after stroke, who were on average at least three months post-stroke and able to walk. Trials were excluded if participants were living in aged care/hospice facilities. Number of participants, age, time since diagnosis, falls history and walking speed were recorded to assess the similarity of the trials.
Trials were included if the experimental intervention included exercise aimed at preventing falls. 11 The control intervention could be no intervention, sham intervention, or another type of exercise. Where trials had multiple intervention arms, only relevant arms were included. The content of intervention, session duration, session frequency, and program duration were recorded to assess the similarity of the trials.
Trials were included if the primary outcome was falls reported as either number of fallers (ie, people experiencing one or more falls) and/or number of falls, and the trial was powered for this outcome. Secondary outcomes of interest were balance; mobility; and quality of life. The timing of the measurements and the procedure used to measure the outcomes were recorded to assess the similarity of the trials. In trials where there was more than one measure for a single outcome, we gave priority to continuous over ordinal or dichotomous measures.
Information about the method (design, participants, intervention, measures) and results (number of participants, and means (SD) or 95% confidence intervals or medians and interquartile ranges of outcomes related to falls, mobility, balance and quality of life) were extracted by two reviewers (LA and PG) with the involvement of a third reviewer where necessary (KS). Data were extracted for later combination using the following hierarchy where possible: between-group difference, within-group differences per group, post-intervention scores per group. Where data were unclear or unavailable, the corresponding authors requested further information via email.
