Abstract
Background
Cardiopulmonary bypass is associated with systemic inflammation during pediatric cardiac surgery and features elevated systemic concentrations of complement, cytokines and chemokines. The objective of this study is to quantify the immunologic efficacy of ultrafiltration (UF) used continuously throughout CPB.
Methods
Pediatric patients were enrolled in a single-arm prospective clinical study (NCT05154864) and received standard cardiac operations, CPB and subzero-balance ultrafiltration (SBUF) with an effluent extraction rate of 30 mL/kg/hr and conventional ultrafiltration (CUF). Paired arterial and UF effluent samples were taken at regular intervals during CPB and the concentration of 33 inflammatory mediators were measured. An exploratory counterfactual analysis assessed whether UF removed a significant amount of mediator mass using area under the curve (AUC), generalized linear mixed-effects models (GLMEM), and median fold change [95% CI] comparisons between observed data (UF) and reconstructed counterfactual data (no UF).
Results
Forty consecutive eligible consenting patients participated; the majority were male (58%), age of 7.3 (1.7 – 39.0) months and weight of 6.7 (4.6 – 14.9) kg. All patients received UF therapy with effluent extraction of 155 (100 – 185) ml/kg during CPB. C3a, C5a, TNF, IL-1α, IL-1β, IL-1Ra, IL-2, IL-6, IL-10, CCL2, CCL3, CCL4, CXCL1, CXCL2, CXCL8, ET1, GM-CSF and TRAIL were detected in the UF effluent. In the counterfactual analysis, only C3a was significantly extracted by UF using AUC (p = 3.5 × 10−12), GLMEM (p = 2.3 × 10−87), and median fold change [95% CI] as C3a showed a decrease of −0.2 [−0.3 to 0] relative to CPB initiation, in the observed data, compared to the counterfactual with a median fold increase of 4.3 [3.1 – 5.3].
Conclusions
Ultrafiltration during pediatric CPB extracts cytokines and chemokines continuously throughout CPB with relatively low effectiveness. In an exploratory counterfactual analysis, C3a was the only inflammatory mediator meaningfully removed from the circulation by UF.
Keywords
Introduction
Cardiac surgery with cardiopulmonary bypass (CPB) is associated with a systemic inflammatory response hallmarked by circulating cytokines and activated complement mediators.1–3 C3a, C5a, TNF, IL-1, IL-6, CXCL8 and the anti-inflammatory mediators IL-1Ra and IL-10 have long been known to be secreted into the circulation, and more recent research has identified C3b and ET1 along with several cytokines such as TRAIL, GM-CSF and chemokines CCL2, CCL3, CCL4 also to be elevated during and following CPB.1–5 Unfortunately, there has been poor consensus on which mediator or groups of mediators are associated with morbid post-operative outcomes following cardiac surgery, although the complement anaphylatoxins C3a and C5a appear to be clinically important in observational data.3,5
Exposure to the non-endothelialized CPB circuit can be associated with post-operative morbidities including volume overload, vasoplegia, cardiopulmonary dysfunction, and other end-organ injuries.1–3 The reaction is primarily driven by the alternative complement pathway as C3 is cleaved to produce the split products C3a and C3b which go on to have a range of effector and amplification functions.6,7 Ultrafiltration during pediatric CPB has been used for decades to mitigate the adverse effects and promote a stable peri-operative period.3,8–10 There are several forms of ultrafiltration that can be grouped as non-continuous forms – modified ultrafiltration used immediately after weaning from CPB – and conventional ultrafiltration during rewarming on CPB or continuous forms used throughout the entire CPB time.3,11 Non-continuous forms can reliability address volume overload and reduce bleeding complications as well as transfusions.3,12–14 Continuous forms, theoretically, have a more potent immunomodulatory function as they sieve small mediators throughout the entire CPB exposure.3,9,15 However, the clinical results of investigations studying various forms of ultrafiltration are mixed, perhaps due to small sample sizes of prospective studies, heterogeneity in ultrafiltration protocols and institution-specific factors. 3
Subzero-balance ultrafiltration (SBUF) is a precise and reproducible method of ultrafiltration (UF) used during pediatric CPB. 16 SBUF was designed to capitalize on a relatively high effluent extraction rate, measured in ml/kg/hr, for immune mediator as well as a pre-determined small negative balance (to prevent tissue edema and volume overload. 16 Further, it is used in unison with CUF, to remove cardioplegia volume, intra-cardiac irrigation fluid and other sources of excess volume. There are no analyses that comprehensively describe the immunomodulatory impact of SBUF or continuous forms of ultrafiltration throughout children’s heart surgery. It is therefore difficult to ascertain any immunologic effect. This lack of functional understanding of the therapy is prohibitive to identifying an optimal approach to enhance recovery for these vulnerable patients. This study aims to comprehensively describe the systemic inflammatory response during pediatric cardiac surgery with CPB and continuous UF, consisting of SBUF and CUF, as then using a counterfactual framework – where the observed data with UF is compared to a reconstructed hypothetical dataset without UF – to explore the immunologic impact of this ultrafiltration technique.
