Abstract
Objective:
The aim of this study was to explore the feasibility of conducting a randomized controlled trial of dynamic Lycra® orthoses as an adjunct to arm rehabilitation after stroke and to explore the magnitude and direction of change on arm outcomes.
Design:
This is a single-blind, two-arm parallel group, feasibility randomized controlled trial.
Setting:
In-patient rehabilitation.
Subjects:
The study participants were stroke survivors with arm hemiparesis two to four weeks after stroke receiving in-patient rehabilitation.
Interventions:
Participants were randomized 2:1 to wear Lycra® gauntlets for eight hours daily for eight weeks, plus usual rehabilitation (
Main measures:
Recruitment, retention, fidelity, adverse events and completeness of data collection were examined at 8 and 16 weeks; arm function (activity limitation; Action Research Arm Test, Motor Activity Log) and impairment (Nine-hole Peg Test, Motricity Index, Modified Tardieu Scale). Structured interviews explored acceptability.
Results:
Of the target of 51, 43 (84%) participants were recruited. Retention at 8 weeks was 32 (79%) and 24 (56%) at 16 weeks. In total, 11 (52%) intervention group participants and 6 (50%) control group participants (odds ratio = 1.3, 95% confidence interval = 0.2 to 7.8) had improved Action Research Arm Test level by 8 weeks; at 16 weeks, this was 8 (61%) intervention and 6 (75.0%) control participants (odds ratio = 1.1, 95% confidence interval = 0.1 to 13.1). Change on other measures favoured control participants. Acceptability was influenced by 26 adverse reactions.
Conclusion:
Recruitment and retention were low, and adverse reactions were problematic. There were no indications of clinically relevant effects, but the small sample means definitive conclusions cannot be made. A definitive trial is not warranted without orthoses adaptation.
Introduction
Studies with children who have spastic hemiplegia caused by cerebral palsy suggest that wearing dynamic Lycra® orthoses as an adjunct to goal-directed training may improve movement and functional goal achievement. 1 This evidence raises the question of whether the orthoses may be effective as an adjunct to rehabilitation in adults with arm impairments after stroke. Arm impairments, which include weakness and sensory loss, restrict independence in activities of daily living and affect stroke survivors’ quality of life. 2
Dynamic Lycra® orthoses are commercially available dynamic braces that use tensile properties of Lycra® to generate torsion, correct muscle force imbalances across joints, optimize muscle length and functional positioning, and provide compression to enhance proprioception and sensory awareness.3,4 However, effectiveness in stroke rehabilitation has not been fully evaluated, despite anecdotal evidence that they are already in use in clinical practice. One single case study of 6 weeks wear in a survivor with long-standing stroke 4 and a crossover trial with 16 stroke survivors 3–36 weeks after stroke onset 3 involving only 3 hours orthosis wear have shown improvements in arm impairment, sensation and functional outcomes after orthosis wear. Evidence is therefore limited to low-quality study designs, and rigorous effectiveness studies are required.
The aim of this feasibility randomized controlled trial was to examine recruitment, retention, adverse events, intervention fidelity, magnitude and direction of difference in outcomes in stroke survivors receiving Lycra® orthoses as an adjunct to usual rehabilitation, compared to those receiving usual rehabilitation only. It also aimed to explore survivor and carer perceptions of acceptability, to inform decisions about a future definitive randomized controlled trial.
Methods
This was a single-blind, two-arm parallel group, feasibility randomized controlled trial. The study was funded by the Chief Scientist Office at the Scottish Government (grant no. CZH/4/1075). Tayside Clinical Trials Unit was responsible for oversight of study conduct and governance, and East of Scotland Research Ethics Committee provided ethical approval (ref. no. 15/ES/0093). The trial was registered with ClinicalTrials.gov (identifier: NCT03063970). Recruitment was conducted between March and December 2017, with final follow-up assessment in March 2018.
Participants were recruited from two acute stroke units in the North East of Scotland. Eligibility criteria were as follows: stroke survivors two to four weeks after unilateral ischaemic stroke; persistent arm activity limitation indicated by Action Research Arm Test (ARAT) scores ⩽45 (maximum = 57); 5 and able to participate in usual rehabilitation. Exclusion criteria were as follows: inability to provide informed consent, previous stroke resulting in residual disability, hemiplegic shoulder pain, Lycra® sensitivity, premorbid arm impairment or already using arm orthoses. A sample of 51 subjects was judged adequate to provide information about potential effects and variance to inform sample size calculations for a future definitive trial. 6
Regular rehabilitation therapists identified potential participants from stroke unit admission records and provided them with study invitation materials. Stroke survivors expressing interest in participation provided written permission to be contacted by a researcher. The study researcher (A.J.) then visited participants, explained the study and obtained informed consent for study participation before undertaking eligibility screening, followed by baseline assessment and randomization.