Analyses were conducted using R version 4.5.0. 14 Meta-analysis could be conducted if there were ≥ two trials. An incidence rate ratio (IRR) was used to pool the total number of falls, relative risk (RR) to pool number of fallers, continuous identical measures used mean difference (MD) and otherwise standardised mean difference (SMD). For all meta-analyses, a random-effects model using an inverse variance method was employed in which the restricted maximum likelihood method was used to estimate the between-trial variability. Pooled summaries were reported with 95% confidence interval (CI).
Heterogeneity between trials was assessed using the I2 statistic. I2 values of 25, 50 and 75% indicate low, moderate and high heterogeneity, respectively. 15 Where there were insufficient trials to pool results for a particular comparison/outcome or where data could not be pooled (due to non-normality, for example), original published summaries are described.
Results
The electronic search strategy identified 12,652 papers. After screening titles, abstracts, and reference lists, 158 potentially relevant full papers were retrieved. 155 papers failed to meet the inclusion criteria, most commonly because the intervention was not targeting falls, falls were not the primary outcome, and the trial was not powered to detect an effect on falls. This left three papers to be included (Figure 1). One of the three papers was the protocol of a randomised trial, but the full trial was able to be sourced from the authors. 16
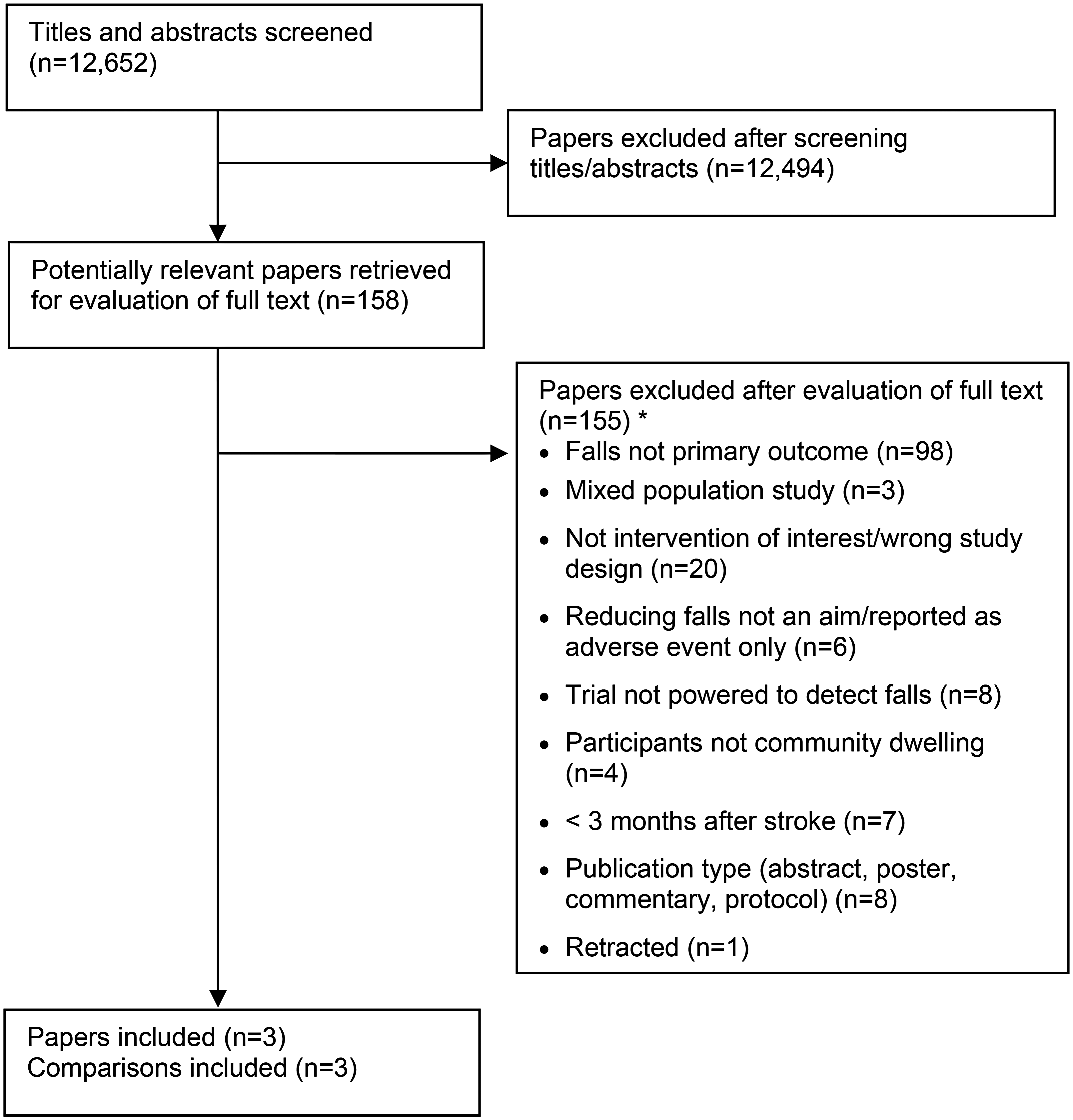
Flow of trials through the review. *Papers may have been excluded for failing to meet more than one inclusion criteria.
The three included trials comprised 677 participants investigating exercise versus no/sham intervention (Table 1).17–19 No trials investigating two different types of exercise were eligible.
Summary of included trials investigating exercise vs no/sham intervention (n = 3).
