Abstract
Objective
To assess the added value of caregiver-mediated exercises combined with telerehabilitation in addition to usual care compared to usual care alone on the self-reported mobility outcome after subacute stroke.
Design
Multicentre, observer-blinded, parallel randomised controlled trial. An off-site researcher allocated treatments using minimisation.
Setting
Four rehabilitation centres in the Netherlands.
Participants
Forty-one patient–caregiver dyads within 3 months poststroke.
Intervention
Eight-week blended care program with caregiver-mediated mobility exercises for 2.5 h per week supported by telerehabilitation and four face-to-face sessions in addition to usual care.
Main measures
Self-reported mobility domain of the Stroke Impact Scale postintervention. Secondary outcomes were functional outcome, dyads’ psychosocial wellbeing, care transition to the community postintervention and after 6 months.
Results
Forty-one dyads (21 intervention, 20 control) were randomised, and 37 (N = 18; N = 19) were analysed following intention-to-treat. The Stroke Impact Scale mobility was not significantly different between groups postintervention (B 0.8, 95% CI –6.8–8.5,
Conclusions
Caregiver-mediated exercises combined with telerehabilitation yielded no differential effect on our primary outcome self-reported mobility. Although the trial is underpowered, current findings are in line with previous trials. Future studies should further explore beneficial effects of caregiver involvement in stroke rehabilitation targeting psychosocial wellbeing.
Introduction
Recovery of independent mobility (e.g. walking and maintaining balance) is a top priority of patients poststroke. 1 In addition, independent mobility determines to a great extent the ability to return home 2 and level of dependence on informal caregivers. It is estimated that approximately 75% of stroke survivors achieve independent mobility. 3 However, a risk of mobility deterioration exists when professional support tapers off.4,5
Intensive, repetitive task training promotes recovery of mobility and independence in ADL in the first 6 months poststroke6,7 and is recommended by clinical guidelines worldwide. Although the optimal dose remains unclear, the evidence suggests that more is better. 8 The recently updated National Institute for Health and Care Excellence (NICE) guideline for stroke in the United Kingdom recommends at least 3 h of individualised motor/functional therapy a day. 9 Unfortunately, patients often do not receive recommended amounts of training as a result of low staffing levels and time spent in non-patient activities. 10
Training of patient-caregivers dyads to perform task-specific exercises together, i.e. caregiver-mediated exercises, has the potential to augment training without increasing costs. 11 In this trial, a dyad refers to a patient and informal caregiver pair. Blended care, i.e. combining face-to-face support with telerehabilitation, may be a promising approach to provide tailored support to dyads and facilitate the transition to self-management at home. 12 Moreover, informal caregivers can provide sustained support after discharge and promote continued activity in the community.
The primary objective of this randomised controlled trial was to assess the added value of caregiver-mediated exercises combined with telerehabilitation in addition to usual care after subacute stroke on patients’ self-reported mobility outcome when compared with usual care alone. Secondary objectives were to assess the differential effects on patients’ functional outcome, dyads’ psychosocial wellbeing and on the care transition in terms of length of stay and preparedness for discharge.
