Abstract

Introduction
Sclerotherapy has been used in the treatment of chronic venous disease (CVD) in its liquid form for more than 160 years. Referring to the chemical ablation of target veins using a sclerosant leading to its transformation into a fibrous cord, it has evolved significantly since its usage in treatments performed by Professors Pétrequin and Soquet, 1 with key developments including detergent sclerosants (e.g. sodium tetradecyl sulphate (STS), polidocanol (POL)) and foam sclerotherapy. Today, both liquid and foam sclerotherapy is among the recommended treatment options for both tributary and truncal veins in national and international guidelines.2–4
Sclerotherapy has seen a revival in popularity due to its ease of use relative to other modalities of CVD treatment. It is a flexible technique that can be used to treat a variety of superficial incompetent vessels ranging from telangiectasia and reticular veins to saphenous trunks. It can also be used to treat venous malformations and deeper veins such as pelvic varicosities. While sclerotherapy, if executed correctly, is an efficient form of treatment with minimal complications, it must not be simply regarded as a simple intravenous injection procedure. Clinicians need to be trained in phlebology and vascular sonography and must be aware of the indications and contraindications for using this treatment. Safe volumes for administration can also vary according to the target vessel and patient characteristics.
This article seeks to highlight the management recommendations from the International Union of Phlebology (UIP), 4 highlighting salient management decisions that clinicians must consider prior to administering sclerotherapy treatment. These include specific patient characteristics, target vessels, and specific techniques to optimise success and avoid complications.
Management recommendations
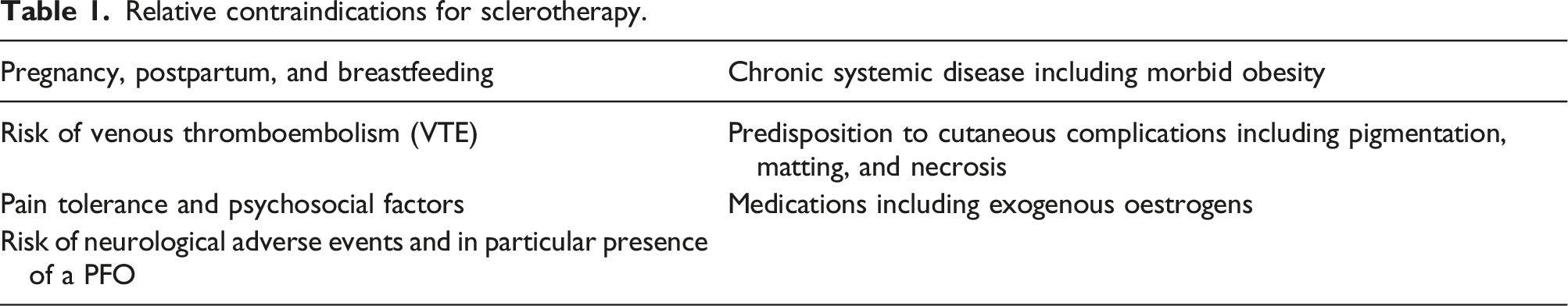
Relative contraindications for sclerotherapy.
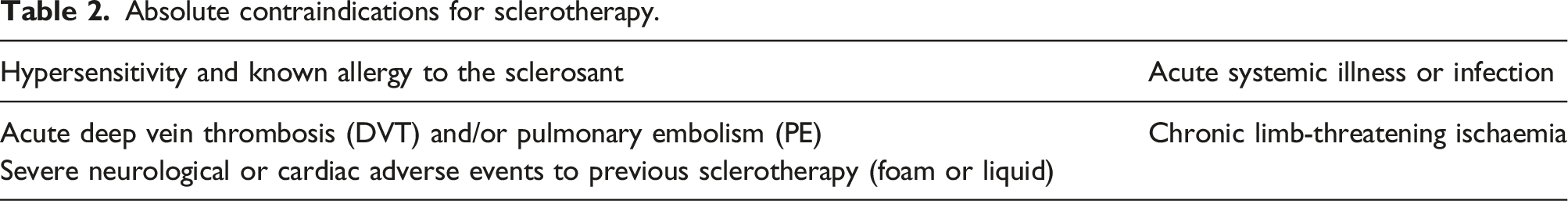
Absolute contraindications for sclerotherapy.
