Abstract
Objective
To analyse the components used in exercise interventions for people with symptoms of neurogenic claudication due to lumbar spinal stenosis and identify components associated with successful interventions.
Data sources
Eligible papers published up to April 2023 from MEDLINE, EMBASE, CINAHL, PEDro, CENTRAL, Web of Science, and trial registry websites.
Review methods
Literature searches were performed by an Information Specialist. We searched for randomised trials evaluating exercise interventions for people with neurogenic claudication symptoms (the primary symptom of lumbar spinal stenosis). Two authors independently performed study selection, data extraction, and quality assessments using the Cochrane Risk of Bias tool Version 2 and the TIDieR checklist for intervention reporting. Details of intervention components were extracted, tabulated, and synthesised using an intervention component analysis approach.
Results
We found thirteen trials reporting 23 exercise interventions delivered to 1440 participants. These featured 60 different components. Most exercise interventions included supervision and flexion-based exercises. Balance exercises were rarely included. Exercise components featured more frequently in successful interventions included stretches, strength or trunk muscle exercises, fitness exercises, especially cycling, and psychologically informed approaches. Interpretation is limited by low study numbers and heterogeneity. No conclusions could be drawn about exercise supervision or dose.
Discussion
Exercise interventions for people with neurogenic claudication typically feature multiple components. Common features such as supervision, lumbar flexion, and aerobic fitness exercises and also less common features such as stretches, strengthening exercises, and psychologically informed approaches warrant consideration for inclusion when designing and optimising exercise interventions for people with lumbar spinal stenosis.
Introduction
Lumbar spinal stenosis is a common spinal condition in older adults.1,2 The cluster of symptoms typically arising from lumbar spinal stenosis is referred to as neurogenic claudication and includes pain, aches, heaviness, and tingling radiating from the spine into the buttocks and legs, made worse by standing and walking and relieved by sitting.
Controversy around the benefits, costs and risks of surgical treatment3,4 has led to a growing interest in non-surgical care including exercise to help people with lumbar spinal stenosis and neurogenic claudication.5,6 Exercise is commonly prescribed 7 based on the theoretical benefits of increased spinal flexibility, enhanced blood flow to spinal nerves, improvements in physical fitness, strength and balance, and benefits for mental well-being, confidence and self-efficacy.8–11 A number of recent systematic reviews12–15 have synthesised data from clinical trials and concluded that exercise as part of multimodal care improves some outcomes. However, these trials delivered exercise interventions with notably different characteristics and comparators. Conclusions drawn from such heterogeneous data may have limited use for clinicians and policymakers wanting to understand how individual or combined exercise intervention components might impact patient outcomes. Furthermore, there is recognition that decision-makers are interested in research evidence to explain how complex exercise interventions work in different contexts and to support real-world decision-making. 16
The concept for this systematic review was shaped by the views of patients with lumbar spinal stenosis and neurogenic claudication and by the views of clinicians. Both have highlighted inconsistent guidance and lack of detail on specific components to guide exercise prescription. The aims of this review detailed in our protocol were to estimate exercise treatment effectiveness and to explore the key components of these exercise treatments. The first point has been addressed by others in systematic reviews and meta-analyses published recently.13–15 We, therefore, undertook this review to synthesise the evidence relating to specific components within exercise interventions for lumbar spinal stenosis and neurogenic claudication. For the purposes of the review, we defined an exercise intervention as, ‘A regular, structured and/or supervised exercise regimen’. This includes individual or group exercises, and supervised exercises or home exercises if following a structured programme, or a mixture.
Objectives
The overall objectives are
to narratively synthesise the existing evidence on exercise intervention components from randomised controlled trials that aim to improve outcomes of symptom severity, disability and walking in people with lumbar spinal stenosis and neurogenic claudication; to tabulate the various exercise intervention components of the included trials; to explore which exercise intervention components may be associated with a successful outcome (i.e. a significant difference in favour of exercise intervention) or unsuccessful outcome (i.e. no significant between-group difference, or significant difference in favour of non-exercise intervention).
Methods
The review protocol was registered with PROSPERO (2019 CRD42019137266). A systematic search was conducted to synthesise data from randomised controlled trials published between 1996 and May 2023. We used the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) 2020 checklist 17 to report the findings.
The eligibility criteria for inclusion of trials are presented in Table 1. Our protocol stipulated an age range of 65 years or older, which we amended after noting several relevant studies involving participants with lumbar spinal stenosis symptoms aged 50 upwards from a preliminary review of the literature.
Eligibility criteria for inclusion of studies.
