Abstract
Objectives
To evaluate the risk of symptomatic venous thromboembolism (VTE) recurrence at 3 months in relation to treatment duration, according to baseline risk factor profiles, in patients with superficial vein thrombosis (SVT) treated with intermediate dose of tinzaparin.
Methods
We performed a pooled analysis on individual data from two prospective studies designed to assess the efficacy and safety of tinzaparin in intermediate dose (131 IU/kg) in patients with SVT. Treatment duration was at the treating physician’s discretion. All patients were followed up for at least 3 months.
Results
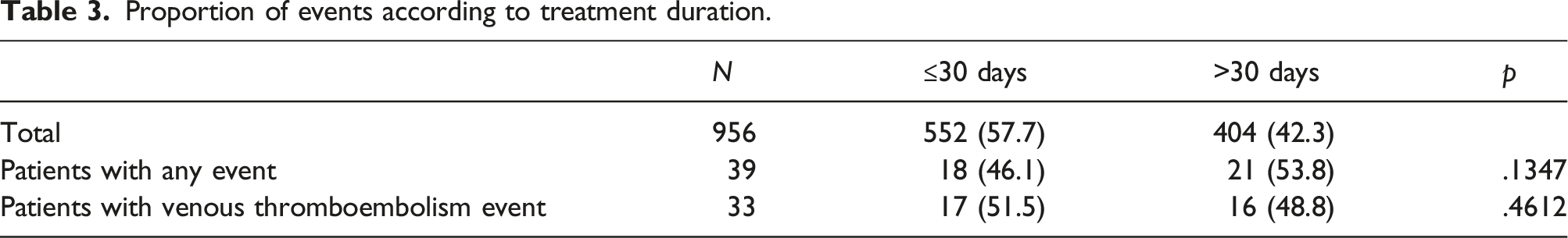
A total of 956 patients (65% female, mean age 58.7 ± 13.7 years) were included. The median treatment duration was 30 days (range, 3–200 days). History of deep vein thrombosis (DVT), location of SVT above the knee, and palpable induration were the only independent factors associated with prolonged treatment duration. During follow-up, 95.9% of patients were event free. Outcomes-related adverse events occurred in 39 (4.1%) patients and their median duration of treatment was 33 days (range, 7–200 days). Recurrent VTE events occurred in 33 patients, including 22 cases of SVT recurrence, 8 cases of DVT, and 1 case of pulmonary embolism. The median time to the event was 29 (6–113) days. Recurrent thromboembolic events were not related to treatment duration as occurred in 17 patients (51.5%) treated up to 30 days and in 16 patients (48.8%) received prolong treatment (p = .46). Length of thrombus at the index event was significantly associated with higher risk for VTE recurrence.
Conclusions
Intermediate dose of tinzaparin for 30 days is an effective and safe treatment for SVT. The risk of recurrent VTE events may be higher in patients with greater amount of thrombus at index event.
Keywords
Introduction
Superficial vein thrombosis (SVT) is a common clinical condition affecting both patients with normal or varicose veins. 1 In most of the cases, SVT is a result of thrombus formation within a varicose or dilated incompetent vein due to venous stasis. 1 Historically thought of as a benign condition treated with nonsteroidal anti-inflammatory drugs (NSAIDs), due to concomitant inflammatory process; however many studies documented the association of SVT with deep vein thrombosis (DVT) and pulmonary embolism (PE).2–5 Nowadays, SVT is considered as a superficial form of venous thromboembolism (VTE) sharing the same risk factors and may occur concomitantly or in sequence with DVT and/or PE.2,5 A meta-analysis by Di Minno et al., reported that concomitant prevalence of DVT and PE with SVT was 18.1% and 6.9%, respectively. 5
Optimal treatment of SVT is not well established because of lack of quality randomized, controlled clinical trials in this patient population. 6 For the treatment of SVT anticoagulants, NSAIDs, elastic compression stockings, and surgery have been proposed. Survey data have shown that management of SVT varies widely.7,8 According to current guidelines, fondaparinux at prophylactic doses for 45 days is recommended (Class I, Level B). 9 Alternately, intermediate dose of low-molecular weight heparins (LMWH) should be considered for the treatment of SVT (Class IIa, Level B). 9
Although anticoagulation therapy is the mainstay of SVT treatment, the optimal dosage and duration of such treatment remains debatable. 6 The most recent Cochrane review on the treatment of SVT precluded a formal meta-analysis regarding anticoagulation with LMWHs due to heterogeneity of the evidence. 10 Several studies have demonstrated that intermediate doses of LMWs for 30 days are efficient to prevent short-term (≤3 months) thromboembolic complications.11–13 Others reported a considerable risk of late (≥3 months) VTE recurrent events justified an extent treatment duration.2,14
We performed a pooled analysis on individual data from two prospective studies designed to assess the efficacy and safety of tinzaparin in intermediate dose in patients with acute SVT of the lower limb. The objective of this study was to evaluate the risk of symptomatic VTE recurrence at 3 months in relation to treatment duration, according to baseline risk factor profiles, in patients with SVT treated with intermediate dose of tinzaparin.
