Abstract
Objective
Does short-term use of pneumatic compression devices (PCD) and off-the-shelf compression alleviate symptoms and improve quality of life in women with lipedema and secondary lipolymphedema?
Methods
Prospective, randomized controlled, industry-sponsored, proof-in-principle study comparing PCD plus conservative care (PCD+CC) to CC alone (control). Adult females with bilateral lipedema and secondary lymphedema were randomized to PCD+CC or CC. Outcome measures were lower limb and truncal circumferential measurements, bioimpedance, and quality-of-life, symptom, and pain intensity questionnaires.
Results
Both groups experienced improvements in leg circumference and bioimpedance with more improvement in the PCD+CC group than the CC group. Pain scores of the SF-36 survey and numerical rating scales were improved in the PCD+CC group. Wong–Baker Faces scores showed trends toward improvement in both groups.
Conclusions
This proof-in-principle study supports conservative management with graduated compression and with or without PCD for improvement in leg circumference, bioimpedance measurements, and pain in patients with lipedema.
Introduction
Lipedema is a connective tissue disorder that affects up to 11% of women. 1 It is characterized by painful, swollen subcutaneous tissue and disproportionate fat accumulation. Patients are often not aware they are affected by this disease; rather, they think they are just overweight or obese. Patients with lipedema often feel symptoms such as heaviness, pain, and easy bruising that impact quality of life. Affected limbs can become so large and heavy that daily tasks such as walking, cleaning, or shopping become impossible.
Lipedema is a progressive disease. There are currently 3 primary stages that describe disease severity: stage 1 involves thickening of subcutaneous tissue and disproportion accumulation of subcutaneous tissue in the extremities (tissue remains smooth and is generally not heavy or swollen); stage 2 is characterized by increased fibrous tissue, leading to a nodular feel in the subcutaneous tissue. Symptomatic lipedema patients at stage 2 and higher almost always have secondary swelling on physical exam and most with evidence of impaired lymphatic function.2–4 Stage 3 involves progression with the formation of lobules of skin and subcutaneous tissue (see Supplemental Table S1).
Patients with lipedema have been shown to have dilated and tortuous lymphatics. 5 Studies have also shown increases in tissue water content with increasing stages of lipedema, consistent with secondary lymphedema. 2 This lipolymphedema (or lipedema with swelling) is a secondary lymphedema, much like venolymphedema, and can be easily distinguished from primary lymphedema as it spares the hands and feet.
There is currently no cure for lipedema, so treatment focuses on symptom management, improving patient-reported outcomes, and prevention of complications. At present, the 2 main courses of treatment include nonsurgical conservative treatment (e.g., comprehensive decongestive therapy (CDT), diet, exercise, and emotional/psychological/social support) and lipedema reduction surgery, a type of lymph-sparing liposuction performed by a surgeon trained in lipedema treatment. The primary goals for treatment include reduction/elimination of inflammation, swelling, and pain; increase in lymphatic flow, which reduces/eliminates excessive fluid and swelling; overall management of the physical impact of lipedema; and quality of life improvements, which can include emotional, psychological/mental, spiritual, and social enhancement in addition to physical management.
Intermittent pneumatic compression devices (PCD) are often used as home-therapy to treat secondary lymphedema or lipolymphedema and may be helpful in preventing the progression of lipedema. Pneumatic compression devices move lymphatic fluid and support the elimination of proteinaceous fluids, thus leading to improved patient-reported symptoms, decreased limb girth and volume, increased elasticity of tissues, and fewer episodes of infection.6–8 Atan et al. found improvements in limb volume, pain, and physical functioning in patients with stage 3 or 4 lipedema after CDT or PCD treatment. 9 Szolnoky reported daily treatment with CDT, PCD, and multilayered short-stretch bandaging performed for 5 days led to significant improvements in leg volume, capillary fragility, and pain in women with lipedema. 10
The purpose of this study is to assess whether short-term use of PCD and off-the-shelf compression are associated with alleviation of symptoms and improvement in quality of life in women with lipolymphedema (lipedema with swelling).
Materials and methods
Study design and population
This is a postmarket, prospective, single-center, open-label, industry-sponsored, proof-in-principle randomized controlled study comparing PCD plus conservative care (PCD+CC) versus conservative care (CC) alone in patients with lipedema and secondary symmetrical lymphedema. The study received ethics approval through Advarra IRB (protocol number PRO00041073). All participants signed the IRB-approved written informed consent before participating in the study. The study is registered on www.clinicaltrials.gov (NCT04213989).
Lipedema diagnostic criteria.

Adapted from Sandhofer M, et al. Dermatol Surg 2020;46(2):220-228.
