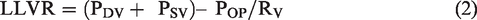Abstract

Introduction
Chronic venous insufficiency (CVI) may be present in up to 50% of the population1,2 and is associated with a significant socioeconomic impact 3 and impairment in quality of life. 4 CVI encompasses a wide spectrum of anatomical and functional abnormalities that ultimately result in elevated ambulatory venous pressures at the ankle.
The physiology and pathophysiology of venous return from the lower limb are conceptually very different from that of the arterial circulation and can be challenging to understand. In arterial disease, a stenosis leads to downstream compensatory vasodilatation until a threshold is reached at which point no further dilatation is possible, and there is a sharp drop in downstream blood flow (critical stenosis). However, in the venous system, return of blood from the lower limb may be influenced by a number of variables such as outflow pressure, volume of inflow and venous resistance. 5
Herein, we aim to highlight parallels between the venous return model of the systemic circulation and that of the lower limb. Using the example of a non-thrombotic iliac vein lesion (NIVL), we describe how the model for systemic venous return originally described by Guyton et al. 6 may be repurposed to facilitate our understanding of the clinical manifestations and treatment options for CVI.
Systemic venous return
The circulatory model experiments of Weber, Starling and Bayliss, Starr and Rawson, and later Guyton have all culminated to our current understanding of venous return (VR) and its importance in regulating cardiac output in both physiological and pathophysiological states. 7 The Guytonian model described a mean circulatory filling pressure, which is the pressure in the vascular system under conditions of circulatory arrest. 6 However, this model includes the peripheral as well as the central venous (cardio-pulmonary) circulation in a composite circuit. The peripheral venous system holds approximately 70% of the blood volume which is distributed between an unstressed volume (volume of blood in veins not under pressure; transmural pressure = 0; e.g. superficial veins in the leg) and a stressed volume (volume of blood in veins under pressure; transmural pressure >0; e.g. deep veins within a closed fascial compartment in the leg). This stressed systemic blood volume creates the pressure gradient from the periphery to the right heart (against the right atrial pressure) that ensures venous return and constitutes the mean systemic venous pressure, the peripheral equivalent of the mean circulatory filling pressure described by Guyton. 8
Briefly explained, in a steady state, cardiac output is equal to VR, which is described by the following Guytonian equation
VR, venous return; PMS, mean systemic venous pressure; PRA, right atrial pressure; RV, resistance to venous return.
This equation describes how VR depends on the gradient between the upstream (pressure in the peripheral vasculature driving blood flow to the heart; PMS) and downstream (right atrial pressure; PRA) pressures as well as resistance to flow as described by Ohm’s law. Raised intra-abdominal pressure, for example during laparoscopic procedures, compresses intra-abdominal veins including the inferior vena cava, increasing RV with a resulting decrease in VR. 9
Repurposing of the Guyton equation for systemic venous return
Just as this formula enables anaesthetists and intensive care physicians to understand and manage the haemodynamic changes and certain ventilatory parameters in altered clinical states such as shock or raised intra-abdominal pressure, we have found that it a useful tool for conceptualising CVI and its management.
When considering venous return from the lower limb, the upstream pressure is equal to the sum of the transmural pressures in the deep venous system and superficial venous system. The Guytonian equation (1) above can therefore be modified for the lower limb as follows
LLVR is the lower limb venous return; PDV is the transmural deep venous pressure; PSV is the transmural superficial venous pressure; POP is the outflow pressure; RV is the resistance to venous return.
The deep veins carry approximately 90% of venous blood in the lower limb, and this stressed volume is, for most part, the sole contributor to the mean systemic filling pressure of the limb. Thus, PMS for the limb is the same as the PDV. The superficial and extra-fascial veins constitute the unstressed volume and have a transmural pressure of zero, i.e. PSV = 0.
The downstream pressure against which the limb veins have to drain is equivalent to the outflow pressure (POP) of the lower limb. This would be the right atrial pressure (PRA) if the intra-abdominal pressure was the same as PRA. However, the intra-abdominal pressure is positive, varies with respiration and is higher in the obese, making the POP higher than the PRA.