Methods
Design
The SeVEN and SeVEN EXTension studies were two prospective observational studies designed to investigate patients with SVT of the lower limbs treated with intermediate dose of tinzaparin.11,12 The studies were performed by hospital- and office-based vascular physicians who regularly treat patients with SVT and are capable of conducting duplex ultrasound (DUS) for confirmation and monitoring of the thrombotic process. The studies were performed in consistent with the principles of the Declaration of Helsinki and protocols were approved by the institutional review board of each hospital. Written informed consent was obtained from all patients.
The study details have been reported.11,12 Briefly, patients were eligible for inclusion if they were ≥18 years of age and if they had symptomatic SVT of the legs, with thrombus length ≥5 cm confirmed by DUS and duration of symptoms less than 10 days. Eligible patients received treatment with intermediate doses of tinzaparin (75% of the therapeutic dose −131 UI/Kg) once daily for at least 14 days, regardless of the baseline risk factors and the clinical and DUS characteristics of the SVT. The duration of the treatment was at the treating physician’s discretion. The expected duration of treatment and any changes in dosage were not specified to the patients at the time of consent for participation to the study. Patients were ineligible if they had a contraindication to received anticoagulant therapy (recent surgery, trauma, or bleeding), if they required anticoagulant therapy at therapeutic dosages (proximal extension of the SVT <3 cm from the saphenofemoral (SFJ) or saphenopopliteal junction (SPJ)), history of DVT and PE within the last 6 months, or body mass index (BMI) > 35 kg/m2. A full list of inclusion and exclusion criteria of both studies is provided in Supplementary Appendix 1.
Patient characteristics prospectively recorded included demographics, comorbidities, risk factors for VTE events, SVT complications along with treatment characteristics, and outcome. According to BMI, patients were categorized in three subgroups: with normal weight (18.5 < BMI <24.9 kg/m2), overweight (25 < BMI <29.9 kg/m2), and obese (BMI >30 kg/m2). Patients were followed for 3 months. Each patient was seen in the office for a 30 days (visit 2) follow-up appointment followed by an office or phone appointment at 90 days (visit 3), and data available from the physical or phone contact were collected. DUS was mandatory in every physical visit for confirmation of SVT, estimation of the thrombotic process including measurement of thrombus length and exclusion of DVT. In addition, patients were seen if any new event occurred between follow-up appointments, and DUS findings and clinical status were recorded.
Outcome measures
Efficacy and safety outcomes were identical for both studies. The primary efficacy outcome was symptomatic DVT and/or PE, extension of thrombus toward the SFJ or SPJ, or recurrence of SVT up to week 12 in relation to treatment duration, in patients with SVT treated with intermediate dose of tinzaparin (see Supplementary Appendix 2 for definitions). The principal safety outcome was major bleeding. Secondary outcomes were to determine risk factors for VTE recurrence. Suspected VTE and bleeding events were objectively confirmed (clinically and with DUS) and reviewed by an adjudication committee.
Risk factors for VTE recurrence
We assessed the influence of the following potential risk factors for VTE recurrence: age; sex; BMI; SVT characteristics (thrombus length, location of thrombus, and distance from the SFJ/SPJ < 3 cm); and chronic risk factors as follows: personal or family history of VTE; cancer; autoimmune disease; soft tissue infection; and reduced mobility (bed rest, hospitalization, long-hour travelling, and work with long hour sitting or standing).
Statistical analysis
Categorical data are presented as counts and corresponding percentages, whereas continuous data are presented as means and ranges. Time to event parameters was measured from the date of diagnosis and was estimated using Kaplan–Meier curves, while the log-rank test was used for assessing statistically significant differences. The chi-square test was used for testing categorical variables, significance was determined at the level of 5%, and all tests were two-sided. Logistic regression analysis was performed to assess potential risk factors related to the duration of treatment (i.e., duration of treatment ≤ 1 month versus > 1 month). In order to investigate for possible confounding factors and adjust for them, a multivariate analysis based on logistic regression was performed. Variables entered in this model were those that statistical significance in the univariate analysis was p < .20. The statistical analysis was performed using SASVR software (SAS for Windows, version 9.4, SAS Institute Inc., Cary, NC).
