Abstract
Objectives
The addition of a varicose veins (VV) module to the existing Swedish National Registry for Vascular Surgery (Swedvasc) and its impact on quality of care were evaluated.
Methods
Vascular departments and private VV clinics were invited to enter data from 2016.
Results
Registrations were approximately 10,000 yearly but dropped to 5390 in 2022 when a fee was introduced for private clinics due to reduced funding. 88% more interventions were reported to Swedvasc 2020 than to the National Board of Health and Welfare. Regions differed in interventions per 100,000 inhabitants/year from 21 to 233 and in preoperative CEAP C4–6 from 30.8%–90.4%. Follow-up was 9.4%. These data contributed to the decision to prioritize the patient group for national guidelines and pathways of care, which will be monitored by Swedvasc.
Conclusions
A national VV registry with high coverage is possible and can contribute to national quality of care. The main challenge is funding.
Introduction
National quality registries for venous interventions can provide real-world data about quality of care for surgeons, patients, and authorities planning delivery of care. In research, data from quality registries can balance the results of randomized controlled trials which are often performed in highly specialized centers; however, this requires a high coverage in order to avoid selection bias. A European registry has been proposed, and preferably with data imported directly from electronic medical records (EMRs).1,2 So far this has not been possible in a larger scale, presumably due to a challenging mix of multiple caregivers in regionally and privately owned clinics and differing systems of EMR and funding. The national registries for interventions for varicose veins (VVs) often have incomplete coverage for the same reasons.3,4
For arterial interventions registries have become part of surgical daily life in several countries, and national coverage can be possible depending on the health care system and the use of social security numbers.5,6 One alternative could thus be to add a VV module to such a registry.
Entering data in a registry can be time-consuming and tedious, and more administration should not lightly be imposed in a health care world burdened by bureaucracy. Thus, in an informal survey about the creation of a national registry for VV in the Swedish National Registry for Vascular Surgery, Swedvasc, some years ago, most vascular surgeons were negative to the idea.
However, with increasing reports of complications after VV treatment and possible inequalities of care between regions, the Swedish Society for Vascular Surgery (SSVS) and Swedvasc steering committees decided to add a VV module to Swedvasc. The aim of this study is to report lessons learnt during the first years after the introduction of the registry and its possible impact on quality of care for the patient group on a national level.
Methods
Creating the registry
All SSVS members and privately owned VV clinics were informed about the project and invited to send suggestions about the content. A reference group was formed with representatives from vascular departments and private VV clinics reviewing the suggested content and form during the process. The VV module of Swedvasc was launched in 2016.
Infrastructure and funding
The registry was added as a new module of Swedvasc in order to use the same IT infrastructure and to make it familiar to Swedish vascular surgeons. Funding was included in Swedvasc´s budget, from the Swedish Association of Local Authorities and Regions (SALAR) and the Swedish National Board of Health and Welfare.
Entering data in Swedvasc is not compulsory by law, neither for arterial nor venous interventions, but it is strongly encouraged and for carotid and aortic interventions the coverage has been 97%.5,6
Eligible cases and entering data
Interventions for superficial venous incompetence can be registered for open surgery, endovenous treatment, and sclerotherapy, including primary VV or secondary VV to venous malformations or post-thrombotic states. Swedvasc encourages entering data also for interventions for cosmetic reasons to monitor the rate of complications, but treatment of teleangiectasias is not supposed to be sole indication, neither treatment of small clusters of VV for cosmetic reasons.
Data is entered manually by a doctor, nurse, or administrative staff.
Form and variables
The VV module has three parts: preoperative, perioperative, and follow-up data, similar to registrations for arterial surgery. The recommended time for follow-up is after 1 year (10–14 months), but it was possible to register at any time as routine varied. As most vascular departments did not follow-up their patients, it is possible to actively choose “We do not intend to do follow-up” and state the reason, such as “Not our routine, patient declined, patient moved to other region, patient deceased.”
Due to a widespread administration fatigue amongst vascular staff, the intention was to limit variables to very few, and typically it will take 2–3 min to complete. An English translation is added as online supplementary material. It was also planned to revise and modify the content of the VV module early, according to lessons learnt after start.