Resistance to venous return (RV) may be influenced by a number of factors including venous stenosis or occlusion, thrombosis and venous wall fibrosis in the post-thrombotic vein (thrombo-fibrotic changes decrease vessel wall compliance, with compliance being inversely proportional to resistance).
The final equation for venous return thus becomes
LLVR is the lower limb venous return; PDV is the transmural deep venous pressure; POP is the outflow pressure; RV is the resistance to venous return.
Haemodynamic response to non-thrombotic iliac vein lesion
This model, as represented by equation (3), can be used to describe the haemodynamic sequelae and resultant clinical changes that may be expected from an NIVL. NIVLs are prevalent in the general population and may increase the risk of future development of CVI. 10 The presence of a NIVL can result in high resistance to venous return (RV), the degree to which being dependent on the severity of the compressive lesion. Subsequently, in order to preserve lower limb venous return (LLVR), the transmural pressure in the deep veins (PDV) rises. If the rise in PDV (created by the stressed volume within the deep veins) is unable to compensate for the high RV, the excess volume is diverted from the deep veins to the superficial veins (the unstressed volume compartment), resulting in a reduction in LLVR. High lower limb venous pressures (PDV), along with dilatation of the superficial veins (unstressed compartment) to accommodate the residual blood volume, result in the loss of the pressure segmentation mechanism in the venous valves 11 and venous stasis in the limb with the clinical manifestations of CVI, including varicose veins.
Superficial venous ablation therapy reduces the unstressed volume reservoir, and the result is an increase in the stressed (deep venous) volume within a constrained fascial compartment. The increased stressed volume causes the PDV to rise and, to some extent, overcome the RV, thus increasing venous return. Compression therapies directly increase the PDV as well as act to reduce the unstressed volume reservoir to achieve the same effect.
Iliac vein stenting often provides symptom relief, even without treatment of superficial vein reflux, 12 by instantly reducing outflow pressure (POP) and resistance to venous return (RV). The clinical evidence underpinning ongoing venous stenting is accumulating; however, further high-quality randomised controlled trial data are awaited, including by experienced teams using dedicated venous stents and intravascular ultrasound guidance.
Venous resistance to blood flow is influenced both by the compliance of the vein wall as well as the external pressure on the vein. The occurrence and severity of PTS is influenced by the extent and anatomical location of the initial deep vein thrombosis, recurrent ipsilateral DVT, residual thrombus after DVT, obesity and older age. 13 After venous thrombosis, there is thickening of the vein wall with a decrease in distensibility (decreased compliance, increased resistance) as has been demonstrated in experimental studies. 14
Raised intra-abdominal pressure in obese patients is an additional contributor to a high POP and has been implicated in CVI. 15 The relationship between intra-abdominal pressure and impairment of venous return has also been previously investigated in an experimental model of a Starling resistor 5 demonstrating that, at higher intra-abdominal pressures (Starling pressure), increasing venous stenosis has little further effect on upstream pressures as the intra-abdominal pressure sets the outflow pressure needed for venous return. In the significantly obese with CVI, POP is raised regardless of whether or not a venous pathology, such as NIVL, is present. In the case of obese patients, stenting of generous deep venous segments (iliac, caval and femoral as required) will reduce the POP and has been reported to provide symptom relief. 12
Summary
The discovery of the high prevalence of the NIVL as a source of functional venous outflow obstruction in the lower limb allows the Guytonian model of systemic venous return to be applied specifically to lower limb venous haemodynamics in CVI. The repurposed equation LLVR = PDV – POP/RV helps explain many phenomena seen with NIVL, PTS and other venous pathologies, and the rationale for deep venous stenting, superficial venous ablation and compression therapies. Other pathophysiological processes in CVI, like the genetic determinants of valve development and thrombophilia, inflammatory responses in the vein wall, body habitus, hydration, prolonged standing and so on doubtless play a role in the development of the final clinical picture. However, these too impact venous return via their effect on the various components of this repurposed equation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.