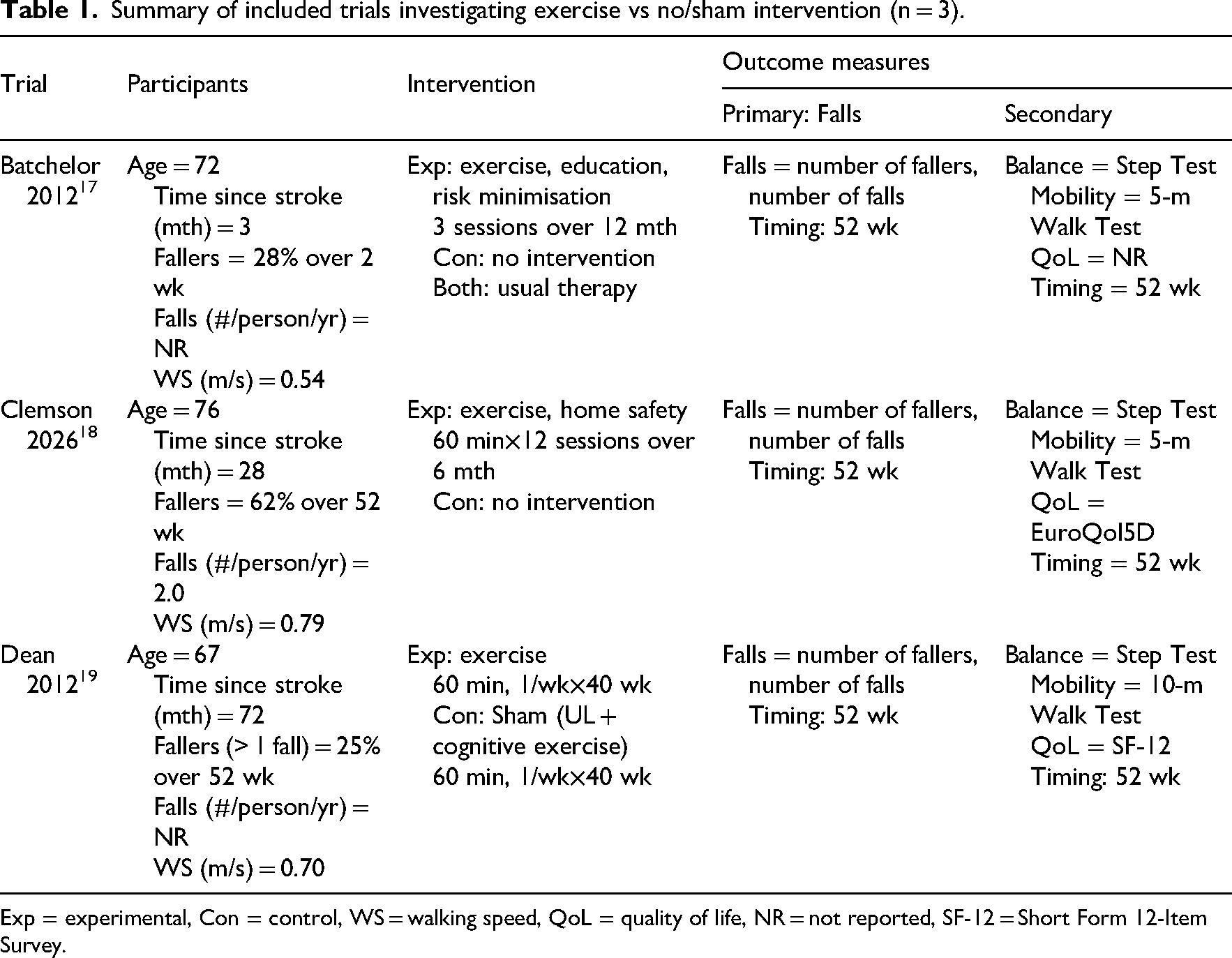
Exp = experimental, Con = control, WS = walking speed, QoL = quality of life, NR = not reported, SF-12 = Short Form 12-Item Survey.