PROM
A PROM (Patient Reported Outcome Measure), VARIShort, was introduced in the registry 2020, for preoperative and 1-year follow-up data. The questionnaire VEINES-Qol/Sym had been validated previously in a Swedish translation, followed by a validation of VARIShort, the Swedish translation of the short-form VVSymQ, which in turn includes items from VEINES-Qol/Sym.7,8 Those units that have started using VARIShort for follow-up usually send it by a letter if a physical visit is not planned.
Monitoring your own data
As with arterial interventions, each practitioner registering in Swedvasc can download data for his or her own clinic at any time, in the form of an excel file. There is also a start page where you can see upcoming 1-year follow-ups and incomplete entries.
Support
Some of the private VV clinics had limited knowledge of quality registries and needed support when starting, which was provided by a dedicated nurse from Swedvasc.
Results
Participating units and coverage
Vascular departments in regional hospitals and privately owned centers for VV treatment were invited to register in the VV module.
Introduction of the VV module varied in speed between regions and clinics, but in some regions, it was a prerequisite for procurement of VV treatment from privately owned VV clinics which then joined early. The number of registrations is seen in Figure 1. Number of registrations in Swedvasc´s module for varicose veins interventions 2016–2022.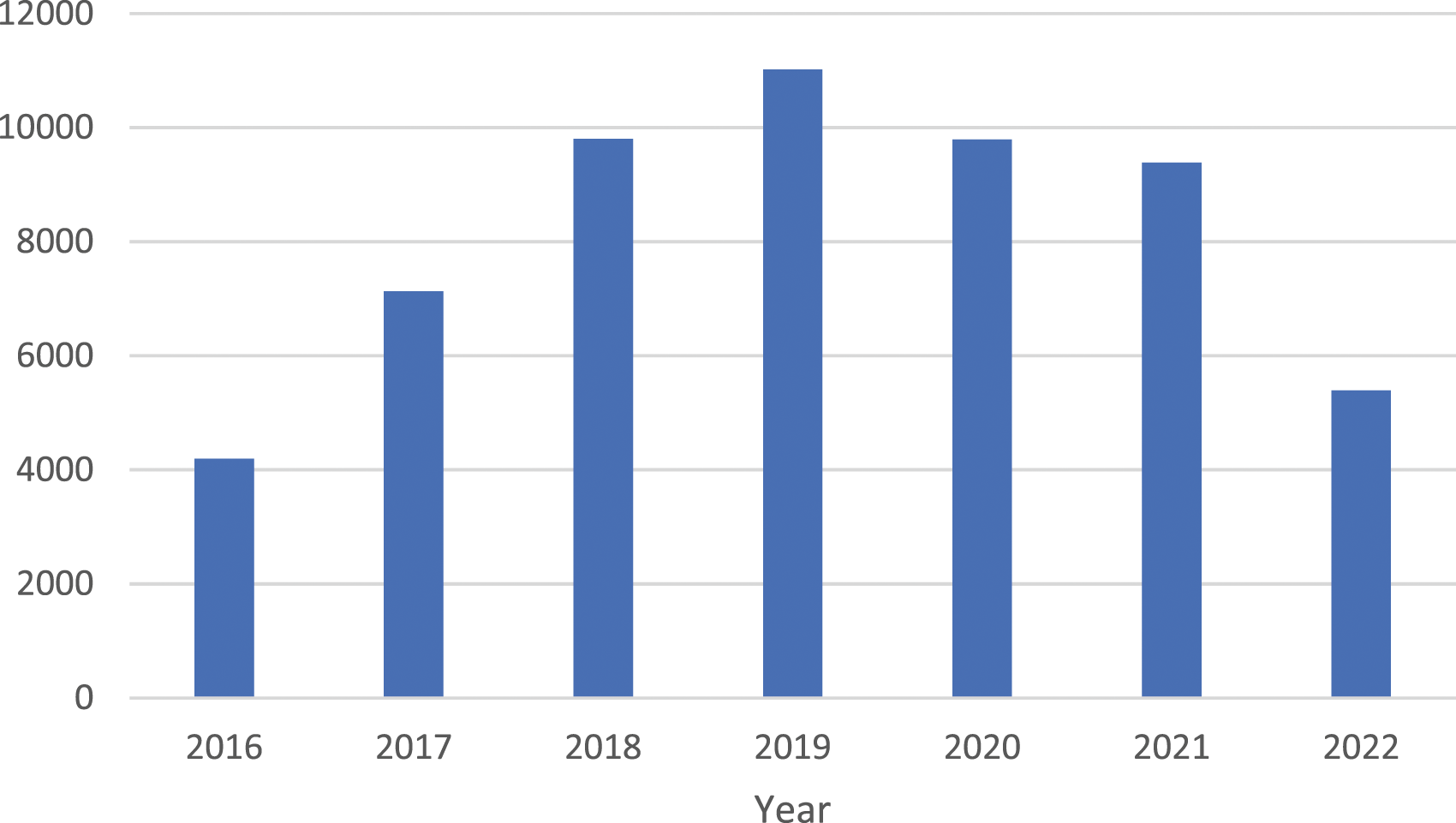
Coverage compared to official data from the Swedish National Board of Health and Welfare increased rapidly and in 2018 it was 188%! As has been reported in a previous study, some of the privately owned VV clinics when questioned stated that they were not aware that reporting to the Board is mandatory by law. 9 Thus, the true number of VV interventions in Sweden is still unknown but has been estimated to be 12, 000/year, if sclerotherapy only is excluded. 9 When a fee was introduced in 2022 for private clinics registrations dropped, mainly for interventions with cosmetic indications. 10
