Abstract
Objective
To estimate the cost-consequence of treating spasticity early with botulinum toxin in the acute stroke unit.
Design
Secondary cost-consequence analysis, using data from a double-blind randomised-controlled trial.
Setting
Single-centre specialised stroke unit.
Subjects and Interventions
Patients with Action Research Arm Test grasp-score of <2 and who developed spasticity within six weeks of a first stroke were randomised to receive injections of: 0.9% sodium-chloride solution (placebo) or onabotulinumtoxin-A (treatment).
Main measures
Resource use costs were calculated for the study. Mean contracture costs for each group were calculated. The Barthel Index and Action Research Arm Test were used to generate a cost per unit of improvement.
Results
There were no significant differences associated with early treatment use. The mean contracture cost for the treatment group was £817 and for the control group was £2298 (mean difference = −£1481.1(95% CI −£2893.5, −£68.7) (
Conclusions
Treating spasticity early in stroke patients at risk of contractures with botulinum toxin leads to a significant reduction in contracture costs. The cost per improvement of Barthel and Action Research Arm Test indicates that the intervention costs less and is more effective.
Trial Registration data
EudraCT(2010-021257-39) and ClinicalTrials.gov-Identifier:NCT01882556.
Introduction
The cost of managing stroke places a significant burden on the individual, families and the economy. 1 These costs significantly increase in stroke patients who develop complications such as spasticity, contractures, pain and pressure sores.2, 3 There is a growing body of evidence that demonstrates that spasticity occurs early following stroke. 4 In patients who do not recover useful arm function, this is likely to lead to contractures, pressure sores and pain. 4
Botulinum toxin is one treatment that can reduce spasticity 5 but it is not routinely offered to acute stroke patients. A possible reason for this could be the perception that this drug is expensive. 6 Apart from one cost-effectiveness study from the results of a randomised-controlled trial, 6 economic studies have either used retrospective cohort analysis 2 or economic modelling using expert opinion.7, 8
We have previously demonstrated that screening and treating spasticity early, on first presentation after the first stroke in patients who have no useful arm function, with botulinum toxin prevents contractures, reduces pain, and does not interfere with functional recovery (EUBoSS Trial). 5 This current paper uses results from this randomised-controlled trial to investigate the cost-consequences of using botulinum toxin early in the management of spasticity following stroke.
Using data from the previous trial we aim to
Identify the major cost drivers in the management of patients with post-stroke spasticity and contractures. Conduct a cost-consequences analysis of botulinum toxin in the management of patients with post-stroke spasticity and contractures.
Methods
This study reports on the cost-consequences of the early use of botulinum toxin in post-stroke spasticity. The trial was approved by North West – Greater Manchester South Ethics Committee Reference number 10/H1003/111. It was registered with EudraCT (2010-021257-39) and at ClinicalTrials.gov-Identifier: NCT01882556.
The protocol 9 and the results related to effectiveness have previously been published. 5 In brief, in this double-blind placebo-controlled study, patients with an Action Research Arm Test grasp-score ⩽2 and who developed spasticity within six weeks of a first stroke were randomised to receive injections of either 0.9% sodium-chloride solution (placebo) or onabotulinumtoxin-A (treatment). In addition to a range of measures of impairment and hand function, the Barthel Index, and Action Research Arm Test were measured at six months post-stroke. 9
Further, details regarding participants’ use of health services were documented at two, four, six and 12 weeks following treatment and at six months post-stroke. These included GP visits, hospital visits and admissions. Current medication use and any changes from discharge were documented. Treatments to manage contractures were also recorded. Patients were encouraged to document such activities in a diary and this additional informal questioning was completed at each review to ensure all information was provided.
Resource use costs associated with the study were summarised into relevant categories and valued in £ sterling using a price year of 2017/18. The costs were determined from national published sources of unit costs British National Formulary, 10 National Health Service reference costs (NHS 2017/2018) and estimated contracture costs. 11
The Barthel Index and Action Research Arm Test were used to generate a cost per unit of improvement in each of the measures. A one-way sensitivity analysis was undertaken (using IBM SPSS 27) to assess the extent of potential changes in the main cost parameters and outcomes of the treatment using the mean difference and lower and upper bounds of the confidence intervals. The 95% confidence interval of net cost and changes in outcomes were used to generate a series of potential scenarios and explore the changes in the estimated cost per unit of improvement.
