Abstract
Objective
To assess the feasibility and, preliminarily, the effectiveness of long-term, personalized gait training using a digital wearable system (Gait Tutor) that provides real-time audio biofeedback to correct or reinforce gait behaviour.
Design
Open-label and non-controlled, with assessments before and after intervention.
Setting
Real-world.
Participants
Twenty persons with Parkinson's disease.
Intervention
Participants performed home-based gait training in their ON medication state for 30 minutes, 3 times per week, for 9 months using a Gait Tutor.
Main measures
We evaluated adherence (% of expected sessions), usability, and, preliminarily, efficacy by assessing the motor performance of the participants before and after the intervention.
Results
Seventeen participants (85%) completed the study, performing an average of 83 sessions. Adherence was higher for persons with an intermediate disease stage (80.5% of expected training sessions), compared to those with a more advanced disease stage (46.2%). All participants reported extremely positive scores on the questionnaire about ease of use and effectiveness (4.37 ± 0.42). The Movement Disorders Unified Parkinson's Disease Rating Scale motor scores remained stable after the training (mean 9 months). In people with an intermediate disease stage, clinical scores and physical capacity tended to improve.
Conclusions
For the first time, this study shows the feasibility of long-term real-world gait training for people with Parkinson's disease, providing preliminary evidence that personalized, technology-driven rehabilitation strategies can be sustained over extended periods and can assist clinicians in objectively assessing gait performance in the real world.
Introduction
Parkinson's disease is the second most common neurodegenerative disorder after Alzheimer's disease (180–200 cases/100,000 people in the 55–64 years range 1 ). Persons with Parkinson's disease suffer from gait and balance impairments, with more than 60% reporting at least one fall per year, 2 significantly affecting their functional independence and quality of life.3,4 There has been a growing interest in easy-to-use and low-cost digital technology, to monitor disease progression outside of clinical settings.5,6 Digital rehabilitation programmes using real-time feedback (auditory and vibrotactile feedbacks are more common with respect to haptic and visual 7 ) of gait parameters recorded by wearable sensors showed promising results in improving mobility in people with Parkinson's disease. 8 In Carpinella et al., 9 Parkinson's disease participants used wearable-based visual and acoustic feedback in 20 gym sessions (3 times a week). The intervention group showed better balance than the controls, with no difference in speed. In another study, a remote training was compared over 1 year with an active control group walking with a pedometer, showing that both groups improved physical activity. 10 A two-month home-based training programme improved symptoms, mobility, and cognition in the early to mild stages. 11 A 5-week tele-rehabilitation was found to be feasible and safe in mild-to-moderate Parkinson's disease. 12 In a European project (CuPiD), a digital gait trainer (smartphone and wearables) was developed (Gait Tutor (GT), mHealthTechnologies srl, Bologna, Italy) and tested in a pilot randomized controlled trial, with 20 participants with Parkinson's disease in the intervention (3 times a week for 6 weeks), and 20 participants with Parkinson's disease as controls. 8 Single and dual task walking speed improved after the training in both groups. Balance improved more in the intervention. No adverse events were reported. A recent study also showed that Gait Tutor's vocal feedback was more effective (more walking distance covered) than conventional rhythmic auditory cueing. 13
Recent studies showed that remote exercise and rehabilitation can improve motor performance in the short term. 14 Nonetheless, since people with Parkinson's disease could likely benefit from continued rehabilitation throughout their disease course, long-term remote rehabilitation should be considered. Based on this, the primary objective of the current work is to assess the feasibility of an extended training programme, using Gait Tutor for 9 months (no studies using digital technologies have yet employed such a period). As a secondary exploratory objective, we will present preliminary effectiveness results.
Methods
Participants
Twenty people affected by Parkinson's disease were recruited between February 2020 and October 2022. The sample was equally divided between two sites in Italy (IRCSS Institute of Neurological Sciences in Bologna) and Israel (Tel Aviv Sourasky Medical Center).
The participants were included if they had received a diagnosis of Parkinson's disease according to the Movement Disorders Society criteria; were in Hoehn and Yahr (H&Y) stage 2–3; were able to walk for 10 m independently; and had a stable levodopa therapy for at least the past 3 months.
Individuals were excluded if they had other pathologies that could potentially alter their walking resistance (e.g. congestive heart failure, disabling lung diseases); had vestibular or visual dysfunction affecting gait and may interfere with visual and/or auditory feedback during walking; had cognitive deficits, assessed by a score of 25 or lower at the Montreal Cognitive Assessment (MOCA); had severe (clinically assessed as potentially interfering with the training programme) freezing of gait (FoG); or were anticipated to have difficulty in carrying out the study protocol. Participants were allowed to continue their usual care, including any physical therapy. The study was conducted with the prior approval of the local ethics committees of each institution (Italy n. 758-2019-DISP-AUSLBO; Israel n. 0127-13-TLV) and all participants gave written informed consent in accordance with the declaration of Helsinki.
Gait Tutor system
The Gait Tutor system is a medical device consisting of three inertial measurement units (IMUs) and an Android-based smartphone with a dedicated app for real-time data processing and vocal feedback generation. The smartphone interface is easy to use, enabling the user to connect three wearable inertial sensors: two sensors are attached to the shoes with Velcro straps, while the third is placed on the chest using an elastic strap (Figure 1).