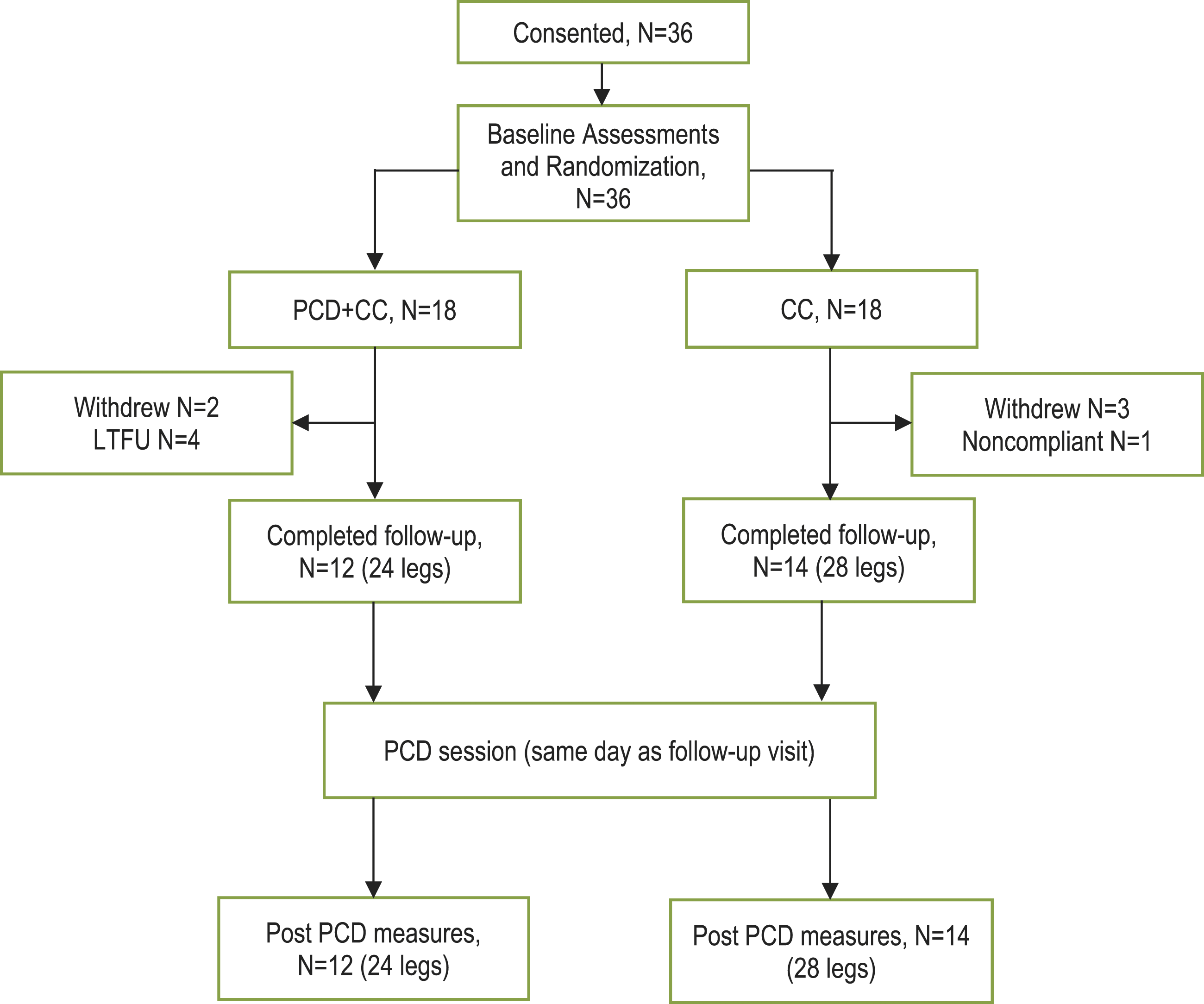
CONSORT diagram of participant flow. CC, conservative care; LTFU, lost to follow-up; PCD, pneumatic compression device.
Assessments
Demographics and baseline characteristics were collected at baseline. Follow-up clinic assessments were initially intended to be performed at 4 weeks but due to the COVID-19 pandemic were modified to be performed up to 12 weeks after treatment initiation.
Primary outcomes included circumferential measurements and patient-reported outcomes (PROs). Circumferential measurements of each leg (at ankle, mid-calf, lower thigh, and upper thigh), hip, and waist were taken at baseline and follow-up using a Gulick II tape measure. Multiple patient-reported questionnaires were completed at baseline and follow-up. The RAND 36-item short form health survey (SF-36) was completed to assess participants’ overall quality of life. Symptoms and pain intensity were assessed using the Patient-Reported Outcomes Measurement Information System (PROMIS) Pain Interference short form 6d, v1.0 and Wong–Baker FACES Pain Scale. Additionally, a 10-point numeric rating scale (NRS) was used to measure perceptions of swelling, heaviness, pain, fatigue, tenderness, and tightness independently for each leg.
There were also 2 predefined exploratory outcomes: bioimpedance measurements and patient-reported mobility. Bioimpedance measurements were performed using the InBody 770 body composition analyzer (InBody USA, Cerritos, California 90703). Bioimpedance measurements included whole body extracellular water/total body water (ECW/TBW) ratios as well as segmental ratios for the right/left arm, right/left leg, and trunk. Mobility was assessed using the PROMIS Mobility 2.0 questionnaire.
Adverse events were also collected at follow-up.
Statistical analysis
This proof-in-principle study was not formally powered for a specific primary hypothesis; thus, no formal power/sample size calculation was performed. The sample size was determined based on a desire to be able to estimate the mean of the primary within group endpoints, using 95% confidence intervals (CI), with the estimation of the endpoint means to have a precision (half width of the CI) of approximately 40% of the observed standard deviation (SD). To achieve these parameters, we determined a target analyzable sample size of 15 participants per group (30 total legs as the observational unit).
Percent change in circumference was calculated as the group change from baseline/total cohort baseline circumference x 100. The within and between group comparative change between time point analyses for the continuous measurements were performed using an analysis of covariance (ANCOVA). The total baseline values derived from the ANCOVA analysis were used to adjust for baseline differences between groups.
Due to the exploratory nature of the study, p values are reported as apropos, point estimates, and 95% confidence intervals are used to guide exploratory analyses for potentially clinically meaningful differences. A p value of <0.05 indicated statistical significance.
Statistical software was R version 4.1.0 or higher (The R Foundation for Statistical Computing. https://www.R-Project.org).
Results
Demographics and baseline characteristics by treatment group.
