Abstract
Objective
The use of cyanoacrylate products (CA) in incompetent perforator vein (IPV) treatment has not been thoroughly examined. The primary objective of this study is to describe the technique of ultra sound guided direct injection of IPV with CA, and secondarily to determine early closure rates and safety of this technique.
Methods
A retrospective analysis of patients undergoing IPV injection at two centres between 2015-2018 was conducted. Demographics, CEAP classification and IPV location were collected. Outcomes were assessed at two follow-up appointments.
Results
A total of 83 perforator vein injections were completed. CEAP classifications include C2 – C6 classes. Location of perforators were posteromedial (6%), femoral canal (9%), paratibial (14%), and posterior-tibial (71%). IPV closure rates were 96.3% at initial follow-up (16 ± 2 days). Closure rates decreased to 86.5% at second follow-up (72 ± 9 days). There were no deep vein thromboses during follow-up. One patient developed septic thrombophlebitis that was successfully managed with antibiotics.
Conclusion
Ultrasound-guided CA glue injection is a simple and low risk procedure that effectively closes incompetent perforator veins.
Keywords
Introduction
While there have been great advances in the treatment of chronic venous disease, the treatment for incompetent perforator veins (IPV) is less well developed. 1 IPV are known to play a significant role in not only the advancement but also the recurrence of lower limb venous insufficiency.2 –5 Despite this, there continues to be no clear consensus regarding the best treatment or indication for treatment for IPV.2,4,6 Treatment of perforators in CEAP class 2 and class 3 patients is debatable, however, clinical practice guidelines of the Society for Vascular Surgery and the American Venous Forum recommend treatment of perforating veins with reflux >500 ms and a vein diameter >3.5 mm located near healed or active venous ulcers. 7 In addition, multiple modalities for the treatment of IPV are found in the SVS and AVF clinical practice guidelines including surgery such as subfascial endoscopic perforator surgery (SEPS), thermal ablation (laser and radiofrequency) and ultrasound (US) guided foam sclerotherapy. 7
At our centers, patients with advanced chronic venous insufficiency (CEAP class 5-6), IPV in the vicinity or underneath an ulcer are treated simultaneous with any superficial long axial reflux. We feel that simultaneous treatment of IPV plus superficial long axial reflux in CEAP class 2-4 patients with a supportive Brodie-Trendelenburg test results in improved symptom control and less need for completion treatments or staged perforator treatments. It occurred to the authors that during cyanoacrylate (CA) based closure of superficial long axial reflux the remaining unused CA glue could be repurposed during the same procedure for closure of pathologic IPV. We therefore discussed this as an off-label option with patients who we deemed to have clinically significant IPV based on the above criteria. The purpose of this study is to describe this technique, as well as investigate the early closure rates, clinical outcomes and complications following US guided direct injection of CA-based products into IPV.
Methods
All patients presenting to the Vancouver General Hospital Vascular Surgery clinic and Victoria Royal Jubilee Hospital Vascular Surgery clinic who received CA embolization of IPV between 2015 and 2018 are included in this study. Data was retrospectively analyzed from clinic electronic medical record databases. Patient demographics, class of venous disease, location of perforator vein treatment, amount of CA used, primary and secondary follow-up outcomes and complication, and need for post-procedure sclerotherapy were collected. Institutional approval for the study was obtained by the University of British Columbia Clinical Research Ethics Board (H20-00946). Retrospective individual patient consent to be involved in this study was not obtained, as a waiver of individual consent was approved by ethics. Individual informed consent was still obtained to receive treatment of IPV.