Results
Between January 2012 and December 2013, 93 participants were randomised and received injections (see Figure 1: CONSORT diagram). The treatment group (
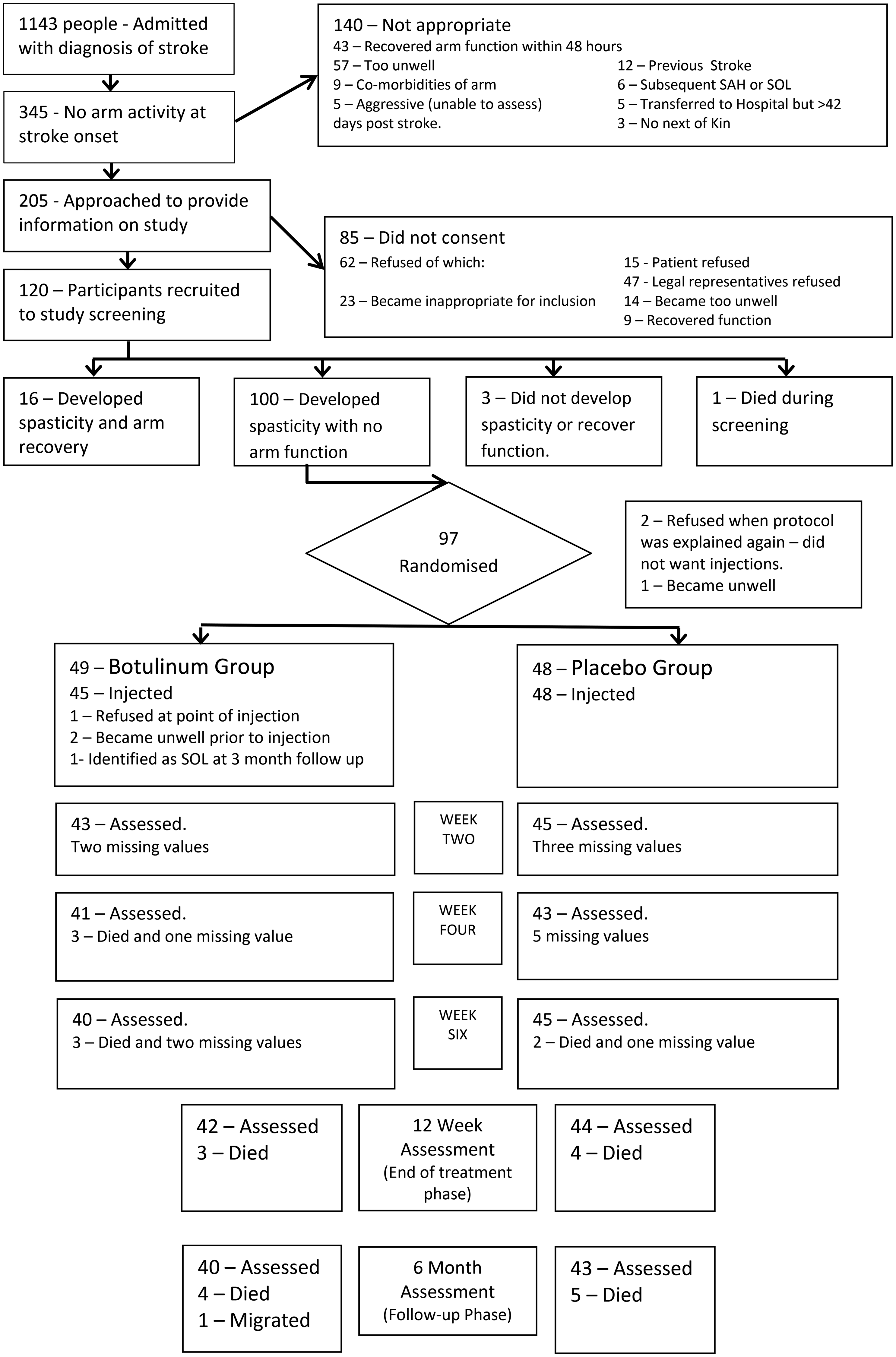
Consort flow chart summarising the flow of participants through the study. The last value was carried forward in cases of missing values. SAH: subarachnoid haemorrhage; SOL: space occupying lesion.
Intervention cost
The costs related to the intervention arm of the trial were summed to give the total cost per participant (NHS costs). Where possible, unit costs for the UK were applied (e.g. Personal Social Services Research Unit, British National Formulary 10 ) to increase generalisability. These are reported in Table 1.
Summary of all intervention costs.