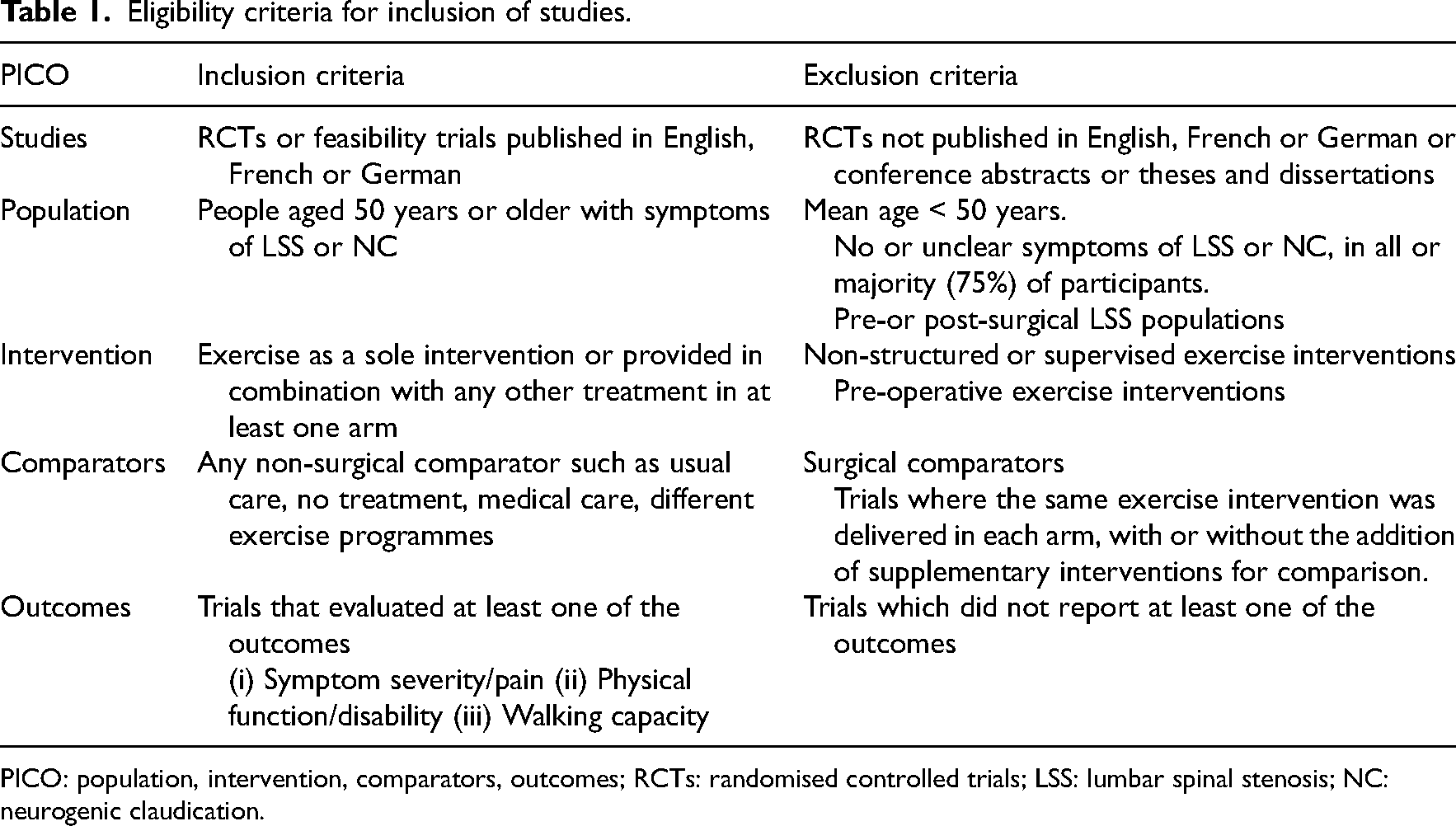
PICO: population, intervention, comparators, outcomes; RCTs: randomised controlled trials; LSS: lumbar spinal stenosis; NC: neurogenic claudication.
Based on previous systematic reviews,18–20 a robust search strategy was developed by an information specialist in health sciences (Supplemental Table S1). Electronic searches for eligible studies were conducted within the databases, including the ClinicalTrials.gov and ISRCTN trial registry websites. For all included studies, we also undertook backward and forward reference searching by hand.
After the screening of titles and abstracts by pairs of authors (CC, EW, or CC, SM) and the exclusion of duplicates, full texts were retrieved and were independently assessed by two authors (as above). Any discrepancies in selecting the studies were resolved through discussion.
