Abstract
Deep vein thrombosis (DVT) of the lower extremities is one of the most common peripheral vascular diseases, with significant complications and sequelae. Metabolomics aims to identify small molecules in biological samples. It can serve as a promising method for screening compounds that can be used for early disease detection, diagnosis, treatment response prediction, and prognosis. In addition, high-throughput metabolomics screening can yield significant insights into the pathophysiological pathways of DVT. Currently, the metabolomic profiles of DVT have yielded inconsistent expression patterns. This article examines the recent advancements in metabolomic studies of DVT and analyzes the factors that may influence the results.
Introduction
Deep venous thrombosis (DVT) is the abbreviation for a condition in which aberrant blood clots form in the lumen of the deep veins, resulting in luminal constriction, obstruction, and extensive collateral venous return. DVT is a typical peripheral vascular lesion in vascular surgeries, causing swelling, pain, ulceration, necrosis, disability, secondary pulmonary embolism (PE), and even mortality in severe cases.1–3 The risk of PE has grown due to the rising incidence of DVT. 4 Venous thromboembolism (VTE) is a collective term for DVT and PE. Consequently, early detection and prevention of VTE remains a crucial public health priority as it is a common cause of hospital-acquired death. 5 However, VTE is not a “pure disease” in that it is subject to interactions with the environment and metabolism, thereby necessitating a thorough understanding of the pathophysiological features of the disease. 6 VTE is a multifactorial disease subject to metabolic and environmental interactions. Thus, primary prevention requires early disease detection. 7 An early and accurate diagnosis of DVT is of the utmost importance. Currently, DVT is primarily diagnosed using a combination of clinical symptoms and signs, imaging, and laboratory investigations. Color Doppler ultrasound has high sensitivity and accuracy, making it an essential diagnostic instrument for DVT.8,9 DVT diagnostic tests consist of the sequential application of clinical decision rules and the D-dimer test,10,11 which, due to their only moderate sensitivity for DVT, 6 make them supplementary diagnostic instruments. In the early stages of thrombosis or when thrombosis has not yet formed, the above-mentioned tests and screening indicators may produce negative results, frequently delaying the correct diagnosis and leading to the progression of the disease without any preventive or therapeutic measures. This, in turn, delays the start of treatments until the middle and late stages of the disease, ultimately affecting disease prognosis. Determining a predictive diagnostic method for DVT and gaining a deeper understanding of the disease's pathogenesis will help to develop more effective preventative and therapeutic measures, especially as one of the most pressing concerns regarding DVT is diagnostic latency.
Several biomarkers, such as microRNA,12,13 selectin,14,15 interleukin-1, 16 interleukin-8, 17 particulates, 18 and other inflammatory markers, 19 have been investigated as predictive diagnostic tools for DVT. However, no identified single substance is sensitive or specific enough to predict the diagnosis of DVT.
Important in the field of biology, metabolomics is the study of the types and quantities of metabolites, or endogenous metabolites, as well as their varying patterns in response to external disturbances. Metabolomics focuses on the metabolic pathways of endogenous metabolites in the whole organism, organ, or tissue. Metabolomics also assesses metabolic changes influenced by intrinsic or extrinsic factors and their change over time. Metabolite analysis of biological fluids has been suggested to more accurately reflect the pathophysiological state of organisms.20,21 Metabolomics includes techniques such as nuclear magnetic resonance (NMR), gas chromatography-mass spectrometry (GC-MS), liquid chromatography-MS (LC-MS), ultra-performance liquid chromatography-MS (UPLC-MS), flow-injection assay-MS (FIA-MS), and capillary electrophoresis-MS (CE-MS).22,23 The metabolome is one of the most detailed and predictive sources of phenotypic information, and assessment of its relation to pathogenesis focuses on changes in small-molecule metabolites that are the substrates and products of various metabolic pathways during disease progression. 24 The metabolome has been utilized to diagnose, identify improved disease biomarkers, and to comprehend the pathogenesis of numerous diseases, including cancer and diabetes.25,26 Additionally, it is used to assess the efficacy of drug therapy and to screen for drug toxicity. 27 Metabolomics makes it possible to detect tens of thousands of metabolites simultaneously in cells, organs, tissues, and biological fluids.28,29 The most commonly used biological fluids in metabolomics are serum and urine, and monitoring the levels of specific metabolites in these fluids has become an important method for early disease detection. 30 The use of metabolomics technology has emerged as a means to better understand disease pathophysiology, 6 and analysis of metabolites in DVT may lead to a better understanding of the disease process, the discovery of biomarkers of the disease, and help in developing an accurate clinical diagnosis. Consequently, the purpose of this article is to provide a summary of the metabolomic profile of DVT. Specifically, this review looks at published studies that have studied the link between certain metabolites and DVT. We then summarize biomarkers and relevant metabolic pathways in DVT patients, as well as factors that may affect the results of the studies.