Resource use analysis is presented in Table 2. There were no significant differences when the costs associated with all prescribed drugs were assessed between groups at discharge, three months and six months. The calculated costs associated with the initial admission and subsequent admissions for both groups were analysed, and no statistically significant difference was identified. The mean total costs associated with hospitalisations were £21,577 in the treatment group and £21,389 in the control group (mean difference £187.6; 95% CI −£3989.3, £4364.5). When all costs were combined, no statistically significant differences were identified (Table 2).
Between-group analysis of resources and costs.
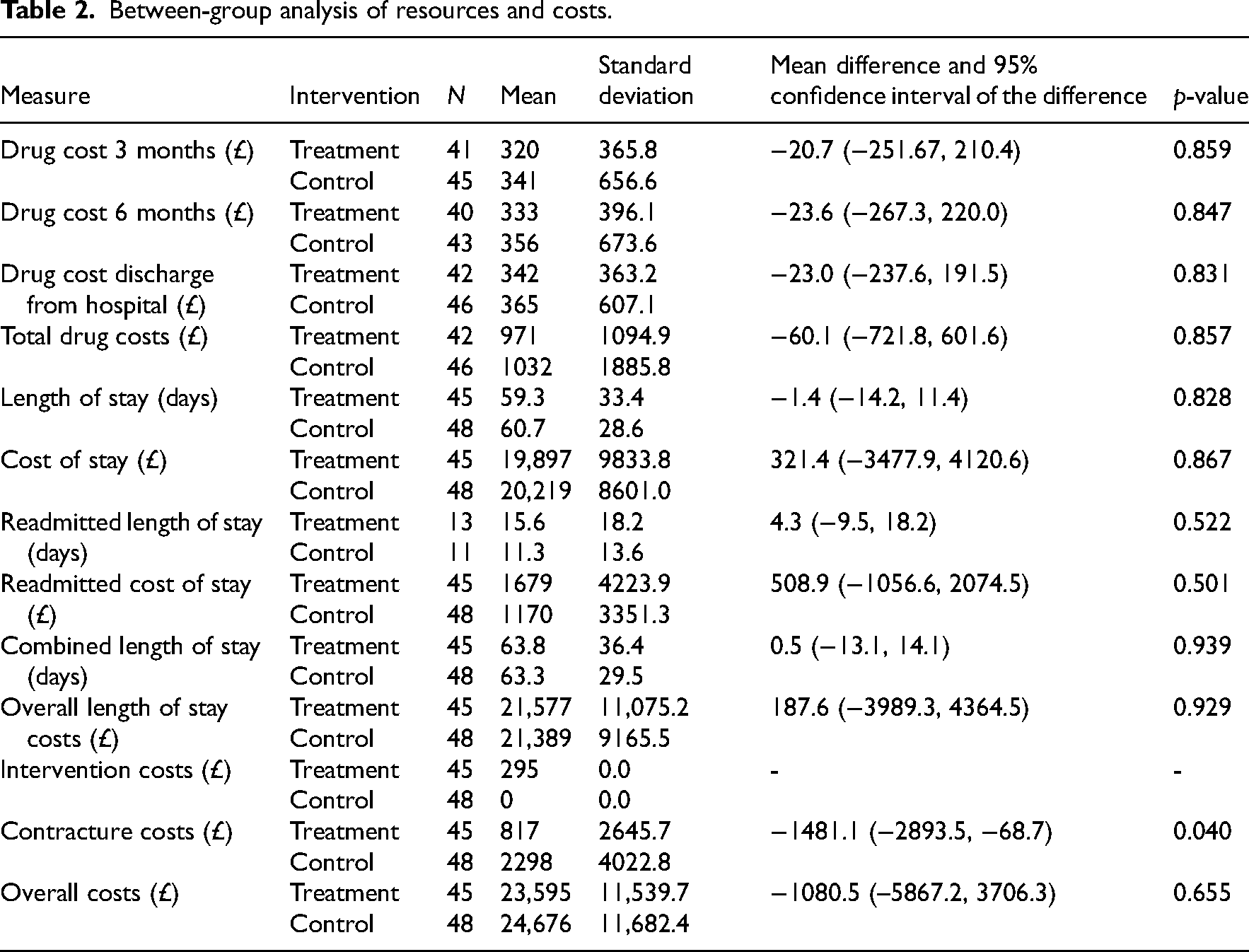
The mean contracture cost for the treatment group was £817 while the costs for the control group was £2298. This was statistically significant (