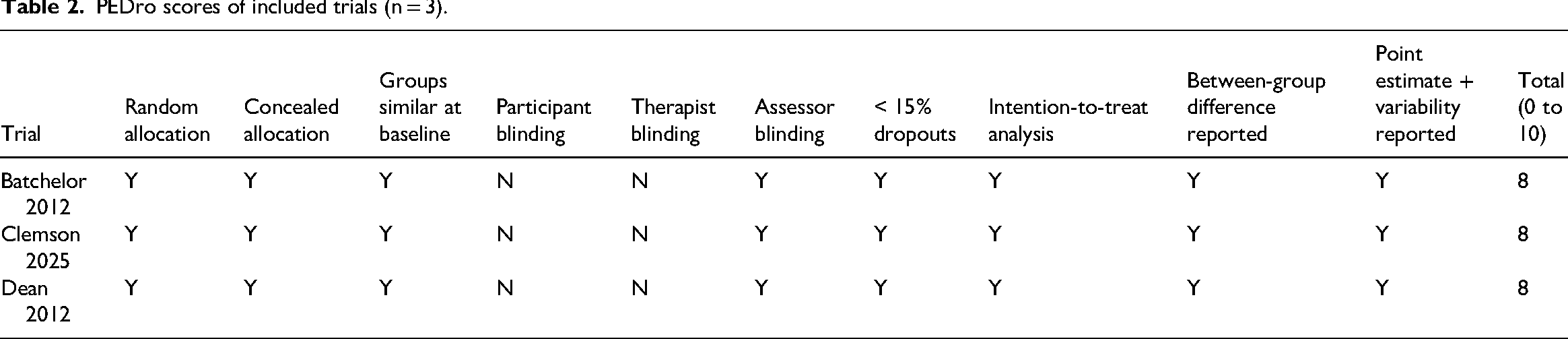
All trials scored 8 out of 10 on the PEDro scale (Table 2).17–19 They randomly allocated participants, reported between-group differences, point estimates and variability data. They had concealed allocation, blinded assessors, less than 15% dropouts, and undertook intention-to-treat analysis. No trials blinded participants or therapists which is difficult due to the nature of the intervention.
PEDro scores of included trials (n = 3).
The mean age of the participants ranged from 67 to 75 years across trials. Mean time since stroke ranged from 3 to 72 months. Mean walking speed at baseline ranged from 0.54 to 0.79 m/s (Table 1).
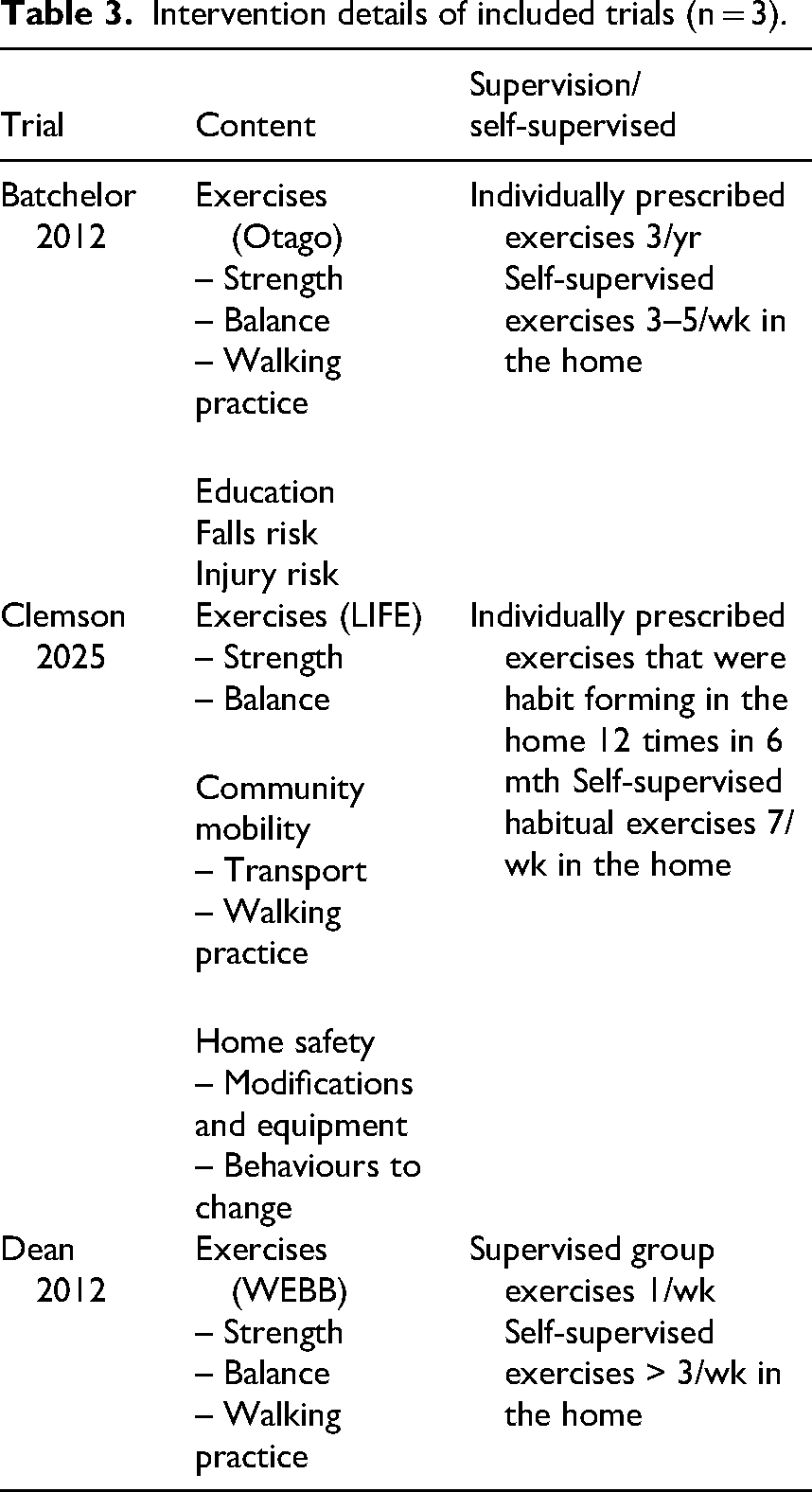
All trials included exercise aimed at reducing falls. See Table 3 for a detailed outline of the intervention for each trial. Two trials had a no intervention control17,18 and one trial had a sham control. 19
Intervention details of included trials (n = 3).
All trials reported the number of falls and the number of people experiencing falls over 12 months. In terms of secondary measures, all trials measured balance with the Step Test 20 and mobility with a timed walk test, and two trials measured quality of life (Table 1).