Methods
Interventions
How metabolomics data changes when experiencing DVT forms our area of research. The analysis method used in the reviewed studies falls under the metabolomics field. Where possible, metabolites linked to DVT should be provided in papers that are suitable for inclusion in the review. Due to the small number of pieces of literature included, there was no time window set for sampling after the DVT presentation.
Search strategy
Database search strategy.

No language restrictions exist. Only case-control studies and animal experiments were considered. We excluded letters to the editor, editorials, reviews, expert opinions, any form of review, cohort studies, case reports, interventional trials, and cross-sectional studies. This review intends to show the metabolomics analyses change when observed in DVT patients or animal models without any means of intervention. Consequently, only case-control investigations were included.
Data extraction
Two literature searches were performed independently, and data was extracted in two stages, with the first stage retrieving literature based on the search formula and the second stage performing literature screening, data extraction, and cross-checking of the selected literature. If disagreements arose, they were resolved through common-issue discussions or by referring them to a third researcher. For literature screening (using EndNote software), duplicate or irrelevant works of literature were eliminated first, followed by a primary screening by reading the titles and abstracts, and then a double screening by reading the full text to select eligible studies. Figure 1 depicts the literature evaluation procedure. Literature selection methodology.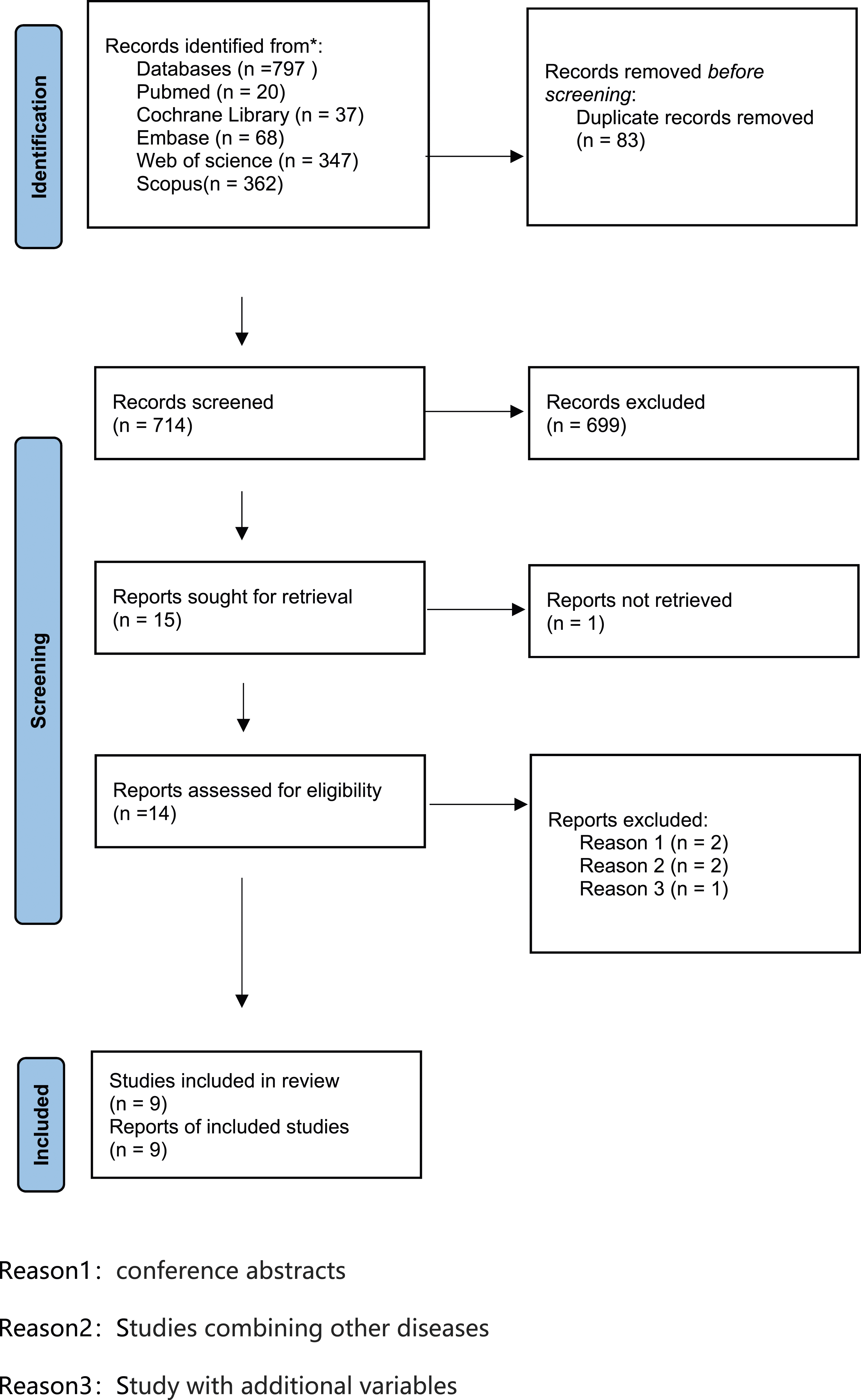
Data summary