Results
Patients
A total of 956 patients were included in the analysis: 296 from the SeVEN study and 660 patients from the SeVEN EXTension study. Details of patients excluded or lost to follow-up are shown in Figure 1. The mean age of the patients was 58.7 ± 13.7 years (range, 19–88 years), whilst 65% of them were female. Overweight or obese were 665 (69.5%), restricted mobility was reported in 166 (17.3%), whereas varicose veins (VVs) were present in 601 (62.8%) patients. The great saphenous vein (GSV) was involved in 556 (58%) of the patients, in 258 (27%) of them in the above knee segment, the small saphenous vein (SSV) in 95 (10%), and the varicose tributaries in 595 patients (62.2%). In 45% of the cases, the thrombotic process involved concomitant superficial axial veins (GSV or SSV) and the varicose tributaries. Almost one-third of the patients (31.2%) had a medical history of thromboembolic events with SVT to be the most common one (27.3%). Among patients, there were 3 cases with active cancer and 3 with a history of cancer (totaling 0.6% of the study population). Study flow diagram. BMI: body mass index; DVT: deep vein thrombosis; PE: pulmonary embolism; SVT: superficial vein thrombosis.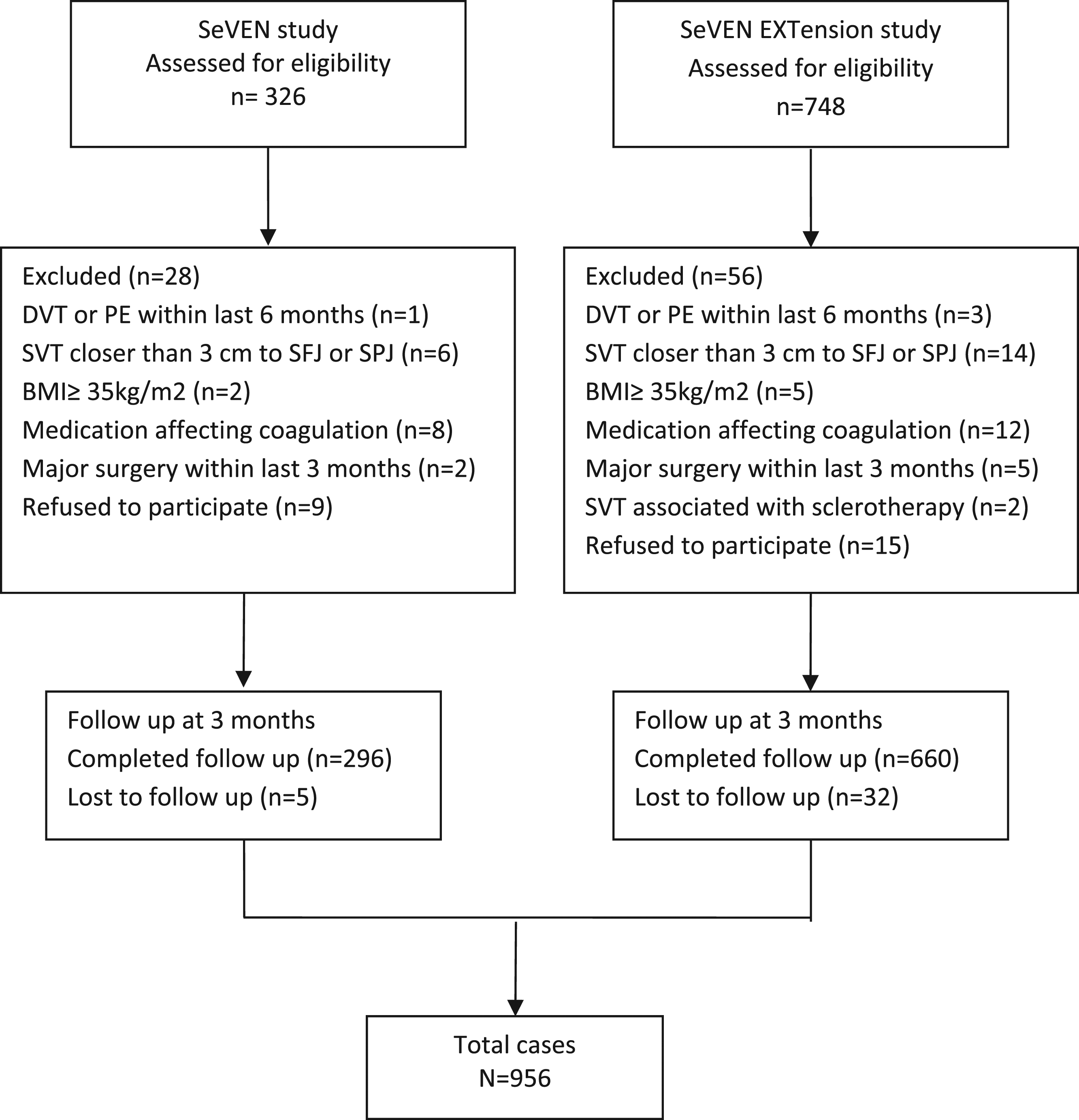
Patient’s demographics and clinical characteristics in relation to treatment duration.