All patients, regardless of CEAP class, underwent formal US evaluation through the vascular lab. Perforator veins were deemed incompetent if at least 3.5 mm in diameter with reflux lasting a minimum of 500 ms as per guideline definitions.7,8 If US assessment revealed combined axial and perforator incompetence, IPV were assessed for clinically significant incompetence utilizing the Brodie-Trendelenburg test. The Brodie-Trendelenburg test is most useful in CEAP classes C2/C3, as C4-C6 patients had different goals of treatment. If varicosities filled within fifteen to thirty seconds upon standing with the tourniquet in place or with manual pressure applied to the incompetent axial vein, it was determined there was an offending perforator requiring treatment. If, however, veins did not fill with manual pressure or tourniquet applied, while radiographically incompetent, this perforator vein did not indicate contribution to reflux, and could therefore be monitored for progression or treated in a staged approach. If patients had a positive Brodie-Trendelenburg test, with supportive US findings of perforator incompetence, CA embolization of the IPV was undertaken. The authors found that the number and size of IPVs did not necessarily correlate directly with the CEAP classification system. All classification levels of Clinical-Etiology-Anatomic-Physiology (CEAP) were eligible for inclusion in the study so long as the above criteria for IPV were met. Patients were treated according to the British Columbia Medical Service Plan guidelines, meaning CEAP 2 patients were eligible for treatment with significant symptoms.
Procedure
Prior to starting the procedure, patients were informed that this procedure is an off-label use of this product and were notified of the risks and benefits. Consent to receive treatment was then obtained. All procedures were completed in a sterile fashion in an ambulatory care setting. The majority of our patients received concomitant superficial long axial reflux and this was carried out percutaneously under local anesthetic in the standard fashion. Any IPVs refluxing directly into the treated axial vein were “paved over” by puncturing into the axial vein distal to the perforator and are therefore not included in the data analysis. We then carried on with US guided direct CA injection of the IPVs. Patient positioning in the supine, prone, or lateral positions was dependent on the location of the perforator and on occasion necessitated repositioning. Tilting of the procedure table into reverse Trendelenburg was utilized to improve percutaneous access into the IPV.
The ultrasound is used to confirm location and incompetence status of the perforator vein. The small footprint of a 13-6 MHz or 10-5 MHz or “hockey stick” probe reduces the distance from the skin puncture to the IPV entry and steepens the approach angle to facilitate accurate puncture of the IPV. To maximize accuracy of needle entry, alcohol prep solution is used in place of sterile gel. This is not critical but does maximize sterility and removes the gel as a barrier to traverse. It is important to accurately mark the origin and orientation of the IPV as it exits the fascia and travels towards the skin. (Figure 1(a)) This determines the angle of