All metabolite data collected from the analyses were extracted from each investigation, and data on study characteristics and metabolomic alterations were extracted for each included study. The principal components of data extraction included the first author’s name, country, study design type, assay content, assay method, metabolites involved, sample size, statistics, Body mass index (Kg·m−2), mean (SD), comorbidities, and medications.
Patient and public participation
As this was a narrative review, neither patients nor the general public were involved in any way.
Results
This review’s literature inquiry was conducted through March 30, 2023. The flowchart of study selection and ultimate inclusion is depicted in Figure 1. Exclusions included duplicates (82 studies), a systematic review, 7 two conference abstracts,31,32 two studies combining other diseases,33,34 and one study with additional variables. 35
Study characteristics.
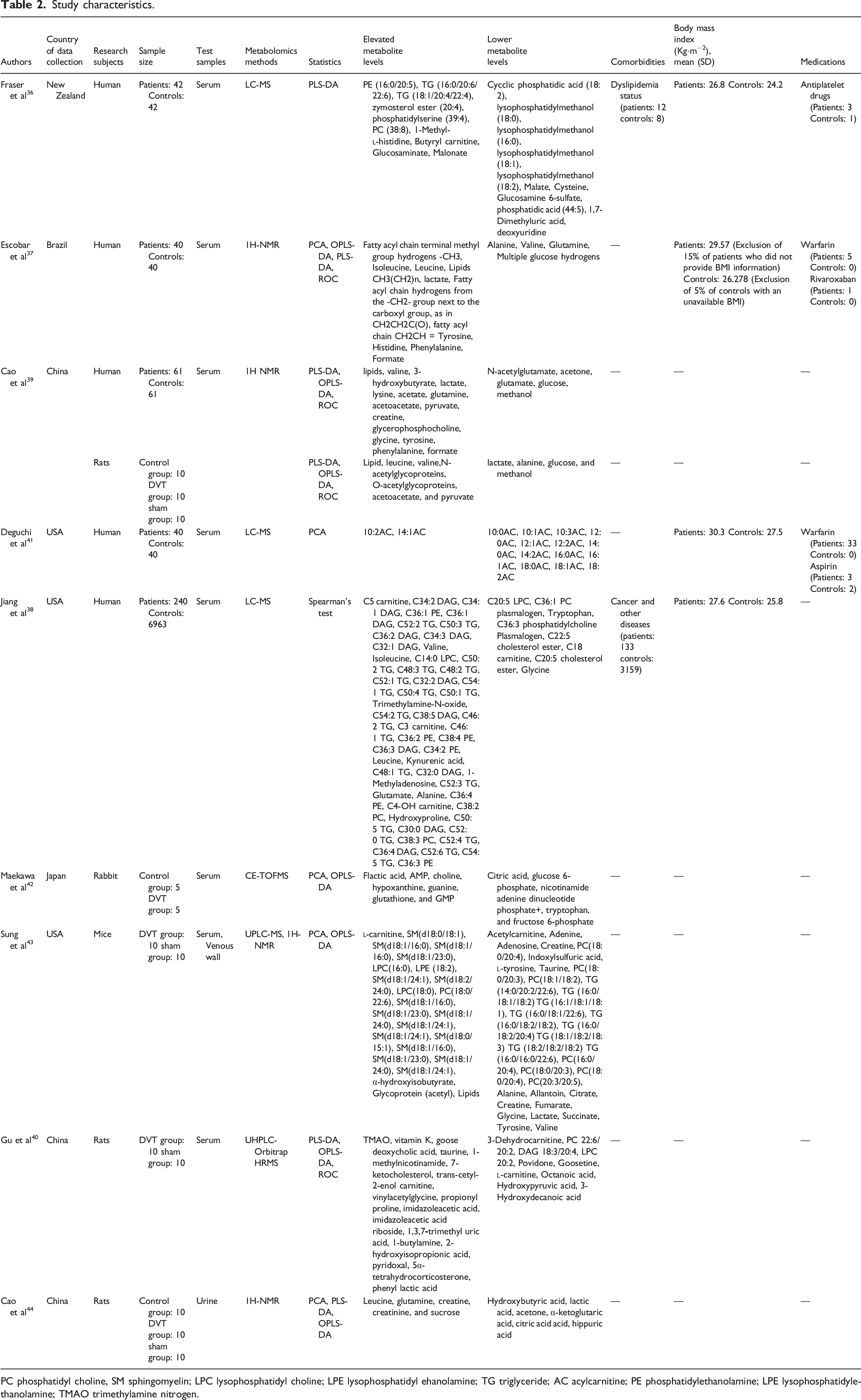
PC phosphatidyl choline, SM sphingomyelin; LPC lysophosphatidyl choline; LPE lysophosphatidyl ehanolamine; TG triglyceride; AC acylcarnitine; PE phosphatidylethanolamine; LPE lysophosphatidylethanolamine; TMAO trimethylamine nitrogen.
Metabolic pathways involved in the study.

Flowchart for metabolomics-based investigations on DVT patients or animals. DVT, deep vein thrombosis; LC-MS, liquid chromatography-mass spectrometry; NMR, nuclear magnetic resonance. Adapted from “metabolomics general overview,” by BioRender.com (2023). Retrieved from https://app.biorender.com/biorender-templates.
Deguchi et al.’s non targeted investigations of acylcarnitines in human serum revealed low levels of several long-chain acylcarnitines in the VTE groups.
41