Effect of exercise vs no/sham intervention on falls
All trials of exercise vs no/sham intervention measured number of fallers in 660 participants and were included in the meta-analysis (Figure 2). There was an estimated 2% reduction in number of people experiencing one or more falls in favour of exercise (RR 0.98 95% CI 0.81 to 1.18, p = 0.84, I2 = 42%) with uncertainty in the estimate as the confidence interval includes the possibility of no effect.

Effect (RR, 95% CI) of exercise on number of fallers (ie, people experiencing one or more falls) by pooling data from three trials (n = 660).
All trials of exercise vs no/sham intervention measured number of falls on 665 participants and were included in the meta-analysis (Figure 3). There was an estimated 16% lower incidence in rate of falls in favour of exercise (IRR 0.84, 95% CI 0.62 to 1.15, p = 0.29, I2 = 31%) with uncertainty in the estimate as the confidence interval includes the possibility of no effect.
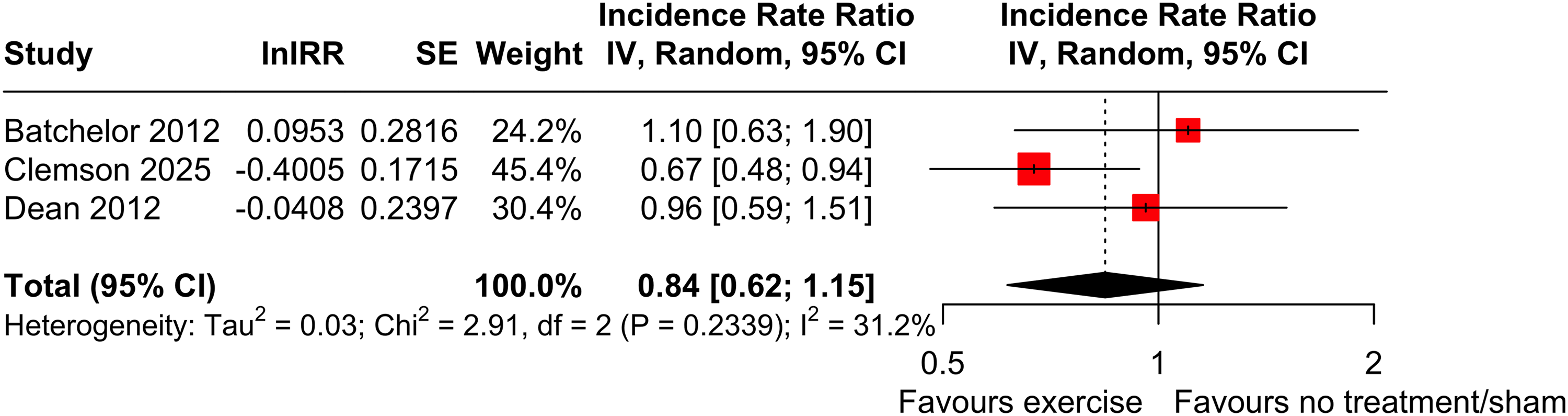
Effect (IRR, 95% CI) of exercise on number of falls by pooling data from three trials (n = 665).