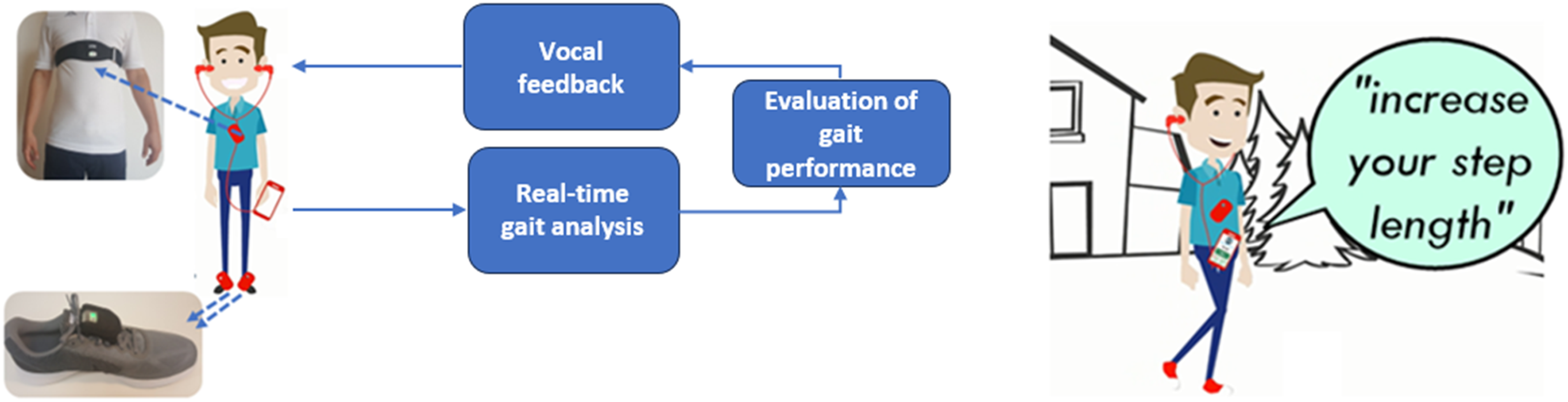
Features of the Gait Tutor system. The system consists of a smartphone, two IMUs worn on the shoes (to estimate gait parameters), and one on the chest (to estimate trunk posture). The device provides real-time audio feedback (e.g. ‘increase your step length’) on selected gait and postural parameters (e.g. step length) based on the observed pattern to correct gait behaviour.
Gait Tutor is a closed-loop biofeedback system that, by analyzing gait in real time, provides appropriate (i.e. based on the analysed gait pattern) vocal feedback to help users improve their gait (e.g. ‘take longer steps’, ‘walk faster’). It is possible to train different walking characteristics (such as cadence, gait speed, asymmetry, and trunk posture during walking) as defined during the clinical assessment. The system is personalized based on the user's optimal walking pattern, recorded during the clinical assessment, and serves as a target therapeutic zone.
Moreover, the mobile app automatically records the frequency and duration of training sessions, eliminating the need for participants to use a diary as is typically required in traditional settings. Clinicians can remotely change the training settings and monitor the results of the gait workouts (e.g. the number of sessions, distance covered, average gait speed). The gait and posture parameters that can be trained (i.e. set for real-time feedback) are detailed in Supplementary Table 1.
Intervention
At baseline, all participants were instructed to perform gait training using the Gait Tutor device at least 3 times a week for 30 minutes, following the American College of Sports Medicine (ACSM) guidelines. 15 The real-world gait intervention lasted 9 months. Each participant was asked to perform the Gait Tutor training in the ON therapy condition (at least 30 minutes after taking one of the usual doses of levodopa) to maximize the motor response of drug therapy. The participants were trained to use the Gait Tutor application, and instructions were given on correctly applying IMUs to shoes. During training, the smartphone was inserted into a pocket of the participant's clothing. During the initial visit of the training period (and during any follow-up visits), Gait Tutor's intervention was personalized: an optimal performance walk was performed to calibrate the Gait Tutor system and serve as a reference target zone for subsequent training. Training parameters were individually set for each participant based on a thorough clinical evaluation aimed at understanding their specific needs. These parameters were then regularly monitored and adjusted during follow-up visits, allowing for a tailored training approach. During the clinical assessments, we also recorded any concurrent physical therapy the participants were undergoing. Additionally, regular videoconference calls were held with the clinical staff involved in the project, including neurologists, physiatrists, neuro-physiotherapists and neuro-physiopathologists.
The recruitment and training programmes of participants at the beginning of the experimental phase of the project were suddenly interrupted due to the COVID-19 global pandemic. The lockdown imposed by governments worldwide affected the participants involved in the study in both countries, who could not carry out training with Gait Tutor according to the protocol. The pandemic conditions in Italy and Israel differed, and each country had its own set of government regulations that evolved following the progression of the global pandemic. Therefore, the training was not conducted in parallel between the two countries: from February 2020 to February 2022 in Italy and from January 2021 to October 2022 in Israel.
Outcomes
This study evaluates feasibility, usability, and, preliminarily, efficacy. The primary outcome of the study is adherence to the Gait Tutor training programme, which is aimed at assessing the feasibility of a long-term tele-rehabilitation programme.
Regarding feasibility, we also report outcomes related to the training sessions that can be automatically derived from the Gait Tutor system: total length of the training period and average measures of training sessions about duration, number of steps, distance covered, and percentage of praising vocal feedback. The latter is the percentage of vocal messages that were provided by the system when the participant had a correct gait pattern (e.g. ‘Well done!’), to reinforce the positive behaviour inside the target therapeutic zone.
Usability (participant satisfaction) was evaluated after the intervention using a 3-item Likert scale, with scores ranging from 1 (‘totally disagree’) to 5 (‘fully agree’) assessing the ease of use of the assistive device, the perceived effectiveness of gait training, and the quality of the professional services (e.g. information, attention) received for using the device.
A preliminary evaluation of efficacy was conducted considering clinical and motor assessments performed on an outpatient basis in ON medication state before (pre-intervention; T0) and after the training period (post-intervention; T1), on average 10 months 12 days ± 49 days after T0. The following secondary outcomes were considered:
Movement Disorders Unified Parkinson's Disease Rating Scale (MDS-UPDRS) Part III Mini-Balance Evaluation Systems Test (MiniBESTest) Berg Balance Scale (BBS) Timed Up and Go (TUG) under single- (st) and dual-task (dt) conditions 10-m walking test (10 MWT).
To further explore efficacy, a set of measures related to gait performance were extracted from the training sessions (duration and percentage of praising vocal feedback) using the initial and final 2 months of training to evaluate the initial and final performance, respectively. The 2-month period was established to ensure that an adequate number of training sessions were conducted for all participants in order to be included in the statistical analysis. This analysis enabled the evaluation of potential improvements or declines in motor performance during remote gait training, providing a complementary assessment with respect to the clinical and motor assessment performed at the baseline and final clinical visits.
