Abstract
Objective
To obtain consensus on management criteria for symptomatic patients with chronic venous disease (CVD; C2–C6) and superficial venous reflux.
Method
We used a Delphi method by means of 36 statements sent by email to experts in the field of phlebology across the world over the course of three rounds. The statements addressed criteria for different venous treatments in patients with different characteristics (e.g. extensive comorbidities, morbid obesity and peripheral arterial disease). If at least 70% of the ratings for a specific statement were between 6 and 9 (agreement) or between 1 and 3 (disagreement), experts’ consensus was reached.
Results
Twenty-five experts were invited to participate, of whom 24 accepted and completed all three rounds. Consensus was reached in 25/32 statements (78%). However, several statements addressing UGFS, single phlebectomies, patients with extensive comorbidities and morbid obesity remained equivocal.
Conclusion
Considerable consensus was reached within a group of experts but also some gaps in available research were highlighted.
Introduction
Over the past decades, numerous effective treatments have been developed for patients with chronic venous disease (CVD). The increased availability of different treatment options has made the management of CVD more challenging. It has resulted in a large worldwide variation in management preferences for treating patients with CVD.1,2 Management strategies are not only based on evidence from the literature but also on the physician’s own experience, availability (and costs) of the equipment and on national healthcare reimbursement systems. However, in clinical practice, management decisions should ideally be influenced mainly by a combination of patient characteristics, clinical findings and results of duplex ultrasound (DUS).1,3,4
Although there are several well-established national and international guidelines,5–8 their recommendations only focus on the treatment of a diseased population rather than an individual patient. 9 Appropriate tools for management strategies incorporating the specific characteristics of an individual patient, as well as the clinical and DUS findings, are lacking in current clinical guidelines.5–8 To bridge the gap between current clinical guidelines and individualized care, expert opinion methods such as a Delphi consensus 10 are desired.