For each included trial, we used the Cochrane Risk of Bias tool version 2 21 to assess the potential for bias across five domains (randomisation, intervention assignment, missing data, outcome measurement, and reporting). We used signalling questions from the Cochrane template to assess the domain-level judgements about the risk of bias and then reached an overall judgement about the risk of bias for each trial. The risk of bias was judged as ‘low’, with ‘some concerns’ and ‘high’. We also assessed the quality of the reporting of exercise interventions using the TIDieR checklist (0–24 score, higher scores mean higher quality reporting).22,23 Scoring was completed by one author (CC) and checked by a second author (EW/SM). Any discrepancies in scoring were resolved through discussion.
A bespoke data extraction form was piloted for five trials (CC/EW) and was then used to extract relevant data from all included trials (CC). Extracted data was verified by a second author (EW/SM). Data such as trial characteristics, study participants, interventions, follow-up periods, outcome measures, and main results were extracted. We also used the domain headings of ‘Why’, ‘What procedures’, ‘What materials’, ‘How’, ‘When and how much’, ‘Tailoring’, and ‘What materials’ from the TIDieR exercise intervention reporting checklist to capture all relevant exercise intervention components.
We used a modified two-step intervention component analysis approach 24 to provide a narrative description of the exercise interventions reported in the trials.
The first step is to develop an intervention component analysis table by tabulating the different components of the exercise interventions and identifying interventions that were successful and unsuccessful.
The second step is to identify and report the exercise intervention components that appear to be important in influencing successful and unsuccessful treatment effects.
Step 1: Tabulation of exercise intervention components
We coded the exercise intervention component data from the included trials to produce an intervention component analysis table. This involved tabulating the information on trial interventions and items of the TIDieR checklist (such as equipment used, and who delivered the interventions), and effects on study outcomes from each included trial, row-wise.
To inform our intervention component coding, we defined ‘supervised exercise’ as exercise programmes undertaken under the guidance of a clinician in an individual or a group setting. We defined ‘structured home exercise’ as exercises performed at home supported by regular review appointments in the clinic.
We categorised water-based exercises and harness-supported walking exercises as ‘spinal unloaded exercises’. Where exercise was delivered as part of a multimodal intervention, the supplemental components were extracted and coded. Any physical or electrical treatments (heat, cold, or electrotherapy) were coded as ‘passive modalities’, and any ‘hands-on’ manipulation or mobilisation techniques were coded as ‘manual therapy’. Exercise programmes underpinned by a psychologically informed/cognitive behavioural approach were coded as ‘psychologically-informed approach’.
We also coded the control interventions of the randomised controlled trials. Epidural spinal injection and/or oral medication were coded as ‘Medical care’; self-management of symptoms as ‘Self-care’ and waiting list/no treatment as ‘No care’.
With various outcome measures used across the included trials, we prioritised outcome reporting as below:
For symptom severity/pain, we prioritised reporting the Zurich Claudication Questionnaire-Symptom Severity score. When not available, we used a visual analogue scale or numerical pain rating scale For physical function/disability, we prioritised reporting the Zurich Claudication Questionnaire-Physical Function scores where available, followed by the Oswestry Disability Index and then other physical function or disability scores For walking capacity, we prioritised objective measures over self-reports.
For follow-up assessments, we defined short-term follow-up as 3 months or less, and medium-to-long-term as any follow-up longer than this. Where multiple follow-ups were reported, we selected the closest available data to 3 and 12 months, respectively.
We extracted treatment effect estimates directly from published tables or text when available. We contacted authors when possible for additional data as required, and we estimated effect sizes (using Review Manager (RevMan), Version 5.4,), from pre- and post-treatment outcome data when between-group differences were not available.
The intervention components were then mapped against intervention success that was coded according to whether or not an intervention was associated with a successful short-term or medium-to-long-term outcome or with an unsuccessful outcome. Exercise interventions with a statistically significant difference compared to a comparator treatment for any of our selected outcomes were coded as ‘successful’ and those with no statistically significant between-group differences, or a statistically significant difference in favour of a non-exercise comparator were coded as ‘unsuccessful’.
Step 2: Interrogation of the exercise intervention components
We mapped the number of successful exercise interventions featuring a component (guided by the domains of the TIDieR checklist) as a proportion of the total number of interventions featuring that component. Common intervention components that were featured in 75% or more of the exercise interventions were highlighted.
We then explored any links between outcomes and the components that featured more commonly in successful and unsuccessful interventions. These relationships were considered in the context of the number and quality of the included trials and differences in trial design/methods, interventions and comparators.