Data analysis
Baseline clinical characteristics were analysed for significant differences between individuals recruited at the two clinical centres using the Mann–Whitney U test. Analyses of participants’ adherence and the efficacy of the intervention were conducted, considering participants who had completed the gait training programme and attended the final clinical assessment.
The primary outcome (adherence) was calculated by determining the ratio between the total duration of the Gait Tutor training sessions performed (excluding training sessions shorter than 5 minutes) and the total duration of the sessions expected to be carried out during the training period (three sessions of 30 minutes per week for the entire training period). The training period was defined as the time between the first and the last session carried out by each participant. A threshold of 75% was arbitrarily chosen a priori to assess the feasibility of the gait training programme.
A pre-post analysis was conducted on secondary clinical endpoints collected at baseline (T0) and at the clinical visit after the 9-month training (T1). Depending on data distribution, the Wilcoxon rank test or paired sample t-test was used. As previously stated, drop-outs were excluded from the analysis. A Spearman correlation analysis was performed to find a possible association between adherence to the gait training programme and the changes in the analyzed outcomes.
To identify those who responded to the proposed intervention, we implemented an exploratory methodology based on studies using wearable digital technologies for rehabilitation after stroke.16,17 To be defined as a responder, the participant should show, considering the average progression of the disease (and related outcomes) in 9 months, an improvement (reduction in MDS-UPDRS III score or increase in walking speed) exceeding the corresponding Minimal Clinical Important Change (MCID).
Horváth et al. reported an MCID of −3.25 points for detecting minimal but clinically pertinent improvement. 18 The MDS-UPDRS III progression reported in the literature is highly variable due to different sample sizes and participant characteristics (e.g. disease severity), and the medication state in which the assessments are performed. The following values are reported: 0.33, 19 1.84, 20 2.21, 21 2.4 22 and 3 23 points/year. Among these values, first, we excluded the study with a sample size that was very small with respect to the other studies (33 participants 19 ). Then, we considered the smallest 1-year progression value (1.86) from the remaining studies to be conservative in identifying responders (i.e. with higher values of progression, lower values of improvements would be needed to be identified as a responder). Hypothesizing a linear increase in MDS-UPDRS III progression, 22 the expected change over a 9-month period was estimated (considering 9/12 of the value). Participants were classified as responders on the MDS-UPDRS motor score if, following the intervention, they exhibited a reduction in the clinical score equal to the sum of the MCID (−3.25 18 ) and the 9-month expected progression (1.395). Responders based on MDS-UPDRS motor score were, therefore, those participants with a change (post – pre) < −1.855 points.
Considering in-lab gait speed, the reported values in the literature for progression in Parkinson's disease were −0.0124, 23 −0.03, 19 −0.034 24 and −0.04 m/s 21 per year, with a certain degree of variability given by the different severity of the analysed participants and by the different instrumented tests performed (e.g. 10MWT or walking on an instrumented mat). In addition, a change of −0.021 m/s was found in real-world settings. 20 Based on the assumption of a linear decrease, 21 the expected change over 9 months was estimated. Participants were classified as responders in terms of gait speed if, following the intervention, they exhibited an improvement equal to or greater than the sum of MCID (0.082 25 ) and the expected 9-month progression (−0.0093) computed from lower value reported in literature. 23 Responders based on 10MWT gait speed were, therefore, those participants with a change (post – pre) > 0.0727 m/s. Furthermore, the overall responders were defined as those participants who were identified as responders concerning both the MDS-UPDRS motor score and the in-lab gait speed.
Results
Clinical characteristics
The clinical characteristics of the Italian and Israeli cohorts are presented in Table 1. Age and disease duration differed significantly between the two centres (P = .031 and P = .040, respectively). The Israeli participants were older and had significantly longer disease duration. The MDS-UPDRS III score was higher in Israel, although this difference was not statistically significant in the two centres (P = .0816). The Italian cohort had a majority of participants with a H&Y score equal to 2, while the Israeli cohort had a majority of participants with a more advanced H&Y score (H&Y = 3). Among all the people recruited, 11 (55%) had a Hoehn and Yahr stage of 2, while the remaining 9 participants had a worse H&Y score (H&Y = 3). At the Israeli site, only male participants were included in the study.
Demographic and clinical characteristics of the participants.
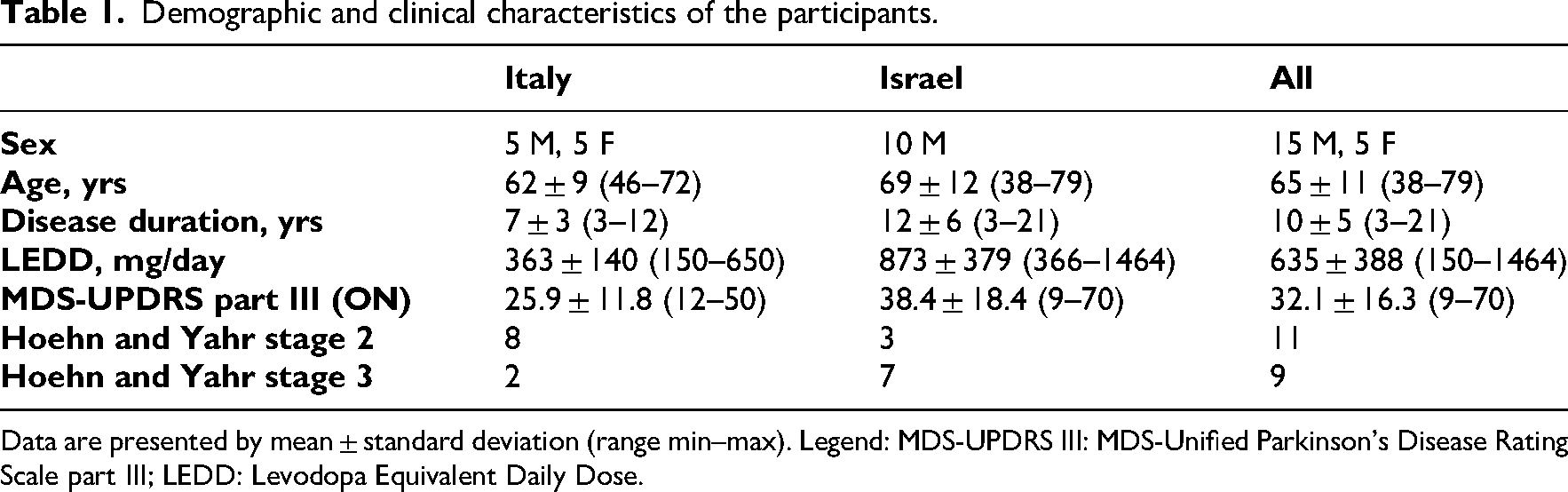
Data are presented by mean ± standard deviation (range min–max). Legend: MDS-UPDRS III: MDS-Unified Parkinson's Disease Rating Scale part III; LEDD: Levodopa Equivalent Daily Dose.