While other studies have provided a more significant number of predictive metabolites using non-targeting methodologies, multiple studies have repeatedly reported elevated levels of phosphatidylcholine-containing metabolic species.36,38 Regarding the expression levels of carnitine,36,38,41 alanine,37–39 choline,36,38,39 and other metabolites, diverse opinions exist. Fraser et al. identified 21 plasma metabolites and calculated the area under the curve for the combination of 21 plasma metabolites to be 0.92 (p = .00174, sensitivity = 0.971, specificity = 0.857), providing compelling evidence for the relationship between metabolites and venous thrombosis.
36
Jiang et al. discovered abnormalities in multiple metabolites, and a particularly abundant metabolite in DVT patients was identified as diacylglycerols. C5 carnitine was found to be substantially associated with the development of VTE after controlling for common influencing factors (age, gender, and disease cause), confirming the findings of Deguchi et al.’s study.
38
Escobar et al. identified a number of metabolites, including lipids, glucose, alanine, glutamine, and valine. By calculating the area under the curve values, it was determined that, of the many metabolites with differences, glutamate and valine were the most significant ones. Additionally, by comparing analyses, it was discovered that the ratio of glucose/lactate and branched chain amino acids (BCAAs)/alanine was altered, which is closely related to their involvement in energy production.
37
Cao et al. identified 20 distinct metabolites in DVT patients and seven metabolites that were altered in both rats and patients. They concluded that serum 1H NMR metabolomics could be used for the clinical diagnosis of DVT and identified several metabolic pathways induced immediately after thrombosis, such as changes in carbohydrate metabolism, lipid metabolism, and amino acid metabolism.