entry into the IPV and determines the ergonomics of comfortably injecting the CA. (Figure 1(b)) Once the angle of entry is determined, the US is then positioned on the side of the patient that allows the operator to visualize the screen while maintaining the angle of entry. (Figure 1(c)) Note that the placement of the US will be determined by the handedness of the operator, the angle of entry, and the location of the IPV on the right or left leg. For example, a right-handed operator injecting a medial calf perforator on the right leg oriented superficial to deep in a cephalad direction would place the US on the patient’s left side. If there are multiple perforators to treat, all with varying directions of origin, the US machine will be moved to maximize ergonomics for injecting the perforator in the appropriate direction while maintaining visualization of the screen.
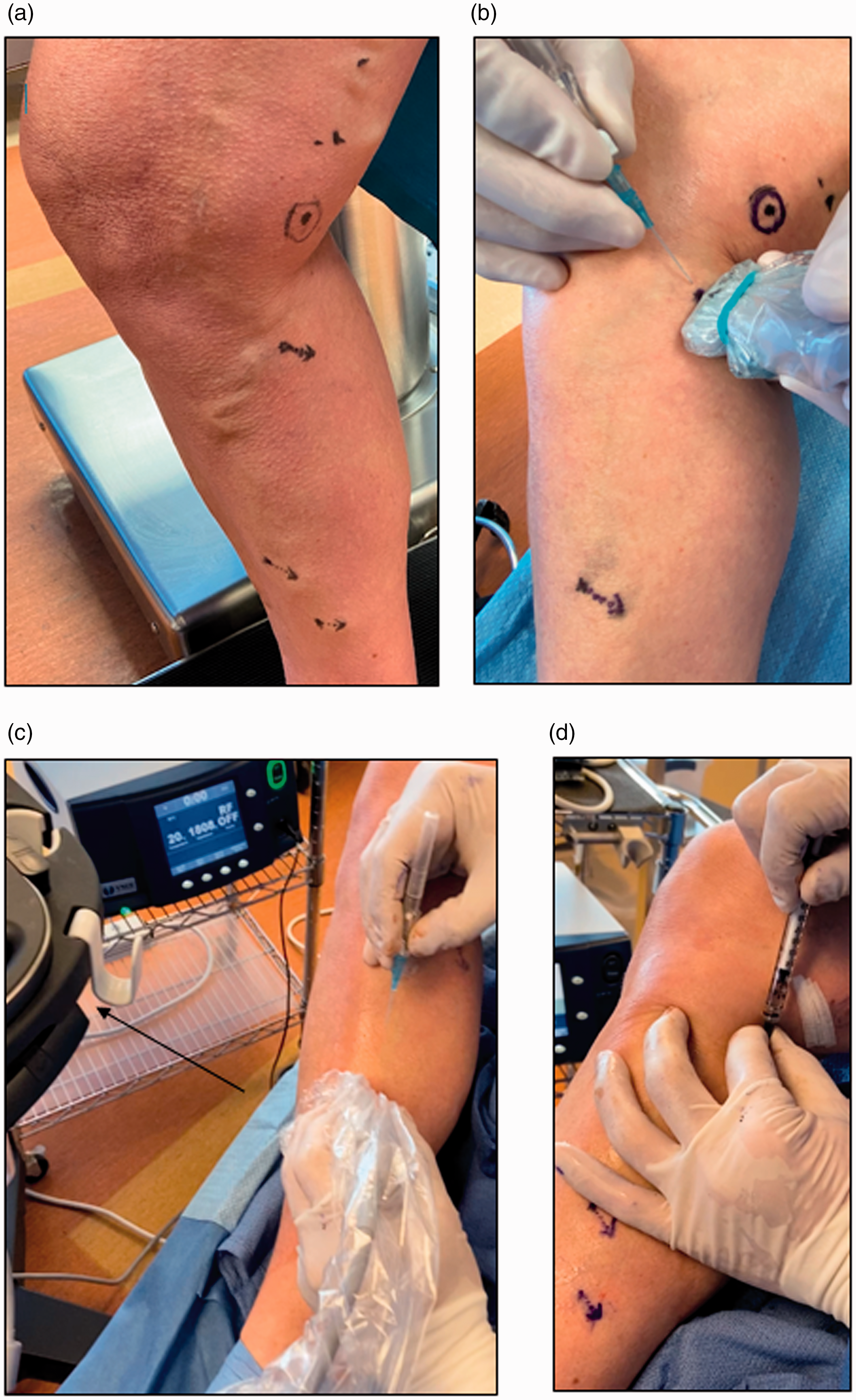
(a) Mark the orientation of the perforator as it exits the fascia and travels towards the skin, to determine entry of the needle. (b) Based on the angle of perforator, needle entry is executed in a deep to superficial direction (c) Positioning of the US machine to allow for comfortable visualization of the screen while maintaining the determined angle of entry. (d) Attachment of pre-filled syringe onto Jelco.
A 1 mL syringe is pre-filled with approximately 0.05 ml of CA/millimeter of IPV diameter. The volume can be adjusted upwards to 0.1 ml of CA/millimeter for longer IPVs. It is important to avoid glass syringes which will polymerize CA.
Approximately 0.5 cc of 1% xylocaine is first infiltrated at the proposed treatment site. A 1 inch 22-gauge Jelco (IV Catheter Jelco Protectiv Safety Straight 22 g × 1”Blue Smiths Medical ASD, Inc Model: 3050 Medex SKU: SMD-3050) is inserted into the IPV under US guidance with transverse or longitudinal views. If deeper IPV are targeted, a longer 1.5-2 inch 22 gauge needle or 4 Fr micro puncture set can be used. The deep vein-perforator junction is routinely attempted to be visualized; however, this can be technically challenging. If visualized easily, the needle is brought to within 5 mm of the junction, if not, the needle is brought just above the fascial level. When in the correct location, the Jelco is slid 2 mm over the needle tip and the needle is withdrawn from the Jelco. Back bleeding from the Jelco confirms intraluminal positioning. The Jelco must be steadied with one hand against the patient so as not to lose positioning of the Jelco tip within the IPV as evidenced by the constant back bleeding from the Jelco. The pre-filled syringe is attached, to the Jelco and the CA is injected swiftly to prevent polymerization of CA at the interface of the syringe and Jelco, and the Jelco tip within the vein. (Figure 1(d)) Once the syringe is emptied, the Jelco is withdrawn and external compression is applied for one minute. US is used to confirm closure of the treated vein. Utilizing compression wrapping post-procedure is not mandatory and was left to the discretion of the surgeon. We did not perform liquid or foam sclerotherapy simultaneous with the initial procedure but rather performed this for clinically relevant superficial varicosities (and not IPV) at the follow-up visits.