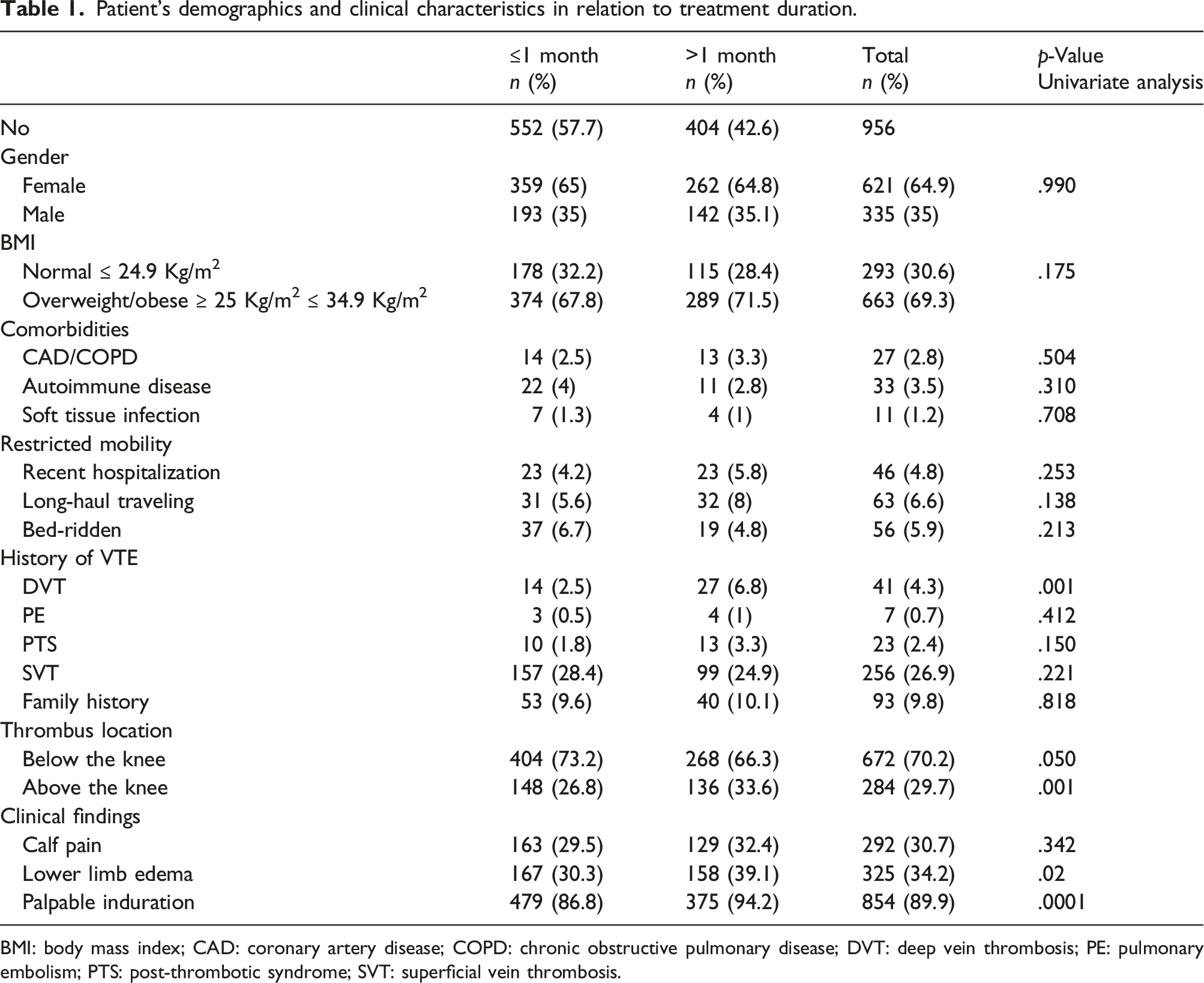
BMI: body mass index; CAD: coronary artery disease; COPD: chronic obstructive pulmonary disease; DVT: deep vein thrombosis; PE: pulmonary embolism; PTS: post-thrombotic syndrome; SVT: superficial vein thrombosis.
Efficacy outcomes
Efficacy outcomes.

DVT: deep vein thrombosis; CRNMB: clinically relevant non-major bleeding; PE: pulmonary embolism; SFJ: saphenofemoral junction; SVT: superficial vein thrombosis.