Preoperative data
Preoperative data for registrations in Swedvasc´s module for varicose vein interventions 2016 – 2022. All variables can be found in online supplementary material.

Perioperative data
Interventions were endovenous ablation (67.0%), sclerotherapy (30.2%), phlebectomies or miniphlebectomies (63.6%), and stripping (2.8%), most often in combination.
Follow-up and complications
Follow-up at any time was registered in 14.1%, 7255 entries, the most common reason stated for no follow-up was “Not routine” in 84.2%.
Follow-up decreased, presumably due to the pandemic. Of interventions performed in 2020, only 9.4% had a follow-up at any time and 7.7% after 1 year. Of these, a total of 54 (6%) had some kind of complications: 9 (1%) had a deep venous thrombosis, 8 (1%) had nerve injury with pain, 29 (3%) with disturbing sensory loss, and 4 (0.4%) with other serious complications such as infection.
PROM
VARIShort was introduced in June 2020. For interventions performed in 2021, only 10% had a follow-up with VARIShort but with a considerable variation (0%–80%).
Differences between regions
The number of VV interventions/100,000 inhabitants >20 years of age was 119 for the country and varied from 21 to 233 between regions (see map in Figure 2). Varicose vein interventions registered in Swedvasc per 100,000 inhabitants > 20 years old/county 2021. With permission from Swedvasc.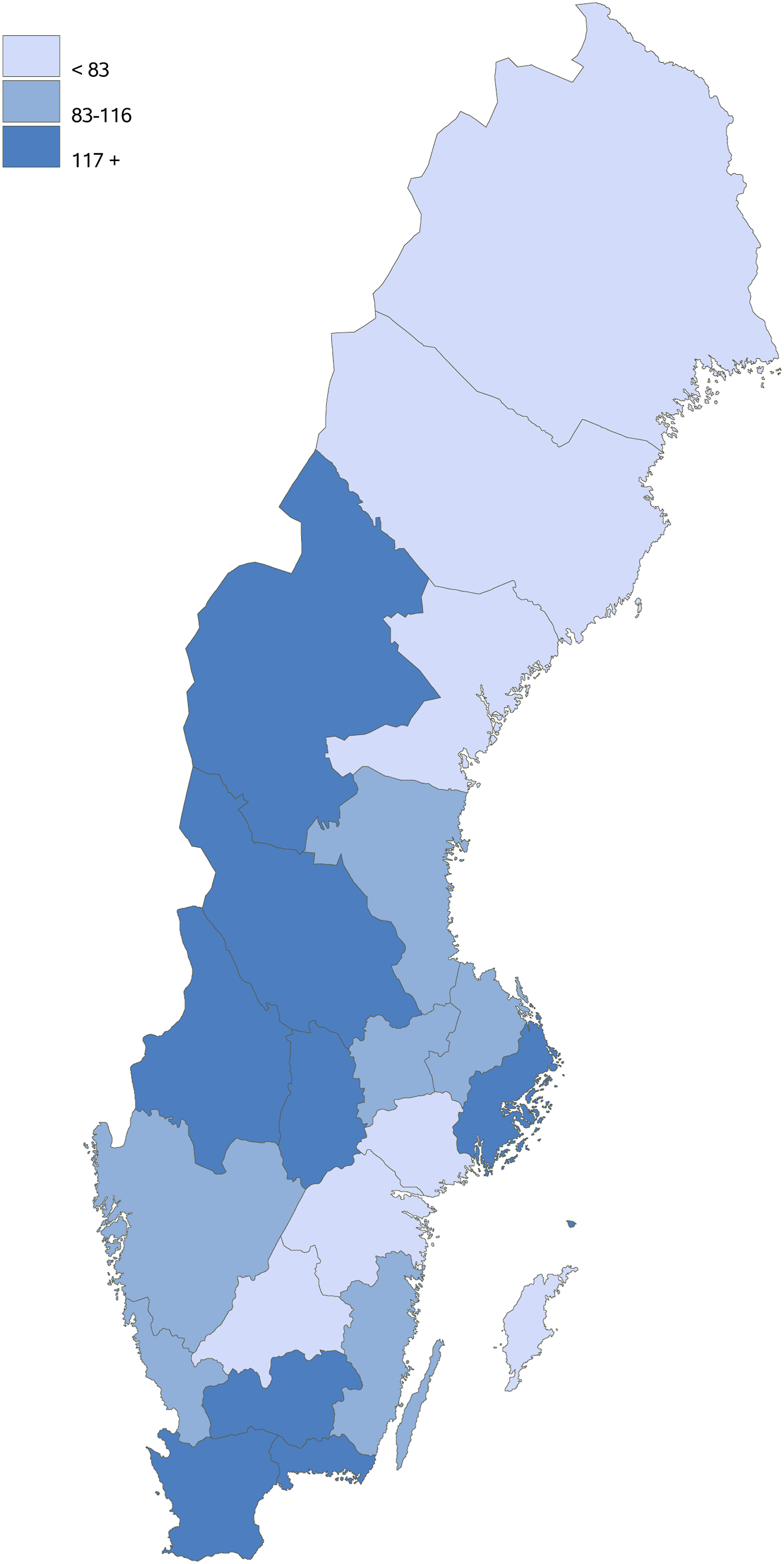
For all registered interventions, 56% were tax-funded, 6% by insurance companies, 33% by the patient, and 5% not declared.
Differences between regions for tax-funded interventions
49.2% of the tax-funded procedures were performed in privately owned clinics with regional commission.