Effect of exercise vs no/sham intervention on balance, mobility and quality of life
All trials of exercise vs no/sham intervention measured balance (Step Test) on 570 participants and were included in the meta-analysis (Figure 4). The experimental group took 0.49 steps (95% CI −0.11 to 1.08, p = 0.11, I2 = 0.1%) more in 15 s than the control group with some uncertainty in the estimate.

Effect (MD, 95% CI) of exercise on balance (step test in # steps/15 s) by pooling data from three trials (n = 570).
All trials of exercise vs no/sham intervention measured
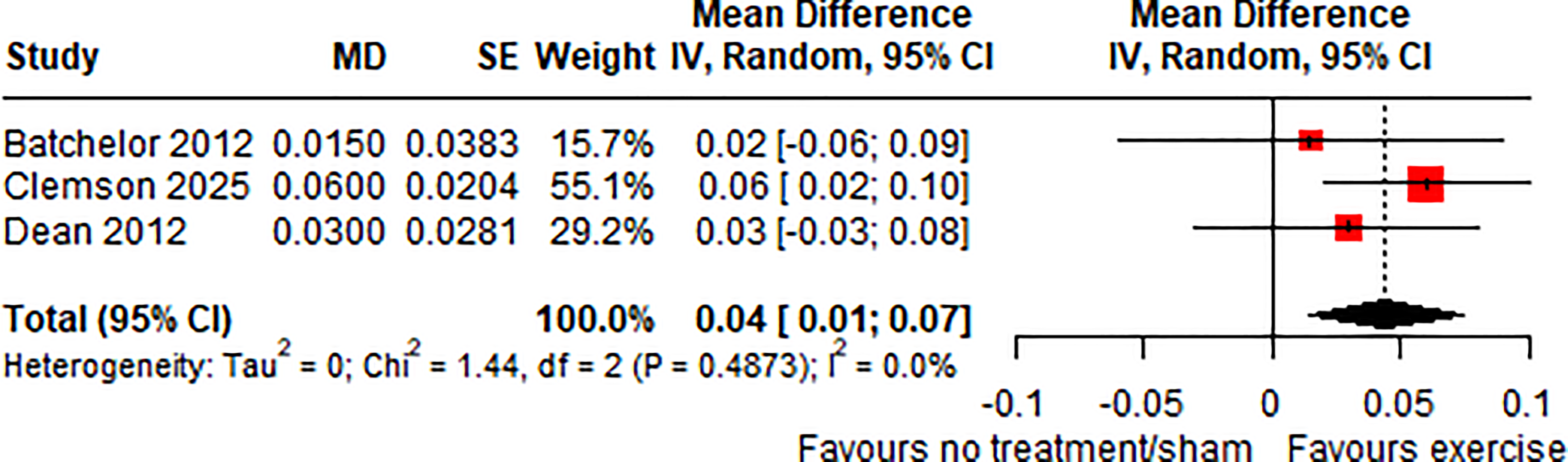
Effect (MD, 95% CI) of exercise on mobility (preferred walking speed in m/s) by pooling data from three trials (n = 561).