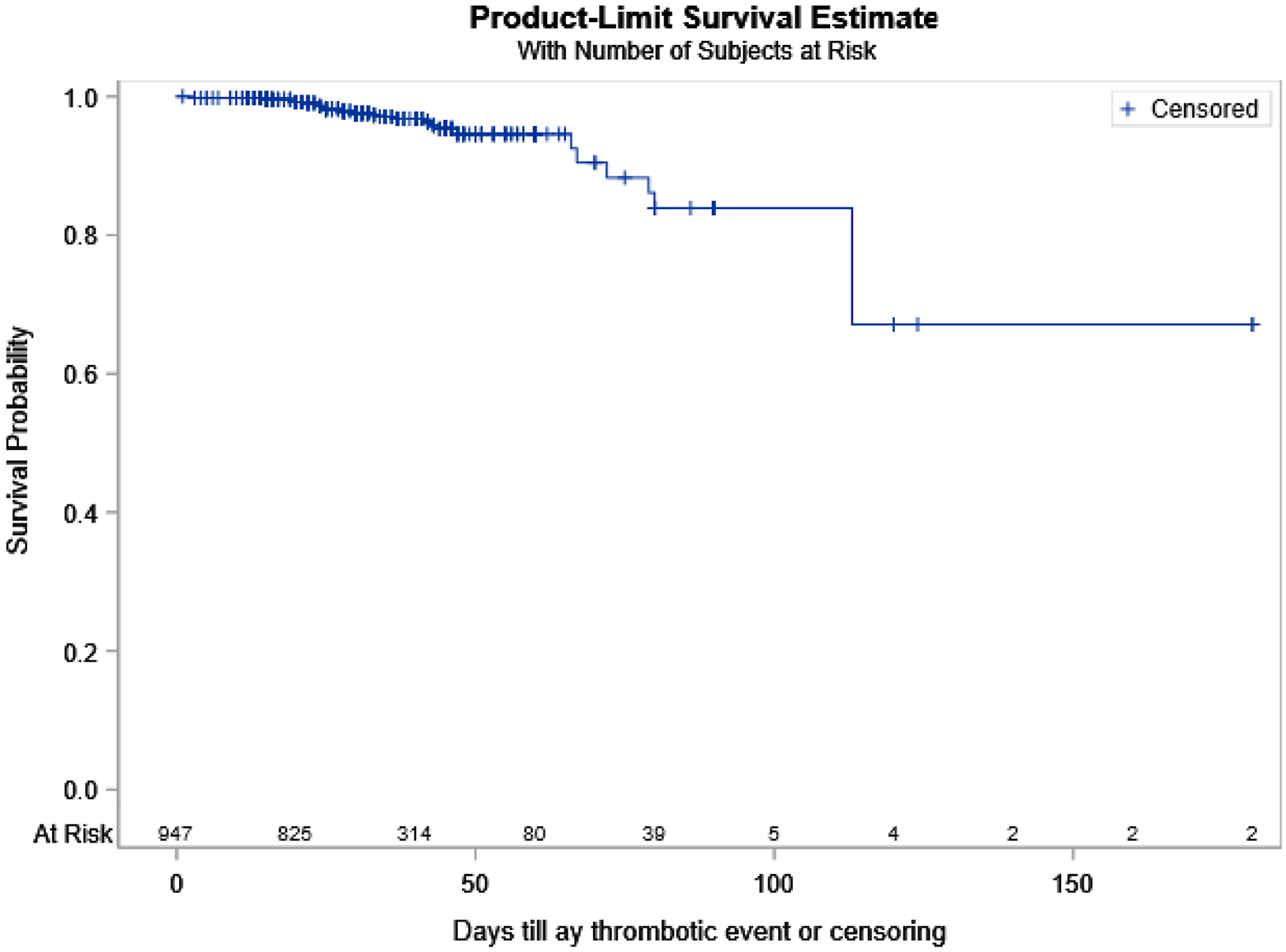
Kaplan–Meier curves for the occurrence of any thromboembolic event.
Proportion of events according to treatment duration.
Risk factors for venous thromboembolism recurrence.
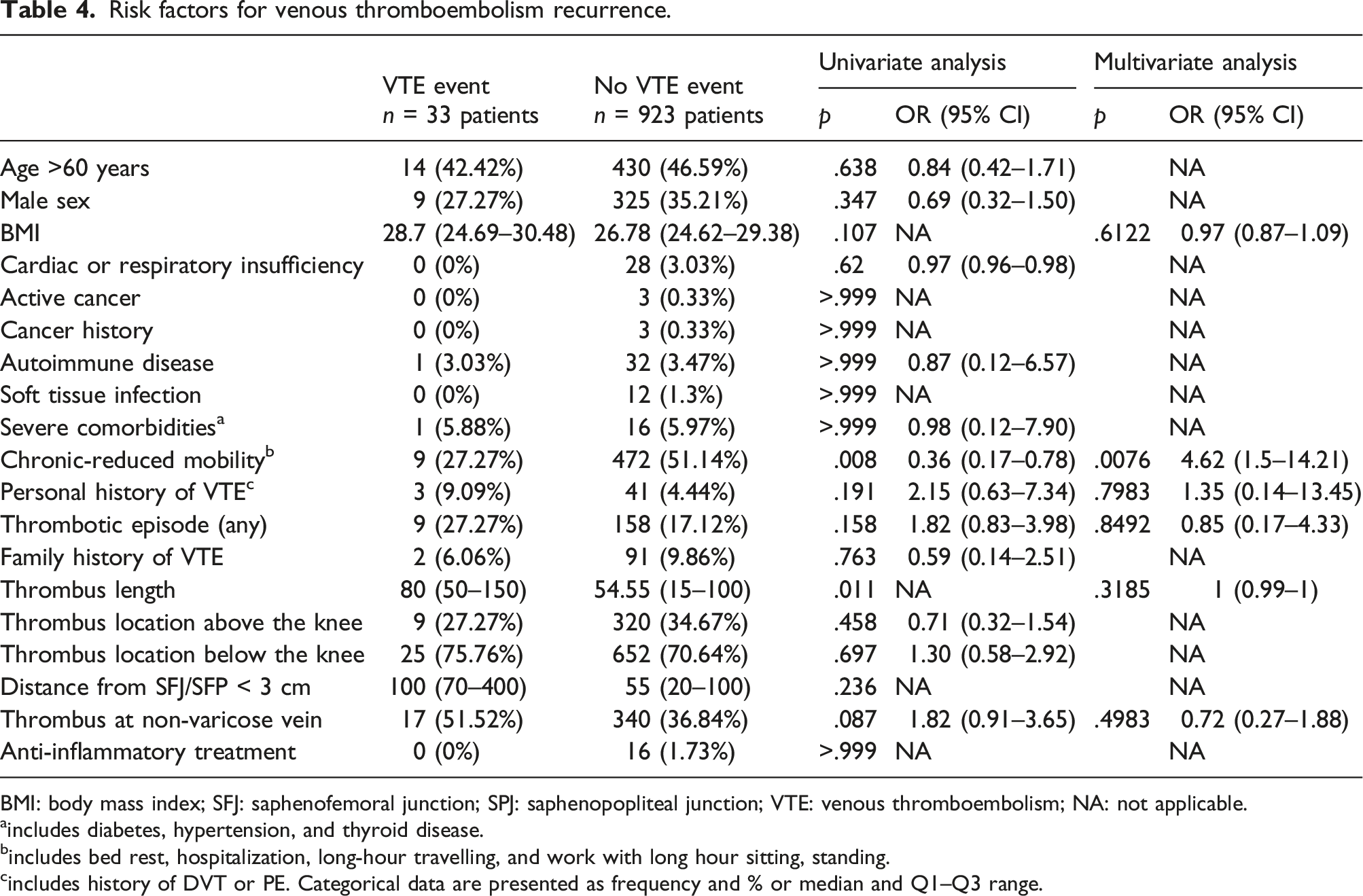
BMI: body mass index; SFJ: saphenofemoral junction; SPJ: saphenopopliteal junction; VTE: venous thromboembolism; NA: not applicable.
aincludes diabetes, hypertension, and thyroid disease.
bincludes bed rest, hospitalization, long-hour travelling, and work with long hour sitting, standing.
cincludes history of DVT or PE. Categorical data are presented as frequency and % or median and Q1–Q3 range.
Discussion
Patients with SVT often are treated with LMWs in various dosages and durations. In this study, we investigated the treatment of SVT with intermediate dose of tinzaparin (131 UI/Kg), in outpatient’s managed by hospital-based and office-based vascular physicians. The results of this study confirm that intermediate dose of tinzaparin for 30 days is an effective and safe treatment for SVT. In addition, length of thrombus at the index event was significantly associated with increased risk of VTE recurrence.