No relevant changes or differences in physical therapy were observed among the participants throughout the study period.
Feasibility and usability
Seventeen participants (85%) completed the study. Three participants (60 ± 12 years; two participants with a H&Y stage of 2 and one with a more advanced H&Y stage (3) withdrew from the study prior to the conclusion of the planned training period. Table 2 presents the results of feasibility and usability for all participants and is divided by disease stage.
Feasibility and usability of the Gait Tutor system. Characteristics and statistics of training sessions performed by participants who completed the training programme are shown.
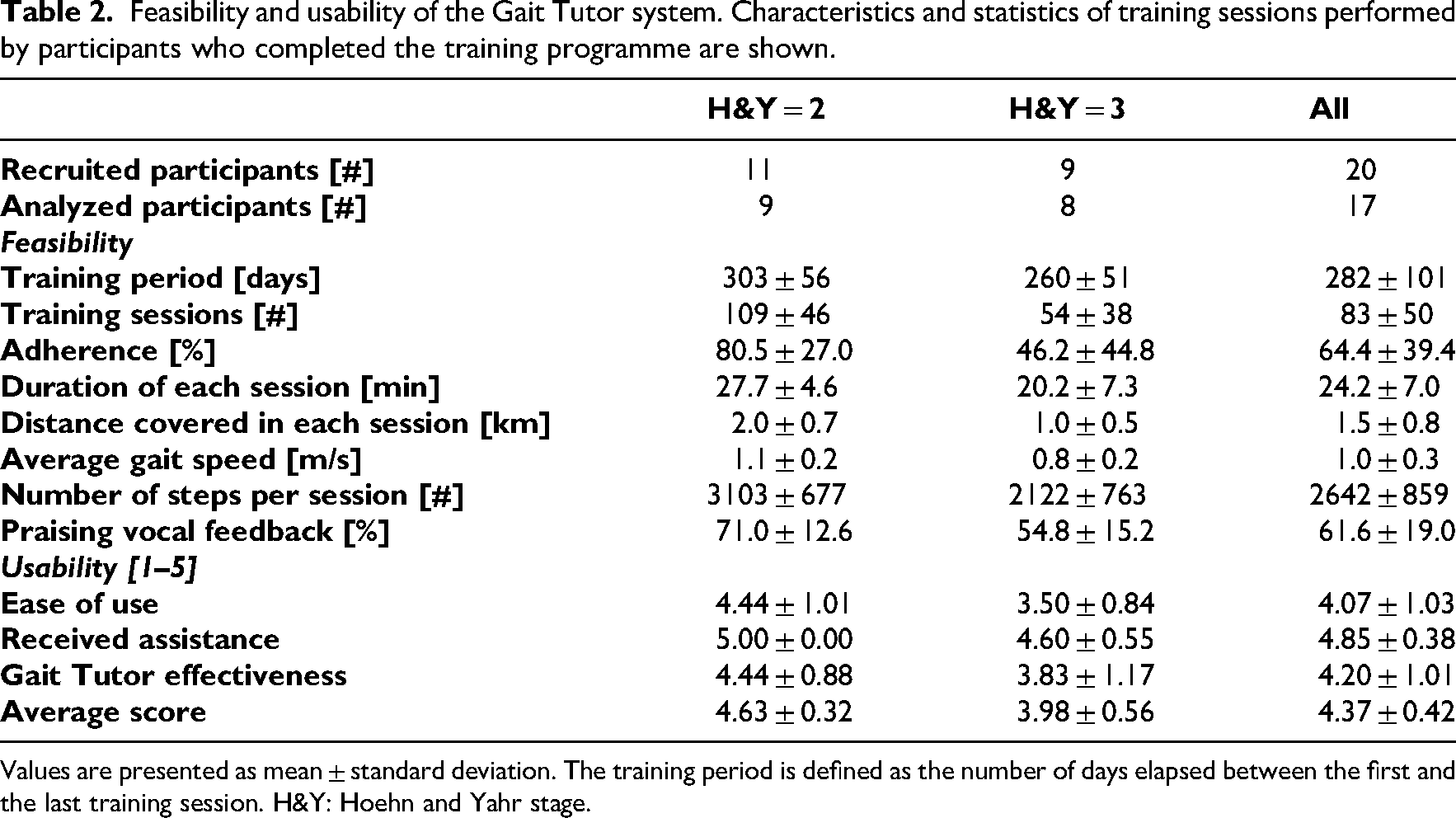
Values are presented as mean ± standard deviation. The training period is defined as the number of days elapsed between the first and the last training session. H&Y: Hoehn and Yahr stage.
Figure 2(a) shows the adherence to training of every participant. The average adherence to the training programme was 64.4%, exceeding the 75% feasibility threshold for participants with a milder stage of the disease (H&Y = 2, showing an 80.5% adherence). The average training period for individuals who completed the study exceeded nine months, and no adverse events occurred while using the Gait Tutor system. As reported in Table 2, participants with a HY stage of 2 performed on average longer training sessions (27.7 minutes) with a higher gait speed, distance covered, and number of steps than those with a more advanced stage (20.2 minutes). The mean percentage of praising vocal feedback was 61% overall. It was 71% and 55% for participants with a HY stage of 2 and 3, respectively. Supplementary Table 2 presents a comprehensive overview of each participant's demographic data, clinical characteristics, and training results.