Two trials of exercise vs no/sham intervention measured
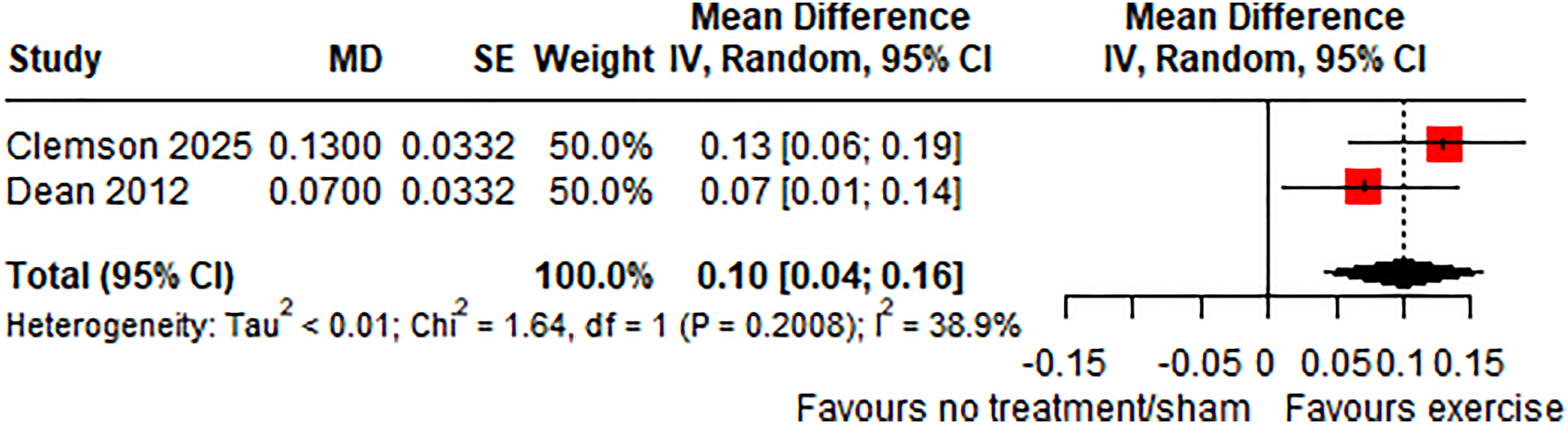
Effect (MD, 95% CI) of exercise on mobility (fast walking speed in m/s) by pooling data from two trials (n = 426).
Two trials of exercise vs no/sham intervention measured quality of life using different measures. One trial 19 used the SF12 and reported no difference (MD 0 out of 100, 95% CI −3 to 3, p = 0.98). Another trial 18 used the visual analogue scale of the EuroQol5D and reported little difference (MD 3 out of 100, 95% CI −1 to 7, p = 0.13).
Discussion
In this review, we found a trend in favour of exercise in reducing the rate of falls after stroke compared with no/sham intervention, although there is considerable uncertainty around the true estimate of effect. Notably, the included trials showed small effects on mobility and balance and no effect on quality-of-life measures, suggesting that falls reduction may not necessarily occur through changes in these outcomes alone.
Overall, our review findings suggest that exercise may reduce the rate of falls after stroke, but that it doesn’t prevent an individual falling. This is similar to previous stroke reviews, although the tighter inclusion criteria used here mean that whilst we included less total participants (677 compared to 1352 in the review by Yang and colleagues) our results may be more accurate.6,21 Yang and colleagues concluded that in future, trials should be included if they measure falls as a primary outcome and collect these data over some time. 22 In our review, we applied this recommendation by including only trials that measured falls over 12 months and were powered for this outcome.