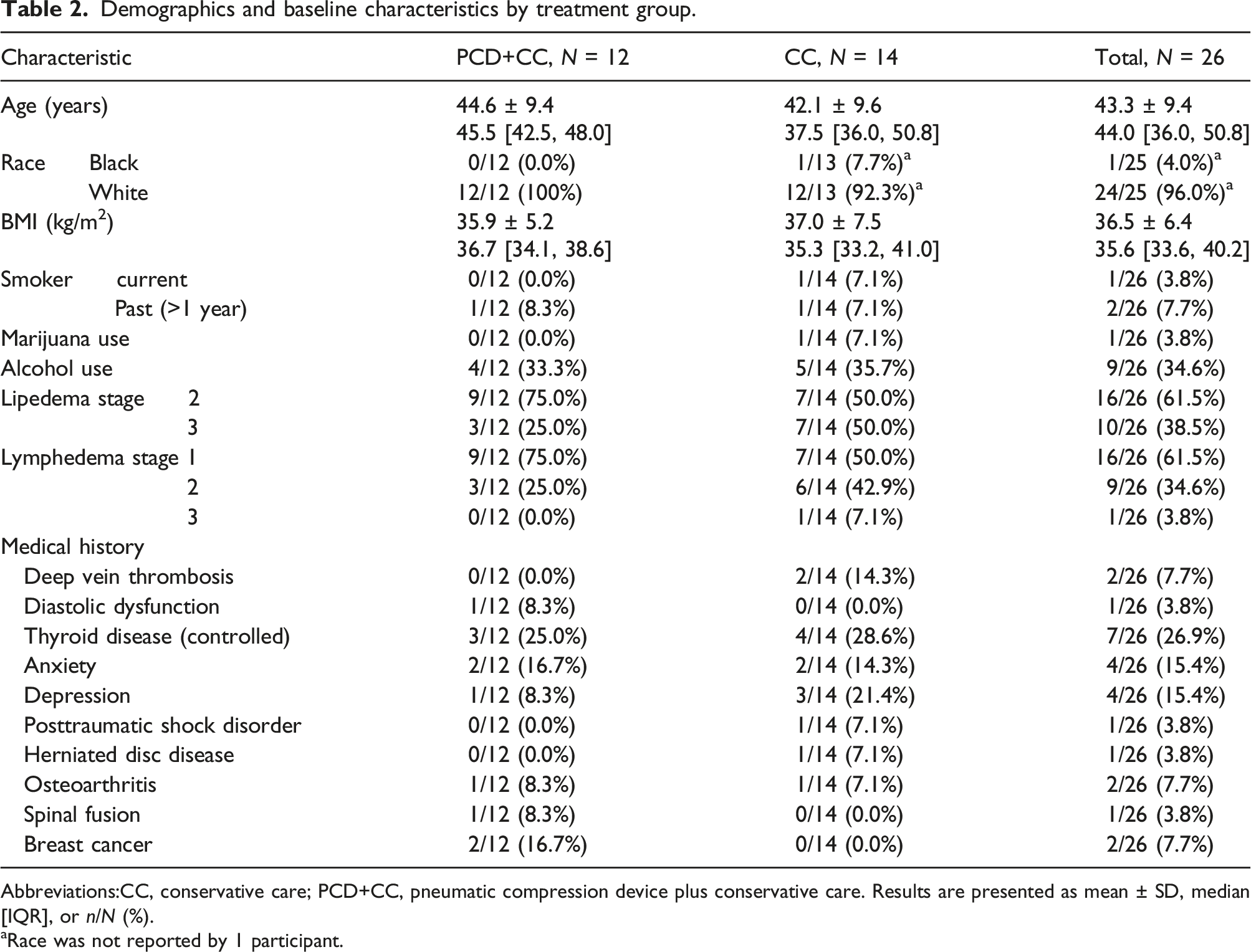
Abbreviations:CC, conservative care; PCD+CC, pneumatic compression device plus conservative care. Results are presented as mean ± SD, median [IQR], or n/N (%).
aRace was not reported by 1 participant.
Change in circumference measurement (cm) from Baseline to Follow-up.
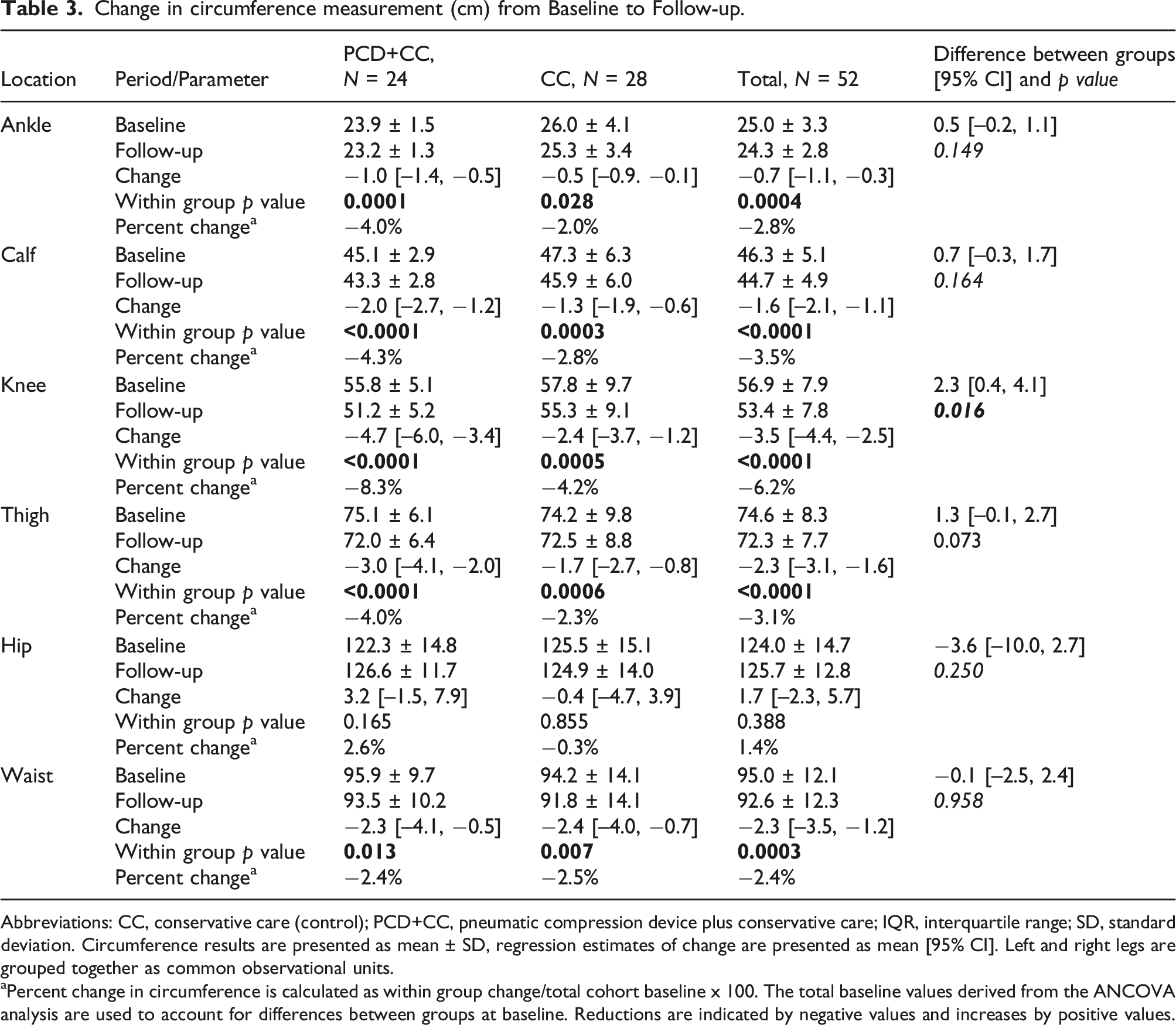
Abbreviations: CC, conservative care (control); PCD+CC, pneumatic compression device plus conservative care; IQR, interquartile range; SD, standard deviation. Circumference results are presented as mean ± SD, regression estimates of change are presented as mean [95% CI]. Left and right legs are grouped together as common observational units.