The distribution of preoperative clinical classes according to CEAP differed between regions within the tax-funded group, with a proportion of C4–6 of 60.8% (30.8–90.4), and the proportion of C6, active ulcer, 8.4% (4.6%–24.7%).
Treatments most often included an endovenous method, in 75.6%, but with regional variation, 18.0%–90.4%. The corresponding figures for regionally and privately owned clinics were 66.0% and 85.4%, respectively.
Follow-up at any time was overall 13.9% with at a variation between regions of 1.0% and 49.9% and a difference between regionally and privately owned clinics, 18.7% and 7.4%, respectively.
Impact on quality of care
As data from the VV registry verified inequalities between regions regarding indications and payment and a low follow-up rate with complication rates unknown, SALAR prioritized the patient group for national guidelines and pathways of care, and these were finalized in 2022 for patients with venous disease of the lower leg including venous ulcers. During the implementation process, the VV registry will be used for monitoring adherence, and a mobile phone solution is planned for follow-up of uncomplicated cases. There are also projects for automatic retrieval of data to the registry from EMR.
Funding of the registry
Funding has been reduced lately. A fee for registrations was introduced for the privately owned clinics in 2022.
Discussion
The brief module for varicose vein interventions added to the Swedish National Registry for Vascular Surgery, Swedvasc, rapidly gained substantial coverage with data demonstrating inequalities between regions regarding indications and payment and a low follow-up rate with complication rates unknown. Data from the registry was used for the creation of national guidelines and pathways of care and will be used for monitoring implementation; thus, the registry has a vital role in improvement of quality of care on a national level. Funding is the main challenge.