Follow-up and monitoring
Initial follow-up was completed within 4 weeks of the procedure, with secondary follow-up approximately 6 weeks afterward. At each appointment a focused history and physical exam was completed to assess for resolution of symptoms and signs of any associated complications. US was used at each follow-up appointment to assess for closure of the IPV and for complications related to extension of glue into the deep venous system. US guided foam sclerotherapy with 1 to 3% sodium tetradecyl foam with air in a 1:3 ratio was used during follow-up in some patients for residual superficial varicosities. If superficial varicosities were symptomatic at follow-up, US was used to first confirm ongoing closure of the previously targeted IPV. If this was recanalized it was considered a failure and would be treated with repeat CA injection.
Data analysis
The primary outcome of this study was successful closure of treated incompetent perforator veins on US assessment and resolution of symptoms. Data was compiled using Microsoft Excel (Microsoft Corporation, Redmond, WA). Results were presented as either percentage of total or mean ± standard error of the mean.
Results
Between 2015 and 2018, a total of 62 patients (55 females, 7 males, mean age 66), 67 legs (33 left, 34 right), and 83 incompetent perforator veins were treated. Information collected displayed in Table 1. Sixty-one out of 62 patients received concomitant long axial vein ablation with CA. The one patient who did not receive simultaneous long axial vein treatment had received treatment for ipsilateral axial varicosities 8 months prior to IPV treatment. Posterior tibial perforators were the most common perforator veins treated (Table 1). The number of perforators treated per patient ranged from 1 to 4. Volumes used ranged from 0.1-0.4 mL per perforator.
Demographics of patients treated with direct perforator injection of cyanoacrylate-based adhesives.

Initial follow-up occurred at an average of 16 ± 2 days, and second follow-up occurred on average 72 ± 9 days post procedure. Outcomes are displayed in Table 2. Fifty-five out of 62 patients presented for initial follow-up, 37 out of 62 patients presented for secondary follow-up. There were no cases of DVT. Superficial thrombophlebitis occurred in 16.4% of patients on initial follow-up and 2.7% of patient had superficial phlebitis at second follow-up. 12.7% and 56.8% of patients received additional sclerotherapy for residual veins at first and second follow-up respectively.
Outcomes of cyanoacrylate adhesive injection into perforator veins.

Immediate closure of treated IPV was 100% at the end of all procedures on the day of treatment. There were no cases in which access into the perforator was not possible resulting in abandonment of the procedure. At first follow-up, 2 patients had recurrence of IPVs yielding a success rate of 96.3% At secondary follow-up, an additional three patients had recurrent IPVs, bringing success rate down to 86.5% (32/37). One patient with an ulcer and recanalization was reinjected with a larger volume of CA and had successful closure of this IPV with ulcer healing afterwards. One patient developed post-operative septic thrombophlebitis. She was managed successfully with antibiotics but refused further CA injection. The third patient had good clinical resolution of varicose veins with asymptomatic perforator recanalization that was not treated. The remaining two patients both had chronic recurrent venous ulcers, deep venous reflux and evidence of patent IPV in the same region where treatment was provided. It was challenging to determine if this was true recanalization of the treated IPV or new development of IPVs in the treated region. One of these patients had two repeated CA injections and was eventually able to achieve ulcer healing. The other patient is a post-thrombotic patient with multiple recurrent IPV and recurrent ulcers. They had several injections with CA with mixed results.