aPercent change in circumference is calculated as within group change/total cohort baseline x 100. The total baseline values derived from the ANCOVA analysis are used to account for differences between groups at baseline. Reductions are indicated by negative values and increases by positive values.
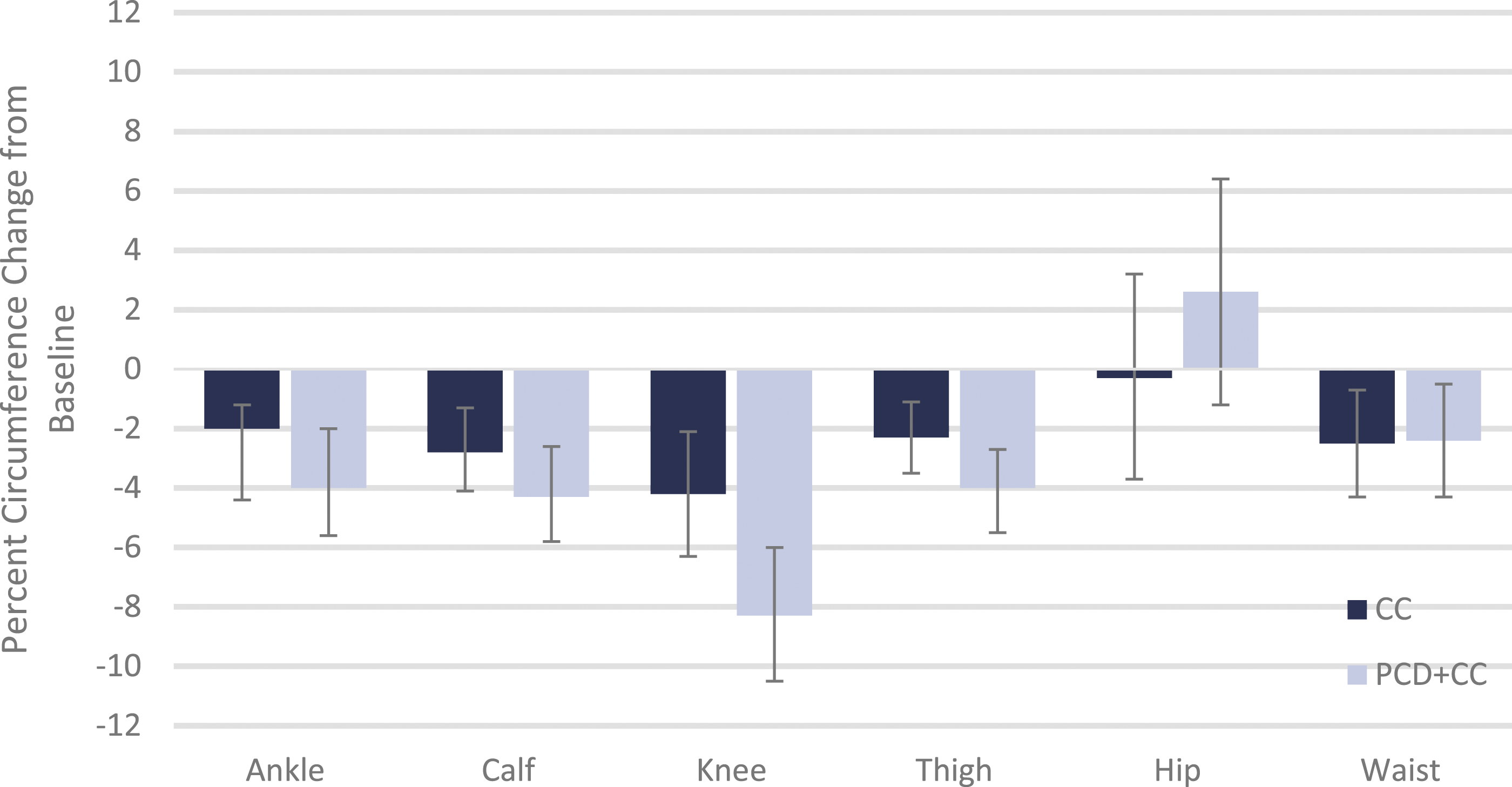
Percent change in circumference from baseline to follow-up. CC, conservative care; PCD+CC, pneumatic compression device with conservative care. Error bars indicate the 95% confidence interval.