Randomization to intervention or usual rehabilitation control groups was undertaken in a 2:1 ratio to optimize information collected about the intervention in this small feasibility study. After screening and baseline assessment, the study researcher entered participant study identification number and minimization information into an electronic password-protected remote, concealed, web-based randomization system developed for the study by Tayside Clinical Trials Unit. Minimization was applied by severity level of activity limitation on ARAT scores. 5 Levels were previously defined by examining Nine-hole Peg Test 7 and ARAT scores together, with scores of severe 0–3 indicating little or no movement, Moderate 4–28 indicating some movement but limited dexterity, defined by inability to place pegs in the Nine-hole Peg Test and mild ⩾29 indicating ability to place one or more pegs in the Nine-hole Peg Test. 8 The computerized system-generated random sequence allocation determined group allocation and the study occupational therapist (L.W.) was then informed by the researcher of participant group allocation.
Demographic data were collected at baseline and included gender, age and time from stroke onset to randomization. Stroke type and lesion location were determined by review of computerized tomography scans with a specialist neuro-radiology consultant. Other data describing participant characteristics included stroke severity, measured by the National Institute of Health Stroke Scale, 9 cognitive function measured on the Montreal Cognitive Assessment, 10 handedness measured on the Edinburgh Handedness Inventory 11 and independence in activities of daily living measured on the Barthel Index. 12
Baseline data were collected by the trained study researcher (zero weeks). At eight weeks, outcome data were collected from all participants and the orthosis was removed from intervention group participants. At 16 weeks follow-up, data were collected to assess long-term duration of any effects. Data were collected by a blinded assessor, a neuro-physiotherapist trained in outcome assessment, in hospital or in participants’ homes if they had been discharged. The nature of the intervention, an orthosis that was worn and applied by staff or patients themselves, meant participants and rehabilitation therapists could clearly see which group the participant was allocated to. This meant that neither participants nor rehabilitation therapists could be blinded to intervention allocation.
Outcomes were as follows:
Data on recruitment, screening and randomization rates collected from stroke unit admission data;
Retention rate at 8 and 16 weeks;
Adverse events reported by participants or staff;
Wear fidelity recorded by participants and their families in daily diaries;
Activity limitation (ARAT); 5
Arm strength (Motricity Index); 13
Grip strength (Jamar Plus digital hand-held dynamometer); 14
Fine manual dexterity (Nine-hole Peg Test calculated in seconds/peg placed); 15
Elbow spasticity quantified by differences in angles of muscle reaction with fast and slow stretch (Modified Tardieu Scale); 16
Light touch perception at fingers, hand, forearm and arm, and proprioception at thumb, wrist, elbow and shoulder (Erasmus Modified Nottingham Sensory Assessment); 17
Patient-reported arm activity (Motor Activity Log 14). 18
Participants randomized to the intervention group were visited by the study occupational therapist to measure the arm, prescribe and fit the orthosis. The occupational therapist was an experienced clinician with more than 20 years of neurological rehabilitation experience. She was trained in orthosis measurement and fitting through attendance at the manufacturer’s training course and had many years’ experience of measuring and fitting orthoses in clinical practice, in collaboration with manufacturers’ orthotists. She also had additional training with the manufacturer’s orthotist in preparation for the study. In addition, the manufacturer’s orthotist provided advice and guidance on measurement, fitting and adaptations as required throughout the study. To ensure measurement and fitting accuracy, the orthotist, or another health professional, assisted during orthotic measurement procedures. Where possible, and according to manufacturers’ instructions, measurements were conducted in the middle of the day to account for fluctuations in oedema first thing in the morning and later in the day.
To optimize cutaneous and proprioceptive sensory stimulation and control of movement, maximum limb coverage by the orthosis was considered appropriate. During the piloting phase prior to this feasibility randomized controlled trial, gauntlet and short-fingered glove dynamic Lycra® orthoses designs were tested for acceptability and feasibility with 17 stroke survivors who met inclusion criteria for the randomized controlled trial. Participants reported difficulties doffing and donning the glove, compared to the gauntlet, with restrictions to dexterity, discomfort and blistering at the thumb web reported with glove wear. It was decided that the gauntlet design was most acceptable and feasible for use in the randomized controlled trial. The gauntlet extended from metacarpophalangeal joints to deltoid insertion, encompassing thumb metacarpophalangeal joint, wrist and elbow joints (Figure 1). The pilot study is in submission for publication elsewhere.

Example of Lycra® gauntlet used in the study.
Several visits for measurement, fitting and adjustment were required, therefore an additional week for gradual increase in wear duration to eight hours per day was incorporated into trial flow, to accommodate adjustments and allow acclimatization to wear. After this week, participants wore the orthosis for eight hours daily for eight weeks, to support motor control and provide continuous sensory stimulation during task practice and performance throughout the day, giving a total intervention period of nine weeks. In the absence of definitive research about wear duration in this population, eight hours per day was determined by the manufacturer’s seven-day wear protocol for Lycra® garments 19 and through discussion with the manufacturer’s orthotist.
The study team made adaptations to the dynamic Lycra® orthoses as necessary throughout the intervention period to ensure fit, comfort and duration of wear while remaining compatible with the study protocol. Participants, nurses, rehabilitation professionals and family members were trained in donning and doffing the orthosis, in checking for oedema or other skin and circulatory problems and received written instruction leaflets to support their training. Piloting highlighted that some participants presented with or developed upper limb and hand oedema. Intervention group participants and their carers were therefore instructed to contact the research occupational therapist immediately should problems of comfort, fit or oedema arise that prevented wear.
