Abstract

Keywords
Introduction
The term chronic venous disease (CVD) represents symptoms and signs that manifest in relation to venous insufficiency. Clinical manifestations may be classified by the Clinical-Etiological-Anatomical-Pathophysiological (CEAP) classification system, 1 with clinical C0 representing no signs related to CVD and C6 referring to the most severe manifestation of ulceration.
Most patients with CVD will also have C1 disease, referring to telangiectasias and reticular veins. While isolated C1 disease often does not result in significant symptoms that are associated with more severe stages of disease, it can still have an impact on patients’ psychological quality of life due to cosmetic issues. For the venous specialist with an interest in cosmetic procedures, microsclerotherapy of telangiectasia and reticular veins is therefore the most common procedure within their scope of practice.
While clinical practice guidelines have provided an overview of treatment options for C1 disease, these have often focused more on C2 to C6 disease when considering the technical aspects of phlebological interventions. Few have addressed the technical aspects of microsclerotherapy.2–4 Given that this is not a simple injection treatment, clinicians must have adequate phlebological training, and this document aims to provide recommendations and technical considerations when offering treatment to patients with C1 disease.
Management recommendations
Prior to intervention, all patients should undergo a comprehensive clinical assessment to identify indications and contraindications for microsclerotherapy. 5 Duplex ultrasound remains the ‘gold standard’ in the diagnosis of all CVD stages. If C1 disease is not accompanied with symptoms consistent with venous disease, duplex ultrasound is not required and investigation of abnormal venous haemodynamics with duplex ultrasound should be reserved for symptomatic patients only. After identifying any underlying incompetent veins and/or proximal reflux, it is recommended that surgical correction of abnormal venous haemodynamics is performed before microsclerotherapy is offered. However, should the identified underlying venous reflux affect an area not involved with C1 disease, elimination of reflux may not be necessary. In these cases, a shared decision-making approach to correcting this incompetence should be employed prior to any offered intervention.
Both liquid and foam sclerosants can be used for microsclerotherapy, with few studies performed to directly compare their relative effectiveness.6–9 Patients should be treated while lying flat and methods to better visualise the feeding vessels should be utilised (e.g. loupes, polarised LED systems, and ultrasound).10,11 Short 25-32G needles should be used, and correct needle puncturing and position can be confirmed by observing drawback of venous blood into the syringe. Injections should be performed in the direction from larger veins to smaller telangiectasias. Disposable syringes with smooth glide plungers should be used to help with maintaining low pressures and consistent flow while injecting, with cautious monitoring advised to ensure intravascular injection and avoid extravasation of sclerosant.
Recommended concentrations for sclerosants used.
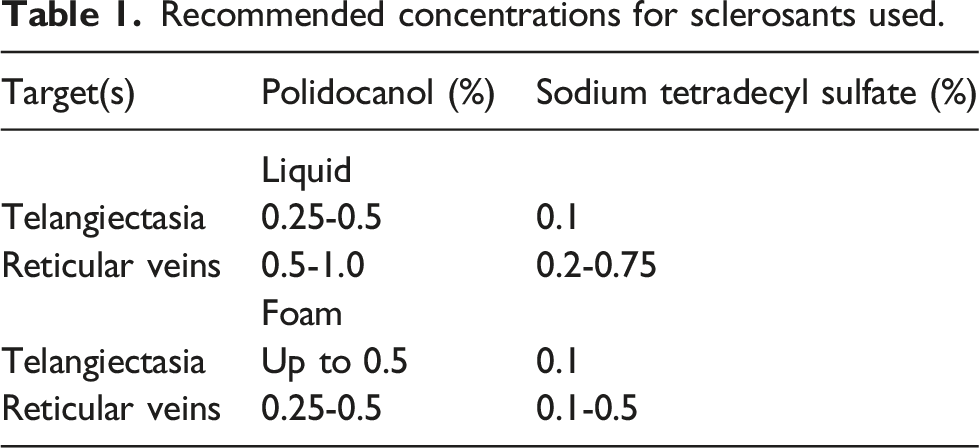
Post-procedurally, it is recommended that class 2 elastic compression (23-32 mmHg) be provided to all patients undergoing microsclerotherapy. There is evidence that post-procedural compression improves clinical outcomes and reduces risk of hyperpigmentation. 17 If indicated, clinicians should also consider offering microthrombectomy to further help reduce the risk of hyperpigmentation.
Discussion
This article provides a one-page clinical practice guideline on microsclerotherapy. It is part of a series of publications for the International Union of Phlebology (UIP) One-Page Guidelines which are aimed at ensuring that patients with venous disease receive timely and appropriate care based on current best evidence and expert consensus. This one-page guide aims to provide an overview of technical considerations that have not been previously addressed in other clinical practice guidelines.2–4
Clinicians are advised to consider performing duplex ultrasound for patients with asymptomatic C1 disease should underlying venous incompetence be suspected. This is supported by evidence from multiple studies which showed underlying venous reflux in the saphenous trunks and/or varicose tributaries in patients presenting with C1 disease. 18 Telangiectasias and reticular veins that are resistant to sclerotherapy have also been shown to be related to a connected perforating vein. 19 However, critics have argued that reflux alone and gravitational effects may not be able to explain all telangiectasia, especially those located proximal to the distal localisations of other CVD skin changes. While it is logical that underlying reflux should be treated to improve success rates of microsclerotherapy, work is clearly needed to further delineate the pathophysiology of C1 disease to clarify if routine duplex scan for underlying reflux is required in all presentations.
This guideline aims to provide clinicians with recommendations on technical considerations to improve C1 treatment success rates. However, while the success of treatment of symptomatic CVD can be measured by the use of both subjective (e.g. reduction in pain) and objective measures (e.g. reduction in swelling/calf diameter, obliteration of vein), C1 disease tends to be asymptomatic with treatment sought for cosmetic reasons. Studies have used various means to determine ‘success’, including observer ratings of images, 20 but these are usually subjective measures which makes direct comparisons between studies difficult.
Once these objective measures for C1 treatment success have been determined, further trials should be performed to compare the relative success and efficacy of the various forms (liquid vs foam), type (polidocanol vs STS), and concentrations. While both foam and liquid sclerosants have been used in microsclerotherapy for C1 disease, limited direct comparisons exist regarding their effectiveness in this specific patient population. Optimisation of concentrations to achieve optimal outcomes while minimising adverse events should also be performed in these studies, ultimately improving treatment outcomes and patient satisfaction.
Footnotes
Author contributions
K.P. and A.H.D. conceptualised the design of the short report and one-page guideline.
E.S. performed the literature review and formulation of the recommendations.
M.T. contributed to the formatting and layout of the one-page guideline ( The one-page guideline.![]()
All authors reviewed the short report prior to submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
Professor Alun Davies on behalf of the UIP.