Taken together, the findings in people after stroke differ from the evidence in healthy older adults, where exercise reduces the number of people who experience a fall. 5 The fact that falls aren’t fully prevented in people after stroke may reflect the high-risk nature of this group. However, it's also important to consider the complexity of post-stroke impairments and the wide range of factors that contribute to falls in this population. 3
Since the last review, a new large, randomised trial (FAST) has provided important evidence in this area. 18 FAST reported a significant reduction in rate of falls and differed from earlier exercise trials in several ways. The intervention focused on embedding exercises into everyday activities, with an emphasis on habit formation, and combined this with targeted home safety and community mobility strategies. Importantly, each component was tailored to the individual's ability, e.g., prioritising home-safety approaches for those with poorer mobility. This tailoring of the intervention may explain why FAST achieved a reduction in falls when earlier trials did not. FAST was informed by the earlier study by Dean and colleagues, which found that exercise delivered in a circuit class and without a home safety component tended to increase falls among people with more severe disability. 19 In that trial, slower walkers trended towards more falls with exercise (IRR 1.47, 95% CI 0.77 to 2.80), whereas faster walkers showed a trend towards fewer falls (IRR 0.58, 95% CI 0.28 to 1.17). This highlights the importance of tailoring the intervention to the individual's capability and level of risk. The FAST results reinforce this and suggest that a tailored approach may be necessary to see meaningful reductions in falls.
This review has both strengths and limitations. A key strength is that the included trials were adequately powered trials and reported the same falls outcomes, enabling the use of published between-group data within the meta-analyses, thereby improving the accuracy of the pooled estimates. Similarly, the trials employed common measures of mobility and balance, allowing the calculation of mean differences in real units that are clinically interpretable. 21 However, the review only included three trials, all conducted within a single country, which restricts the generalisability of the findings. Additionally, although the trials were generally high quality (scoring 8 out of 10 on the PEDro scale), the nature of the interventions meant that blinding of participants and therapists was not possible, introducing potential bias.
In conclusion, while our review highlights ongoing uncertainty about the overall effect of exercise on falls after stroke, the emerging evidence suggests that exercise (particularly when embedded into daily routines and includes home-safety elements) can reduce the rate of falls. Future studies should consider stratified approaches that reflect the diversity of disability levels and the multifactorial nature of falls after stroke.
Clinical messages
Current evidence suggests exercise can reduce the rate of falls but not the number of individuals experiencing falls.
Clinicians need to consider the individual after stroke and their risk factors for falls, tailoring the intervention to individual ability.
Supplemental Material
sj-docx-1-cre-10.1177_02692155261441558 - Supplemental material for Exercise to reduce falls in Community-Dwelling people after stroke: A systematic review with meta-analysis
Supplemental material, sj-docx-1-cre-10.1177_02692155261441558 for Exercise to reduce falls in Community-Dwelling people after stroke: A systematic review with meta-analysis by Katharine Scrivener, Ingrid Lin, Louise Ada, Natasha A Lannin, Petra L Graham, Elisabeth Preston, Alena Haines, Sally Day, Lindy Clemson and Catherine M Dean in Clinical Rehabilitation
Footnotes
ORCID iDs
Author contributions
KS: Conceptualisation, Methodology, Investigation, Data curation, Writing – Original Draft
IL: Investigation, Data curation, Writing – Original Draft
NL: Writing – Review & Editing
PG: Data curation, Formal analysis, Writing – Review & Editing
LA: Data curation, Writing – Original Draft
EP: Writing – Review and Editing
LC: Writing – Review and Editing
SD: Investigation, Writing – Review & Editing
AH: Methodology, Investigation, Writing – Review & Editing
CD: Writing – Review and Editing
Funding
The authors received no financial support for the research or publication of this article. The following authors acknowledge funding support: Ingrid Lin was supported by an Australian Government Research Training Program (RTP) Scholarship via Macquarie University; Natasha Lannin was supported by a Heart Foundation fellowship (#106762).
Declaration of conflicting interests
The review authors (KS, IL, NL, EP, SD and LC) acknowledge their involvement as authors in one of the included trials, with CD authoring two of the included trials.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