Participants developing oedema were provided with a graduated compression oedema glove to wear overnight and with the dynamic Lycra® orthoses. This was to reduce the likelihood of oedema preventing dynamic Lycra® orthoses wear, or the onset of skin and circulatory problems. Where oedema occurred, the occupational therapist would provide the oedema compression glove and monitor oedema, fit and comfort. If oedema glove provision was not appropriate or not effective, she would next involve the study orthotist to advise on adaptations to the dynamic Lycra® orthosis. When this happened, the orthosis was sent for adjustment and the participant restarted the intervention and continued wear until the eight weeks total duration of wear was complete.
After randomization, both groups received an information leaflet about general care of the hemiplegic arm. Intervention group participants were also asked to complete a diary to record daily frequency and duration of wear. During the eight-week intervention period, the occupational therapist telephoned all participants at four weeks to explore progress, identify problems and maintain interest in the study. Apart from the telephone call at four weeks, the control group received no additional intervention. Both groups received usual rehabilitation, typically 45 minutes of occupational and physiotherapy per weekday. For participants discharged home during the study, rehabilitation typically comprised one session each of physiotherapy or occupational therapy per week.
Rehabilitation therapists were asked to complete diaries to record usual rehabilitation duration and content for intervention and control group participants. To record usual therapy in a standardized way, therapists were provided with a list of therapy interventions, identified in the pilot study as representing usual practice, and asked to record time spent in each per session and number of repetitions. Interventions included activities of daily living, range of movement, task training, weight-bearing, hands on therapy, stretching and positioning, strengthening, mental practice and mirror therapy.
Data analysis