Methods
We conducted a multicentre, observer-blinded randomised controlled trial with two parallel groups (trial registration NTR7664, 11 December 2018). The study protocol with the acronym Allied Rehabilitation using caregiver-Mediated exercises combined with telerehabilitation for Stroke (ARMed4Stroke) was extensively described and published previously. 13 No protocol changes were made after trial commencement, and no protocol violations were found during monitoring visits. The Medical Ethics Review Committee of VU Medical Centre approved the study protocol on 30 April 2019 (2019.081 — NL67357.029.18), and all participants gave written informed consent. Recruitment started in September 2019 and ended in September 2022, with the last follow-up measurement in January 2023. The current report of results follows the CONSORT statement. 14
Patients and caregivers were recruited from four rehabilitation centres in the Netherlands during in- or outpatient care: (a) Reade Centre for Rehabilitation and Rheumatology, Amsterdam; (b) Roessingh, Centre for Rehabilitation, Enschede; (c) Sint Maartenskliniek, Nijmegen; and (d) Vogellanden, Zwolle. Eligibility was determined in the first week of admission by a rehabilitation physician or physiotherapist. Inclusion criteria were as follows: aged ≥18 years; willing to participate and able to understand Dutch on a sufficient level to understand instructions; complete self-reported questionnaires; and provide written informed consent. Additional inclusion criteria for patients were ≤3 months poststroke according to the WHO definition 15 excluding subarachnoid haemorrhage, living independently pre-stroke, planned to be discharged home or living at home, able to follow instructions (Montreal Cognitive Assessment >21) and willing and able to appoint a caregiver. Caregivers were defined as persons close to the patient (e.g. partner, family and friend) who were not practicing healthcare professionals nor were they paid for their efforts. Caregivers were eligible if they were mentally and physically able to support the patient, as determined by a trained physiotherapist during an intake exercise session. Exclusion criteria for both were a serious comorbidity that interfered with the intervention (e.g. lower extremity fracture) or not being medically stable. After baseline assessment by a blinded observer (MM), an off-site researcher (EW) randomly allocated eligible dyads to the Armed4Stroke intervention in addition to usual care or to usual care alone using an online randomisation module with a minimisation algorithm. Covariates in the minimisation were as follows: (a) centre; (b) inpatient vs outpatient; (c) age ≥70 vs <70; and (d) functional ambulation categories <5 vs 5. The off-site researcher informed the physiotherapist about the assigned intervention. Physiotherapists and dyads could not be blinded to treatment allocation.
The Armed4Stroke intervention was an 8-week progressive, task-specific exercise program focused on mobility in addition to usual care after subacute stroke. Mobility training was supervised by an informal caregiver, supported using blended care with a minimum of four face-to-face sessions scheduled every other week and asynchronous support via the online telerehabilitation platform Telerevalidatie.nl. 16 The physiotherapist composed a tailor-made exercise program in face-to-face consultation with dyads. Dyads were asked to perform exercises together for a minimum of 5 × 30 min per week and were encouraged to train outside of usual training hours and in weekends. During caregiver-mediated exercises, dyads had access to their tailored exercise program with exercise videos, personal instructions, mobility milestones and a messaging environment for asynchronous consultation via the telerehabilitation platform. During face-to-face sessions, patients and caregivers were trained and educated; treatment fidelity was discussed; barriers or facilitators were addressed; progress was evaluated; novel goals were identified; and the exercise program was modified accordingly.
Patients in both study arms received usual mobility exercise therapy following guidelines from the Royal Dutch Society for Physical Therapy. 17 Multidisciplinary teams and supporting staff were instructed that content, frequency, and duration of regular therapies should be scheduled as usual in both arms. Patients in the control group did not have access to the Armed4Stroke module on the telerehabilitation platform, and no explicit attention was given to caregiver involvement. All participants kept an 8-week diary to monitor compliance, fall incidents and estimate treatment contrast. Participants reported daily mobility-related exercise minutes with an informal caregiver, with a nurse, in therapy and self-directed exercise time. In addition, telerehabilitation use and fall incidents were recorded weekly.
Outcomes were assessed by a blinded observer at baseline (≤3 months poststroke), 8 weeks (postintervention) and 6 months follow-up. The primary outcome was the self-reported mobility domain of the Stroke Impact Scale 3.0 after 8 weeks. The mobility domain consists of nine questions about perceived ability to maintain balance, make transfers, walk and climb stairs. Questions are rated on a 5-point Likert scale, and a domain score (0–100) is calculated. The Stroke Impact Scale has excellent psychometric properties. 18 Secondary outcome measures were described in our study protocol, including references. 13 Secondary outcomes to establish effects on patients’ functional outcome were the other domains of the Stroke Impact Scale, Rivermead Mobility Index, Functional Ambulation Category, 6 min walking distance, 5 m comfortable walking speed, Motricity Index Leg Score, Berg Balance Scale, Nottingham Extended ADL Scale, Community Ambulation Questionnaire and modified Rankin Scale at 8 weeks and 6 months follow-up; and the mobility domain of the Stroke Impact Scale at 6 months. Differential effects in dyads’ psychosocial wellbeing were evaluated at 8 weeks and 6 months follow-up using the Stroke Self-efficacy Questionnaire and EuroQol-5D for patients, modified Caregiver Strain Index, Care-related Quality of Life Instrument (seven dimensions and Visual Analogue Scale) for caregivers, General Self-efficacy Scale, Hospital Anxiety and Depression Scale, PROMIS Computer Adaptive Tests for anxiety and depression, Fatigue Severity Scale and the general functioning subscale of the McMaster Family Assessment Device for both patients and caregivers. Differential effects in the care transition were evaluated at 8 weeks and 6 months follow-up using the Transition Preparedness Scale for patients and Preparedness for Caregiving Scale for caregivers and using length of stay defined as the number of days between hospital admission and discharge home.