Discussion
This study illustrates a new method which can be employed in the treatment of IPVs. This method demonstrates promising short-term results of IPV closure with low rates of recanalization and minimal complications. In follow-up US of recurrent perforators, it can be difficult to be certain of the mode of failure whether it reflected recanalization or a new IPV in the severe post thrombotic patients. Sometimes residual CA glue can be seen lining the wall of a previously treated perforator vein on US indicating recanalization at the same location of CA injection. Regardless, closure rates in this study are comparable to other similar publications on this method of IPV treatment. 6 Two of our 5 patients with recurrent IPV had deep venous reflux and chronic ulcerations, with one additional patient having post-thrombotic syndrome. These results reflect the higher failure rate in patients with deep venous reflux. Although this subset of patients is prone to failure, recanalized IPV can still be successfully re-treated with CA. Given our observation that CA ablation of IPV is safe at the volumes injected, we now dose the aliquot of CA injected into the IPV to 0.1 ml/millimeter diameter of IPV to a maximum of 0.5 ml to reduce recanalization rates. Future studies should investigate IPV treated with higher volumes.
The safe and successful treatment of superficial long axial reflux using CA has been well-established.6,9 –13 In contrast, the utility of CA in IPV is less well studied. There are two other series in the literature reporting success rates ranging from 76-100%2,6
There are some notable differences in our technique compare to these two series. We use direct Jelco IV access rather than the 7 Fr sheath system seen in Toonder et al.’s work. 6 We find the delivery of CA into the perforator vein is more precise with the 22-gauge Jelco. Our method also injected a lower volume of glue material inside the perforator (0.1 cc-0.4 cc vs 0.7 cc) without a higher rate of DVT or other complications.
Prasad et al. investigated the treatment of IPV with concurrent CA adhesives and foam sclerotherapy injections. Their results are promising showing 100% occlusion rates in all perforators at both 3- and 6-month follow-up. 2 Furthermore, 14 out of the 83 treated limbs were followed to 12 months, still exhibiting 100% occlusion. Although the results of their work suggest recanalization of treated IPV is unlikely, in our experience, recanalization is possible even after complete closure with some recurrent IPV. Prasad et al. attribute their high occlusion rates to the combined treatment of the strong adhesive properties of CA and the concurrent foam sclerotherapy of overlying subcutaneous collaterals. 2 In our study, foam sclerotherapy is only administered during the follow-up visits. It is possible that concurrent foam sclerotherapy contributes to lower recanalization rates as well as maintaining closure however this was not exclusively investigated in this paper.
Needle tip placement is another difference between Prasad et al.’s work and this study. By routinely attempting to narrow the distance of needle tip placement to within 5 mm of the deep vein-perforator junction, compared to 3-5cm in Prasad et al.’s method, we were able to treat long segment of the IPV and achieve near complete closure of the entire IPV length. With that said, Prasad et al. still saw complete closure of IPV even if only short segments of the IPV-superficial vein junction were treated. We share the concern of deep vein extension of CA into deep veins cited by Prasad et al particularly with injection into direct perforating veins. However, we did not appreciate this complication at the time of the initial injection nor at the follow up visits. We believe placing the Jelco tip within the perforator but at no closer than 5 mm from the perforator-deep vein junction delivers the CA to the precise location of the anatomic pathology and without extension into the deep system. For indirect perforators, we found that larger volumes of CA were necessary given the larger capacitance of the intramuscular sinuses found in continuity with the indirect perforators. In some indirect IPV cases, injection was completed into the sinus itself.
Considering this is an off-label use of CA, it is interesting to compare this method of IPV treatment with other modalities already used in common venous practice. One of the earliest papers investigating US-guided foam sclerotherapy for IPV treatment by Thibault and Lewis reported successful closure of IPV at 6 months post treatment in 83.7% of patients. 14 Since then, multiple papers have shown various occlusion rates of IPV dependent on methods of treatment. 2 A review of 68 patients undergoing US-guided sclerotherapy for IPV showed a 75.4% occlusion rate at mean follow-up of 20.1 months. 15 Other literature on the use of foam sclerotherapy further indicate that multiple treatments of the same IPV are required to successfully achieve complete closure. 16 Based on our results in conjunction with Toonder et al. and Prasad et al.’s it appears primary occlusion rates are higher in IPV treated with CA compared to US-guided foam sclerotherapy. However, given the lack of existing data looking at occlusion rates over 6 months for IPV treated with CA, drawing a conclusion of this magnitude may be premature and requires longer follow-up. While foam sclerotherapy is often used for perforators, the major advantage of CA over foam is operator control. Unlike foam sclerotherapy, CA solidifies instantly at the injection site, thus minimizing the risk of systemic embolization. In the authors experience, US sclerotherapy for IPV treatment has lower closure rates compared with CA. Another advantage of CA over foam sclerotherapy is the elimination of compression post procedure. Some of our authors do administer a few days of compression in patients with large varicose veins to prevent phlebitis, but we do not consider this mandatory for success of CA ablation of IPV.