Descriptive analysis, calculating number and percentage, was conducted for recruitment, retention and adverse event rates. Proportion of missing data was examined to inform outcome measure selection for a definitive trial. Definitive hypothesis testing was not conducted; however, between-group comparison was undertaken to explore difference in magnitude and direction of change. Analysis was by intention-to-treat. Primary analysis involved group comparison for change in outcome between baseline, and 8 and 16 weeks, adjusted for baseline scores and severity of activity limitation, derived from baseline ARAT scores. Continuous outcomes were assessed for normality, and relevant transformations were made. Repeated measures analysis was performed, and outcomes were assessed by multiple linear regression to give mean difference (95% confidence interval (CI)). Continuous data that could not be transformed to normal distribution were translated to binary outcomes and analysed using logistic regression. Categorical data were assessed using logistic regression to provide an odds ratio (95% CI).
Post-study interviews
A total of 12 orthosis group participants agreed to participate in interviews at 16 weeks with their carers or family members, to explore experiences of study participation. Interviews were guided by a topic guide (Supplemental material), examining benefits, limitations, acceptability and feasibility of orthosis wear and experiences of trial participation. Transcripts were read, coded and thematically analysed by two study researchers (A.J. and J.H.M.) experienced in qualitative methods.
Results
A total of 43 participants were recruited. Age ranged between 23 and 86 years, of which 24 participants (56%) were male. Participant characteristics and baseline scores for each group are presented in Table 1. The 2:1 randomization strategy worked, and groups were similar at baseline.
Participant characteristics.
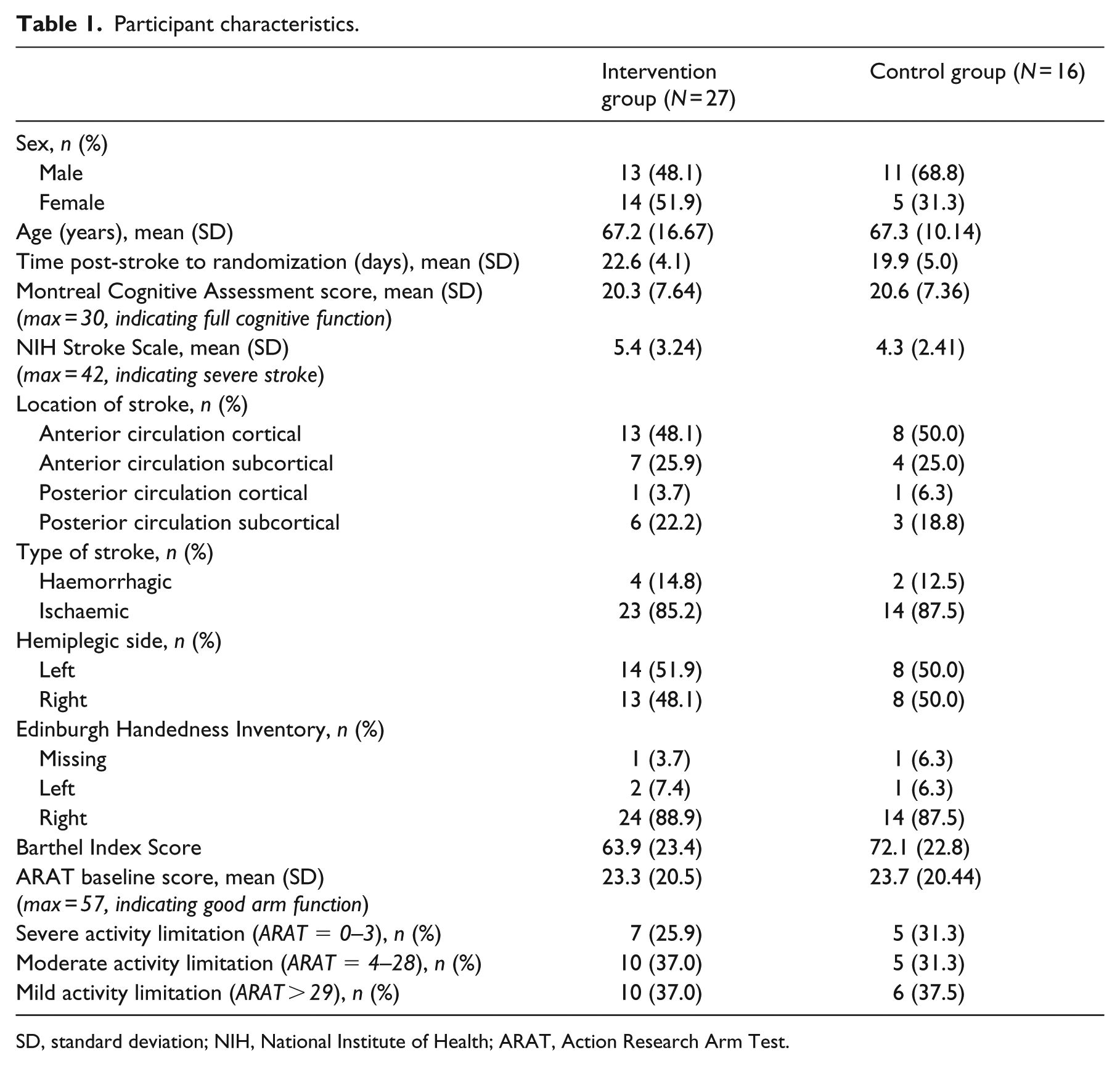
SD, standard deviation; NIH, National Institute of Health; ARAT, Action Research Arm Test.
Participant flow through the study is presented in Figure 2. Median recruitment rate per month was 4 (range = 1–8) and the sample size of 43 was 84% of the anticipated target (
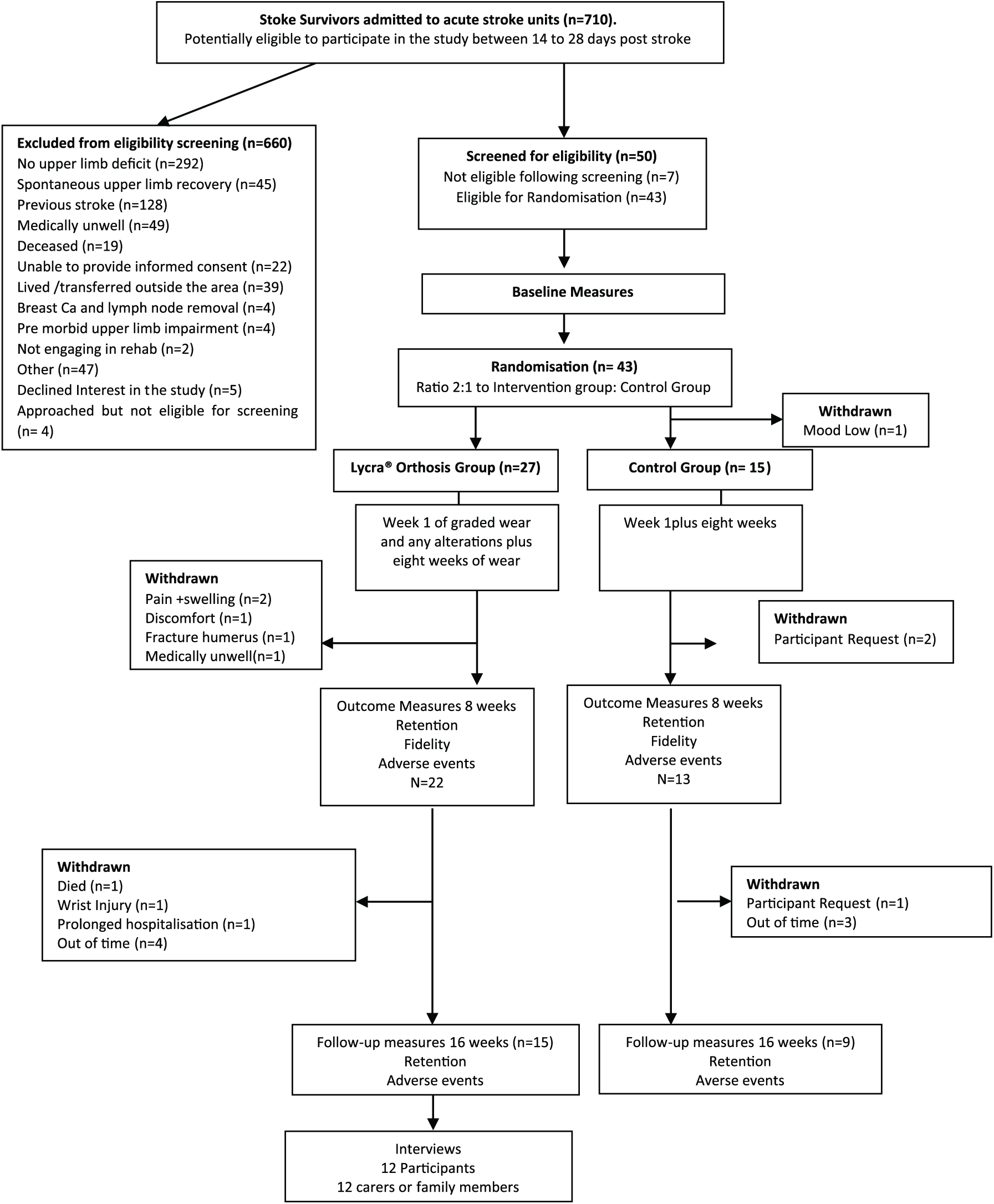
Participant flow through the study.