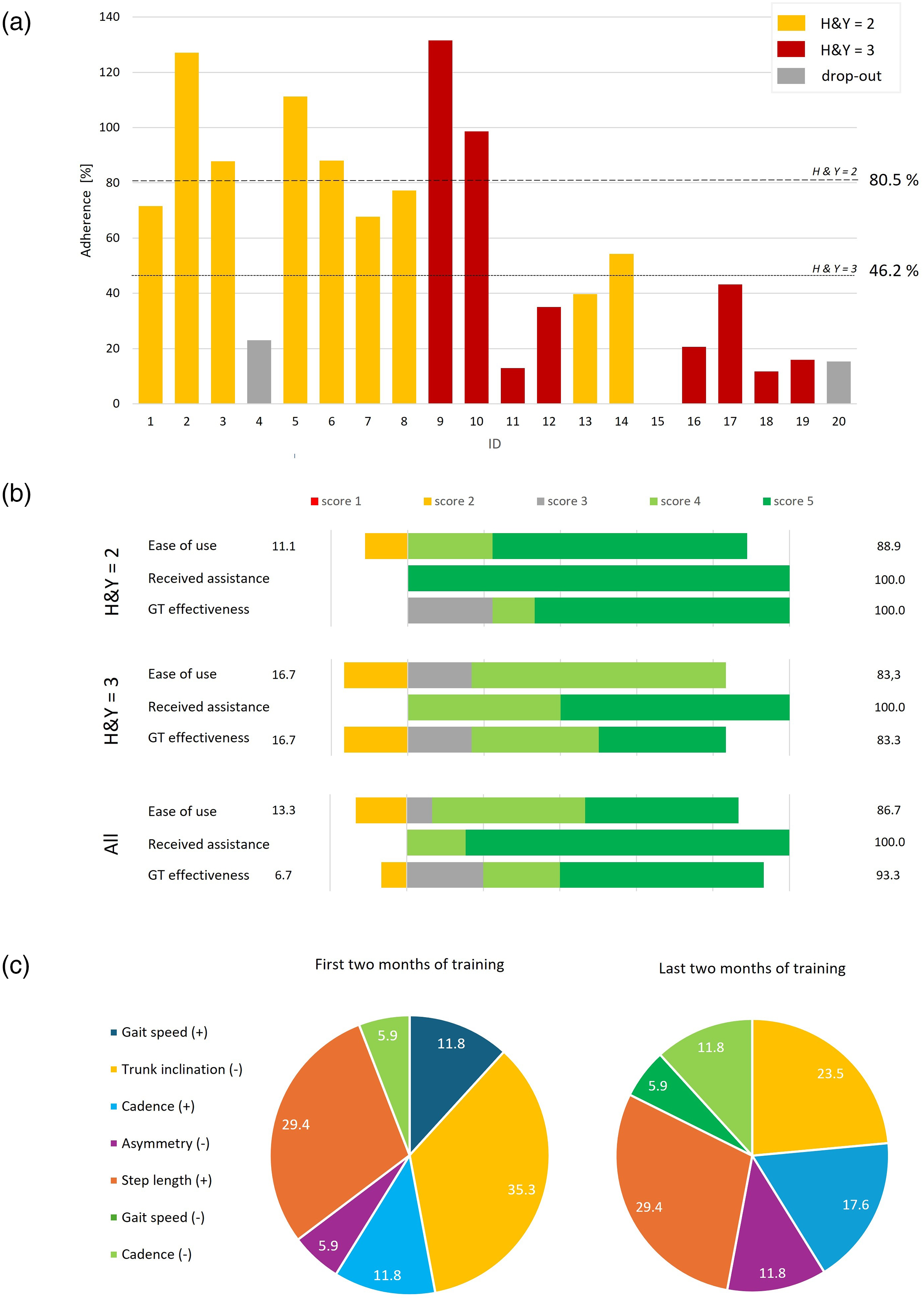
(a) Adherence to the training of all participants from both sites (1–10 Italian participants, 11–20 Israeli participants). The percentage of average adherence considering individuals with HY = 2 and HY = 3 who completed the gait training programme is shown as a dashed line. Participants with HY = 2 and HY = 3 are shown as yellow (light grey in the printed version) and red (dark grey) bars, respectively. Drop-outs are represented as grey (medium grey) bars (if they did not complete the training programme) or as white spaces (if they did not perform any training session, e.g. participant #15)). (b) Bar plot related to the 3-item Likert scale scores for ease of use, received assistance, and Gait Tutor perceived effectiveness by participants who completed the training programme. The percentage of negative (1,2) and positive scores (3–5) are reported on the right and the left, respectively. H&Y: Hoehn & Yahr stage. (c) Distribution of the provided biofeedback messages to the participants during the first two (left) and last two (right) months of training. The ‘+’ (‘−’) symbol indicates feedback aimed at increasing (decreasing) the specified parameter.
The scores assigned by the participants to the satisfaction questionnaire completed at the end of their training are reported in Figure 2(b). Individuals with more severe clinical features (H&Y = 3) reported lower values for ease of use and effectiveness. It should be noted that some of the satisfaction questionnaire scores are missing (see Supplementary Table 3).
The percentage of feedback messages that were provided by the system in the first and last two months of the intervention is reported in Figure 2(c). In the first two months, the most common feedback, considering all participants, was based on the correction of posture (35.3%). This decreased to 23.53% in the last phase, where the feedback on increasing step length was the most common one.
Clinical, physical capacity, and gait training outcomes
Pre-post values of clinical scores, physical capacity, and outcomes related to training sessions are presented in Table 3, divided by disease stage.
Pre–post evaluation of participants who completed the gait training programme, divided by different Hoehn and Yar stages. Duration of the training sessions and the praising vocal feedback were extracted using the initial and the last two months of training (related to T1 and T2 evaluations, respectively).