Routine ultrasound guidance is recommended when treating tributary varicosities, perforating veins, recurrent varicose veins, and saphenous trunks. For telangiectasia, sclerotherapy may be administered under direct vision, while for reticular veins, direct vision or ultrasound guidance is recommended. Additionally, fluoroscopic-guided sclerotherapy for venous malformations and pelvic varicosities may be performed.
Determining the maximum volume of foam to be used in one session should be based on individual risk–benefit analysis, the target vessel in question, and the presence of a patent foramen ovale (PFO). When treating saphenous trunks or accessory tributaries with open outflow paths to central circulation, higher volumes of more than 4–10 mL should be avoided; however, larger volumes can be used for smaller tributaries if the venous trunks have been previously ablated. Volumes of foam required can be also reduced by technique optimisation strategies such as using tumescent anaesthetic to reduce the diameter of the target vessel. If a patient has a known PFO, the treating clinician is advised to weigh the individualised benefits and risks of performing sclerotherapy prior to performing the procedure.
Vessel type in relation to sclerotherapy type and concentration.


The one-page guideline.
Discussion
This article provides a one-page clinical practice guideline summarising the indications for sclerotherapy and safe volumes for treatment. It is part of a series of publications for the International Union of Phlebology (UIP) One-Page Guidelines which are aimed at ensuring that patients with venous disease receive timely and appropriate care based on current best evidence and expert consensus.
Worldwide, sclerotherapy is becoming one of the most widely used treatments for varicose veins due to its ease of use, lower complication burden compared to other treatment modalities, and cost-effectiveness. 5 Clinical effectiveness of ultrasound-guided foam sclerotherapy has also been shown to be acceptable in a recent meta-analysis, with low rates of recurrence and recanalisation, 6 with foam being more effective than liquid sclerotherapy. 7 There still remains debate on whether the form and/or concentrations of the sclerosant used has an impact on the effectiveness of treatment, with a study showing equal effectiveness between 1% and 3% POL in treatment of the saphenous veins, 8 but others observing better clinical outcomes when using higher concentrations. 9 Further trials should aim to clarify this uncertainty to advise the development of future guidelines.
Complications most commonly include thrombophlebitis, pain, pigmentation, and bruising, but these are usually minor and can be transient. More severe complications include deep venous thrombosis and neurological symptoms such as migraines or transient visual changes, but these are usually rare if care is taken to determine the risk profile of each individual patient and tailor the sclerotherapy treatment according to this risk–benefit analysis. Additionally, if caution is exercised to minimise the concentrations and volumes of sclerosant used during treatment, risk of these significant complications is further reduced. 10
For all vascular specialists who routinely utilise sclerotherapy for the treatment of the spectrum of CVD, this article and summary document should act as a reminder of the need for individualised risk assessment and tailoring of treatment to avoid a ‘one-size-fits-all’ treatment strategy. This one-page guideline summarises the various considerations that vascular specialists need to keep in mind when faced with a patient presents to their practice with symptoms related to CVD. Referral to this document and application of the management principles outlined will hopefully help specialists improve their patients’ clinical outcomes and avoid significant complications related to sclerotherapy treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
Professor Alun Davies on behalf of the UIP.
Contributorship
K.P. and A.H.D. conceptualised the design of the short report and one-page guideline. H.B.M., B.B-H, S.G., and D.C. contributed to the literature review and formulation of the recommendations. M.T. contributed to the formatting and layout of the one-page guideline and wrote the initial draft of the short report. All authors reviewed the short report prior to submission.