Daily orthosis wear was recorded in diaries by intervention group participants, carers or health professionals. In total, 20 (77%) diaries were returned, of which only 5 (25%) had entries for the planned 56 days of the intervention. Of those, three participants (60%) reported data for every day. Across returned diaries, median duration of daily wear was 8 hours (range = 3–9) over 28 (range = 0–56) days.
Usual rehabilitation was poorly recorded, with only 17 of 43 (40%) diaries returned across the sample, with 12 diaries (44%) returned for the intervention group and 4 (33%) for the control group. The median (range) number of entries completed by rehabilitation therapists per participant was 6.5 (1–44), and for the control group, this was 4.0 (2–6). Most diaries reported time spent rather than repetitions of usual therapy interventions, and most reported only a small selection of the identified interventions. Of those usual therapy interventions reported by both groups, median (range) minutes spent by intervention and control groups respectively, were as follows: range of movement, 15 (3–60) versus 10 (5–20) minutes; task training, 10 (15–2) versus 30 (0–30) minutes; hands on therapy, 10 (3–40) versus 5 (0–5) minutes; stretching and positioning, 20 (3–60) versus 10 (10–15) minutes; strengthening, 12.5 (5–20) versus 7.5 (5–10) minutes; and mental practice, 20 (10–30) versus 5 (0–5) minutes.