RAND SF-36 score changes from baseline to follow-up.
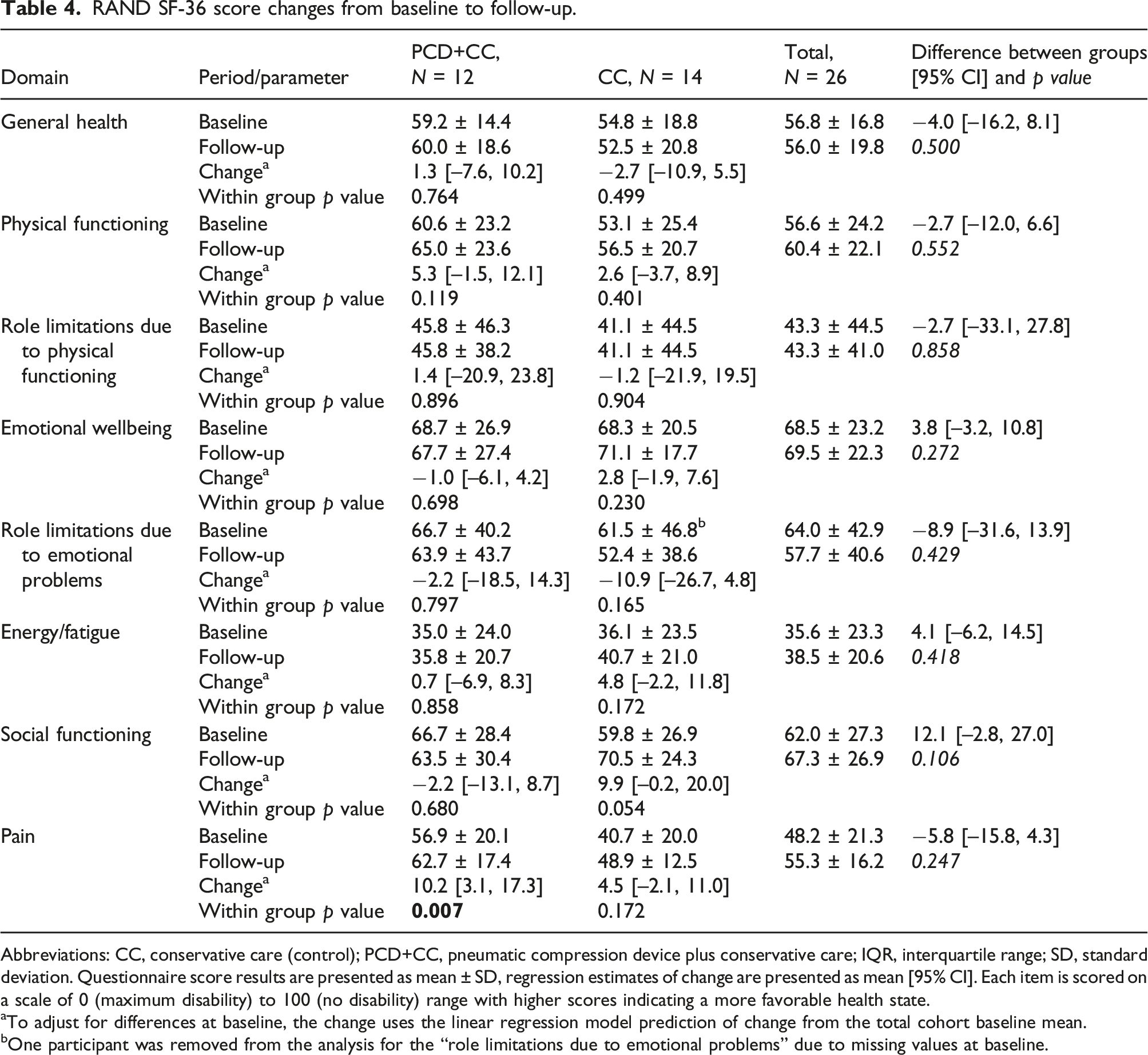
Abbreviations: CC, conservative care (control); PCD+CC, pneumatic compression device plus conservative care; IQR, interquartile range; SD, standard deviation. Questionnaire score results are presented as mean ± SD, regression estimates of change are presented as mean [95% CI]. Each item is scored on a scale of 0 (maximum disability) to 100 (no disability) range with higher scores indicating a more favorable health state.
aTo adjust for differences at baseline, the change uses the linear regression model prediction of change from the total cohort baseline mean.
bOne participant was removed from the analysis for the “role limitations due to emotional problems” due to missing values at baseline.
PROMIS pain interference, PROMIS mobility, and Wong–Baker FACES.