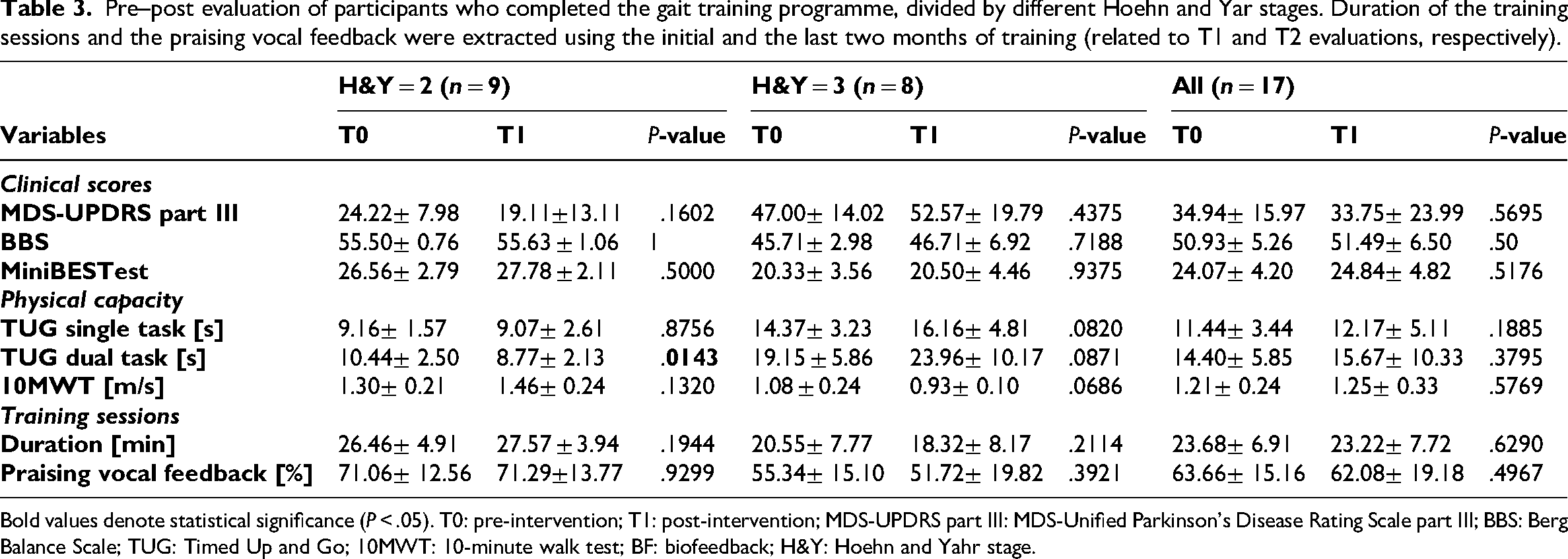
Bold values denote statistical significance (P < .05). T0: pre-intervention; T1: post-intervention; MDS-UPDRS part III: MDS-Unified Parkinson's Disease Rating Scale part III; BBS: Berg Balance Scale; TUG: Timed Up and Go; 10MWT: 10-minute walk test; BF: biofeedback; H&Y: Hoehn and Yahr stage.
Considering all participants (i.e. H&Y = 2 & H&Y = 3), clinical scores, physical capacity, and training sessions, statistics were not significantly different between the two time points. MDS-UPDRS III showed an average reduction of 1.19 points.
People with a H&Y score of 2 showed a general improvement trend in both clinical and physical capacity outcomes, with a significant improvement (i.e. reduction) in dual-task TUG time. An average improvement was seen in the MDS-UPDRS motor score that, although not significant, exceeded the minimally clinically important difference (MCID) for improvement. 22
In contrast, in people with a more advanced Parkinson's disease stage (H&Y = 3) there was an average worsening of the MDS-UPDRS III motor score (although not statistically significant). No significant differences were found, but a general decline trend was present, except for the BBS and the MiniBESTest.
Adherence was 64.4% on average but no significant correlations (P> 0.05) were identified between adherence to the gait training programme and the changes in all outcomes (clinical, physical capacity, and gait training). Clinical scores and training session outcomes for each participant are reported in Supplementary Table 4.
Representative results of gait training are reported in the Supplementary Material: Supplementary Figure 1 shows the duration of gait training sessions and the corresponding gait speed achieved in each trial by two representative participants with different disease stages and stable medication regimes over the training period; Supplementary Figure 2 shows training data from an Italian participant with gait asymmetry, step height, and trunk posture inclination as the training target parameters selected by the clinician during the initial assessment; Supplementary Figure 3 reports the distribution of the personalized biofeedback messages played over the whole training period for an Italian participant.
Responders to intervention
By analysing the change in MDS-UPDRS score and gait speed achieved during the 10MWT, a total of nine participants (53%) were classified as responders on the MDS-UPDRS score, and six (35%) were classified as responders on gait speed (Figure 3). Furthermore, five participants (29%), all of whom had a H&Y of 2, exhibited a response to both the MDS-UPDRS motor score and gait speed. Finally, six participants (67%) with a H&Y stage of 2 improved the MDS-UPDRS motor score or gait speed. The outcomes achieved by each participant are detailed in Supplementary Table 5. It should be noted that for one participant, there was missing data for MDS-UPDRS III; for two participants, there was missing data on gait speed; in those cases, it was impossible to perform this analysis (Figure 3).