Descriptive data for baseline, and 8- and 16-week outcome scores (mean (SD)), median (range) are shown in Table 2. By 8 and 16 weeks, the control group demonstrated higher scores on all measures. Change from baseline (zero weeks) and exploratory logistic regression analysis are shown in Table 3. ARAT, Nine-hole Peg Test and Modified Tardieu Scale spasticity angle data were skewed and could not be transformed to normal distribution. Data were therefore translated to binary outcomes, defined as improvement in one or more ARAT severity level; improvement from unable to able to place nine pegs; and change to 0 on Modified Tardieu Scale spasticity angle (defined as R2 angle of catch on slow stretch minus R1 angle of catch on fast stretch), indicating no spasticity.
Outcome scores at baseline (0 weeks), 8 weeks and 16 weeks: mean (SD), median (range).
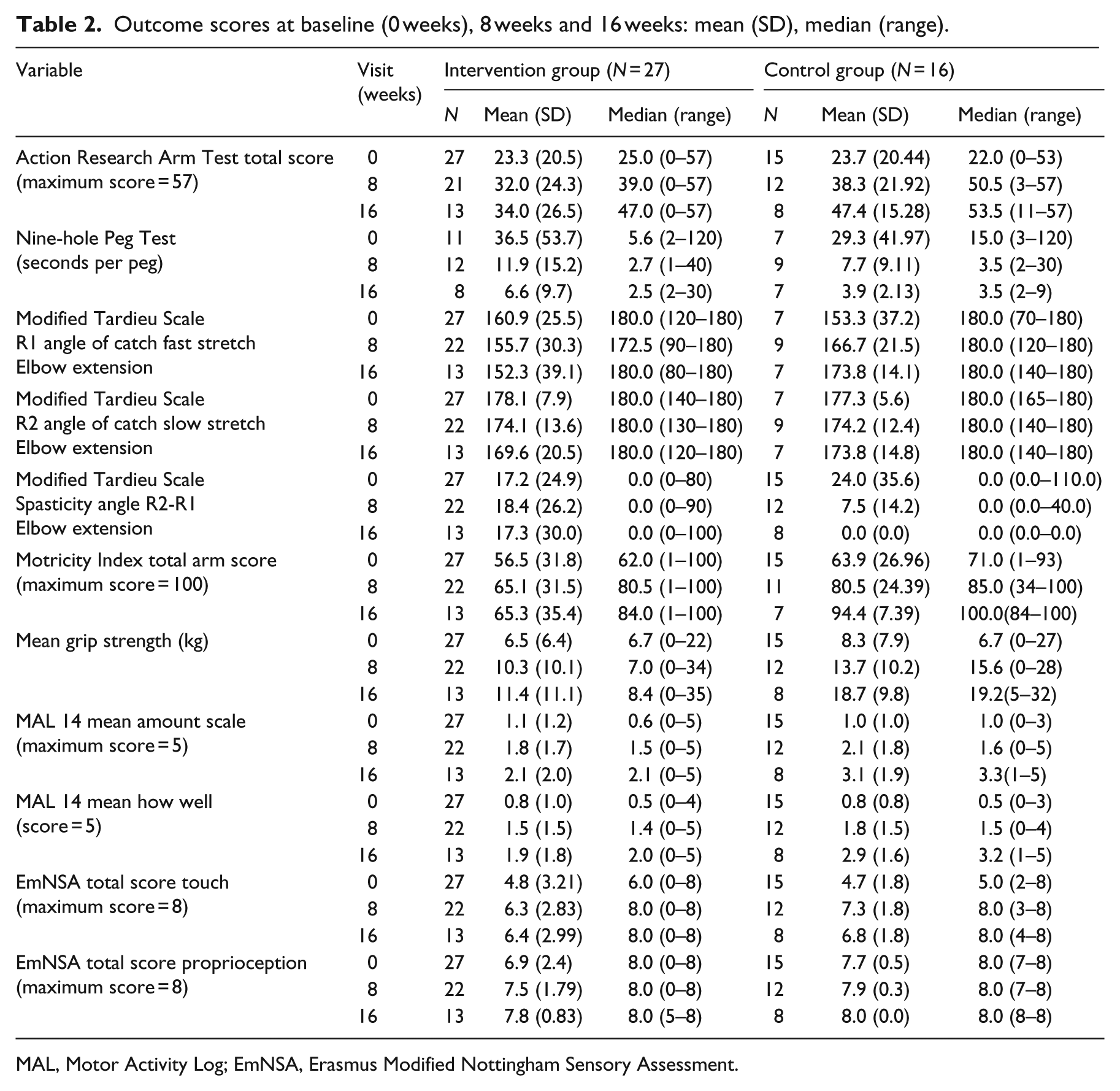
MAL, Motor Activity Log; EmNSA, Erasmus Modified Nottingham Sensory Assessment.
Between-group differences in change, 0–8 and 0–16 weeks: adjusted odds ratio/mean difference (95% CI).
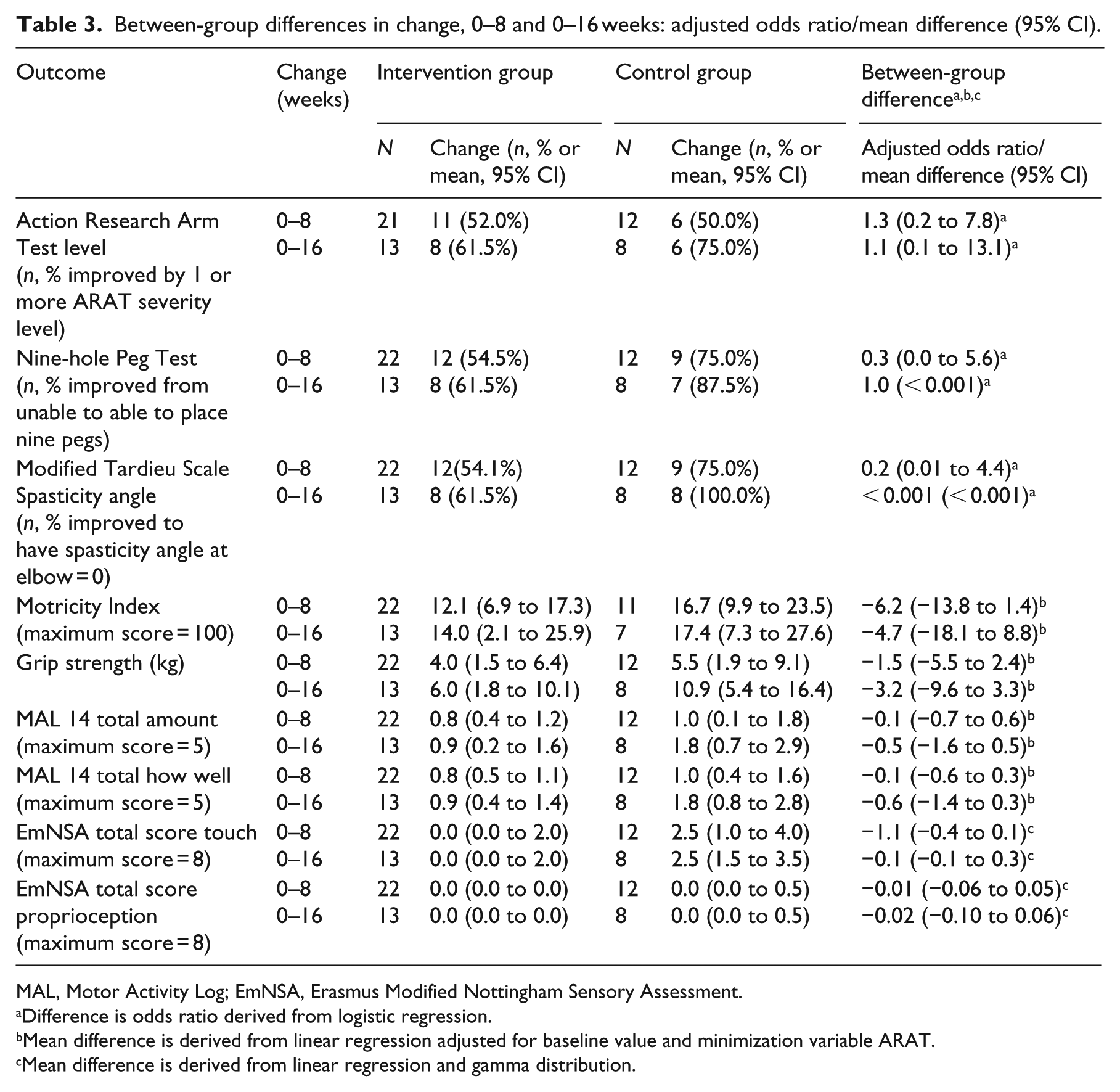
MAL, Motor Activity Log; EmNSA, Erasmus Modified Nottingham Sensory Assessment.