Results
A total of 10,353 records were identified from electronic searches and a further 11 records from hand searches. Of 78 publications shortlisted for full review, 13 trials (14 published reports) were included (Figure 1). Reasons for excluding shortlisted trials are provided in Supplemental Table S2.
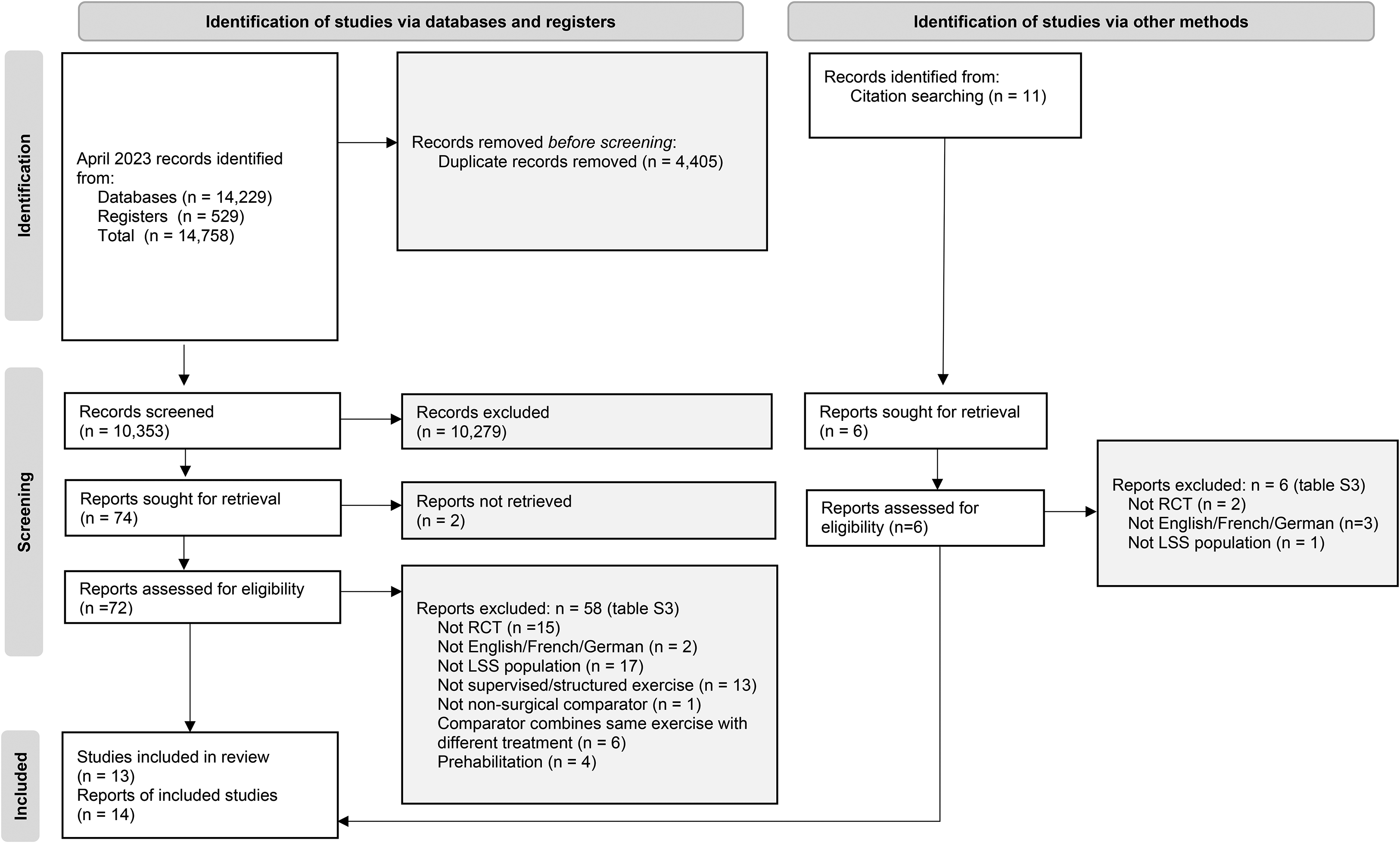
Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) flow diagram.