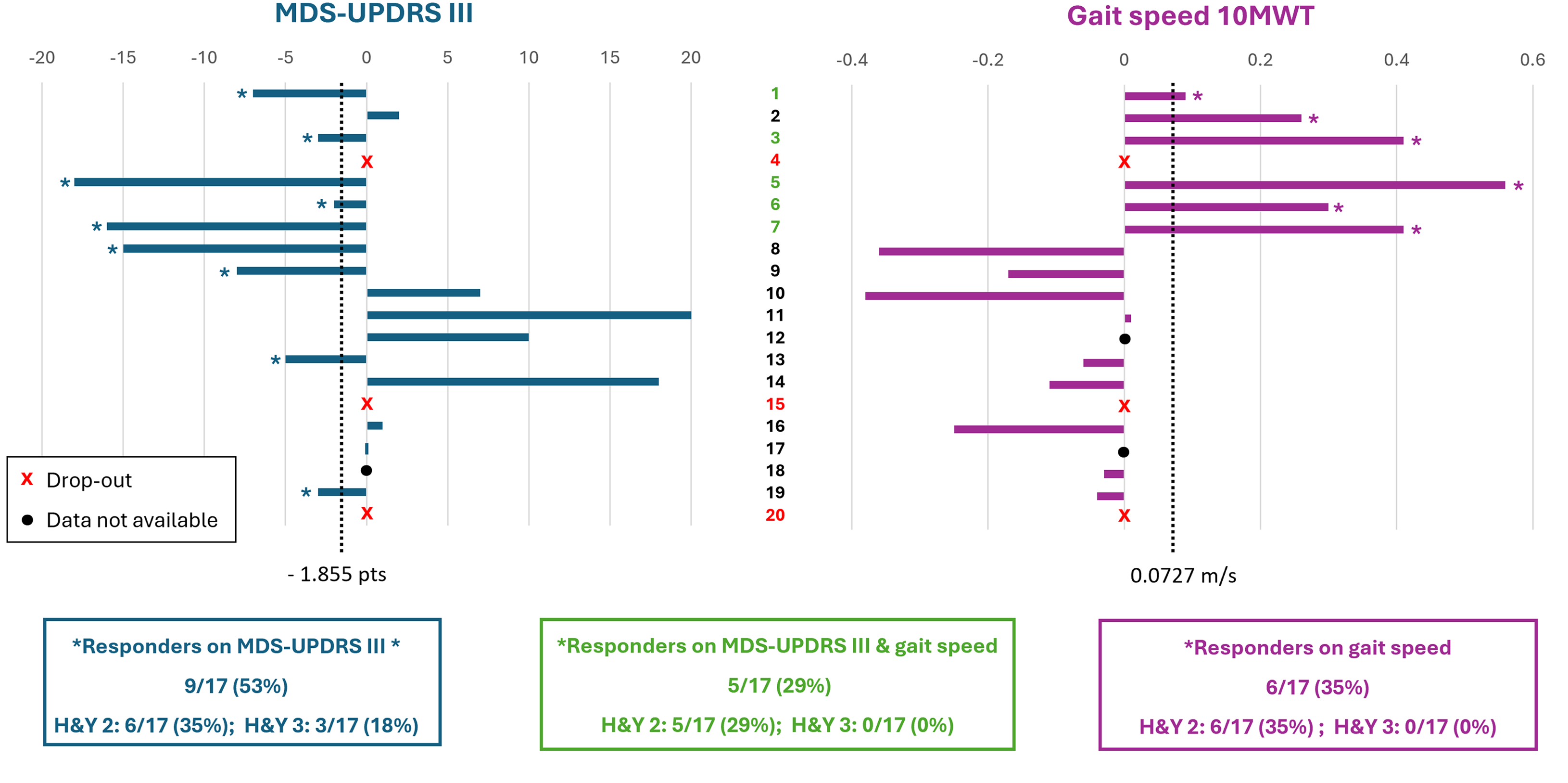
Responders on MDS-UPDRS III and in-lab gait speed, excluding participants who dropped out. Each bar represents the difference between pre and post values of MDS-UPDRS III (left side) and gait speed achieved in the 10MWT (right side). An asterisk at the end of a bar indicates that the participant met the criterion for a clinically meaningful change (responder): participants were considered responders if the change in MDS-UPDRS III was lower than −1.855, and if the change in gait speed was greater than 0.0727 m/s. The thresholds used to assess responsiveness are shown as vertical dotted lines. An ‘x’ denotes a participant who did not complete the 9-month intervention period (dropout), while a black dot indicates participants with missing data for the specific outcome.
Discussion
While previous literature reported on the feasibility and effectiveness of using mobile health technology to monitor Parkinson's disease symptoms 26 and deliver rehabilitation at home, 8 this is the first study to investigate the feasibility of using a wearable system for several months as a form of tele-rehabilitation during daily living. Adherence, the primary outcome, was 64.4% on average. It met the predefined target of 75% among people with a H&Y stage of 2 (80.5%), while it was lower in more advanced Parkinson's disease participants (46.2%). Eighty-five per cent of participants could complete the programme, with an average training duration over 9 months. The overall adherence is comparable to other tele-rehabilitation studies. Bianchini et al. 12 reported over 85% (70%) participants doing more than 60% (80%) of the planned self-conducted tele-rehabilitation sessions (5 weeks, 30 minutes, three sessions per week). Zajac et al. 27 achieved 86.4% of the planned sessions in a music-based walking intervention (4 weeks, 30 minutes, five sessions per week). Ellis et al. showed an average use of three days per week over one year for a mHealth exercise programme. 10 Lutzow et al. 28 reached a 74.5% adherence in a long-term training (6–8 months) using a smartphone app, remotely supervised by a therapist. As a note, the adherence differed in the two countries. In Italy, the average adherence exceeded the target, with three participants doing more training than planned. In contrast, the average adherence among Israeli participants was below the target. This discrepancy could be attributed to the older age and higher disease severity of the Israeli cohort and, possibly, to the period in which the project was carried out, with an overlap with the COVID-19 pandemic, which posed significant difficulties in recruiting and in completing continuous training, especially during the initial period with lockdown measures. Furthermore, gait training was performed in different periods with different seasonality in the two countries. As a limitation, this was not taken into account in the analysis.
No adverse events occurred using the system and usability responses were generally extremely positive, but individuals with more severe clinical features (H&Y = 3) reported lower satisfaction with ease of use and lower effectiveness.
Although the primary objective of the project was to assess the feasibility of the intervention, the efficacy for an intermediate stage of disease (H&Y = 2) was promising. Parkinson's disease has a progressive motor deterioration 29 and recent literature showed that the MDS-UPDRS motor score increases linearly 22 between 0.33 19 and 3 points per year. 20 Thus, achieving a stable clinical state after 9 months can be regarded as a promising result. The average MDS-UPDRS motor score was in fact stable, decreasing (not significantly) by 1.19 points. Focusing on intermediate participants (H&Y = 2), the average decrease (improvement) of MDS-UPDRS motor score was higher, 5.11 points (more than the corresponding MCID 18 ). In the higher disease stage, there was an opposite trend, with an average decline (an increase of 5.57 points). Interestingly, there was a significant reduction in the time to perform a dual task Timed Up and Go (by 1.67 seconds; Table 3), showing a possible effect of the closed-loop biofeedback system in improving gait automaticity. 8 The training with vocal feedback could, in fact, be envisioned as dual-task training.