Statistical Analyses
Success of observer-blinding was tested by comparing actual with guessed allocation using Cohen's κ statistic. Baseline differences between groups were evaluated using Chi-squared tests, independent samples
Results
A total of 41 dyads were recruited (Figure 1: CONSORT flowchart) and randomised to the Armed4Stroke group (N = 21) or usual care alone (N = 20). Low agreement was observed between blinded guesses from the assessor and actual treatment allocation (κ=0.219,
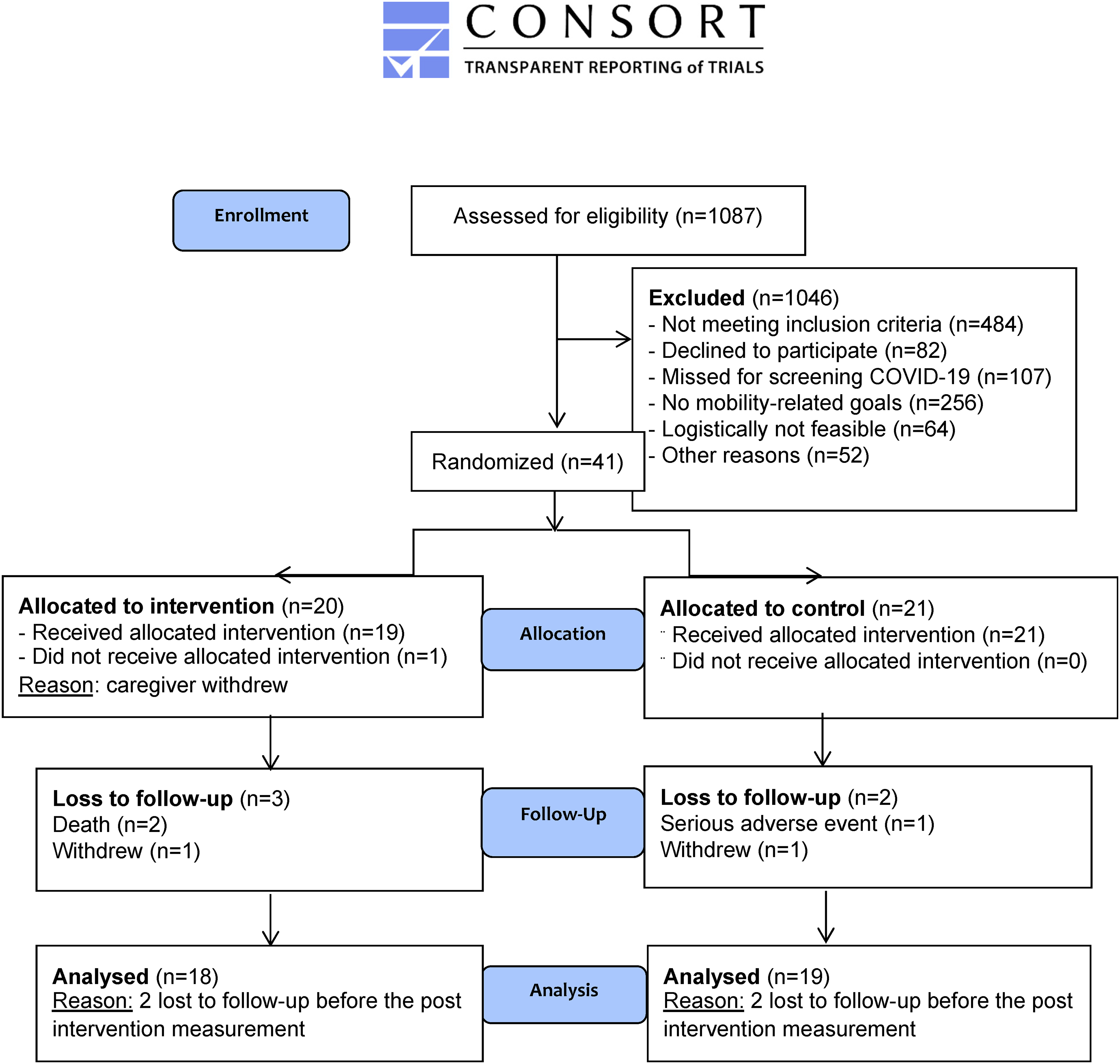
CONSORT 2010 flow diagram.