Participant characteristics, interventions and comparators, follow-up time points, risk of bias, and intervention reporting quality of the included trials are presented in Table 2. Ten trials25–35 had two treatment arms, and the remainder had three arms.36–38 The included trials involved a total of 1440 participants with a mean age of 65 years (standard deviation 9), of whom 985 were allocated to a supervised or structured home exercise intervention. More than 50% of the trial participants were female (802/1440, 56%). Gender-related data was not available in one trial. 36 The included trials were conducted in various countries across the continents of Europe/United Kingdom (5), Asia (4), and America (4). All trials were published within the last 17 years, with 10/13 trials published in the last 10 years. The sample size ranged from 24 to 435. Only four trials had a total sample size of above 100. In 9/13 trials, two or more groups received supervised or structured home exercise interventions, resulting in 23 exercise intervention arms overall.
Summary of included trials: participant characteristics, treatment category comparisons, follow-up time points, risk of bias, and intervention reporting quality.

NC: neurogenic claudication, LSS: lumbar spinal stenosis; Ex: exercise; MT: manual therapy; PM: passive modalities; EA: education and advice; Med: medical care; ESI: epidural steroid injection; HE: supplementary home exercises; PI: psychologically informed approach; SS: symptom severity; PF: physical function; WC: walking capacity; ST: short-term; MLT: medium- to long-term; SS: symptom severity; PF: physical function; WC: walking capacity; NPRS: numeric pain rating score; ODI: Oswestry Disability Index; VAS: visual analogue scale; ZCQ-SS: Zurich Claudication Questionnaire symptom severity scale; ZCQ-PF: Zurich claudication questionnaire physical function scale; SWT: shuttle walk test; JOA: Japanese Orthopaedic Association; BPI: Brief Pain Inventory.
Konno et al. 39
Cochrane Risk of Bias tool composite scores: categories low risk, medium risk, and high risk of bias.
TIDieR checklist for exercise intervention reporting: score 0–24, higher scores mean higher quality reporting.
Components that featured in ≥ 75% exercise interventions of the included trials were supervised exercises (18/23), land-based exercises (18/23), some form of lumbar lordosis reducing/ flexion-based exercises (17/23), and some form of aerobic fitness exercises (walking, cycling, or general fitness, 18/23).
Around 50% of the exercise interventions were reported to be developed based on theoretical rationale (14/23); and involved sessions that lasted between 30 and 60 minutes (14/23); and were delivered in a hospital/clinical setting (13/23) by physiotherapists (12/23). Other personnel such as chiropractors, physiotherapy assistants, exercise/fitness instructors, or physicians were less commonly involved.
The majority of exercise interventions were performed at least twice a week (21/23). Around half lasted a period of 6 weeks (12/23) and the majority lasted between 3 and 11 weeks (18/23).
Supervised exercises were delivered once to twice weekly and home exercises prescribed daily or twice daily. With home exercises, the frequency of contact with clinicians varied widely, with one study 27 reporting a mean of three participant-clinician contacts, and another study 29 scheduling up to 13 contacts for review of exercises and delivery of adjunctive manual therapy.
Over half of the exercise interventions (15/23) were ‘multi-modal’ in nature, combined with manual therapy, passive modalities, medical treatments, education, or psychologically informed approaches. Studies published in the last 5 years29–35,37 were less likely to include passive modalities and more likely to include a psychologically informed approach.
Risk of bias assessments
The domain-level judgements about the risk of bias are as follows: The risk of bias arising from the randomisation process was low in the majority of the trials (10/13). Only five trials were judged to be of low risk for intervention assignment. Four were of high risk and the other four trials had some concerns. Six trials were at high risk of missing outcome data. Three trials had some concerns while only four trials were of low risk. In measuring outcomes, the majority of trials (10/13) were at low risk of bias. Three trials had some concerns. Eight trials were at low risk of bias in reporting the results while five trials had some concerns.
Overall, only three trials were judged to be at low risk of bias (Table 2). Seven trials were judged to present some concerns in at least one domain, while three trials were at higher risk of bias with some concerns in more than one domain.
Reporting of exercise intervention elements
The quality of intervention reporting varied from a lowest score of 5/24 to a highest of 21/24 on the TIDieR checklist (Table 2). All studies were rated as 0/2 for reporting exercise intervention modifications. Reporting of intervention procedures, and delivery frequency, duration, location, personnel and equipment were variable, with the majority (10/13 to 13/13) reporting these elements partially (1/2) or fully (2/2). Only 1/13 trials was rated 2/2 for full reporting of exercise tailoring and only 2/13 were rated 2/2 for full reporting of assessment of treatment fidelity. In contrast, all trials scored 2/2 for reporting a brief title/ description of the exercise intervention and 9/13 scored 2/2 for reporting a rationale/theory/goal underpinning the intervention.