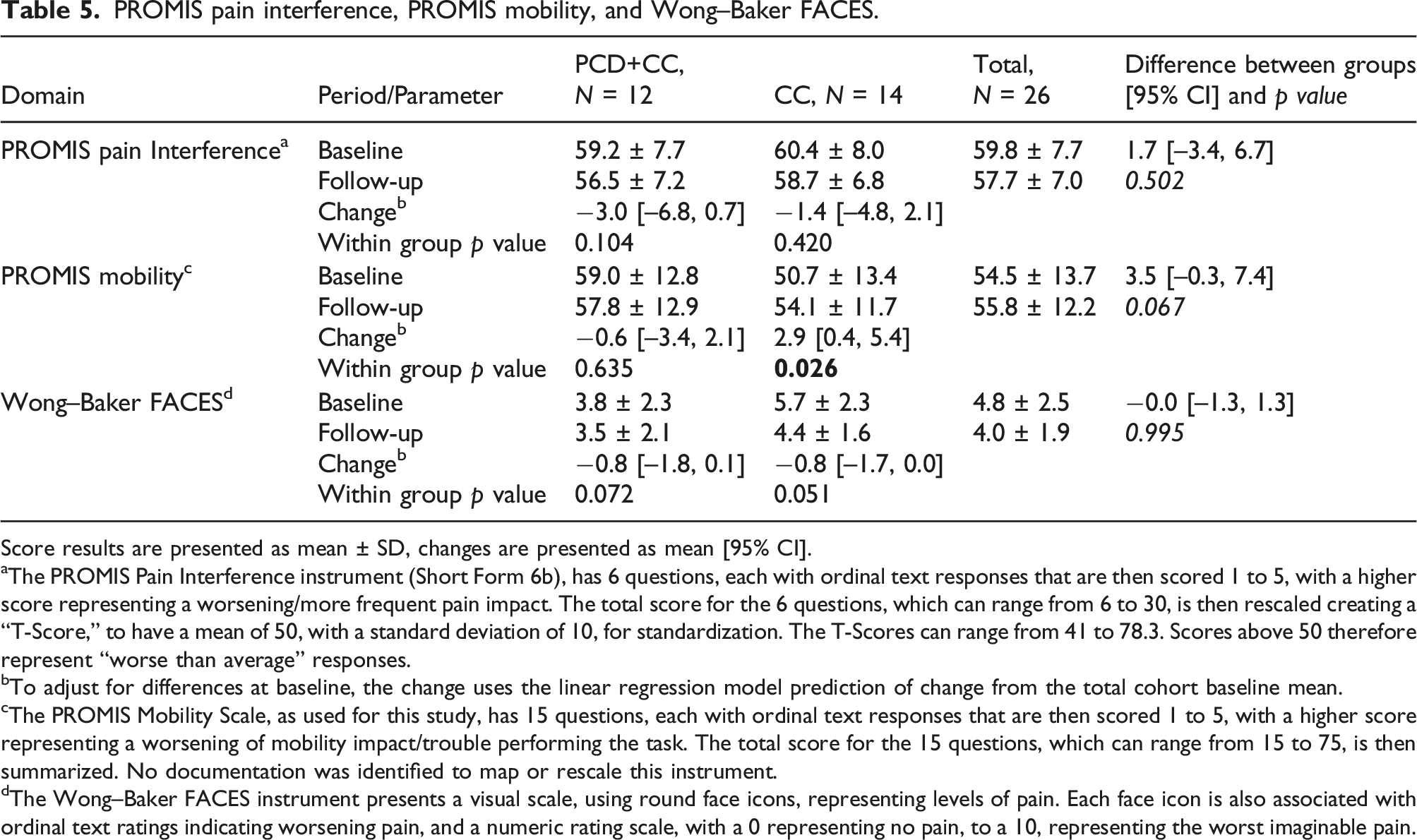
Score results are presented as mean ± SD, changes are presented as mean [95% CI].
aThe PROMIS Pain Interference instrument (Short Form 6b), has 6 questions, each with ordinal text responses that are then scored 1 to 5, with a higher score representing a worsening/more frequent pain impact. The total score for the 6 questions, which can range from 6 to 30, is then rescaled creating a “T-Score,” to have a mean of 50, with a standard deviation of 10, for standardization. The T-Scores can range from 41 to 78.3. Scores above 50 therefore represent “worse than average” responses.
bTo adjust for differences at baseline, the change uses the linear regression model prediction of change from the total cohort baseline mean.
cThe PROMIS Mobility Scale, as used for this study, has 15 questions, each with ordinal text responses that are then scored 1 to 5, with a higher score representing a worsening of mobility impact/trouble performing the task. The total score for the 15 questions, which can range from 15 to 75, is then summarized. No documentation was identified to map or rescale this instrument.
dThe Wong–Baker FACES instrument presents a visual scale, using round face icons, representing levels of pain. Each face icon is also associated with ordinal text ratings indicating worsening pain, and a numeric rating scale, with a 0 representing no pain, to a 10, representing the worst imaginable pain.
Overall, the NRS ratings ranged from no change to significant improvements from baseline for both groups (see Supplementary Table S2). Assessments were done separately for each leg. On the left leg, statistically significant improvements from baseline were observed in the PCD+CC group in pain and tightness, and in the CC group for fatigue and tightness. On the right leg, statistically significant improvements from baseline were observed in the PCD+CC group in tightness, and in the CC group for fatigue. None of the differences for either leg were statistically significant between groups.
Leg bioimpedance measurements were significantly improved from baseline to follow-up for both treatment groups but there was not a statistically significant difference between treatment groups. There were no significant changes in the whole body, trunk, or arms for either treatment group (see Supplementary Table S3).
Summary of general study findings.

NS, nonsignificant difference from baseline; S, significant difference from baseline; * trend toward significant difference from baseline (p < 0.1); +, improvement; -, worsening.
aMeasurements at ankle, calf, knee, and thigh.