Total self-reported exercise minutes during 8 weeks.

IQR = interquartile range. *
Baseline characteristics are presented in Table 2. Patients had an average age of 62 years (SD 10.2), while the caregivers’ mean age was 59 years (SD 11.5). Approximately 56% of our patients were male, and almost 15% of them were not born in the Netherlands. Most patients (90.2%) were recruited during inpatient rehabilitation, participated with their partner (95.1%) and were on average 56 days (SD 16.6) poststroke. A significant between-group difference was observed in stroke classification (
Baseline characteristics.
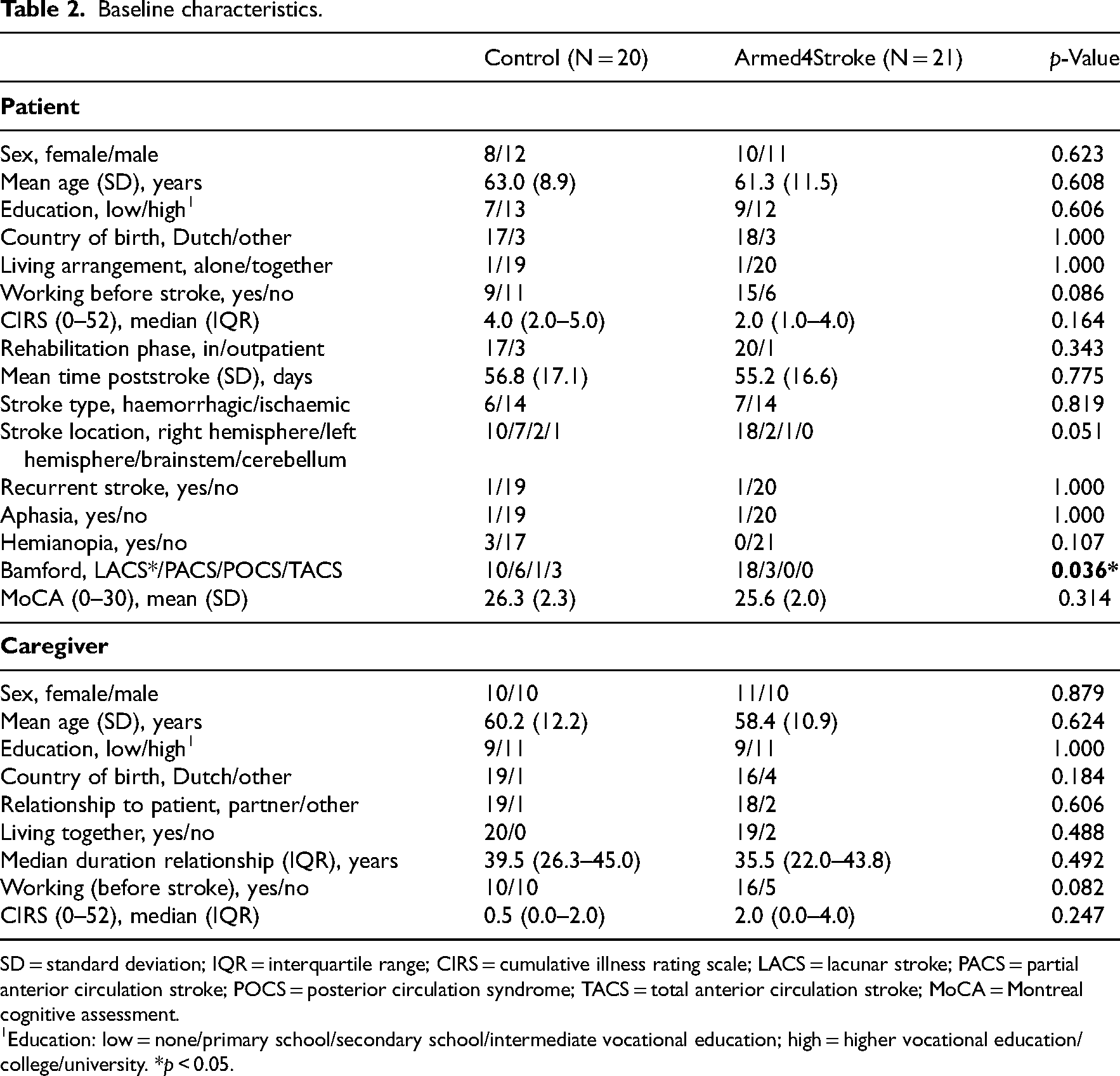
SD = standard deviation; IQR = interquartile range; CIRS = cumulative illness rating scale; LACS = lacunar stroke; PACS = partial anterior circulation stroke; POCS = posterior circulation syndrome; TACS = total anterior circulation stroke; MoCA = Montreal cognitive assessment.