The change in Barthel Index scores between baseline and six months was lower in the control group (mean 7.3 (SD 6.0)) compared to the treatment group (mean 8.1 (SD 5.0)). The mean difference was 0.8 and not statistically significant (95% CI −1.5, 3.3,
Sensitivity Analysis
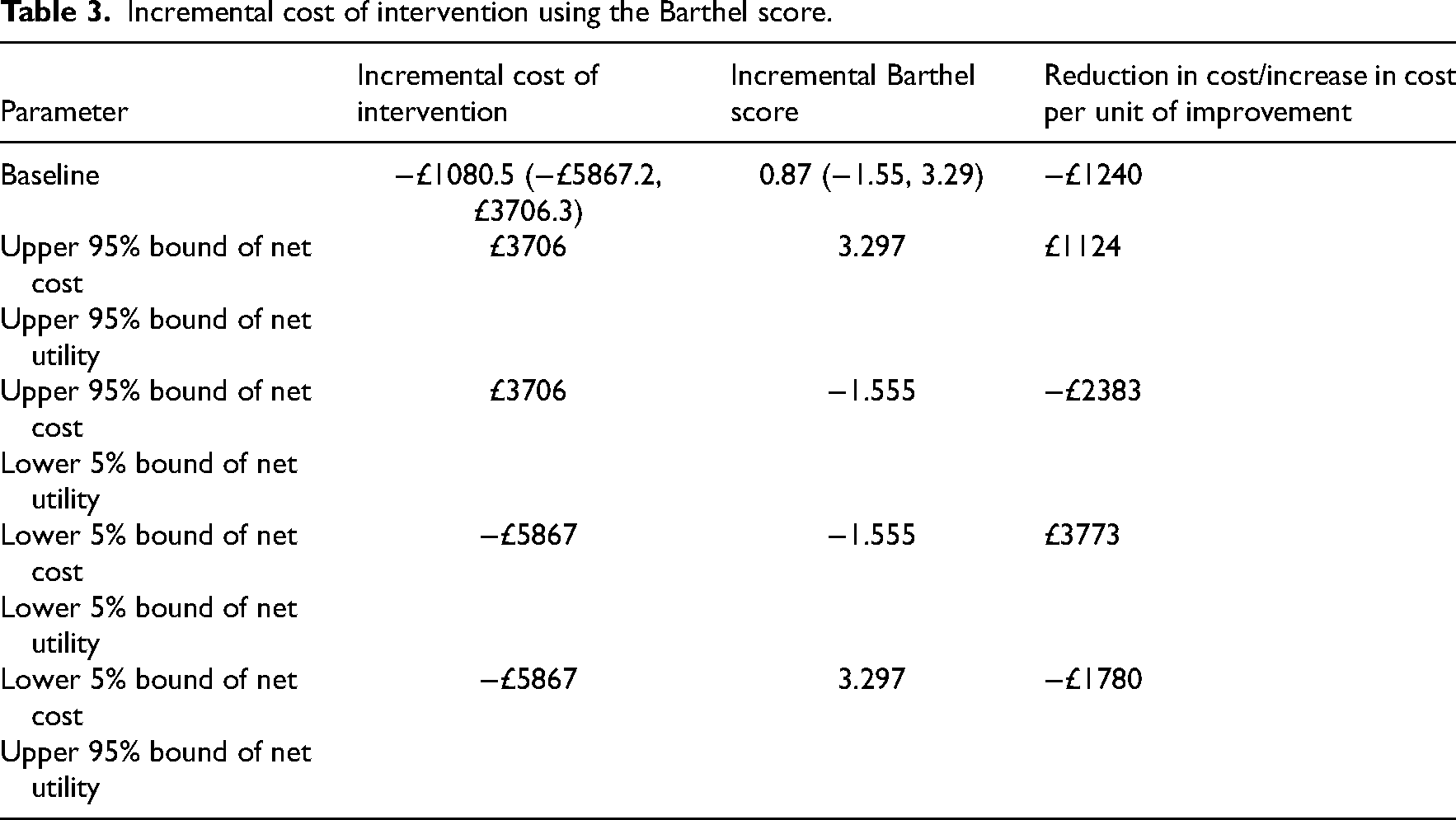
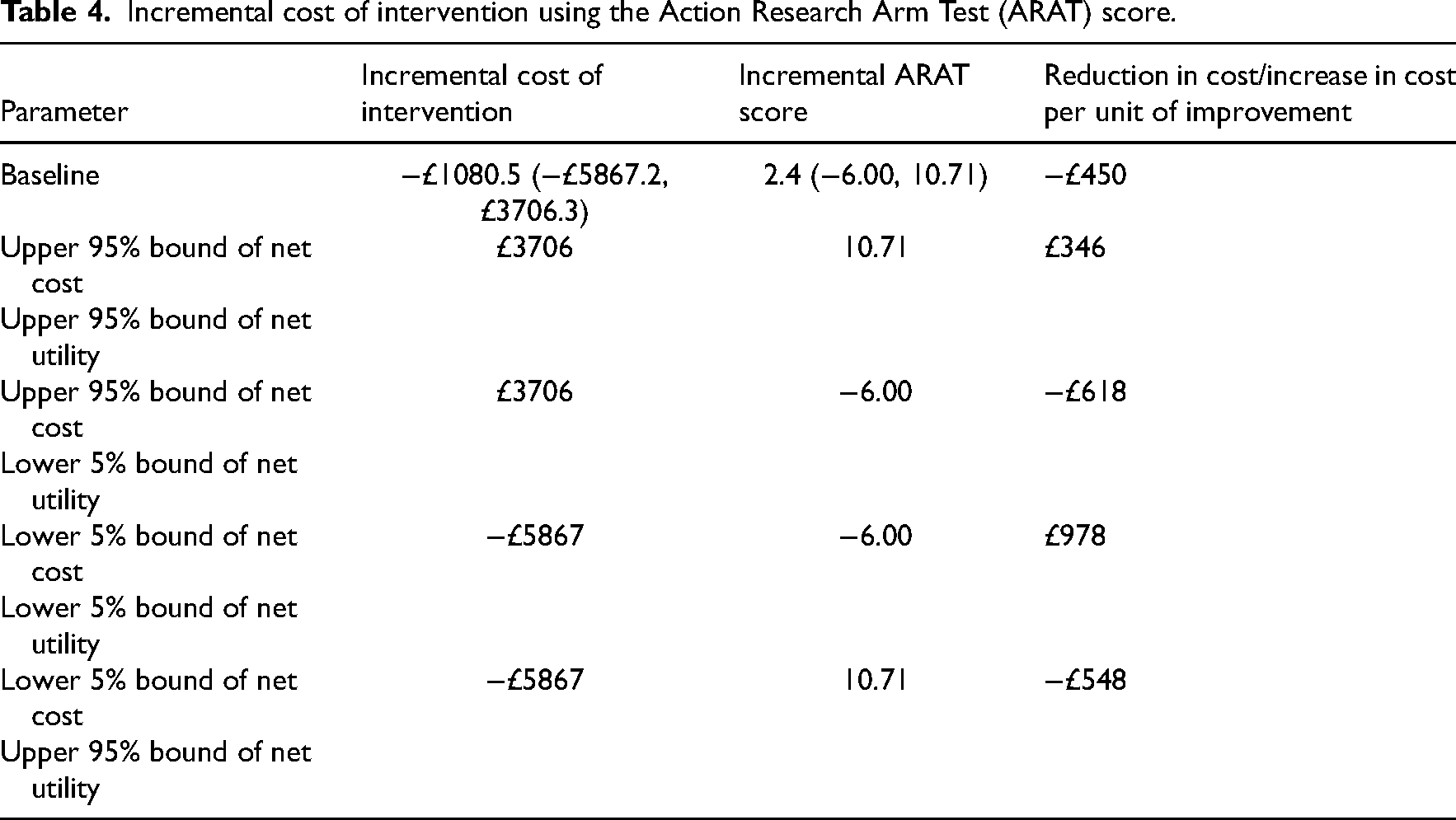
A one-way sensitivity analysis was undertaken to assess the extent of potential changes in the main cost parameters and outcomes of the treatment using the mean difference, lower and upper bounds of the confidence intervals for the Barthel score (Table 3) and the Action Research Arm Test (Table 4).
Incremental cost of intervention using the Barthel score.
Incremental cost of intervention using the Action Research Arm Test (ARAT) score.
When the base case results of the costs of the intervention are used with the base case results of the Barthel scores, the cost per unit of improvement was −£1240 indicating that the intervention costs less and is more effective.
When the lower 5% bound of net cost (−£5867) is used with the upper 95% bound of net utility (3.297), then the intervention is again seen as costing less and more effective (−£1780) (see Table 3). When the base case results of the costs of the intervention are used with the base case results of the Action Research Arm Test scores, the cost per unit of improvement was −£450 indicating that the intervention costs less and is more effective.
When the lower 5% bound of net cost (−£5867) is used with the upper 95% bound of net utility (10.71), then the intervention is again seen as costing less and more effective (−£548) (see Table 4).