Methods
Study design
This is a pre-specified analysis of a single-center single-arm prospective clinical trial (NCT05154864 on ClinicalTrials.gov) investigating the relationship between circulating inflammatory mediators and post-operative clinical outcomes in pediatric patients (<30 kg) undergoing cardiac surgery with CPB and continuous SBUF with effluent extraction rate of 30 mL/kg/hr and standard CUF.5,16 The outcomes of interest in this analysis are inflammatory mediators (complement, cytokines, chemokines, and adhesion molecules) concentrations in a time series up throughout CPB time. Following the descriptive analysis, a counterfactual framework was used to estimate the inflammatory mediator dynamics if SBUF was not used and test for statistically significant immunomodulatory impact. Written informed consent was obtained from substitute decision-makers for all participants under a protocol approved by the IWK Health Centre Research Ethics Board (#1024869) on November 21, 2019 which was maintained throughout the duration of the study.
Study participants
Patients were prospectively enrolled and completed the study protocol between August 2020 and June 2021 at the IWK Health Centre in Halifax, Nova Scotia, Canada. Exclusion criteria included: absence of written consent, known severe hematologic abnormality, genetic syndrome with severe multi-organ involvement, immunodeficiency syndrome and severe liver disease.
Data collection
Patient baseline demographic and clinical information were recorded from medical records. Arterial blood samples (post-oxygenator arterial outlet sampling line) and paired UF effluent samples (effluent line immediate to device outflow) were drawn when full flow CPB was achieved (CPB initiation), every 30 minutes until 120 minutes and then every 60 minutes thereafter until CPB weaning (CPB cessation). The blood samples were collected in EDTA tubes, were centrifuged for 10 minutes (500 × gravity), and the resulting plasma was extracted and underwent a second centrifugation for 20 minutes (2500 × gravity) to yield a platelet-free plasma. The platelet-free plasma and clear effluent was aliquoted, flash-frozen in liquid nitrogen and stored at −80°C.
CPB conduct
CPB prime composition was according to institutional standard of care. Heparin, sodium bicarbonate, calcium chloride, magnesium sulphate, furosemide, and mannitol were administered at standard weight-based doses. Sanguineous prime was used for patients <10 kg and BUF was used to normalize metabolic parameters and hemoconcentrate the volume to approximately 500 mL. 17 Crystalloid prime was used for those >10 kg and retrograde autologous priming was used as indicated by the multidisciplinary team. A Liva Nova S5 TM CPB System with phosphorylcholine coating (48-40-00, London, UK), Terumo FX05 (weight <10 kg) or FX15 (weight >10 kg) oxygenators (1CX*FX05RE/1CX*FX15 E, Tokyo, Japan) and Terumo Capiox® Hemoconcentrator HCO5 (1CX*HC05S, Tokyo, Japan) were used.
Subzero-balance ultrafiltration technique has been previously published. 16 Five percent of the calculated cardiac output was directed to the hemoconcentration device via dedicated roller pump once full flow was reached and was maintained until the time of weaning. Effluent extraction was controlled at 30 mL/kg/hr, and physiologic fluid replenishment was at 25 mL/kg/hr, targeting a negative balance of −5 mL/kg/hr. Direct removal of cardioplegia or field irrigation volume by CUF was additional to this continuous SBUF rate.