Difference is odds ratio derived from logistic regression.
Mean difference is derived from linear regression adjusted for baseline value and minimization variable ARAT.
Mean difference is derived from linear regression and gamma distribution.
The proportion of participants improving ARAT severity level zero to eight weeks was higher in the intervention group (
The assessor was unblinded in 13 cases (31%), despite instructing participants not to disclose group allocation. All measures were fully completed at every assessment, indicating that missing data would not be problematic in a definitive trial.
Qualitative findings
A total of 12 intervention group participants and their carers were interviewed by a study researcher (A.J.) (Supplemental Appendix 1). Many participants reported improved confidence, support, limb awareness, limb alignment, strength, and control of muscle tone and movement (see Supplemental Appendix 1 for illustrative quotes). Other participants reported movement and circulation restriction, skin irritation and worsened oedema. Regardless of experiences, participants were content to complete outcome measures and comply with study steps to support the research. Study documentation and researcher explanations were perceived as clear. Clarity about study involvement, acceptance of screening, randomization and measurement was notable, as was appreciation of their personal contribution as participants. Carers of these participants were supportive of the study and some noted positive outcomes from wear, specifically in participant confidence undertaking certain activities, in providing support, reducing tone and increased awareness of the affected limb. They also perceived disadvantages from orthosis wear as it could not be worn during certain exercises, in dirty tasks or any task involving water.
Discussion
This feasibility randomized controlled study of dynamic Lycra® orthoses in stroke showed a definitive multicentred randomized controlled trial to examine effectiveness could be feasible, but recruitment and retention strategies require adaptation. However, there was little indication that the orthoses might improve outcomes more than usual care, and adverse reactions were reported. Progression to randomized controlled trial may therefore not be warranted.
At 6% recruitment rate was low, but congruent with arm rehabilitation studies recruiting soon after stroke. 8 , 20 Because study researchers recruited participants in collaboration with local staff, we know most eligible participants were recruited. We anticipated recruiting around five participants per month over 10 months; however, slow recruitment, particularly in autumn months, meant this figure was lower. Consequently, we ran out of time to undertake 16-week assessment for 4 intervention and 3 control participants, contributing to low retention at 16 weeks. A multicentre randomized controlled trial would require recruitment through local clinical staff, which would make recruitment slower, necessitating realistic calculation of required sites, and a long recruitment period. During autumn, many stroke survivors were too ill to participate, which aligns with studies showing increased rates of major stroke subtypes in autumn. 21 Our findings therefore provide a realistic estimation of recruitment and study timescales, from which to calculate sites for a definitive trial, while accounting for seasonal variation in recruitment.
Study retention at 81% and 75% at eight weeks for intervention and control groups was low and had this been a definitive trial, would have threatened study validity. 22 , 23 Attrition due to ill-health was inevitable and requires consideration in sample estimation for a randomized controlled trial. Withdrawal with adverse intervention responses was problematic. Early pilot work showed problems with skin marking and oedema; therefore, we introduced adaptations – oedema gloves and gauntlet lengthening to reduce problematic oedema, and silk liners to prevent skin irritation. Despite subsequent adaptations, problems led to withdrawals. Other studies report adverse events of prolonged orthotic wear. Although involving rigid splints, in line with this study, incidents included skin breakdown, oedema and discomfort. 24 , 25 Risks of adverse events have to be balanced against intervention benefit; however, potential benefits of Lycra® orthoses remain uncertain. Reconsidering orthosis design and wear duration will be crucial before a definitive randomized controlled trial.
Three control group participants requested withdrawal because of losing interest, a known phenomenon when there is no control intervention. 20 , 26 We considered sham gauntlets, however previous research in multiple sclerosis found these required elastic characteristics, acting to an extent as an intervention. 27 However, given effects on retention of no active control intervention, an acceptable but inert control intervention should be reconsidered.