Discussion
We have previously demonstrated that the early use of botulinum toxin reduced spasticity and contractures after stroke and that the effects lasted for approximately 12 weeks. 9 In this study, we have demonstrated that the additional treatment with botulinum toxin does not lead to an increase in cost associated with managing these patients.
This indicates that the early use of botulinum toxin for post-stroke spasticity can be cost-saving to the NHS when assessed in terms of gain in the activity of daily living (Barthel Index) and arm function (total Action Research Arm Test). This result contrasts with previously reported cost-effectiveness data that suggested that the base case incremental cost-effectiveness ratio for botulinum toxin type A plus therapy was £93,500.
6
However, it is possible that the magnitude of difference can be explained by the differences in the two studies:
The previous study measured response to treatment using the Modified Ashworth Score (an invalidated measure of spasticity),
12
which has previously been shown to underestimate treatment effects.
13
The previous study treatment was initiated in patients who are likely to have established spasticity and/or contractures (mean time to treatment 46 weeks post-stroke).
6
This contrasts with the current study where treatment was initiated in patients who presented with spasticity prior to contractures being established (mean time to treatment 14 days post-stroke).
5
The previous study primarily focused on improving function using a combination of therapy and botulinum toxin
6
and in the current study,
5
the primary focus was to prevent contractures. In the current study, all patients had an Action Research Arm Test of 0 to 2 at injection,
5
whereas in the previous study 45% of the participants had an Action Research Arm Test >3.
6
There are two main limitations in this study. The first is that the study had a small sample size and was not powered to assess cost-effectiveness. As a result, some data was not recorded and is therefore absent from the analysis. For example, the number of therapist contacts following discharge to the community was not recorded. A further limitation is that this study has only been able to assess the health costs rather than additional social costs. While the study recorded details about the number of paid social care services participants received, unpaid family care was not recorded. Secondly, the patients were not monitored longer term, so we have had to estimate the costs associated with treating contractures. Currently, evidence suggests contractures are likely to be common
6
and are often not managed well. The costs for long-term management of contractures can be high. It may involve treating repeated infections with antibiotics, the treatment of pressure sores and surgery for debridement or to reduce discomfort and allow for basic hygiene. For the purposes of this study, we have used a mean cost for the long-term management of contractures of £9193.
11
We feel that this is a realistic cost that includes the full direct and indirect costs of contracture management.
This study is the first randomised-controlled study using botulinum toxin early to analyse the potential cost-effectiveness of trying to manage contractures. It provides data that suggests that the perceived expense of botulinum toxin early after a stroke before any contractures have developed may help reduce health costs longer term. Clinicians, often subconsciously, make decisions based on the cost of treatment on a daily basis. This is a form of rationing services which clinicians are often unaware they make. It is uncomfortable for clinicians who want to be patient-centred but must balance this ideal, with remaining service-centred to ensure patient flow through their service. We hope that this study helps to ease just one of the many cost-based treatment decisions that clinicians must make.
Treating spasticity early, in stroke patients at risk of contractures, with botulinum toxin does not lead to a significant increase in cost associated with managing these patients. Future-powered studies should now focus on the long-term cost-effectiveness associated with treatment involving botulinum toxin and ensure all social costs are included to allow for a more meaningful result.
Clinical messages
The early use of botulinum toxin, as soon as spasticity is identified, appears to be cost-neutral.
Mean contracture costs for the treatment group were £817 while the costs for the control group were £2298 (
Both base case results for cost per improvement of Barthel and Action Research Arm Test scores indicate that the intervention costs less and is more effective.
Footnotes
Acknowledgements
Grateful thanks to all the stroke survivors, their relatives and carers who made this study possible. Many thanks to Professor Steve Sturman, Mr Brinton Helliwell and Dr Sissi Ispoglou for their assistance during the initial study.
Author contributions
CL and ADP conceived the study, CL acquired the funding with input from ADP, CL designed the study, acted as a blinded assessor and coordinated the study. IH itemised the resource costs and carried out statistical analysis and with input from CP interpreted the results. CL wrote the first draft of the report with IH assisting with the result section. All authors reviewed and agreed to the final draft.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This paper summarises independent research funded by the National Institute for Health Research (NIHR) under its Research for Patient Benefit Programme (PB-PG-0808-16319). The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health. Allergan provided the drug used and an unrestricted educational grant to support this study. They had no role in the study design, data collection, data analysis, data interpretation, or writing of the report and this publication.
