Abstract
Objective
Lower extremity varicose veins are a common chronic venous disorder, affecting approximately 23% of adults globally. Although endovenous thermal ablation, particularly radiofrequency ablation (RFA), has become the preferred treatment, post-procedural deep vein thrombosis (DVT) remains a concern. The necessity of pharmacologic prophylaxis following RFA remains controversial. This study aimed to assess the efficacy and safety of short-term prophylactic rivaroxaban for DVT prevention after RFA.
Methods
In this single-centre, randomized controlled trial, patients with lower extremity varicose veins undergoing RFA, with or without Trivex-assisted phlebectomy, were recruited between August 2020 and December 2024. Participants were randomized to receive either rivaroxaban (10 mg daily for 5 days postoperatively) or standard preventive care including early ambulation and compression. All patients were followed for 1 month, with DVT incidence evaluated by B-mode ultrasonography.
Results
A total of 298 patients were enrolled. No DVT events occurred in the rivaroxaban group, while four cases (2.7%) were observed in the standard care group (p = .04). No major bleeding events were reported. Minor bleeding occurred in 4.7% of the rivaroxaban group and 2.7% of the control group (p = .35).
Conclusions
Short-term prophylactic rivaroxaban significantly reduced DVT risk following RFA, with no significant increase in bleeding complications. These findings support its potential role in optimising perioperative thromboprophylaxis for varicose vein patients undergoing thermal ablation.
Introduction
Lower extremity varicose veins are one of the most prevalent chronic venous disorders, affecting 2–73% of adults globally.1–4 Previous studies have demonstrated that minimally invasive techniques achieve comparable clinical efficacy to conventional surgery.5,6 As a result, thermal ablation methods, particularly endovenous laser ablation (EVLA) and radiofrequency ablation (RFA), have become the preferred treatment modalities for this condition. The increasing adoption of these techniques has been accompanied by a growing incidence of procedure-related complications, with heat-induced thrombosis (EHIT) emerging as one of the most serious concerns.7,8 EHIT occurs when thrombi propagate from the ablation site into the deep veins, substantially increasing the risk of deep vein thrombosis (DVT). 9 These complications not only increase healthcare costs but also prolong recovery, placing additional burdens on patients and healthcare systems. Consequently, pharmacologic thromboprophylaxis has been proposed to mitigate these risks and improve postoperative safety. Nonetheless, concerns persist regarding the bleeding risks associated with anticoagulation, including surgical site bleeding, subcutaneous hematoma, and, in rare cases, intracranial hemorrhage. As a result, the necessity of routine pharmacologic prophylaxis after endovenous thermal ablation remains the subject of ongoing debate.
Low-molecular-weight heparin (LMWH) is widely used for thromboprophylaxis to reduce the risk of DVT. However, its use is associated with several limitations, including the need for parenteral administration, local injection site discomfort, and potential risk of injection site infections. Rivaroxaban, an oral direct factor Xa inhibitor, has demonstrated both efficacy and safety in preventing DVT after hip arthroplasty.10,11 Moreover, evidence suggests that a once-daily 10 mg dose of rivaroxaban effectively prevents DVT without significantly increasing the risk of bleeding. Nonetheless, the necessity of pharmacologic prophylaxis following thermal ablation for varicose veins remains controversial. Therefore, this study aimed to evaluate the safety and efficacy of rivaroxaban for DVT prevention after thermal ablation of varicose veins.
Methods
Trail design
This prospective, single-centre, randomized, single-blind controlled trial was conducted at Dongguan Tungwah Hospital between August 2020 and December 2024. The study utilized a two-group design with a 1:1 allocation ratio. A total of 298 patients with lower extremity varicose veins scheduled for radiofrequency ablation (RAF) were randomly assigned to one of two groups. Ethical approval was granted by the institutional review board, and the trial was conducted in accordance with the Declaration of Helsinki. Prior to enrolment, all participants provided written informed consent, including permission for data publication. All enrolled patients underwent standardised radiofrequency ablation at 55 W. Patients scheduled for great saphenous vein (GSV) ablation, with or without adjunctive Trivex-assisted phlebectomy, were eligible for inclusion. Patients with pre-existing venous thrombosis or ongoing anticoagulant therapy were excluded. Ultimately, 298 eligible patients were randomly assigned into two groups. A received oral rivaroxaban 10 mg daily for five consecutive days postoperatively, while Group B received standard care comprising early ambulation and compression therapy.
All participants were followed up for 1 month, during which subcutaneous induration, pain, hematoma, lower limb edema and ultrasonographic findings were recorded.
Ablation procedure
All patients received standardised radiofrequency ablation under consistent operative conditions.