The optimum duration and dosage of LMWHs were investigated in numerous studies.6,11–15 In the STEFLUX study, a randomized controlled study, patients were randomized to receive either parnaparin 8500 IU (intermediate dose) for 10 days followed by placebo for 20 days or 8500 IU for 10 days followed by 6400 IU for 20 days (intermediate doses) or 4250 IU (prophylactic dose) for 30 days. 11 Of 664 randomized patients, recurrent VTE events occurred in 15.6% of the patients with the 10 day intermediate dose, in 1.8% with the 30 day intermediate dose, and in 7.3% with the 30 day prophylactic doses. The study concluded that, an intermediate dose of parnaparin for 30 days is superior to either a 30 day prophylactic dose or a 10 day intermediate dose. At 90 day follow-up period, the incidence of new VTE events was on average 7.5% and similar across the study groups. In accordance to that, our analysis showed a 3.4% incidence of recurrent VTE events with no difference between groups (3.07% in patients with 30 day treatment duration versus 3.96% in patients with prolong treatment). A recent meta-analysis by Duffett et al., reported that longer duration of treatment with LMWH (30–42 days) was not associated with significant lower incidence of VTE events compared to shorter duration of treatment (< 30 days) (10.0 events per 100 patient-years, 95% CI: 5.3–16 vs 18.3 events per 100 patient-years, 95% CI: 8.3–31.1, respectively). 6 These observations indicate that prolonged treatment with LMWH does not ensure better results on recurrent VTE events.