39
Our initial review of metabolic pathway abnormalities and some associated metabolites in DVT patients is shown in Figure 3. Summary of metabolic pathway alterations in deep vein thrombosis patients. Increased amounts of metabolites are indicated by the color red.TCA cycle = tricyclic acid cycle. Adapted from “Cancer Metabolism in Nutrient-Replete Conditions,” by BioRender.com (2023). Retrieved from.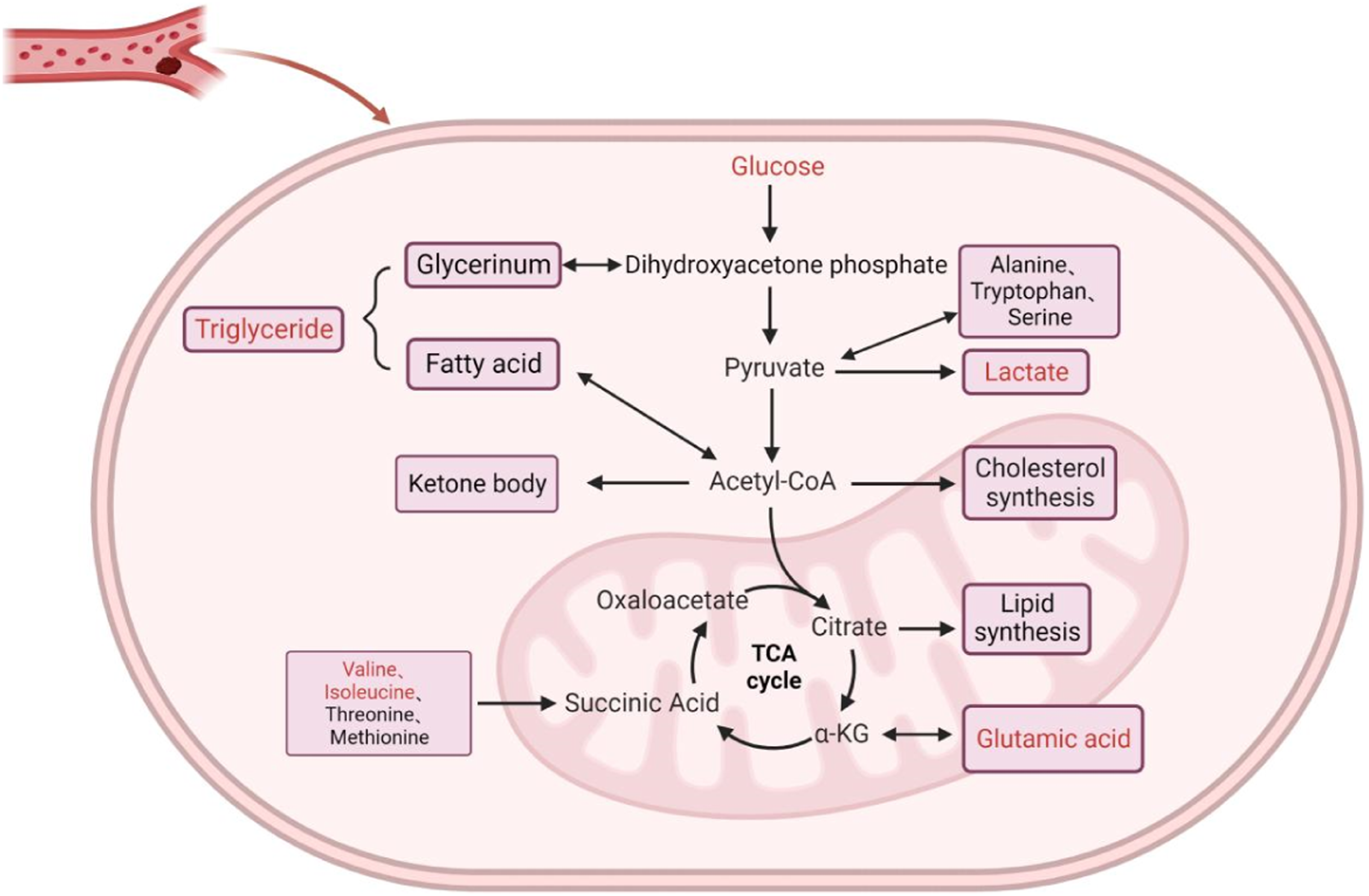
Several animal studies have found a significant correlation between lactate and DVT, although the results are inconsistent. Maekawa et al. identified lactate as the most prevalent metabolite in a rabbit model of thrombosis, suggesting that elevated levels of lactate may indicate active glycolysis of erythrocytes or other thrombus cellular components. 42 Sung et al. identified that DVT mouse sera contained varying concentrations of adenosine, adenine, and intermediates of the tricyclic acid cycle (TCA), including citrate, succinate, and fumarate. Several metabolites, including acetylcarnitine, adenosine, and ceramide, were also identified in the venous wall; however, the serum metabolites were not consistent with the metabolites of the venous wall. 43 Although less studied, in the urine of DVT rats, Cao et al. found that 3-hydroxybutyric acid, lactic acid, acetone, ketoglutaric acid, citric acid, and hippuric acid were among the metabolites with downregulated levels compared to control rats. 44 Despite the differences between human serum analysis and animal studies, animal studies provide more metabolites and novel research ideas and indicate exciting differences based on analytic samples, such as the differences in metabolites present in the venous wall of the DVT model. Consequently, future research can combine the detection of metabolites in serum, urine, and the venous wall to increase the sensitivity and specificity of metabolite detection.
Discussion
Metabolomic characterization of DVT
Globally, DVT has a high prevalence and a negative impact on quality of life. 45 In this narrative review, amino acids and lactic acid were discovered to be the most prevalent chemical subclasses associated with DVT. Purine metabolism, carbohydrate metabolism, lipid metabolism, amino acid metabolism, and energy metabolism are the primary pathways implicated in both DVT patients and animals. Our review contributes to guiding future research planning, specifically studies that seek to advance our understanding of the DVT disease pathogenesis and indicators from a metabolomics perspective. Further research is required to improve our ability to diagnose DVT in clinical settings.