Changes in circumference after a single PCD session at follow-up are presented in Figure 3 (and in Supplementary Table S4). There were statistically significant improvements in circumference at all locations for both treatment groups ranging from −0.3 cm at the ankle to – 1.9 cm at the thigh. The percent change in circumference was similar between groups after a single session (less than 2% difference) with the CC group having marginally higher percent improvements than the PCD+CC group. Percent change in circumference from pre to post PCD session at follow-up. CC, conservative care; PCD+CC, pneumatic compression device with conservative care. Error bars indicate the 95% confidence interval.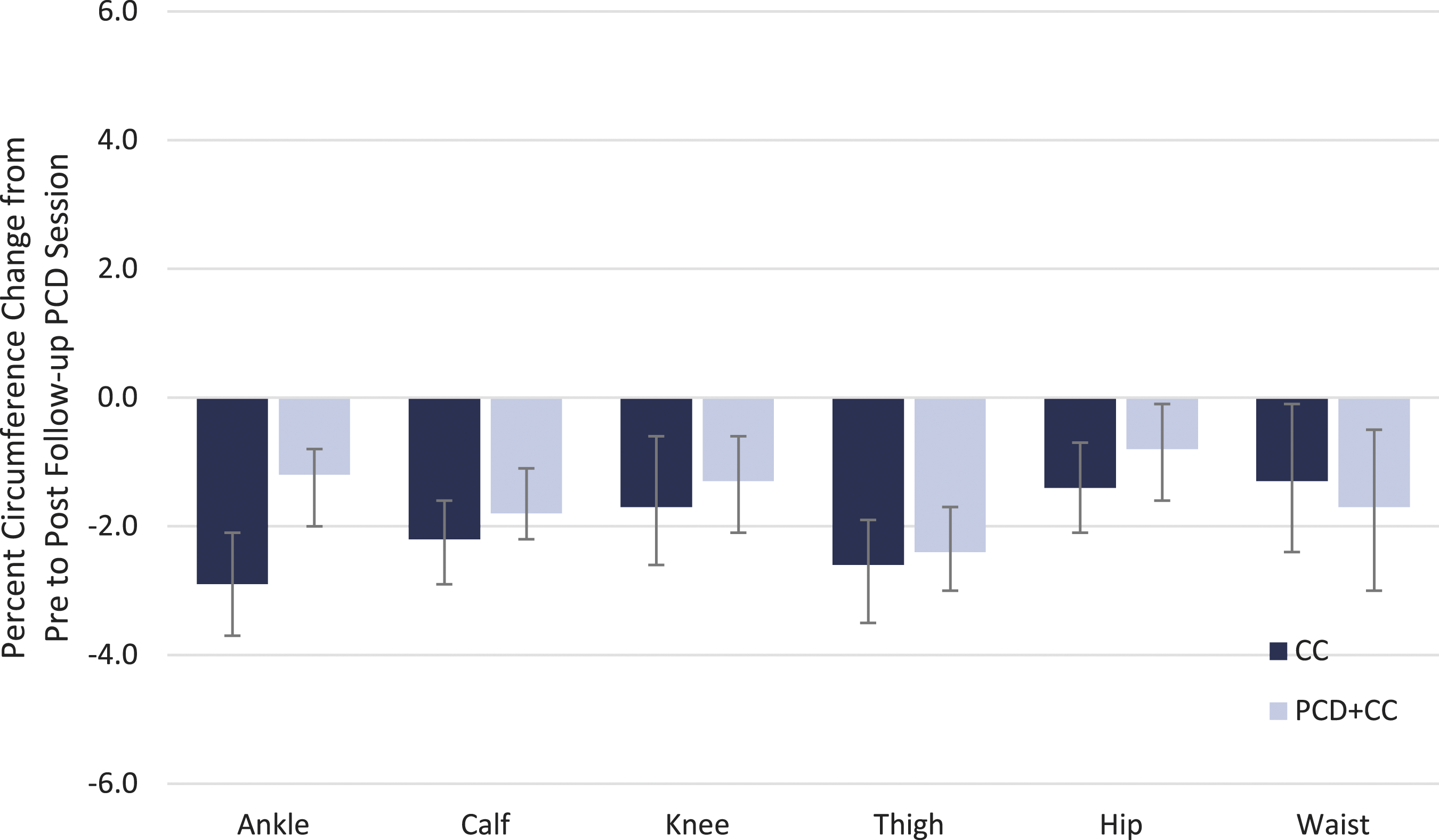
Changes in bioimpedance after a single PCD session at follow-up are presented in the Supplementary Materials (Table S5). Both the legs and arms demonstrated statistically significant improvements after a single PCD session for both treatment groups. Whole body and trunk improvements approached statistical significance for the PCD+CC group only (p = 0.097 and p = 0.052, respectively) with the change for all participants combined showing a statistically significant difference. There were no statistically significant differences between the groups.
One CC participant reported a complication of a cellulitis episode that lasted 1 week but did not require hospitalization.
Discussion
We conducted a randomized controlled proof-in-principle study of objective and subjective outcomes between women with bilateral lipedema and secondary lymphedema undergoing approximately 4–12 weeks of PCD+CC with those undergoing CC only. Additionally, we examined changes in circumference and bioimpedance measurements after a single session of PCD in all participants at follow-up. Our results support the benefits of PCD as an adjunct treatment to CC alone. Both treatment groups showed significant improvements from baseline in circumference and leg bioimpedance measurements with the PCD+CC group more improved than the CC group. It is interesting to note that in the PCD+CC group, we observed an increase in hip circumference, which we hypothesized was due to proximal movement of the lymphatic fluid from the legs to the hip following PCD treatment. We anticipate that the hip circumference would be decreased over time as the excess fluid is processed by the body.