In the present analysis, the majority of the patients were middle-age overweight women with a history of VVs; almost one-third of the patients had the GSV above the knee involved and a medical history of previous thromboembolic events, representing a cohort of patients with various risk factors, relevant to other studies.1,16 The majority of patients received treatment for approximately 30 days, and the only factors associated with prolonged treatment were a history of DVT, presence of thrombus above the knee, and persistence of symptoms such as palpable induration. History of past VTE events and SVT involving the GSV above the knee or SSV in the calf have been associated with increased risk of recurrent thrombotic complications, justified a prolonged treatment.14,16–18 The CALISTO trial, in which the treatment duration was 45 days (fondaparinux 2.5 mg versus placebo), was criticized for its long treatment period making the treatment of any SVT to be not cost-effective. Goldman et al., reported that, such duration of treatment may be reasonable only in patients with severe symptoms, thrombosis close to SFJ/SPJ, or in recurrent disease. 19 Others recommended that, in patients with extensive SVT (>5 cm in length) and with high thromboembolic risk, such as those with recurrent SVT, or SVT close to the junction with the deep veins, or related to malignancy or thrombophilia, may receive a therapeutic or intermediate anticoagulant dose for a longer period, or alternatively, be switched to prophylactic anticoagulation after 30–45 days of initial treatment, for a total of 3 months of anticoagulant treatment. 14
In our study, we were able to confirm that length of thrombus at the index event was associated with increased risk of VTE recurrence at 3 months. In accordance to that, a recent published prospective observational study (INSIGHTS-SVT study) in which patients with SVT were treated with different anticoagulant regimes and doses reported that thrombus length appeared to increase the risk of recurrent VTE events by 4% per centimeter (HR 1.04; 95% CI 1.02–1.06; p < .001). 20 The risk of recurrent thromboembolic events may be higher in patients with greater amount of thrombus. The importance of thrombus length should be systematically investigated in future SVT studies in order to confirm this finding. In addition, a dedicate revised DUS protocol for SVT diagnosis and follow-up, including length of thrombus, by board-certified physicians would offer more accurate monitoring and treatment strategies.
Our study failed to identify others known risk factors for VTE recurrence such as age, male sex, obesity, cancer, and previous episode and/or family history of VTE.2,11,16,21–23 Surprisingly, chronic-reduced mobility was found to have a protective role in VTE recurrence. These results may be the consequence of confounding factors, such as therapeutic management and thus more aggressive and prolong treatment were prescribed in patients with risk factors for VTE recurrence.
Several issues about the treatment of SVT still require further investigation to clarify disparities in the current real-life clinical practice.7,8 Patients with SVT are commonly treated with LMWH, fondaparinux, and direct oral anticoagulants (DOACS) in variable doses and duration. The SURPRISE study, a non-inferiority RCT study, investigated the role of low-dose oral rivaroxaban (10 mg) in comparison with subcutaneous prophylactic dose of fondaparinux (2.5 mg) in patients with SVT treated for 45 days. 24 The primary efficacy outcome (composite of symptomatic DVT, PE, progression or recurrence of SVT, and all-cause mortality at 45 days) was non-significantly higher (3%) in the rivaroxaban group than in the fondaparinux group (2%; p = .003 for non-inferiority). The authors concluded that rivaroxaban 10 mg for 45 days is a simple and effective alternative to injectable anticoagulant treatment; however, higher rates of VTE and bleeding complications might be expected with such a treatment. 24 Kearon et al., investigated the efficacy and safety of rivaroxaban 10 mg once daily compared with placebo for isolated SVT in a phase three, multi-center, RCT of 85 patients. 25 Thrombotic complications were reported in 2.3% of the patients receiving rivaroxaban compared with 11.9% receiving placebo (absolute risk reduction = 9%; 95% CI: −22 to 5.9%). This study was terminated early due to slow recruitment rates. Although favorable results in reduction of thrombotic complications, SVT is not a licensed indication for rivaroxaban at present. Future large RCTs comparing low dose of DOACS with placebo or with intermediate dose of LMWH are needed to clarify the role of DOACS in the treatment of SVT. One of the major strengths of our study is that we used individual patient data. Furthermore, the SeVEN and SeVEN EXT studies had the same study protocol: inclusion and exclusion criteria were identical and SVT was symptomatic and diagnosed with a standardized whole leg DUS exploration and followed for 3 months. Patients were recruited in the same country and were treated in the same way (intermediate doses of tinzaparin once daily for at least 14 days; and duration of treatment was at the treating physician’s discretion). To ensure uniformity and to reduce bias, efficacy and safety outcomes in both studies were centrally adjudicated by the same committee whose members were blinded to treatment allocation and outcome status.