Preoperative evaluation
All patients underwent duplex ultrasonography and D-dimer testing prior to surgery. High ligation of the GSV was performed if the SFJ diameter exceeded 10 mm.
Radiofrequency ablation
Before the procedure, written informed consent was obtained from all participants. Because many patients in this study require phlebectomy so procedures were carried out under spinal anaesthesia with patients placed in the supine position. Under ultrasound guidance, a 6F vascular sheath (Midrary, China) was introduced into the saphenous vein. The tip of the ablation catheter was positioned 2.0 cm distal to the SFJ. Next, tumescent anaesthesia (1 mg adrenaline in 3000 mL saline) was delivered along the vein using a 20-gauge needle under ultrasound guidance. Once correct catheter positioning was confirmed (ECO, China), radiofrequency energy (55 W) was applied for 6 s while the catheter was gradually withdrawn. For patients with varicose tributaries, adjunctive phlebectomy was performed using the TRivex system (inaVein, LLC, USA) following ablation. Post-surgery, elastic bandages were applied first, followed by graduated compression stockings. Moreover, early ambulation was actively encouraged. In addition, one group received rivaroxaban 10 mg daily for five consecutive days, while the other group received foundational DVT prophylaxis measures, including early ambulation, ankle pump exercises, and avoidance of unnecessary dehydration therapies, while excluding the use of anticoagulant medications. All patients were followed for 1 month, during which pain, subcutaneous haematoma, induration, lower limb swelling, and ultrasound findings were systematically recorded.