Results of syntheses
Step 1: Tabulation of exercise intervention components
Details of individual exercise interventions are presented in the intervention component analysis (Supplemental Table S3). Of the 23 exercise interventions, 20 were supervised programmes; 13 were multimodal (five combining exercise with passive modalities; two with manual therapy; two with both; two with manual therapy plus education/advice, one with education/advice plus a psychologically-informed approach, and one with manual therapy plus education/advice plus a psychologically informed approach). Over half of the supervised programmes25,26,28,31–33,35–37 were also supplemented with self-directed home exercises.
Comparator interventions were varied: Four studies used comparators of no care or self-directed care. Two studies used medical care that included epidural steroid injection, one of which compared medical care combined with exercise to medical care alone. The remaining comparators consisted of different structured or supervised exercise interventions with or without supplemental interventions.
From the 13 trials, eight exercise interventions from seven trials29,30,32,33,35–38 reported in favour of exercise for one or more short-term or medium-to-long-term outcome and were coded as ‘successful’.
Fifteen interventions from 10 of the trials25–28,30–34,36,37 did not report in favour of exercise for our predefined short-term or medium- to long-term outcomes and were therefore coded as ‘unsuccessful’. Of these, one trial 31 that combined exercise with medical treatment reported findings in favour of the control intervention of medical treatment without exercise.
In the eight successful exercise interventions, stretches featured in 7/8 compared to 5/15 in unsuccessful interventions. Strength and/ or trunk muscle exercises featured in 7/8 successful interventions, compared to 8/15 of unsuccessful interventions. Fitness exercises including general fitness, walking or cycling exercises featured in 7/8 successful compared to 10/15 unsuccessful interventions with cycling featuring in 5/8 successful interventions compared to just 2/15 unsuccessful interventions. In addition, exercises performed in flexed positions (cycling or spinal unloading) featured in 5/8 successful interventions compared to 5/15 of the unsuccessful interventions. Lumbar flexion exercises featured in a similar proportion of successful (5/8) and unsuccessful (11/15) interventions. Supplementary treatment components of manual therapy, passive modalities, and supplementary home exercises featured in similar proportions of successful and unsuccessful interventions. Psychologically informed approaches featured in (2/8) successful interventions but in none of the unsuccessful interventions; advice and education featured in 3/8 successful interventions compared to 1/15 unsuccessful interventions; and medical care featured more commonly in unsuccessful interventions (5/15) compared to successful interventions (1/8).
The treatment effects of various exercise intervention components varied across the three outcomes of symptom severity, physical function, and walking capacity. Effects on these individual outcomes are tabulated in Supplemental Tables S4 and S5, respectively.
Step 2: Interrogation of exercise intervention components
The proportion of exercise interventions featuring a specific component that was classed ‘successful’ was greatest for interventions drawn on patient/public opinion during development (2/2); using a personalised approach (3/4); duration of 12-week period (1/1); exercise combined with a psychologically informed approach (2/2); inclusion of strength exercises (4/7); cycling (5/7); use of a static bicycle (5/8); and delivered by chiropractors (2/2).
When considering each of the three outcomes of interest individually, intervention components more likely to feature in interventions reporting improved symptom severity were cycling exercises (4/7 for short-term success); intervention components more likely to report improved walking capacity were cycling exercises (4/7 for short-term success), and a psychologically-informed approach (2/2 for longer-term success). No components were identified with improvements in physical function.
The proportion of exercise interventions classed as unsuccessful that featured a specific component was greatest for self-directed care (3/4); home- or community-based exercise programmes (3/4); exercise programmes delivered by physiotherapy assistants (2/2) or exercise instructors (2/2); exercise programmes supplemented by medical care (5/6), use of traction machines (2/2), electrotherapy equipment (7/7) or hot packs (3/4); and exercise programmes supported by adherence strategies such as exercise logs/pedometers/check-in appointments/telephone calls (6/7).
Discussion
We identified 13 randomised controlled trials that provide evidence about exercise interventions for people with lumbar spinal stenosis and neurogenic claudication. We systematically explored the exercise intervention components that were commonly used and those that featured more frequently in trials with successful outcomes.
The intervention component analysis approach allowed us to highlight commonly used components that might warrant further attention in exercise interventions for this population. These include stretches, muscle strength and control exercises (general and core), and aerobic fitness exercises especially if performed in spinal flexion such as cycling. It also allowed us to identify potentially important components that are infrequently prioritised such as balance exercises. In contrast, traditional systematic reviews offer limited detail to guide clinicians and patients affected by lumbar spinal stenosis. Indeed, inconsistent terminology referring to exercise-based interventions (including terms such as rehabilitation, multimodal care and supervised-, directed-, home- and self-directed exercise), and questionable approaches to synthesising effectiveness data have exacerbated our understanding of the fragmented evidence concerning effective exercise components.