Education: low = none/primary school/secondary school/intermediate vocational education; high = higher vocational education/college/university. *
The absolute mean values (SD) of our primary and secondary outcomes at different time points and between-group results of the linear mixed model and generalised estimating equations analyses corrected for baseline score are presented in Table 3. No significant differences were observed between groups for the primary outcome measure, Stroke Impact Scale mobility, after 8 weeks (B 0.8, 95% CI −6.8–8.5,
Results of the intention-to-treat analyses corrected for the baseline score.
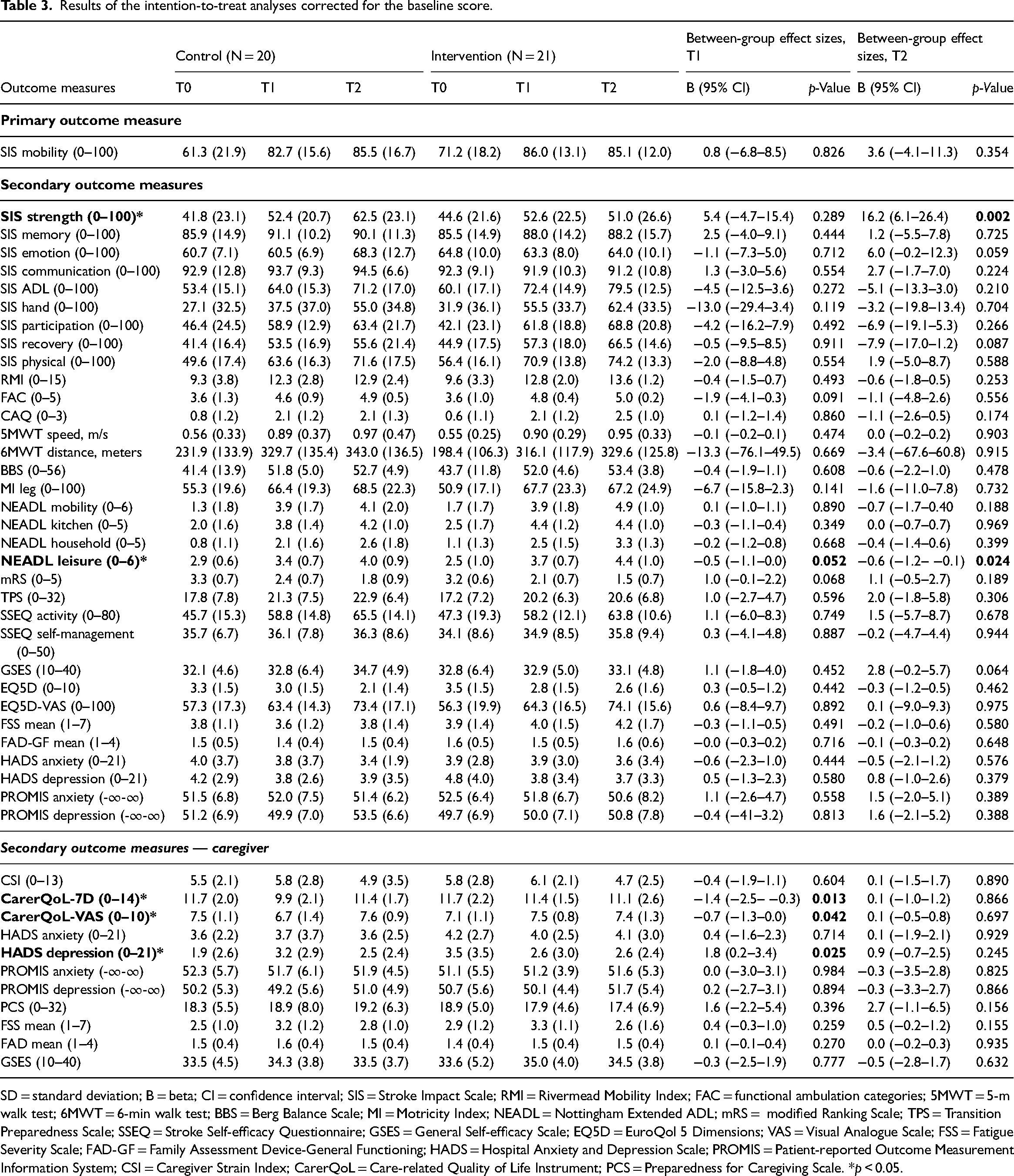
SD = standard deviation; B = beta; CI = confidence interval; SIS = Stroke Impact Scale; RMI = Rivermead Mobility Index; FAC = functional ambulation categories; 5MWT = 5-m walk test; 6MWT = 6-min walk test; BBS = Berg Balance Scale; MI = Motricity Index; NEADL = Nottingham Extended ADL; mRS = modified Ranking Scale; TPS = Transition Preparedness Scale; SSEQ = Stroke Self-efficacy Questionnaire; GSES = General Self-efficacy Scale; EQ5D = EuroQol 5 Dimensions; VAS = Visual Analogue Scale; FSS = Fatigue Severity Scale; FAD-GF = Family Assessment Device-General Functioning; HADS = Hospital Anxiety and Depression Scale; PROMIS = Patient-reported Outcome Measurement Information System; CSI = Caregiver Strain Index; CarerQoL = Care-related Quality of Life Instrument; PCS = Preparedness for Caregiving Scale. *
After 8 weeks, no significant differential effects were observed in patients’ functional outcome or psychosocial wellbeing. We did observe significant beneficial effects in caregivers’ psychosocial wellbeing, following the Care-related Quality of Life Instrument (seven-dimension score
Discussion