The aim of the present study was to achieve an international Delphi-based consensus on management criteria for patients presenting with venous symptoms, clinical signs of CVD, and superficial venous reflux confirmed by DUS.
Methods
Expert panel
Experts were selected from a group of physicians who already participated in our recently published worldwide survey on management strategies in patients with CVD (C2–C6).
1
Only those with at least one scientific publication in the field of phlebology in a peer-reviewed journal and at least 10 years of experience in treating patients with CVD were eligible for the present study. They had to be performing ultrasound-guided foam sclerotherapy (UGFS), endovenous thermal ablation (EVTA) and phlebectomies themselves. Two investigators (SKvdV and MGRDM) selected the expert panel. For this selection, they aimed at having a reasonable distribution between different specialties (vascular or general surgeons, dermatologists, angiologists, phlebologists) and continent or country of clinical practice. Based on the selection criteria, 25 eligible experts were invited by email. They were asked to judge on several statements in view of obtaining consensus over the course of three rounds, according to the Delphi method between March 2015 and November 2015 (Figure 1). In case of non-responding, the investigators sent two email reminders in each round.
Selection of participants for the Delphi consensus.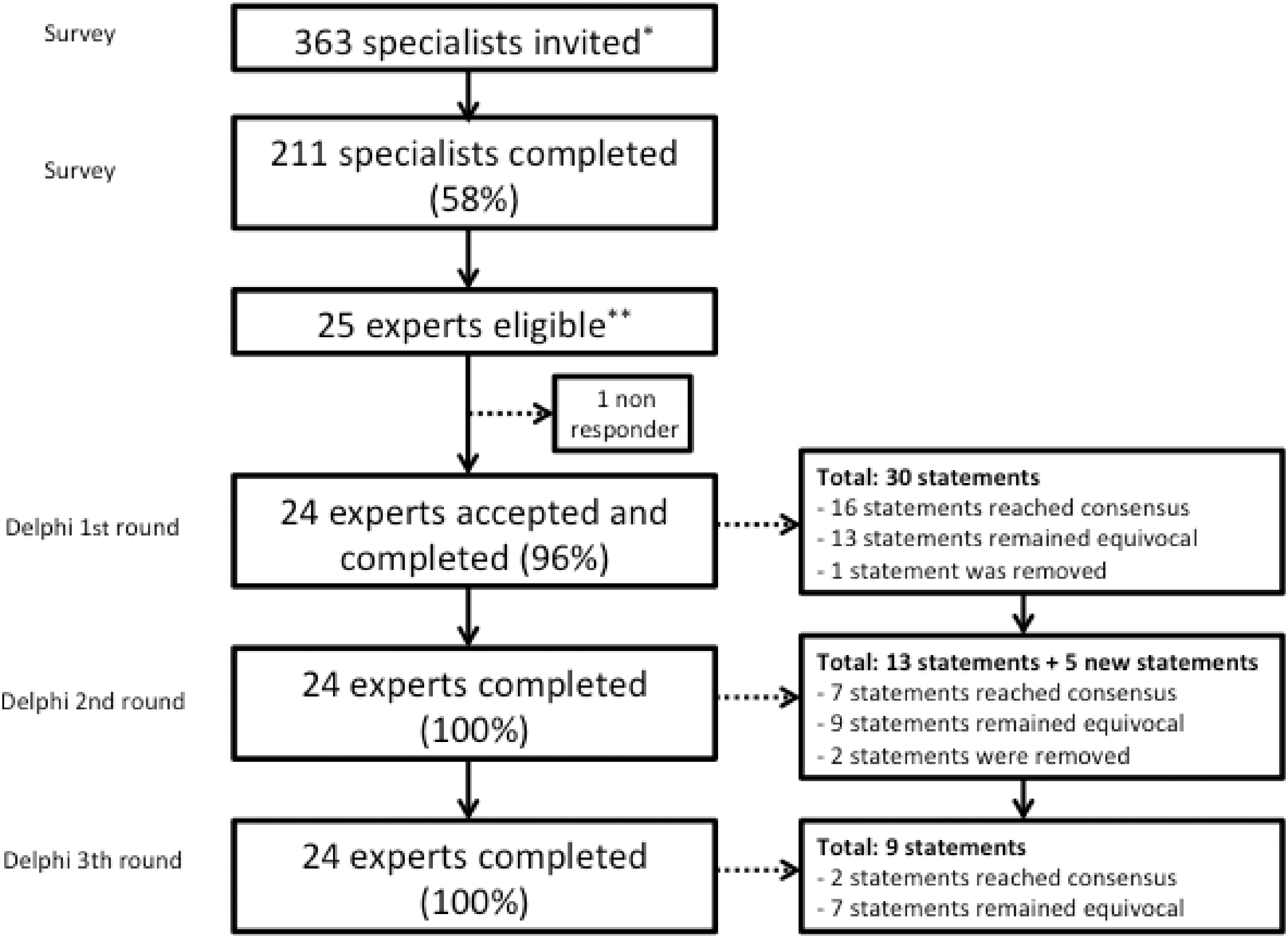
Informed consent was not performed because no patients were involved in this study.
Delphi consensus procedure
Definition of terminology used for the statements of the Delphi consensus.

SFJ: saphenofemoral junction; SPJ: saphenopopliteal junction; BMI: body mass index.
Participants were asked to anonymously rate each statement on a 9-point scale, by ticking boxes with marks between 1 and 9, where a score of 1 denoted complete disagreement and a score of 9 indicating full agreement (Figure 2). If at least 70% of the ratings for a specific statement were between 7 and 9, it was concluded experts consented on agreeing with the statement. Vice versa, if ≥70% were between 1 and 3, it was concluded they consented on disagreeing with the statement. This cut-off level was based on previous literature.13,14 Any other distribution of ratings was valued ‘equivocal’. Participants also had the opportunity to add a comment in the ‘remark(s)’ box accompanying each statement. All this information was used for preparing the subsequent round of the Delphi consensus.