Four recent randomised controlled trials could be considered key sources contributing to the body of evidence in this field.29,32,33,35,37 Despite notable differences in interventions and comparators, systematic reviews have pooled data from all but the most recent of these trials. One recent systematic review 14 concluded that manual therapy with supervised exercise improves short-term walking capacity, and provides small improvements in pain and symptom severity compared with self-directed or group exercise. Data from one of the key trials 37 was, however, omitted without explanation, and data from exercise-based interventions were combined with data from other non-surgical interventions such as acupuncture under the heading of ‘rehabilitation’. Two other systematic reviews used pooled headings of ‘directed exercise + manual therapy versus self-directed or group exercise’, 12 and ‘combined therapies versus home/group exercises’. 13 These headings were used to encompass exercise interventions delivered in different formats (supervised or unsupervised, group or individual) and combined with different supplementary treatments (manual therapy, education, psychologically informed approach). Furthermore, these trials used very different comparator interventions (medical care, self-directed exercise, or structured home exercise with regular contact with a clinician). Because of such intervention heterogeneity, a recent Cochrane review 15 chose not to meta-analyse the data. Instead, the authors undertook a narrative review of recent trials,29,32,37 updating findings from a previous review to conclude that there is moderate-quality evidence for a multimodal approach that includes manual therapy and exercise, with or without education.
The fourth key trial that we identified 40 has been published after these systematic reviews were undertaken and adds to the growing evidence of exercise benefits in people with lumbar spinal stenosis and neurogenic claudication. This is a high-quality randomised controlled trial evaluating the effectiveness of a personalised and supervised group exercise programme combined with a psychologically informed approach compared to best practice physiotherapy.
The majority of exercise interventions in the trials we reviewed relied on theoretical rationales proposed by earlier authors, and/or on evidence from other populations. These rationales include theory-driven recommendations of flexion-based/lordosis-reducing exercises and trunk muscle control exercises for relieving the posture-related symptoms of lumbar spinal stenosis and neurogenic claudication.8,41,42 However, little or no detail was provided to explain the selection of specific exercise intervention parameters and components.
The value of a psychologically informed approach is now recognised in the management of chronic back pain. 43 Whilst only two recent exercise interventions for people with lumbar spinal stenosis and neurogenic claudication incorporated a psychologically informed approach and education, both were classed as successful interventions, reporting sustained improvements in walking capacity. In a population that is known to have poor activity levels 44 and high levels of depression, 45 psychologically informed approaches may be another important factor in optimising exercise prescription.
The majority of exercise interventions in the included trials were supervised and performed at least twice weekly over a duration of at least 6 weeks. Whilst no conclusions could be drawn about the impact of exercise dose and supervision on intervention success, these parameters are consistent with the literature in other musculoskeletal conditions that suggests twice-weekly supervised strength and fitness exercises are most likely to provide a meaningful improvement in pain and function. 46
Strengths of this review include a comprehensive search of the literature on exercise interventions for lumbar spinal stenosis and neurogenic claudication and the use of an intervention component analysis approach. Instead of combining the data from heterogeneous trial interventions, we have undertaken a detailed exploration of components within different exercise interventions. We have used valid and recognised tools for evaluating the methodological quality and intervention reporting of included trials to provide additional context for our findings.
Our findings should be interpreted in the context of several considerations:
Firstly, readers should be aware that the key messages of this review are based on a limited number of available trials of varying methodological and reporting quality, with some inconsistency in findings, and where individual components sometimes feature in only one or two trials. By including evidence only from randomised controlled trials that fit clearly defined population, intervention, comparator and outcome reporting criteria, we sought to optimise the quality and relevance of data included in our synthesis, but less stringent inclusion criteria may have captured additional information. For transparency, a table of shortlisted trials that were excluded is presented (Supplemental Table S2). Because there are so few high-quality randomised controlled trials to draw on currently, it is likely that our interpretations of exercise intervention components may change as more evidence is obtained in the future.
Secondly, it is possible that trials published before 1996 may have been missed. However, hand searches of reference lists from the included trials and systematic reviews did not identify any additional relevant studies. Whilst several published protocols for potentially relevant ongoing or planned trials47–50 were identified through grey literature, we were only able to contact the authors of one of these 49 for which no results were available.
Finally, the coding of interventions as ‘successful’ or ‘unsuccessful’ relied on treatment effect data. This should be taken into consideration when interpreting our exploration of associations between intervention components and effectiveness.