Measurement bias potentially threatened our findings. The assessor, a physiotherapist also working in rehabilitation, was unintentionally unblinded at work six times. She was unblinded seven more times because participants disclosed group allocation. Educating participants more about maintaining blinding and complete assessor independence will be vital in future.
Although low-quality design, previous Lycra® orthoses studies 3 , 4 suggest improved spasticity, proprioception and arm function; therefore, we anticipated some indications of benefit from wear. We found only slightly improved odds of improving ARAT severity level by eight weeks. Examining change in relatively arbitrary ARAT severity levels was somewhat crude but necessary because ARAT data were severely skewed. The approach probably masked important details, given unadjusted ARAT scores (Table 2) suggest higher outcome scores in the control group. Odds ratios and adjusted mean differences on other measures were neutral or favoured controls. There was therefore no suggestion of potential benefit of Lycra® orthoses. Although we did not set decision rules about progression to trial, some indication of benefit would be necessary to justify progression. Small sample size, large variability and lack of definitive hypothesis testing means caution in interpretation has to be applied, but our data imply that benefits from Lycra® orthoses are unlikely, thus informing our judgement about progression to randomized controlled trial.
Previous literature and our pilot work suggested that these orthoses may be beneficial. 3 , 4 The dynamic properties of Lycra® were assumed to enhance sensory awareness and joint alignment, potentially enhancing practice frequency and duration, to improve arm function. 3 , 4 However, indications are they may act as static splints, immobilizing rather than facilitating function. A Cochrane Review examining stretch for treatment and prevention of contracture in neurological conditions 24 shows that static orthoses have little or no impact on arm joint mobility or spasticity, and function, reflecting our findings. If Lycra® orthoses are to be pursued as an intervention after stroke, they need to be explicitly dynamic. 28 Undertaking a definitive randomized controlled trial is, thus, probably not warranted without significant intervention redesign.
Although participants reported enhanced body schema and confidence, it would be ethically unacceptable to further investigate an intervention that causes adverse reactions and poorer physical outcomes, despite
This was, to our knowledge, the first feasibility randomized controlled trial of these orthoses in arm rehabilitation after stroke; however, there were several limitations. The occupational therapist undertaking measurement and prescription was trained; however, errors could have occurred. Furthermore, limb dimensions often changed between first measurement and fitting because of oedema onset, and the difference may have caused some adverse reactions. The study is limited by low retention rates, lack of blinding of healthcare professionals and participants, and by assessor unblinding. We accounted for baseline severity in analysis; however, subgroup analysis based on severity would provide more information about which survivors might respond best. Because of attrition, subgroups were too small to make meaningful conclusions and should be examined in a future study. Limited information about wear fidelity and usual rehabilitation activities, which may both have influenced outcome, should also be addressed in future studies. We were primarily interested in clinical outcomes in this study; however, motion analysis with and without the dynamic Lycra® is also useful to better understand the effects of the orthosis on movement trajectories and to inform future orthosis design. We did collect that data and will report it in a future publication.
This study indicates that a multicentre randomized controlled trial examining the effects of Lycra® orthoses on upper limb impairment and activity limitation could be feasible if recruitment and retention issues were addressed. However, the study underlines the importance of scientifically evaluating interventions already in use in clinical practice, given that we found skin and circulatory problems attributable to the orthoses were problematic. The orthoses may also be detrimental for recovery of arm function, possibly acting as rigid splints. Thus, assumptions in rehabilitation practice and within previous literature, that orthoses act dynamically to facilitate movement, can be challenged. Further research should therefore explore precise motor and sensory mechanisms of action. Adaptation of orthosis design must also be undertaken to prevent adverse events and to ensure they dynamically facilitate movement and function. Progression to full-scale trial is therefore not yet warranted.
Clinical messages
Adverse reactions in this study were high, and retention was difficult and low.
There was little indication that Lycra® orthoses might benefit arm recovery outcomes; however, definitive conclusions cannot be drawn.
Supplemental Material
Supplemental_Material – Supplemental material for Dynamic Lycra® orthoses as an adjunct to arm rehabilitation after stroke: a single-blind, two-arm parallel group, randomized controlled feasibility trial
Supplemental material, Supplemental_Material for Dynamic Lycra® orthoses as an adjunct to arm rehabilitation after stroke: a single-blind, two-arm parallel group, randomized controlled feasibility trial by Jacqui H Morris, Alexandra John, Lucy Wedderburn, Petra Rauchhaus and Peter T Donnan in Clinical Rehabilitation
Footnotes
Author contributions
J.H.M., L.W. and P.T.D. initiated, designed and obtained funding for the study; J.H.M. had overall responsibility for the conduct of the study and for monitoring progress, in collaboration with L.W. and A.J.; A.J. and J.H.M. designed the qualitative component of the study and undertook qualitative analysis; P.R. and P.T.D. designed the statistical analysis which was undertaken by P.R.; and J.M. and A.J. wrote the manuscript and all authors contributed to critically revising it.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Chief Scientist Office at the Scottish Government (Grant Number CZH/4/1075).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