An important aim for a national VV quality registry is a high coverage grade. Arterial interventions are only performed in regional hospitals which all report to the Swedish National Board of Health and Welfare and have well-established EMR systems. Thus, it is possible to control external and internal validity for arterial interventions.5,6 Treatments for VV have been outsourced in Sweden in a varying degree from the regional hospitals to privately owned clinics of which some do not report to the Board as described previously; thus, it is not possible to calculate coverage. However, before the pandemic and the introduction of a fee, an estimated coverage was approximately 90%. 9
An important factor for the acceptance of the registry is that Swedish vascular surgeons are used to register in Swedvasc, and as the VV module has few variables, it became easier for vascular surgeons to accept the extra work. On the other hand, it will be more difficult for research project that will need more data about the patients. At start, regional vascular departments were slow in starting to register interventions for VV, but this has now become routine in most regional hospitals, and an impression is that the registry has raised awareness of the patient group amongst vascular surgeons.
Automated data retrieval to EMR would facilitate for users and presumably improve coverage. Two regional pilot projects were underway but stopped due to planned changes of EMR systems. When the VV module is revised, coding of variables to facilitate data retrieval would be feasible.
The low rate of follow-up means that results and complication rates in Swedish clinical reality are practically unknown. In some departments, there was already a good infrastructure for the PROM VascuQol used for peripheral arterial disease that could also be used for VARIShort, and follow-up for VV was easier. In others, VascuQol is still not used in the majority of cases, which made it difficult to introduce another PROM. An increasing number of clinics are now doing follow-up by letter, and this will hopefully further improve with a planned mobile phone solution for follow-up of uncomplicated cases.
The main challenge for any registry is the funding. Swedish registries are dependent on a financing model with a yearly funding continually adjusted by the authorities involved, and this makes development of the registries difficult to plan, subsequently a revision of the VV module has been postponed. In the planned revision, the VV module will be adapted to the national pathway of care, and if budget allows it also changed according to suggestions from users and prepared for automated retrieval of data from EMR.
When funding was reduced and expenses for the private clinics increased, a fee for these clinics was introduced, at present approximately €1000 yearly, and some private clinics chose not to continue entering data in the registry. Another argument proposed for not participating in Swedvasc was the time spent entering data. However, time expenditure will be further reduced with the planned mobile phone solution for follow-up.
As variables from Swedvasc will be used by the regions to monitor the implementation of national guidelines and pathways of care, funding will hopefully be adapted, and coverage grade improves for tax-funded interventions. Ideally, also interventions for cosmetic indications should be compulsory to register, as treatments for cosmetic reasons remain relatively unregulated in Sweden.
National guidelines are not easy to implement as has been reported previously, and there are challenges in Sweden as well. 11 We hope to be able to report Swedvasc´s contribution to this after a few years and hopefully with an improved situation for the patient group.
Conclusions
The brief module for varicose vein interventions added to the Swedish National Registry for Vascular Surgery, Swedvasc, rapidly gained substantial coverage with data demonstrating inequalities between regions regarding indications and payment and a low follow-up rate with complication rates unknown. This contributed to the prioritization of the patient group for national guidelines and pathways of care, where Swedvasc will also be used for monitoring adherence, and a mobile phone solution is planned. Funding is the main challenge and when a fee was introduced, willingness to register in the private sector diminished.
Supplemental Material
Supplemental Material - Implementation of a varicose vein module added to Swedvasc, the Swedish National Registry for vascular surgery
Supplemental Material for Implementation of a varicose vein module added to Swedvasc, the Swedish National Registry for vascular surgery by Lena Blomgren in Phlebology.
Footnotes
Acknowledgments
I would like to thank past and present members of the steering committee of Swedvasc who have made the VV registry possible and the clinicians who made the effort to register their interventions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scandinavian Research Foundation for Varicose Veins and other Venous Diseases (SFÅV) and by ALF funding from Region Örebro County.
Ethical statement
Guarantor
LB.
Contributorship
LB conceived the study, gained ethical approval, analyzed data, and wrote the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