EHIT severity was classified into five grades according to the criteria proposed by Kabnick et al.
12
(Figure 1). Furthermore, DVT was categorised as either proximal or distal. Proximal DVT affected the femoral or popliteal veins, while distal DVT involved the posterior tibial, gastrocnemius, or peroneal veins. Classification of EHIT. Level 0: Thrombosis is confined to the superficial vein, with no extension to the saphenofemoral junction (SFJ). Level 1: Thrombosis extends to the SFJ but does not involve the femoral vein. Level 2: Thrombosis partially extends into the femoral vein, involving less than 50% of the lumen. Level 3: Thrombosis extends into the femoral vein, with luminal involvement exceeding 50%. Level 4: Thrombosis results in complete occlusion of the deep vein.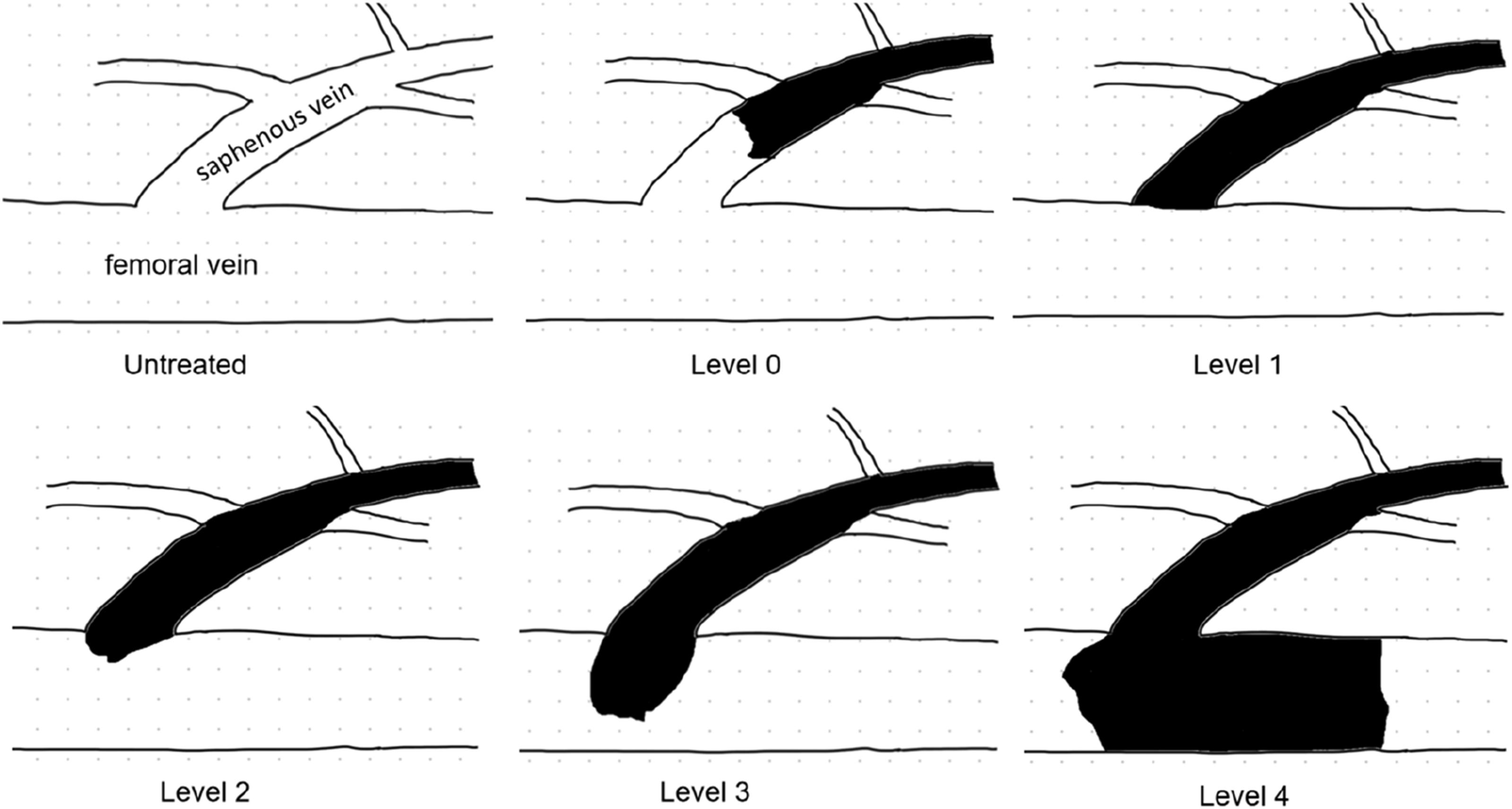
Statistical analysis
Patient characteristics and outcomes were presented as mean ± standard deviation (SD) for continuous variables and as counts and percentages for categorical variables. Between-group comparisons were performed using the chi-square test for categorical variables and the independent t test for continuous variables. All statistical analyses were conducted using PASS version 20.0 (NCSS, USA). A p-value <.05 was considered statistically significant.
Result
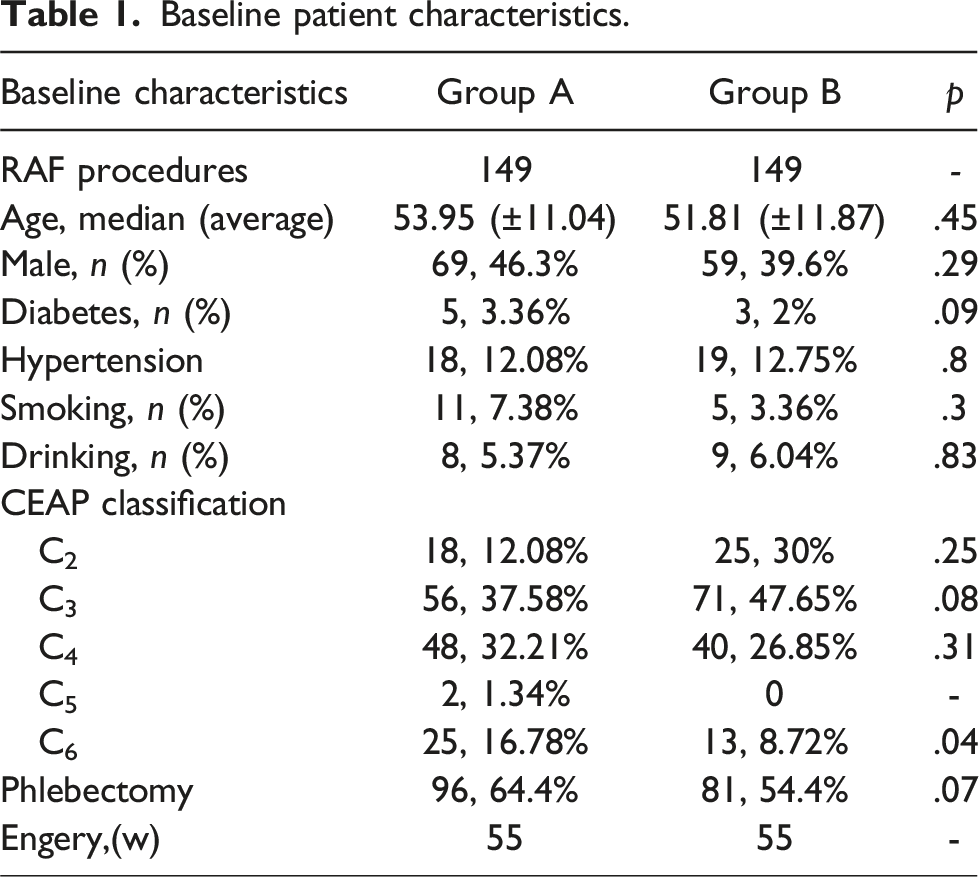
Baseline patient characteristics.