Despite these limitations, our analyses provide some interesting insight into specific exercise intervention components that might show more promise than others, depending on the outcome of importance.
Further research to confirm effective exercise intervention components that target specific outcomes might be warranted to refine future exercise treatments for people with lumbar spinal stenosis and neurogenic claudication. To this end, any future exercise intervention trials should carefully plan, evaluate and report exercise component details.
Clinical messages
For patients with symptomatic lumbar spinal stenosis:
Common exercise components are supervision, lumbar flexion, and aerobic fitness exercises. Additional components that improve patient outcomes are stretches, strength/trunk muscle exercises, and cycling exercise for aerobic fitness.. Adding a psychologically informed approach might help. Balance exercises warrant more research.
Competing interests statement
CC, SL, EW, and CS are the authors of two studies included in this review.
Supplemental Material
sj-docx-1-cre-10.1177_02692155231201048 - Supplemental material for Exercise treatments for lumbar spinal stenosis: A systematic review and intervention component analysis of randomised controlled trials
Supplemental material, sj-docx-1-cre-10.1177_02692155231201048 for Exercise treatments for lumbar spinal stenosis: A systematic review and intervention component analysis of randomised controlled trials by Christine Comer, Esther Williamson, Suzanne McIlroy, Cynthia Srikesavan, Sally Dalton, GJ Melendez-Torres and Sarah E Lamb in Clinical Rehabilitation
Supplemental Material
sj-docx-2-cre-10.1177_02692155231201048 - Supplemental material for Exercise treatments for lumbar spinal stenosis: A systematic review and intervention component analysis of randomised controlled trials
Supplemental material, sj-docx-2-cre-10.1177_02692155231201048 for Exercise treatments for lumbar spinal stenosis: A systematic review and intervention component analysis of randomised controlled trials by Christine Comer, Esther Williamson, Suzanne McIlroy, Cynthia Srikesavan, Sally Dalton, GJ Melendez-Torres and Sarah E Lamb in Clinical Rehabilitation
Supplemental Material
sj-xlsx-3-cre-10.1177_02692155231201048 - Supplemental material for Exercise treatments for lumbar spinal stenosis: A systematic review and intervention component analysis of randomised controlled trials
Supplemental material, sj-xlsx-3-cre-10.1177_02692155231201048 for Exercise treatments for lumbar spinal stenosis: A systematic review and intervention component analysis of randomised controlled trials by Christine Comer, Esther Williamson, Suzanne McIlroy, Cynthia Srikesavan, Sally Dalton, GJ Melendez-Torres and Sarah E Lamb in Clinical Rehabilitation
Supplemental Material
sj-docx-4-cre-10.1177_02692155231201048 - Supplemental material for Exercise treatments for lumbar spinal stenosis: A systematic review and intervention component analysis of randomised controlled trials
Supplemental material, sj-docx-4-cre-10.1177_02692155231201048 for Exercise treatments for lumbar spinal stenosis: A systematic review and intervention component analysis of randomised controlled trials by Christine Comer, Esther Williamson, Suzanne McIlroy, Cynthia Srikesavan, Sally Dalton, GJ Melendez-Torres and Sarah E Lamb in Clinical Rehabilitation
Supplemental Material
sj-docx-5-cre-10.1177_02692155231201048 - Supplemental material for Exercise treatments for lumbar spinal stenosis: A systematic review and intervention component analysis of randomised controlled trials
Supplemental material, sj-docx-5-cre-10.1177_02692155231201048 for Exercise treatments for lumbar spinal stenosis: A systematic review and intervention component analysis of randomised controlled trials by Christine Comer, Esther Williamson, Suzanne McIlroy, Cynthia Srikesavan, Sally Dalton, GJ Melendez-Torres and Sarah E Lamb in Clinical Rehabilitation
Supplemental Material
sj-docx-6-cre-10.1177_02692155231201048 - Supplemental material for Exercise treatments for lumbar spinal stenosis: A systematic review and intervention component analysis of randomised controlled trials
Supplemental material, sj-docx-6-cre-10.1177_02692155231201048 for Exercise treatments for lumbar spinal stenosis: A systematic review and intervention component analysis of randomised controlled trials by Christine Comer, Esther Williamson, Suzanne McIlroy, Cynthia Srikesavan, Sally Dalton, GJ Melendez-Torres and Sarah E Lamb in Clinical Rehabilitation
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Christine Comer is funded by a National Institute (NIHR) Clinical Lectureship ICA-CL-2017-03-015) for this research project. This publication represents independent research. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