Example of a statement showing consensus on agreement (84% of marks between 7 and 9).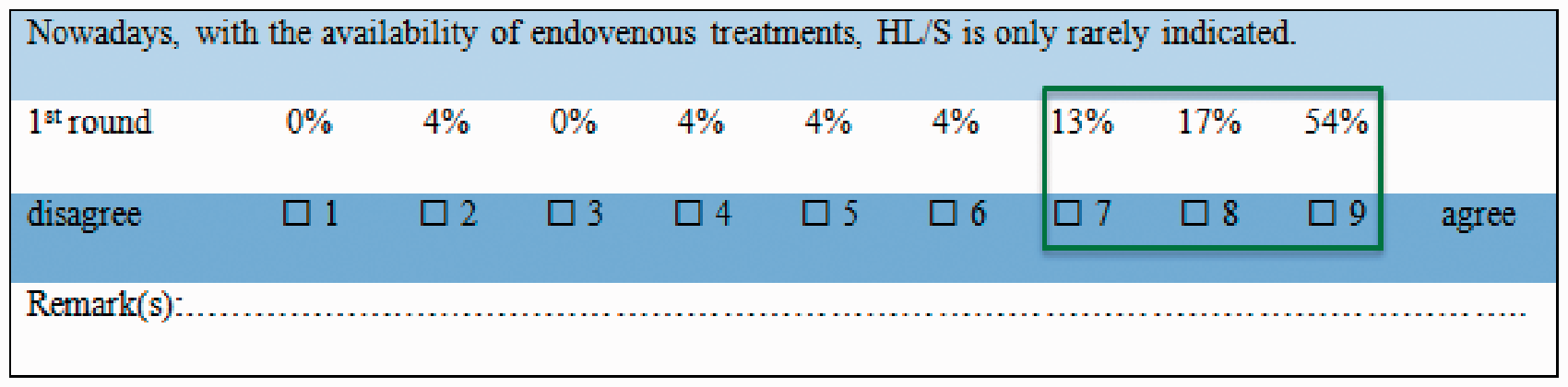
In the second round, experts received a full report of the first round, including a compilation of ratings for all statements of the first round, and all the remarks of their colleagues, provided anonymously. Then, they were asked to rate again those statements that had remained equivocal in the first round. Statements were reformulated and new statements were added based on the remarks of the experts in the previous round. For instance, in the first round, we had included older age as a patient characteristic, which might influence management. However, participating experts suggested age was not the main factor, but rather extensive comorbidities, and therefore the statement was adapted as such. Eventually, the second round contained 18 statements. In the second round, we also attached a summary of the present literature regarding each statement.
The same procedure was followed for the third (and last) round, which contained only nine statements to be rated. Again, participating experts could consider the results of the second round and the remarks of their colleagues.
In case of one or more missing items, the expert was contacted by email to retrieve the missing answer.
Results
Experts
In total, 24 out of 25 contacted experts accepted to participate: 15 vascular surgeons, two general surgeons, four dermatologists, two angiologists and one phlebologist from Europe (17/25), Australia (1/25), North-America (4/25) and Central- or South-America (3/25). Experts were working in private practice (13/25), general hospital (4/25) or university hospital (7/25). All these experts completed the first, second and third Delphi round (25/25). The majority of experts (16/25) had over 20 years of experience in treating patients with CVD.
Delphi consensus
Results of Delphi consensus.
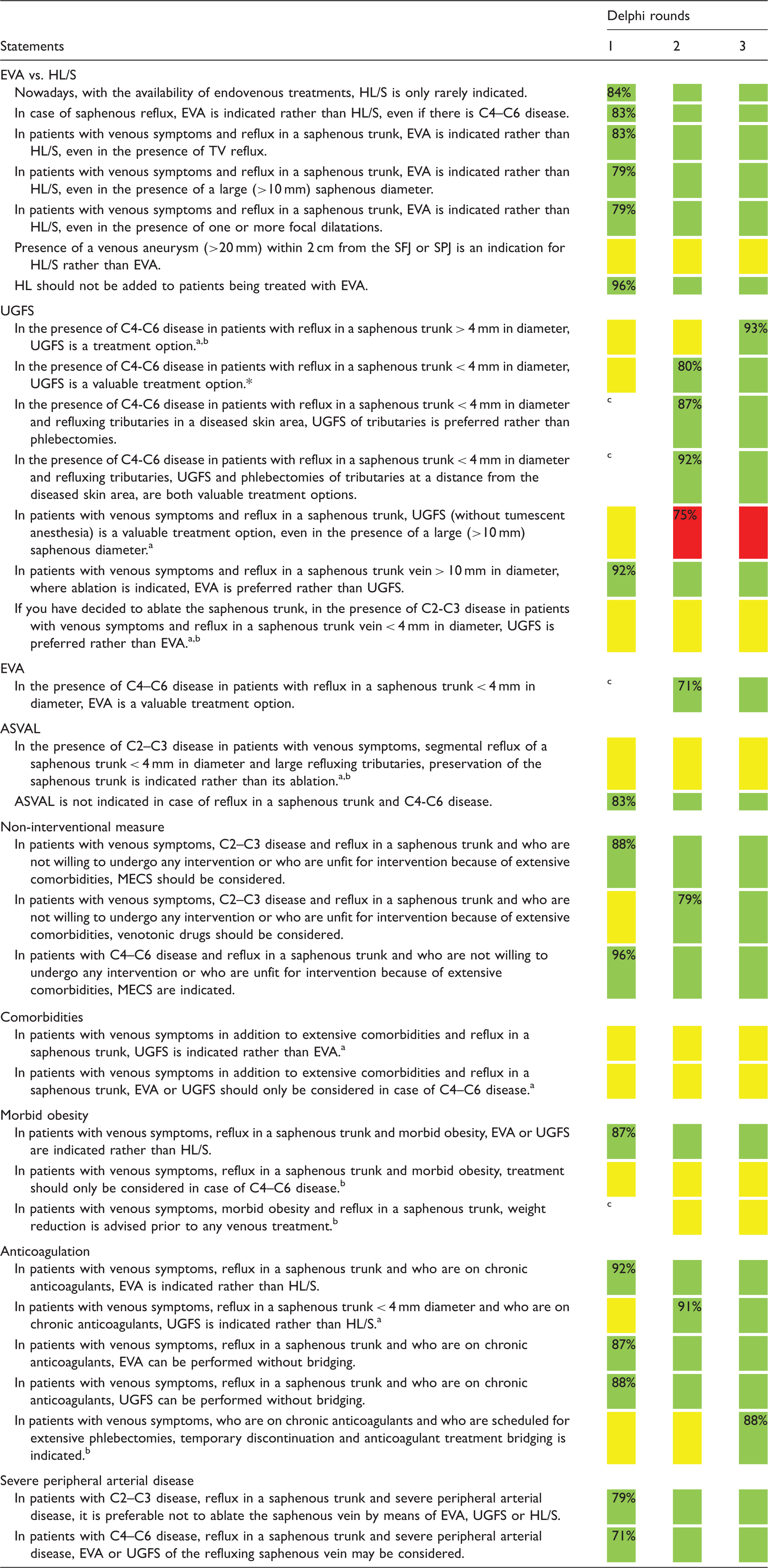
EVA: endovenous ablation (including thermal and non-thermal non-tumescent techniques, excluding UGFS); HL/S: high ligation with stripping; SFJ: saphenofemoral junction; SPJ: saphenopopliteal junction; UGFS: ultrasound-guided foam sclerotherapy; ASVAL: ‘ambulatory selective varicose ablation under local anaesthesia’ (= single phlebectomies without treating the saphenous trunk); MECS: medical elastic compression stockings.
 Consensus on agreeing with the statement.
Consensus on agreeing with the statement.
 Consensus on disagreeing with the statement.
Consensus on disagreeing with the statement.
 Equivocal (no complete agreement nor disagreement).
Equivocal (no complete agreement nor disagreement).
Statement was reformulated after first round.
Statement was reformulated after second round.
Statement was added after remarks of experts in the first round.