IPV treated with endovenous radiofrequency ablation (RFA) show a similar range in occlusive results as sclerotherapy. 2 A pilot study completed in 2009 investigating the feasibility of utilizing RFA showed initial occlusion rates of 64% at 3-month follow-up. 17 This study launched further developments and research into this treatment method. Unfortunately, more recent analysis of IPV treated with RFA yield only moderate results with perforator vein closure rates ranging from 65-95% success.18 –20 The authors feel the RFA technique has significant limitations compared to CA for treatment of IPVs for several reasons. Firstly, RF ablation with the ClosureRFS utilizes a 6 Fr x12cm stylet and bulky handle making precise application of thermal transfer within the perforator challenging compared to the precision of the Jelco tip (Figure 2). Secondly, RFA requires tumescence of the entire treatment area, if one is to perform it under local anesthetic, while CA only needs a small amount of local anesthetic at the skin level. If there is significant movement of the tip outside the perforator, the heat sink effect is lost and the patient will experience significant discomfort. Also, if RFA energy does not contact the entire circumference in large (eg. 4 mm) perforators, making failure likely. For this reason, multiple applications for several minutes within the same perforator is recommended by the manufacturer. In general, our authors find RFA treatment time longer and more technically cumbersome when compared to US guided CA injection.
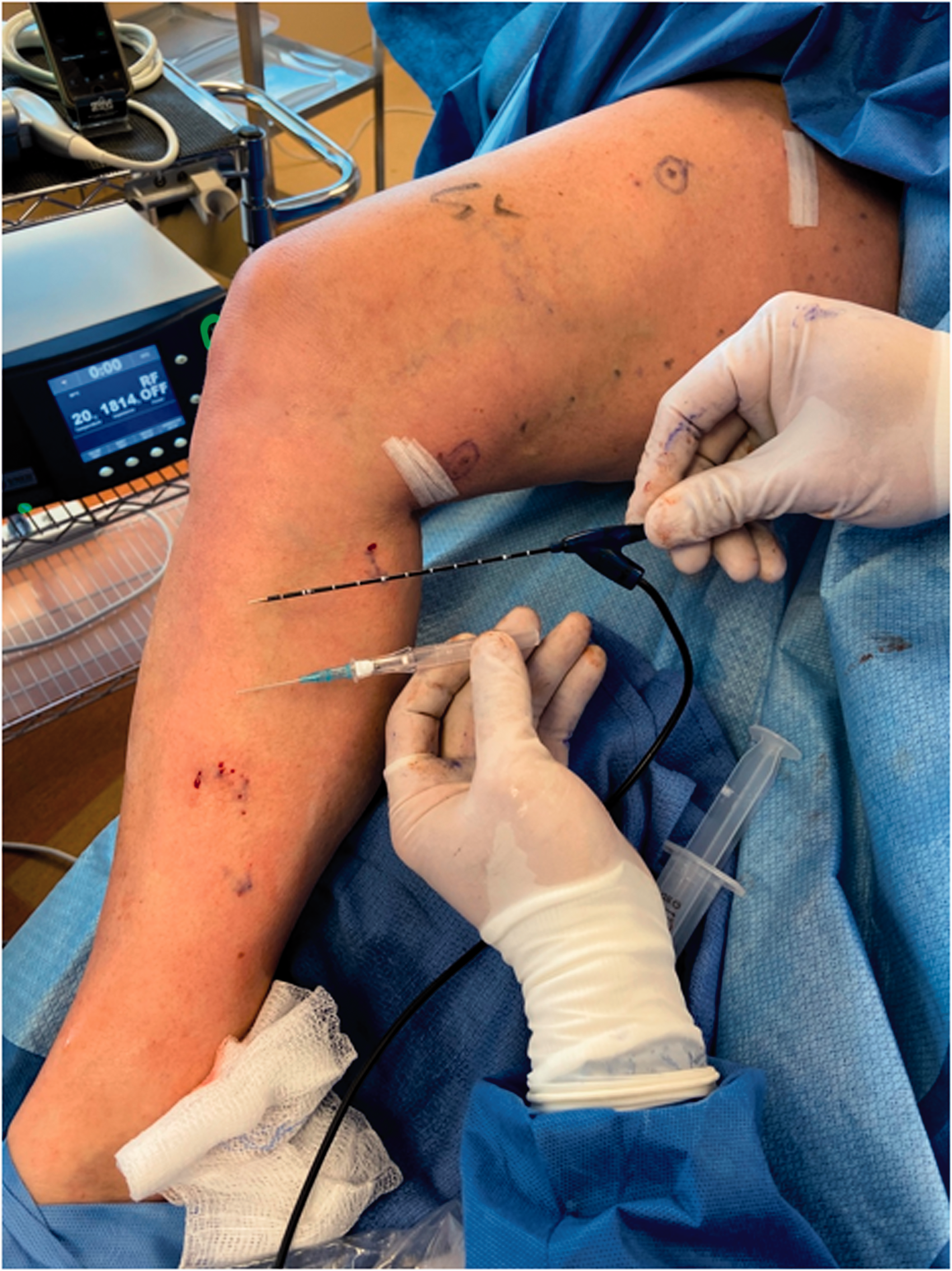
Size comparison between 6 Fr Venefit™ radiofrequency ablation probe versus 22-gauge angiocatheter used for direct injection.
When treating IPV with any method, possible complications must be taken into consideration. Complications of US guided sclerotherapy include, but are not limited to, allergic reaction, superficial thrombophlebitis, septic thrombophlebitis, DVT, and rarely localized skin necrosis.5,15,21 We demonstrated septic thrombophlebitis in one patient. Since CA is a non-absorbable foreign body, sepsis can be potentially difficult to eradicate. Our patient was successfully managed with intravenous antibiotics without needing excision of the CA or phlebitic segment. The