The main limitations of our study are the retrospective nature and the potential selection and treatment bias of the included studies. Treatment duration was at the treating physician’s discretion, and patients were treated by hospital- and office-based vascular physicians. Nevertheless, it represents the current practice followed by vascular physicians in our country. Future studies should investigate the effectiveness of intermediate doses of LMWHs in reducing VTE recurrent events in comparison with placebo or with other anticoagulants. Another limitation is that, although patient assessment was performed under a standardized protocol, different vascular physicians performed DUS, and this may have some impact in the outcomes. Efforts were made to minimize the possibility of inter- and intraobserver variability with the use of a standard ultrasound protocol in the assessment of researcher-reported outcomes. Additionally, all outcomes were objectively confirmed (clinically and with DUS) by an adjudication committee blinded to treatment outcomes. Galeandro et al., 26 developed a three-dimensional (3D) software for ultrasound evaluation of lower limbs venous system that could reduce the risk of such bias and future studies might benefit of that. This software allows physicians to create a 3D venous map of both the hemodynamics and the morphology of the veins in the lower limbs that is more comprehensive and reliable than the standard report. Regarding our study population, we feel that it is a representative cohort of SVT patients with different risk factors for VTE recurrence. Finally, with the telephone follow-up at third visit, underreporting of few events cannot be excluded.
Conclusions
Intermediate dose of tinzaparin for 30 days is an effective and safe treatment for SVT. The risk of recurrent VTE events may be higher in patients with greater amount of thrombus at index event. Future studies should identify subgroup of patients that is likely to benefit from longer treatment duration.
Supplemental Material
Supplemental Material—Risk of recurrent thromboembolic events according to treatment duration in patients with superficial vein thrombosis treated with intermediate dose of tinzaparin
Supplementary Material for Risk of recurrent thromboembolic events according to treatment duration in patients with superficial vein thrombosis treated with intermediate dose of tinzaparin by Christos Karathanos, Stavros K Kakkos, Georgios Georgiadis, Christos Ioannou, Spyros Vasdekis, Dimitrios Chatzis, Panagiotis Latzios, and Athanasios D Giannoukas On Behalf of the SeVEN Collaborators in Phlebology.
Footnotes
Acknowledgments
The authors would like to thank LEO Pharmaceutical Hellas S.A. for the support in order to make possible the undertaking of this study.
Authors’ note
The following are the SeVEN and SeVEN EXT collaborators: (1) Athanasios Giannoukas, Christos Karathanos, Miltiadis Matsagkas, and Konstantinos Spanos, Department of Vascular Surgery, University Hospital of Larissa, Greece; (2) Konstantinos Nikolakopoulos and Stavros Kakkos, Ioannis Tsolakis, Department of Vascular Surgery, University Hospital of Patras, Greece; (3) Georgios Georgiadi and, Miltos Lazarides, Department of Vascular Surgery, University Hospital of Alexandroupolis, Greece; (4) Georgios Papadopoulos and Christos Ioannou, Department of Vascular Surgery, University Hospital of Heraklion, Crete, Greece; (5) Sotiria Mastoraki and Spriridon Vasdekis, Department of Vascular Surgery, ‘‘ATTIKON’’ University Hospital, Athens; (6) Chrysostomos Maltezos, Department of Vascular Surgery, ‘‘KAT’’ Hospital, Athens, Greece; (7) Georgios Trelopoulos, Vascular Surgery Unit, Department of Cardio-Thoracic Surgery, ‘‘Papanikolaou’’ Hospital, Thessaloniki, Greece; (8) Dimitrios Chatzis—private vascular surgeon, Ioannina, Greece; (9) Panagiotis Latsios—private vascular surgeon, Kozani, Greece; (10) Ioannis Papakostas—private vascular surgeon, Ioannina, Greece; (11) Konstantinos Goumas—private vascular surgeon, Agrinio, Greece; (12) Dimitrios Alexandropoulos—private vascular surgeon, Patra, Greece; (13) Christos Rigopoulos—private vascular surgeon, Athens, Greece; (14) Triantafillos Iordanidis—private vascular surgeon, Kozani, Greece; (15) Michalis Peroulis—private vascular surgeon, Ioannina, Greece; (16) Vasileios Saleptsis—private vascular surgeon, Larissa, Greece; (17) Athanasios Siafakas—private vascular surgeon, Athens, Greece; (18) Konstantinos Seretis—private vascular surgeon, Athens, Greece; (19) Ilias Tzortzis—private vascular surgeon, Kalamata, Greece; (20) Dimitrios Likopoulos—private vascular surgeon, Veria, Greece; (21) Christos Papasideris—private vascular surgeon, Athens, Greece; (22) Apostolos Saliveros—private vascular surgeon, Athens, Greece; (23) Ioannis Tzorbatzoglou—private vascular surgeon, Athens, Greece.
Contributorship
CK was responsible for the data analysis, literature search, writing, and review of the study. AG was responsible for the concept, design, data analysis, and review of the study. KMN, GG, CM, SV, GT, GP, KS, ML, MM, IT, SM, SK DCH, PL, IP, KG, DA, CR, TI, MP, VS, AS, DL, CP, AS, and ITZ collected the data. All authors reviewed, amended, and agreed the final draft. CK and AG drafted the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was performed in a manner consistent with the principles of the Declaration of Helsinki.
Informed consent
All patients have been informed about the nature of the study and have provided written informed consent.
Guarantor
CK.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