The main finding of this multicentre, randomised controlled trial is that our intervention yielded no significant differential effect on the primary outcome, patients’ self-reported mobility following the Stroke Impact Scale after 8 weeks, when compared to usual care alone. Within the subgroup of patients recruited during inpatient care, we found no clear benefits with respect to the care transition to the community. We did observe an added value on patients’ independence in leisurely activities after 6 months and caregivers’ psychosocial wellbeing at 8 weeks in line with previous trials.20–22
Caregiver-mediated exercises combined with telerehabilitation in addition to usual care did not result in an improvement of patients’ self-reported mobility after 8 weeks when compared with usual care nor after 6 months follow-up. Despite the lower number of subjects recruited than foreseen in the study protocol, we found no trend in the futility analysis. The neutrality of findings is supported by the absence of beneficial effects on secondary functional outcome measures, with the exception of independence in leisurely activities at 6 months. These findings are in line with several other caregiver-mediated exercise studies poststroke,20,21,23 but runs contrary to another small sampled trial by Galvin et al. 24 The effect estimates reported by Galvin et al., however, were not adjusted for baseline imbalances and may be overly optimistic. Our hypotheses were based on beneficial effects of augmenting task-specific training. Although it should be acknowledged that estimations of treatment contrast were based on self-report and not on the actual number of repetitions of task-specific training, the exercise time did not differ significantly between groups, which may explain the neutral results. Cluster randomisation could prevent issues of contamination in future studies. In addition, informal caregivers may already provide training in usual care. To achieve sufficient contrast, a higher dose may be required, or our protocol may be complemented with other resource-efficient strategies, such as self-directed, 25 circuit-class 26 or peer-to-peer training. Finally, future trials should consider sensors to objectively measure activity and record both scheduled and active therapy time.