authors recommend peri-procedural antibiotics for CA injection in patients with open ulcers. With respect to concerns of leaving a permanent foreign body in patients, while this has been discussed in the literature, it has not been explicitly investigated in IPV treatment. The low volume of CA used in IPV treatment compared to axial vein treatment should be taken into consideration. Our reported range was 0.1-0.4 mL, this is in comparison to the average 1.5-2.0 mL used in axial vein treatment.
The gravest complication of direct CA injection is extension of product into the deep venous system. In this study these events were not observed. Prasad et. al had 4 cases of extension of product into the deep venous system, however none of these situations results in clinical DVT. This is however considered a major complication and requires surveillance. Overall, the development of DVT is rare.6,15 –19
Although we report 100% initial success in this series, it is important to understand these patients were selected for this treatment based on technical criteria outline in this study. As with learning any new skill, there is an initial learning curve with respect to technique and volume of product to use. Our authors feel the learning curve is similar to that of treating IPVs with US guided foam sclerotherapy and is dependent on experience with US guided vein treatments. In total this technique averages approximately 5-15 minutes for IPV treatment. The authors feel treatment success is dependent on the preparation, set up, vein mapping and marking the direction of the perforator on the skin rather than the actual performance of the injection.
Our study does have limitations given its retrospective nature. It is important to recognize that secondary follow-up was on average around 3 months post-procedure. Given the short follow-up window, interpretation of this data should be limited to the fact that this is a technically feasible procedure which is safe and shows promising early results. Results pertaining to long term success remain to be seen in future studies and are paramount in determining success of this novel treatment method. Prospective collection of venous severity scores and quality of life assessments should be included in future studies.