The experts’ opinion on several statements remained equivocal after three rounds. This was the case for statements addressing different indications for UGFS, ASVAL, extensive comorbidities and morbid obesity. The experts stated that it depended on the type and extent of the patient’s comorbidities and therefore no definitive conclusion on preferred management could be made. Regarding morbid obesity, the experts would definitely prefer EVA over HL/S, but there was no agreement on limiting treatment to those patients with skin changes (C4–C6). Moreover, based on their experience, the participants were convinced that advising weight reduction unfortunately was unsuccessful in the majority of obese patients.
Throughout the different rounds of the Delphi consensus, three statements were removed, two because of redundancy after reformulating other statements and one because of lack of relevance following the experts’ remarks (Supplementary Table 1).
Discussion
The present study showed that considerable consensus could be reached within a group of selected experts in the field of treatment of superficial venous disease, by means of a Delphi consensus procedure. This may help physicians to make supported and individualized choices for their patients with CVD (C2–C6).
Not surprisingly, experts’ recommendations regarding EVA were in line with current guidelines on the care of patients with varicose veins and associated CVD.5–8 EVA was considered the preferred treatment for most symptomatic patients with CVD (C2–C6) and a refluxing ST > 4 mm in diameter. The expert panel did not recommend an alternative treatment for specific DUS findings such as terminal valve reflux, a large (>10 mm) ST diameter or the presence of one or more focal dilatations of the ST (Table 2). EVA has now globally become the preferred treatment for ST reflux as is reflected in our results. Although EVA (thermal or non-thermal) has largely replaced HL/S in many countries, it should be acknowledged that, whenever equipment for EVA is not available, HL/S (ideally using tumescent anesthesia, in agreement with recent guidelines 6 ) remains a valuable treatment option. While patient-reported outcomes favor EVTA, long-term efficacy of HL/S and EVTA are very similar.15,16
UGFS is usually considered as the second best option of the minimally invasive treatments for abolishing ST reflux.5–8 In the present consensus approach, the experts considered UGFS to be just ‘a’ treatment option if ST diameter is ≥4 mm, and only agreed on categorizing it ‘a valuable treatment option’ for ST’s with small diameters < 4 mm. This discrepancy illustrates that ST diameter may be used as a relevant criterion to distinguish between different treatment options, 1 which is in line with the European guidelines forsclerotherapy. 17
ASVAL is not yet a worldwide-accepted strategy, and this was reflected in the Delphi consensus. Contrarily to what the investigators had been suggesting, the experts could not agree on particular criteria for implementing ASVAL so far. This is somehow to be expected as only 42% of the experts had ever used ASVAL in their clinical practice (data not shown) and the evidence on the subject is still limited.11,18,19 Future research should point out whether ASVAL can be applied in all patients with varicose veins or in a selected varicose vein population only.