According to current investigations, amino acids are the most prevalent subclass of compounds detected in DVT metabolomic studies. However, it is worth noting that in the various reviewed studies, amino acids such as alanine, valine, and glutamate have been differentially detected.
Alanine is a non-essential amino acid that can be synthesized in organisms through a variety of pathways. In particular, it is converted to pyruvate via the gluconeogenesis pathway, and pyruvate can also be converted to alanine. 46 Alanine is both a nitrogen (N) donor and a nitrogen (N) acceptor obtained via transamination of pyruvate with various amino acids. 47 Several studies have demonstrated that serum levels of alanine metabolism in DVT patients and rats are inconsistent and that changes in alanine levels are closely related to its participation in multiple metabolic pathways,37–39,43 including gluconeogenesis, bile acid synthesis, fatty acid synthesis, and amino acid metabolism. There are few studies on alanine and its relationship with DVT, but as an essential substance in multiple metabolic pathways, it should be seriously considered.
Valine is a BCAA that promotes normal development, repairs tissues, regulates blood glucose, supplies energy to the body, stimulates muscle protein synthesis, and inhibits muscle catabolism. 48 Ingestion of BCAAs has been shown to substantially increase human platelet activity and arterial thrombosis in mice, with the valine catabolic pathway playing a significant role in BCAA-induced platelet activation. 49 The thrombosis-specific serum metabolite had a valine AUC of 0.73, making it one of the most influential metabolites. 37 Although the sensitivity and specificity of the detection assay alone are not very high, leucine and isoleucine are other BCAAs, 50 which were also found to be elevated in DVT patients. Furthermore, BCAAs have been used as independent predictive molecules to distinguish cerebral vein thrombosis patients from healthy controls; 51 thus, a combined assay to detect BCAAs may help to identify a panel of candidate biomarkers for the diagnosis of DVT.