An important confounder of this study is that 98% of patients received concurrent axial vein treatment, making resolution of symptoms difficult to decipher between a combination of both axial and perforator treatment or one over the other. According to Stuart et al.’s work investigating the natural history of IPV in the setting of axial vein treatment in 1998, there are up to 80% of IPVs which become competent after surgical stripping of the long axial vein. 22 While Stuart et al’s work does indicate that axial treatment may reverse IPVs, there are some facts about this study which need to be taken into consideration in comparison. Firstly, we refined our patient selection based on Brodie-Trendelnburg testing and only treated patients with IPV below the level of axial vein reflux. This differs from Stuart’s study where IPVs were counted with the leg in dependent positions. Additionally, patients included in their study received high ligation at the sapheno-femoral junction with associated stripping of the long and/or short saphenous vein plus multiple phlebectomies. This level of intervention would surgically remove any source of overflow into calf perforators. Our patients received only minimally invasive CA axial treatment without concomitant phlebectomies. As such, our patients would have had some remaining superficial varicosities with the potential for persistent, yet reduced, capacitance for overload of the IPV. We believe the Brodie-Trendelenberg test is important for identifying clinically important IPVs that may persist after treatment of long axial reflux. Secondly, the definition of success is different in our study as success was defined anatomically, based on complete closure of IPV following treatment, not reversal of incompetence. It is possible that some of the IPVs treated in this study may have reverted to being competent such that treatment was not clinically indicated. However treatment of long axial veins and IPVs were completed in one procedure, therefore this was not investigated in this study. Regardless, there have been other authors who cite that surgical correction of axial vein reflux does not abolish all IPV, and residual IPV is a risk factor for worsening venous disease despite axial treatment, particularly in cases of venous ulcers.23 –25 Nevertheless, we acknowledge this series may represent some degree of overtreatment of IPV with the exception of CEAP class 5-6 CVI. The selection criteria for staged versus concomitant IPV treatment continues to evolve for many practitioners who treat venous disease.
Conclusion
US guided direct injection of CA into clinically relevant IPV is a simple ambulatory procedure that is safe and effective for the purpose of occluding pathologic perforator veins. This treatment can be applied to all anatomic variants of IPV across a broad spectrum of venous disease. There is a small recanalization rate at early follow-up, however, injections can be repeated in the same location if the IPV fails to close. This technique can be adopted into most practices with expertise in chronic venous insufficiency. Further studies are needed to elucidate the long-term outcomes to define the role of this modality for treatment of IPV in patients with chronic venous insufficiency both with and without co-axial venous treatment.
Footnotes
Acknowledgement
Portions of this study were presented at the 2018 Canadian Society for Vascular Surgery Annual Meeting in Montreal, QC (September 28-29 2018) and the 2019 Western Vascular Society Annual Meeting in Maui, HI (September 28 – October 1 2019).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Joel Gagnon is a medical consultant for Medtronic. There is no financial contribution to the author or the authors institution from Medtronic which may benefit or lose financially from the results, conclusions, or discussion presented in the manuscript. Dr. Shung Lee has received proctoring fees from Covidien/Medtronic. There is no financial contribution to the author or the authors institution from Covidien/Medtronic which may benefit or lose financially from the results, conclusions, or discussion presented in the manuscript. All other authors have no conflicts of interest to disclose with respect to this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Institutional approval for the study was obtained by the University of British Columbia Clinical Research Ethics Board (H20-00946). Individual patient informed consent was not obtained to be involved in this study, waiver of individual consent was approved by ethics.
Guarantor
I Dr. Alexa Mordhorst will be the primary corresponding author and guarantor for this project and its originality.
Contributorship
Dr. Alexa Mordhorst: Collection, analysis, and interpretation of data, writing of the manuscript, critical revision of the manuscript, and agreement to be correspond author. Dr. Gary Yang: Analysis and interpretation of the data, statistical analysis of the data, critical revision of the manuscript, agreement of corresponding author choice. Dr. Jerry Chen: Analysis and interpretation of the data, critical revision of the manuscript, critical revision of statistical analysis, approval of the manuscript, agreement of corresponding author choice. Dr. Shung Lee: Conception and design of the study, analysis and interpretation of the data, critical revision of the manuscript, agreement of corresponding author choice. Dr. Joel Gagnon: Conception and design of the study, analysis and interpretation of the data, critical revision of the manuscript, agreement of corresponding author choice.