Evaluating the response to the training, nine participants (53%) improved MDS-UPDRS III and six participants (35%) improved the 10 m walking test speed. Five (29%) were responders for both outcomes (all with H&Y = 2). Six intermediate participants (67%) improved the MDS-UPDRS motor score or gait speed. Three participants (37%) with a H&Y stage of 3 showed only an improved MDS-UPDRS III score.
What emerges is that the system's usage, acceptability, and effectiveness are higher in the intermediate stage (H&Y = 2). People with a more advanced disease stage (H&Y = 3) may have found more difficulties in using the system for a long time. As Parkinson's disease progresses, in fact, individuals often experience problems with walking, balance, falls, freezing, and dyskinesia.30,31 This limitation related to advanced stages suggests that the system and the training protocol should be adapted to improve adherence and efficacy in the most sever participants We hypothesize that offering a less demanding version (e.g. simpler feedback and adaptable gait targets based on user preferences and needs) may enhance their adherence. Moreover, these results are consistent with the idea that the sooner, the better in Parkinson's disease rehabilitation. 32 In addition, it would be interesting to apply this as a form of prehabilitation (preventive approach focused on improving physical function before significant worsening occurs aiming to minimize disability and improve quality of life) in people in relatively mild stages of Parkinson's disease.
While many gait impairments are common in Parkinson's disease, they may vary from one participant to the next. Indeed, the distribution of the provided messages, changing between the first and last two months of intervention (Figure 1(c)), shows the personalization that can be obtained. This is related both to the selection of the parameters by the clinician, tailored to each participant, and to the performance of each participant, who can find it easier or more difficult to walk within the target zone for a specific parameter.
In addition to rehabilitation, analyzed in detail here, the system enables the objective assessment of gait impairment outside the clinical setting, which can assist clinicians. Based on an analysis of training duration and distance walked, we hypothesize that all the training sessions were conducted outside the home environment, representing a novel approach compared to traditional systems.
The main limitation of the study is its exploratory nature, with its open-label and non-controlled design and small sample size. A randomized control trial should be conducted on a larger and more suitable sample size, 22 with the inclusion of an active-control comparison group, to build on the results presented here and test the effectiveness of the system. 33 In addition, participant perception should be assessed using a more detailed Likert scale in future studies, in order to collect more specific feedback (e.g. ease of perceiving the vocal feedback, perceived usefulness of the training, and overall engagement).
To conclude, we provided preliminary evidence that Gait Tutor is an easy to use, safe, and, preliminarily, effective system to provide long-term remote gait training over an extended period, particularly in the intermediate stages of the disease, which is in line with the beneficial effects on motor symptoms and medication dosage of long-term physiotherapy in Parkinson's disease. 34 Using this system, as part of a comprehensive long-term management of Parkinson's disease, has the potential to provide ongoing benefits. To our knowledge, this is the first study showing the feasibility of digital gait tele-rehabilitation over nine consecutive months. Further studies are needed to identify the optimal training period and the factors that can influence individual responsiveness.
Clinical messages
Long-term real-world remote gait training is feasible in people with Parkinson's disease.
Adherence to training was higher in those people with Parkinson's disease at an intermediate disease stage.
Physical capacity tended to improve in people with Parkinson's disease at an intermediate stage.
Supplemental Material
sj-docx-1-cre-10.1177_02692155251355880 - Supplemental material for A personalised, gait training telerehabilitation programme for people with Parkinson’s Disease: A long-term pilot study
Supplemental material, sj-docx-1-cre-10.1177_02692155251355880 for A personalised, gait training telerehabilitation programme for people with Parkinson’s Disease: A long-term pilot study by Giovanna Lopane, Ilaria D'Ascanio, Mattia Corzani, Valeria Petrone, Giovanna Calandra-Buonaura, Fabio La Porta, Pietro Cortelli, Eran Gazit, Marina Brozgol, Pablo Thumm Cornejo, Nir Giladi, Alberto Ferrari, Carlo Tacconi, Lorenzo Chiari, Luca Palmerini and Jeffrey M Hausdorff in Clinical Rehabilitation
Footnotes
Acknowledgments
ID and LP during the preparation of the article were funded in part by the Complementary National Plan PNC-I.1 ‘Research initiatives for innovative technologies and pathways in the health and welfare sector’ D.D. 931 of 06/06/2022, DARE – DigitAl lifelong pRevEntion initiative, code PNC0000002, CUP: (B53C22006450001). The other authors have no acknowledgements to report.
ORCID iDs
Author contributions
Giovanna Lopane: conceptualization, investigation, methodology, project administration, supervision, visualization, writing – original draft; Ilaria D'Ascanio: data curation, formal analysis, investigation, methodology, visualization, writings – original draft; Mattia Corzani: data curation, formal analysis, investigation, writing – review and editing; Valeria Petrone: data curation, investigation, writing – review and editing; Giovanna Calandra-Buonaura: writing – review and editing; Fabio La Porta: writing – review and editing; Pietro Cortelli: project administration, supervision, writing – review and editing; Eran Gazit: data curation, investigation, writing – review and editing; Marina Brozgol: data curation, investigation, writing – review and editing; Pablo Thumm Cornejo: data curation, investigation, writing – review and editing; Nir Giladi: writing – review and editing; Alberto Ferrari: writing – review and editing; Carlo Tacconi: writing – review and editing; Lorenzo Chiari: conceptualization, project administration, supervision, writing – review and editing; Luca Palmerini: conceptualization, investigation, methodology, project administration, supervision, visualization, writing – original draft; Jeffrey M Hausdorff: conceptualization, investigation, methodology, project administration, supervision, visualization, writing – review and editing.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was realized with the contribution of Ministero degli Affari Esteri e della Cooperazione Internazionale. The Israel Ministry of Health also supported this work.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Luca Palmerini, Lorenzo Chiari, and Carlo Tacconi are co-founders and own shares of mHealth Technologies s.r.l., that commercializes the Gait Tutor system used in this study. Carlo Tacconi is CEO of mHealth Technologies s.r.l. All other authors declare no competing interests.
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy and ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