Glutamate, an acidic amino acid, is one of the most abundant amino acids and plays a crucial function in nutrition, metabolism, and signal transduction. 52 In addition to playing a crucial role in hemostasis and thrombosis, glutamate has been identified as a regulator of platelet activation; activated platelets release glutamate, and glutamate increases agonist-induced platelet activation. 53 Not surprisingly, glutamate was identified as one of the most influential metabolites in distinguishing thrombogenic groups from controls. 37
As a medically essential amino acid, carnitine contributes to energy production and fatty acid metabolism. 54 Studies on humans and animals indicate that carnitine levels are elevated in the serum of DVT groups. Carnitine also binds to fatty acids to generate acylcarnitine, which is then shuttled into mitochondria to participate in oxidative metabolism. 55 Deguchi et al. found in a targeted study of acylcarnitine with low levels of several long-chain acylcarnitines that acylcarnitine inhibits factor Xa and reduces venous thrombosis. 41 This provided a solid foundation for subsequent research, and it can be hypothesized that DVT causes disruption of the pathway that converts carnitine to acylcarnitine and that perturbation of this pathway inhibits the formation of acylcarnitine, resulting in a decrease in carnitine accumulation and acylcarnitine production. 43 With consistent results, Sung et al. identified that carnitine was significantly more abundant (a 67-fold change) in the sera of DVT animals.
Lactic acid is a carboxylic acid compound that has been mistakenly believed to be the end product of glycolysis. 56 It participates in numerous biochemical processes, such as energy regulation, wound healing, and muscle regeneration after ischemia.57–59 Lactate decreases anticoagulation time similarly, 60 and Maekawa et al. identified that lactate was the most prevalent metabolite in venous blood and thrombi and that elevated levels of lactate may indicate active glycolysis of erythrocytes in the cellular component of thrombi. 42 Lactate exacerbates the inflammatory response,61,62 and inflammation has been considered a risk factor for DVT and thrombosis, wherein the resulting damage to the venous wall can result in systemic inflammatory responses.63,64 The presence of elevated lactate in DVT could imply that DVT is an inflammatory disease; indeed, lactate is a regulator of macrophage metabolism, and lactate levels are markedly elevated during inflammation.65,66 More definitive studies are required to confirm the specific metabolic mechanism of lactate in DVT, but it is expected to be a valuable therapeutic target as a plasma biomarker of the disease.
Lipids, including triglycerides and phospholipids, are one of the essential nutrients required by the body, as they provide the body with energy, provide essential fatty acids, and are a component of the body’s cellular tissues. Lipids are associated with inflammation; lipid mediators play an essential role in the resolution of acute inflammation, 67 and the production of lipid imbalances leads to chronic inflammation and exacerbates this condition. 68 The majority of chronic pathologies that are currently understood are typified by a persistent surge in inflammation. It has been observed that there is no significant correlation between the duration of symptoms and the acute or chronic inflammatory response in DVT. 63 Therefore, it is crucial to recognize the role that lipids play in this persistent inflammatory response in DVT. Lipid changes were observed in DVT, and testing of human and animal DVT sera revealed elevated lipid levels.37,39,43 Elevated lipid levels have been suggested to be linked to an increased risk of developing venous thrombosis. 69 Nevertheless, a meta-analysis concluded that there was insufficient evidence linking significantly increased lipid levels to the risk of venous thrombosis. 70 Lin et al. concluded from a genetic standpoint that the three classical lipids (low-density lipoprotein (LDL), high-density lipoprotein (HDL), and triglycerides (TGs)) had no significant causal relationship with DVT. 71 Consequently, the relationship between lipids and DVT is presently ambiguous and contradictory across studies, and additional research is required to examine the relationship between specific lipid substrates and DVT.
Despite the discourse of lipid levels and the risk of DVT, according to epidemiology, elevated TG levels are a biomarker for cardiovascular risk. 72 The relationship between TGs and DVT is currently the subject of an increasing number of studies, with higher TG levels being a risk factor for the development of lower extremity DVT. 73 Similarly, a meta-analysis revealed that patients with venous thrombosis had higher mean TG levels than controls. 74 In our included studies, TGs were also among the metabolites with elevated serum levels in DVT patients.36,38 Current research suggests that TG contributes to venous thrombosis by accelerating the activation of prothrombin, factor X, and factor VII via its influence on the coagulation and fibrinolytic systems.75,76 Nevertheless, studies into the links between TGs and DVT should continue.