Outcome data.
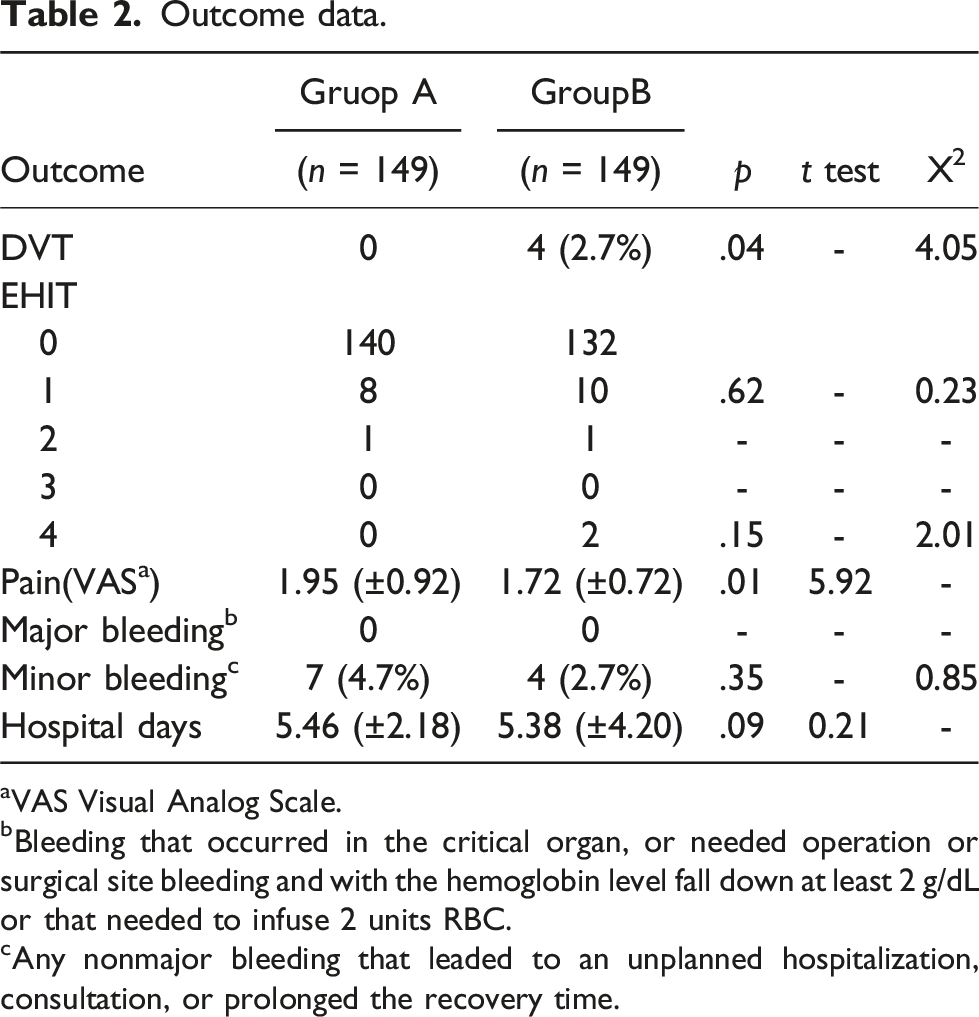
aVAS Visual Analog Scale.
bBleeding that occurred in the critical organ, or needed operation or surgical site bleeding and with the hemoglobin level fall down at least 2 g/dL or that needed to infuse 2 units RBC.
cAny nonmajor bleeding that leaded to an unplanned hospitalization, consultation, or prolonged the recovery time.
Distribution of DVT cases in group B.

CFV: Common femoral vein, CIV: Common iliac vein, EIV: external iliac vein, PV: Popliteal vein, PTV: posterior tibial vein, PerV: peroneal vein, CMV: calf muscular veins.