At the end of the first round of the Delphi consensus, older age appeared to be less relevant than we initially had hypothesized and it was therefore converted into extensive comorbidities as suggested by the participants from the second round onwards. Indeed, clinical studies demonstrated that the feasibility and safety of EVTA and tolerability of UGFS were similar in patients older or younger than 75 years.20,21 In contrast, studies regarding venous treatments in patients with (extensive) comorbidities are completely lacking. Although the investigators thought that minimizing the extent of procedures in patients with comorbidities and/or morbid obesity would be good clinical practice, experts did not reach consensus on this issue. This lack of consensus could partly be explained by the fact that many different combinations of comorbidities can be thought of in these cases and the extent of comorbidities was not defined in the statements. The experts seemed to be more unanimous regarding strategies in patients under chronic anticoagulant treatment and those with severe peripheral arterial disease.
Although we found considerable consensus about treatment strategies in different clinical classes and DUS findings, statements addressing patients’ characteristics remained largely equivocal in the Delphi consensus. This clearly highlights the lack of knowledge in the field of personalized medicine and implies that more research regarding patient characteristics, and how these are influencing treatment outcomes, is needed. In this way, future guidelines may include more tools for proper stratification of patients with CVD. Obviously, when defining individualized treatment strategies in patients with CVD, physicians should also consider patient’s preference, as well as the impact of the disease on Health-Related Quality of Life (HRQoL), the estimated risk of deterioration of CVD and local healthcare resources.
Limitations
This study has several limitations. The statements may have been incomplete, but this was minimized because they were based on a prior worldwide survey among 211 specialists in CVD. 5 Also, this study focused on the treatment of patients with venous symptoms, clinical signs of CVD, and superficial venous reflux in the ST and tributaries, and therefore our results are not generalizable to all patients with CVD (e.g. patients with a pathological perforating vein nearby a venous ulcer or patients with groin recurrence). Selection bias among the eligible experts might have occurred because the majority of the participants were working in Western Europe. However, the response rate was almost perfect. In the absence of clear guidelines for a Delphi consensus, we chose the cut-off value of 70% as acceptable, because of the relatively mild impact of CVD on HRQoL, contrarily to decision-making in more life-threatening diseases in which a higher cut-off may be desired. In addition, there is no standard method for calculating sample sizes for a Delphi study, but there is evidence that expert panels of 20 can reach a valid consensus. 22
Conclusion
In conclusion, in the present investigation about management strategies in patients with symptoms and signs of CVD and superficial venous reflux, the experts agreed on most of the strategies and their opinions largely reflected international guidelines. Nevertheless, it appeared to be more difficult to reach consensus on the influence of certain patient-related characteristics. More research is needed in the field of personalized medicine, in order to further optimize phlebologic care for all our patients presenting with CVD.
Footnotes
Acknowledgements
The authors thank all experts who took part in the Delphi consultation: Lena Blomgren, Kornelia Böhler, Alberto Caggiati, Dumitru Casian, Alessandro Frullini, Kathleen Gibson, Jean-Luc Gillet, Manj Gohel, Evi Kalodiki, Mark Malouf, Uldis Maurins, Mark Meissner, Nick Morrison, Alfred Obermayer, Alvaro Orrego, Pierre Ouvry, Eberhard Rabe, Lars Rasmussen, Stefanie Reich-Schupke, Guillermo Rossi, Roberto Simkin, Ted van den Broek, Marc Vuylsteke, Steven Zimmet.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Contributorship
SKvdV designed study, Delphi consultation, analysed data, drafted paper. RRvdB designed study, drafted paper. OP designed study, critically reviewed paper. TN designed study, critically reviewed paper. MGRdM designed study, Delphi consultation, analysed data, drafted paper.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