Immunoanalysis
Luminex immunoanalysis of patient samples was completed with a Bio-Rad Bio-Plex® 200 System (Hercules, United States). Thirty-three pre-specified human inflammatory factors were analyzed in serum samples while select mediators (C3a, C5a,TNF, IL-1α, IL-1β, IL-1Ra, IL-2, IL-6, IL-10, CCL2, CCL3, CCL4, CXCL1, CXCL2, CXCL8, GM-CSF, TRAIL and ET1) were measured in the effluent according to their small molecular mass via ThermoFisher C3a Simplex Kit (EPX010-12282-901, Waltham, United States), Millipore Sigma Human Complement Magnetic Beat Panel 1 (HCMP1MAG-19K-05, Burlington, United States), Millipore Sigma Human Complement Magnetic Beat Panel 2 (HCMP2MAG-19K-06, Burlington, United States), BioTechne R&D Systems Human XL Cytokine Luminex Performance Panel (FCSTM18-21, Minneapolis, United States), BioTechne R&D Systems Human Magnetic Luminex Assay (LXSAHM-05, Minneapolis, United States) and BioTechne R&D Systems Human Magnetic Luminex Assay (LXSAHM-01, Minneapolis, United States). Bio-Rad Bio-Plex® ManagerTM Software 6.2 (Hercules, United States) was used to complete the data acquisition and used Logistic - 5PL regression for all analytes. All assays were conducted according to the manufacturer’s instructions. Mediator concentrations were standardized via the global median ratio method to correct for inter-assay variability; where sample x from mediator assay a was corrected by ([mediator] xa * ([mediator]global median) / [mediator]assay a median.
Statistical analysis
Formula for mediator analysis during CPB.

The estimated patient circulating volume is 90 mL/kg for neonates, 80 mL/kg for infants and 70 mL/kg for children. xg/ml: measured mediator concentration in pg/ml or ng/ml or ug/ml. AUC: area under curve. UF: ultrafiltration effluent including SBUF and CUF.
An exploratory counterfactual framework was used to estimate the mediator concentration dynamics if SBUF was not used.18,19 Observed circulating mediator mass and counterfactual patient circulating mediator mass – where the cumulative mediator mass extracted in the SBUF effluent is assumed to remain with the patient – over the CPB time were included in this counterfactual analysis (see Table 1). The difference in counterfactual mass and observed mass were calculated for each mediator and time point. Under this framework, the differences between observed (UF) and counterfactual (no UF) was assessed by three complementary approaches: area under curve, generalized linear mixed effects models and fold change. The area under curve (AUC) quantifies the longitudinal mediator exposure throughout the CPB time and was measured by trapezoidal integration of the individual time series.20,21 The AUC of observed and counterfactual groups were compared by Wilcoxon rank sum test. Generalized linear mixed effects models (GLMEM) were used for each mediator mass using a CPB time and SBUF interaction term. A statistically significant interaction term indicates that SBUF removed a statistically significant amount of mediator mass throughout the CPB time in this counterfactual framework. Each model featured a Gaussian family distribution with random intercept for the grouping variable by patient ID to account for related samples for each patient during CPB. Log transformations significantly enhanced the model quality assessed by residuals and hat matrix for leverage due to non-linear relationships between variables. Model R2m was calculated to quantify the variance explained by fixed effects and R2c to describe variance explained by fixed and random effects while coefficient p-values were calculated using Satterthwaite’s method.22,23 Models were assessed by reviewing the normalcy of residuals, presence of leverage points by hat calculations and outliers by Cook’s distance. Multicollinearity were assessed by variance inflation factor (VIF) with severe multicollinearity defined as VIF >10. The fold change for each inflammatory mediator concentration, in the observed and counterfactual data, between time CPB initiation and CPB cessation was calculated by ([mediator] CPB cessation – [mediator] CPB initiation ) / [mediator] CPB initiation and expressed as median fold change with [95% CI] estimated by 1000 nonparametric bootstrap samples with adjusted percentile interval. 24 The observed and counterfactual mediator fold changes were compared by Wilcoxon rank sum test. All statistical analyses were performed using R (version 4.4.2, R Foundation for Statistical Computing, Vienna, Austria). Statistical significance was α = 0.05.
Results
Patient demographics and clinical data
A total of 49 consecutive patients were screened during the study period and 40 patients (82%) gave informed consent to participate. Nine patients (18%) were not enrolled due to a genetic syndrome with severe multi-organ abnormalities (n = 3, 6%), weight over 30 kg (n = 3, 6%), logistical process restraints (n = 2, 4%) and refusal to participate (n = 1, 2%). All 40 enrolled completed data collection and are included in this analysis with no exclusions.