Discussion
This study evaluated the efficacy and safety of rivaroxaban in preventing DVT following RFA for lower extremity varicose veins. Rivaroxaban prophylaxis was initiated immediately after the procedure. Notably, no DVT or major bleeding events were observed in Group A, indicating that rivaroxaban may offer effective and safe thromboprophylaxis, consistent with its well-documented efficacy in orthopedic surgeries. 13
Varicose veins of the lower extremities are highly prevalent, affecting 2–73% of the global adult population. Consequently, the demand for effective and minimally invasive treatment options continues to grow. Compared with conventional surgery, minimally invasive techniques offer distinct advantages, including reduced tissue trauma, faster recovery, and lower perioperative blood loss. Moreover, previous studies have demonstrated that minimally invasive techniques achieve clinical outcomes comparable to those of conventional surgery. 5 Currently, thermal ablation has become one of the most widely used minimally invasive techniques, owing to its efficient closure of incompetent veins via thermal effects on venous walls. However, thermal ablation carries a potential risk of post-procedural DVT, particularly at the saphenofemoral junction. 7 Furthermore, emerging evidence indicates that the presence of varicose veins itself may independently increase the risk of DVT and other venous thromboembolic events. 14 Therefore, administering short-term prophylactic anticoagulation following thermal ablation represents a rational and evidence-supported strategy to mitigate thrombotic risk.
Rivaroxaban has demonstrated superior efficacy compared to LMWH in preventing DVT after knee and hip arthroplasty, without significantly increasing bleeding risk. 15 Recent evidence suggests that pharmacologic thromboprophylaxis after varicose vein surgery may increase bleeding risk if extended beyond 10 days, indicating that the optimal duration likely falls between 5 and 10 days. 16 Therefore, in this study, rivaroxaban was administered for 5 days following ablation, and no unexpected bleeding events were observed. Moreover, patient adherence to oral rivaroxaban was excellent throughout the study period. Previous studies reported a low incidence of EHIT, ranging from 0% to 1%, which led to skepticism about the necessity of routine pharmacologic prophylaxis.17,18 However, more recent data indicate that EHIT incidence in the absence of anticoagulation ranges from 1.7% to 6%,19–21 which is consistent with the 2.7% incidence observed in this study. Considering the high prevalence of varicose veins, the absolute burden of EHIT remains clinically meaningful. DVT represents a serious complication of ablation procedures, with potential consequences for patients’ health, occupational productivity, quality of life, and financial well-being. In conclusion, the findings support the rational use of rivaroxaban for DVT prevention after varicose vein surgery, given its favourable safety profile and patient-centered benefits.
This study has some limitations that warrant consideration. First, some cases of EHIT may have been missed during the follow-up period. Multiple procedural factors are known to influence EHIT occurrence, including CEAP classification, mean vein diameter, frequency of phlebectomy, and the follow-up intervals.19,20,22 Notably, the optimal positioning of the ablation fiber relative to the SFJ remains uncertain. Some studies suggest that positioning the fiber 2–3 cm distal to the SFJ may help reduce EHIT risk. However, newer-generation radial laser fibers allow precise and targeted ablation at the SFJ. In fact, many experienced vascular surgeons advocate direct ablation at the SFJ to minimise varicose vein recurrence. In this study, we followed a standard protocol maintained a 2 cm distance from the SFJ. Further studies with larger cohorts are needed to validate this clinically relevant issue.
Secondly, our study was performed at a single center, conducting it across multiple centers with a larger sample size could potentially provide more precise results.
Thirdly, our study limitation of omitting VTE risk assessment scores. Implementing such scoring criteria to stratify patient groups more precisely could potentially yield more robust findings.
Conclusions
This study indicates that short-term rivaroxaban (10 mg daily for 5 days) is a safe and effective option for preventing EHIT and DVT following RFA, with or without phlebectomy. No unexpected bleeding events were observed, and patient adherence was excellent. These findings support the use of rivaroxaban for thromboprophylaxis in this setting; however, larger multicentre studies are warranted to further validate these results.
Footnotes
Acknowledgements
I am grateful to the entire Collective of Departments for giving me access to the resources that made this project possible. A big thanks goes out to Mr Luo in particular, for the opportunity to work together on such a meaningful project. Finally, I turn to all my partners, without who I would never have been able to finish this dissertation.
Declaration of Conflicting interest
The authors declare no conflict of interest with respect to the research, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grant Dongguan Social Development Technology Project (20211800901432).
Ethical approval
The study protocol was reviewed and approved by the Ethics Committee of DongGuan Tungwah Hospital (Approval No. 2021-KY-024). All participants signed written informed consent forms.
Guarantor
LG.
Contributorship
Conception and design: Luo G, Zhag TS; Analysis and interpretation: Luo G, Zhang TS, Ge XC, Ye PQ, Yan N; Data collection: Zhang TS, Ge XC, Ye PQ, Yuan ZD, Zhong YB, Huang ZP; Writing the article: Zhang TS; Critical revision of the article: Luo G, Yan N; Final approval of the article: Luo G, Zhang TS, Yan N; Statistical analysis: Zhang TS; Overall responsibility: Luo G.