We additionally hypothesised that active involvement of caregivers and providing blended care support would empower dyads and improve psychosocial wellbeing as well as the care transition to the community. 13 We found no beneficial effects on patients’ psychosocial wellbeing; however, our study yielded positive differential effects in caregivers’ wellbeing, namely, improved quality of life and reduced depressive symptoms after 8 weeks. Despite the multiple-comparison problem for these secondary outcomes, the results are in line with previous caregiver-mediated exercises trials poststroke.20,21 Moreover, the majority (70%) of studies investigating mobility-directed, caregiver-mediated exercises reported improved caregiver outcomes in terms of psychological wellbeing or reduced burden. 27 Improved psychosocial wellbeing of caregivers may be linked to their active involvement in rehabilitation care, resulting in more insight and knowledge, improved skills to support the patient, feelings of empowerment and reduced uncertainty. Future caregiver-mediated exercise studies poststroke should explore more in-depth how caregivers experience their involvement in rehabilitation care before proceeding to a larger trial to validate these findings.
Our final secondary objective was evaluated within a subgroup of patients recruited from inpatient care. We hypothesised that active caregiver involvement and provision of blended care support would smooth the care transition to the community by improving preparedness for discharge and reducing length of stay. 13 We found no evidence to support our hypotheses with no significant differences in dyads’ preparedness for discharge. We did observe an important non-significant difference of 1 week in length of stay favouring the Armed4Stroke group, which could potentially reduce costs of care. The lack of significant effects may be explained by variation in length of stay between settings and patients corresponding to the relatively high standard deviations observed. Although these findings should be interpreted with caution, we recommend that future trials are sufficiently powered and include an economic evaluation to explore this further.
This study reports findings from a multicentre, randomised controlled trial with a rigorous study design, and the selection of outcome measures follows international consensus statements.
28
However, several study limitations should be acknowledged. First, this study is underpowered as a result of COVID-19 restrictions. We observed some positive trends on secondary outcome measures, namely, the functional ambulation categories, modified Ranking Scale and Nottingham Extended ADL leisure scale after 8 weeks. At 6 months, a trend was observed towards better perceived recovery and patients’ self-efficacy. However, these trends show considerable variability and should be interpreted with caution. Second, our primary outcome measure may suffer from a ceiling effect and may be insufficiently sensitive to quantify change in mild stroke patients.
29
Future mobility-directed studies should consider 6 min walking distance or 10 m comfortable walking speed measurement as a primary outcome measure. Third, a significantly higher proportion of lacunar strokes was seen in the intervention group. These patients may have had a slight prognostic advantage, but may also have suffered more from a ceiling effect. Fourth, patients were recruited at arbitrary time points poststroke, which could have resulted in a type II error. Fifth, our patients were relatively young and mostly recruited from inpatient clinics, limiting generalizability to other settings. However, diversity in ethnic background and gender in this sample supports the external validity. Finally, further investigation is required to study the long-term effects of our program.
The Armed4Stroke program did not result in improved self-reported mobility. Involving caregivers in task-specific training did not lead to a higher perceived burden compared to usual care. Involvement of caregivers in rehabilitation may improve caregivers’ psychosocial wellbeing.
Footnotes
Acknowledgements
We would like to thank all patients, informal caregivers and staff involved in recruitment and treatment in the participating centres. Thank you for dedicating your time and efforts and for staying motivated during the pandemic, a particularly difficult time to run a trial, treat patients, involve caregivers and experience the consequences of stroke. We would specifically like to thank Esther Kret, Judith Vloothuis, Frank Berenpas, Lianne de Langen, Peter Elich, Judith Fleuren, René Brunsveld, Nancy Brookhuis, Michelle Bernhardi, Nanny Steinmeijer, Nelleke Kooiman and Josien Kosse for their valuable input, energy and motivation. In addition, we would like to thank Birgit Lissenberg-Witte from the epidemiology and data science department of the Amsterdam UMC for her statistical support during our unplanned interim analysis. Finally, we would like to thank our interns Evi Dekker and Jet Kruithof who provided invaluable support in recruiting our final patients and preparing our dataset for analyses.
Author Contributions
All authors conceived the study and were involved in protocol development. GK was penwriter, applied and received funding and RN gained ethical approval. MM and CN were involved in patient recruitment. MM analysed the data and wrote the first draft of the manuscript. CN, EP, RN, JB, GK and EW made a substantial contribution to the analysis and interpretation of data. All authors reviewed and edited the manuscript and approved the final version.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Armed4Stroke project (grant number: 858001102) funded by the Royal Dutch Society for Physiotherapy Scientific College Physiotherapy (KNGF-WCF)/Netherlands Organisation for Health Research and Development (ZonMw). WCF and ZonMw had no role in the design of the study, collection and analysis of data, or in the preparation of the current manuscript.