In lymphedema patients, Kim et al. showed that the normal limb had an ECW/TBW ratio of 0.38 whereas the contralateral affected arm had an ECW/TBW ratio of 0.39. 14 This was highly clinically meaningful and was associated with a 2-cm circumference difference in the limb. Bioimpedance is accurate, reproducible, and is independent of measuring errors that a tape measure can introduce. Zhang et al. also studied lymphedema with bioimpedance measured ECW/TBW ratio and observed an ECW/TBW ratio in the healthy limb of 0.3748 compared to ECW/TBW ratio of 0.3756 in the limb with lymphedema (difference = 0.008). 13 We saw smaller changes, some as small as 0.001 in the ECW/TBW. A change of 0.001 would equate to 12.5% of the change observed between a limb with or without lymphedema, which may be appreciated symptomatically but also may not be a clinically meaningful change to the patient.
We believe this is the first controlled study that shows the benefits of off-the-shelf graduated compression garments in lipedema without the use of short stretch compression wraps. Previously, Atan et al. conducted a randomized study of CDT with short-stretch wraps verses PCD plus exercise or exercise alone in lipedema patients and found significant changes in multiple domains of the RAND SF-36 survey. 9 In contrast, we only found statistically significant improvement in the pain domain in the PCD+CC group in this study. There are several notable differences in population demographics between the studies. Atan’s study population was older, had higher BMI, and more severe lipedema with lower (greater disability) baseline SF-36 domain scores than our population. A non-randomized study by Szolnoky et al. 10 used the Wong–Baker FACEs score to evaluate pain and found significant improvement in patients undergoing PCD and manual lymphatic drainage. However, they did not document patient demographics or staging criteria, and their baseline pain scores indicated more pain at baseline than our population. In our study, both the PCD+CC and CC groups showed improvements in pain that approached statistical significance at follow-up, which may have reached significance with a slightly larger sample size. Although we observed some improvements in the PROMIS pain interference scores, the differences from baseline were not statistically significant. Our PCD+CC group did show statistically significant improvement in pain scores on the NRS scale.
We believe we are the first to report PROMIS mobility scores in a lipedema population. Our PCD+CC group only marginally improved and the CC group worsened with the difference between groups approaching statistical significance. With a larger population, it is likely that the PROMIS would prove to be a validated valuable assessment for use in patients with lipedema.
The addition of a single PCD session in all participants gave us an opportunity to compare circumference and bioimpedance outcomes for participants who were naïve to PCD treatment with those who had undergone previous PCD treatment. We observed slightly greater (but not statistically significant) improvements in circumference for the CC group after this single session than we saw in the PCD+CC group. However, the improvements in the PCD+CC group over time were greater than those seen in either group after a single session. Arm and leg bioimpedance measurements were statistically improved in both groups after the single PCD session but the clinical significance of the differences is unclear.
The study strengths include use of both objective (circumference and bioimpedance) and validated patient-reported (SF-36, PROMIS, Wong–Baker Faces) measures in a population that has not been adequately evaluated in the past. The analyses were performed using the ANCOVA method to adjust for differences in baseline values between the participant groups. We also controlled the educational aspect of the conservative treatment by providing at least one educational session with a CLT for all participants. Adequate instruction by a CLT is an important part of clinical care and the authors recommend it for all patients with lipedema.
Several limitations of our study were caused by the COVID-19 pandemic. Our study initially was intended to follow-up all participants after 4 weeks of treatment. However, due to the pandemic, the follow-up visits were extended to up to 12 weeks after treatment initiation to align with standard of care follow-up visit timing and minimize the number of times the participants needed to attend clinic visits. This led to a follow-up period that was inconsistent across the population relative to the time of treatment initiation. Furthermore, the pandemic may have contributed to the early withdrawal of some participants, which may have influenced the study outcomes since we planned for a rather small sample size to start with. Because of the small sample size, these results should be considered suggestive, not conclusive. There were several outcomes that approached but did not meet statistical significance that may have reached the threshold with a slightly larger population. Future studies with larger sample sizes are needed to provide additional insights.
Our study was also conducted at a single-center suburban private practice with limited patient diversity. Potential recruitment bias in our study may have affected the baseline prevalence of pain 15 and could have skewed the results of our study. Access to the PCD in the current real world, resource-limited healthcare setting may be more restricted. Again, studies with larger, more diverse populations are needed.
Conclusion
This study shows conservative management with graduated compression and PCD significantly improves the leg circumference and bioimpedance measurements in patients with lipedema even after a single session. Leg pain, fatigue, and tightness also show significant improvement in both treatment groups suggesting that, at a minimum, women with lipedema should be prescribed compression garments. The RAND SF-36 quality-of-life and the PROMIS mobility measures showed improvements that did not reach statistical significance in this small study. Further research into nonsurgical treatments of lipedema is needed with larger sample sizes to confirm our findings and see if the quality-of-life improvements reach statistical significance.
Supplemental Material
Supplemental Material - Effect of pneumatic compression device and stocking use on symptoms and quality of life in women with lipedema: A proof-in-principle randomized trial
Supplemental Material for Effect of pneumatic compression device and stocking use on symptoms and quality of life in women with lipedema: A proof-in-principle randomized trial by Thomas Wright, Crystal D. Scarfino, and Ellen M. O’Malley in Phlebology
Footnotes
Acknowledgements
We would like to thank Marc Schwartz for the statistical analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TW has been paid as a speaker for Tactile Medical. EMO was paid by Tactile Medical to assist the lead author with the writing and preparation of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was support by Tactile Medical through a 1-year lease of the InBody 770; a grant to the site for investigator and coordinator time; IRB fees; data management, statistical, and medical writing support.
Ethical approval
This study was approved by Advarra IRB (protocol number PRO00041073)
Guarantor
TW takes responsibility for the accuracy and appropriateness of this article.
Contributorship
TW researched literature, conceived the study, developed the protocol, interpreted data, and contributed to the manuscript writing. CDS was involved with gaining ethical approval, patient recruitment and data collection. EMO wrote the first draft of the manuscript and contributed to data interpretation. All authors reviewed approved the final version of the manuscript.
Trial Registration
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