In general, patients with elevated blood glucose or diabetes have elevated plasma procoagulant levels and reduced fibrinolytic capacity, and the induction of altered coagulation factor quantification can cause a hypercoagulable environment and increase the risk of thrombosis. 77 Indeed, a higher blood glucose concentration is considered a risk factor for venous thrombosis. 78 Hyperglycemia enhanced the incidence of DVT in patients with traumatic lower extremity fractures, revealing a correlation between blood glucose and venous thrombosis. 79 In contrast, a cohort study of a large sample from northern Sweden found no evidence of an association between diabetes and VTE risk. 80 Both elevated fasting glucose levels in the non-diabetic population and self-reported diabetes were not associated with an increased risk of venous thrombosis, according to a MEGA analysis. 81 Patients with DVT had both elevated and decreased blood glucose levels, as determined by metabolite testing.36,39 Therefore, it is inconclusive whether blood glucose levels are associated with DVT, and future investigations into the extent of the contribution of blood glucose levels to the thrombotic milieu in large eligible populations should be considered.
Factors affecting the metabolic profile of DVT
Despite the fact that a number of studies have identified DVT metabolomic profiles, it remains challenging to obtain a standard set of serum metabolic markers for screening or diagnosis of DVT. In terms of both clinical parameters and experimental techniques, a variety of factors can influence the metabolomic profile. Age, gender, smoking status, BMI, the presence of other comorbidities, and heparin use are the primary clinical parameters. Patients with DVT are substantially older, more likely to be male, have a higher prevalence of comorbidities (i.e., cancers), are more likely to be overweight, and smoke significantly more cigarettes than the general population. 38 Additionally, BMI and estrogen use can affect thrombosis. 37 Therefore, when interpreting DVT metabolomic results, these factors should be thoroughly considered. Aspects of the experimental technique may depend on collection vessels, hemolysis, delay at room temperature and freeze-thaw cycles, collection time, storage conditions, etc. Metabolomics analyses (particularly MS) are sensitive to variability in sample preparation and handling; therefore, every metabolomics-based study should describe sample collection and handling in detail and design and implement standard operating procedures. Such methods are necessary for controlling and minimizing experimental variability. Furthermore, it is unknown whether the time of thrombosis has an impact on the results; thus, the time of thrombosis should be accounted for in studies in order to reduce experimental error. T1 Mapping MR can provide intrinsic and objective parameters of thrombus formation, allowing the clinician to ascertain the actual age of thrombus formation. 82 The combination of T1 Mapping and MR may enhance the precision of experimental results. Therefore, a comprehensive evaluation of the aforementioned factors during metabolomics analysis will increase the accuracy of the results.
Conclusions
Multiple metabolites were statistically correlated with DVT in this narrative review, with altered pathways in carbohydrate metabolism, lipid metabolism, amino acid metabolism, and energy metabolism all being linked to DVT. However, individual metabolites are poor predictors; their detection value increases when considered in conjunction with other biomarkers in a multifactorial model. Changes in metabolites contribute to a greater comprehension of DVT pathogenesis and disease screening, which may aid in the early detection of DVT disease. Notably, due to the lack of consistency among researchers in selecting and evaluating candidate biomarkers, it is rare to see studies that measure sensitivity and specificity. 83 Although a set of specific metabolites is typically identified in a single study, it appears difficult to obtain consistent biomarkers from multiple studies. Some studies have yielded inconsistent results, as metabolites such as glucose, alanine, valine, glutamine, lactate, and glycine have exhibited inconsistent performance across studies. Therefore, there is a need for comprehensive studies to evaluate the differentiating metabolites in the serum, urine, and venous walls of various organisms in order to identify a number of valid biomarkers for clinical applications.
Footnotes
Acknowledgments
We appreciate the Natural Foundation of Inner Mongolia Autonomous Region (Item number 2020MS08083), China, for funding this study. The mapping of this study was made possible by Mingliang Shi, who is also acknowledged by the authors. We would like to thank Prof. Meifeng Zhu for his contribution in touching up the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Natural Science Foundation of Inner Mongolia Autonomous Region (Item number 2020MS08083).
Ethical statement
Guarantor
Liu Yang.
Contributorship
Weiguang Jiang and Liu Yang devised the protocol, conducted a literature search, selected the studies, extracted the data, and drafted the initial draft. Yongkang Dang and Xuechao Jiang contributed to the incorporation of the studies, interpretation of the data, and revision of the manuscript. Lan Wu and Xiangyang Tong assisted with the refinement of the diagrams and tables, while Jianquan Guo and Yongtao Bao oversaw the entire manuscript. All authors have reviewed the manuscript and consent to its publication.