Clinical parameters (n = 40) no. (%); median (IQR).
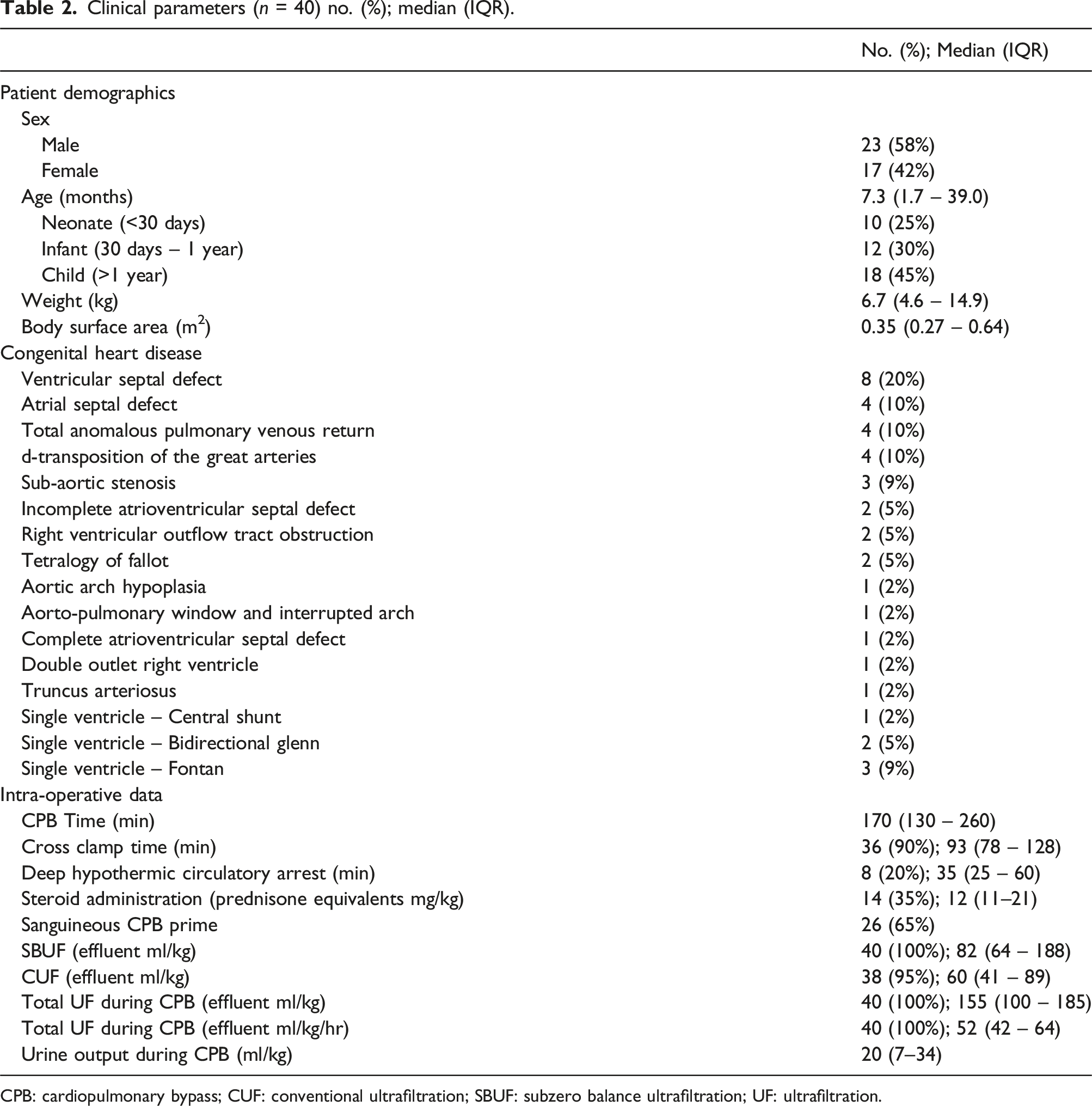
CPB: cardiopulmonary bypass; CUF: conventional ultrafiltration; SBUF: subzero balance ultrafiltration; UF: ultrafiltration.
Inflammatory mediator profiles during CPB and UF
Inflammatory mediator concentration changes through CPB and UF (n = 40); median (IQR).
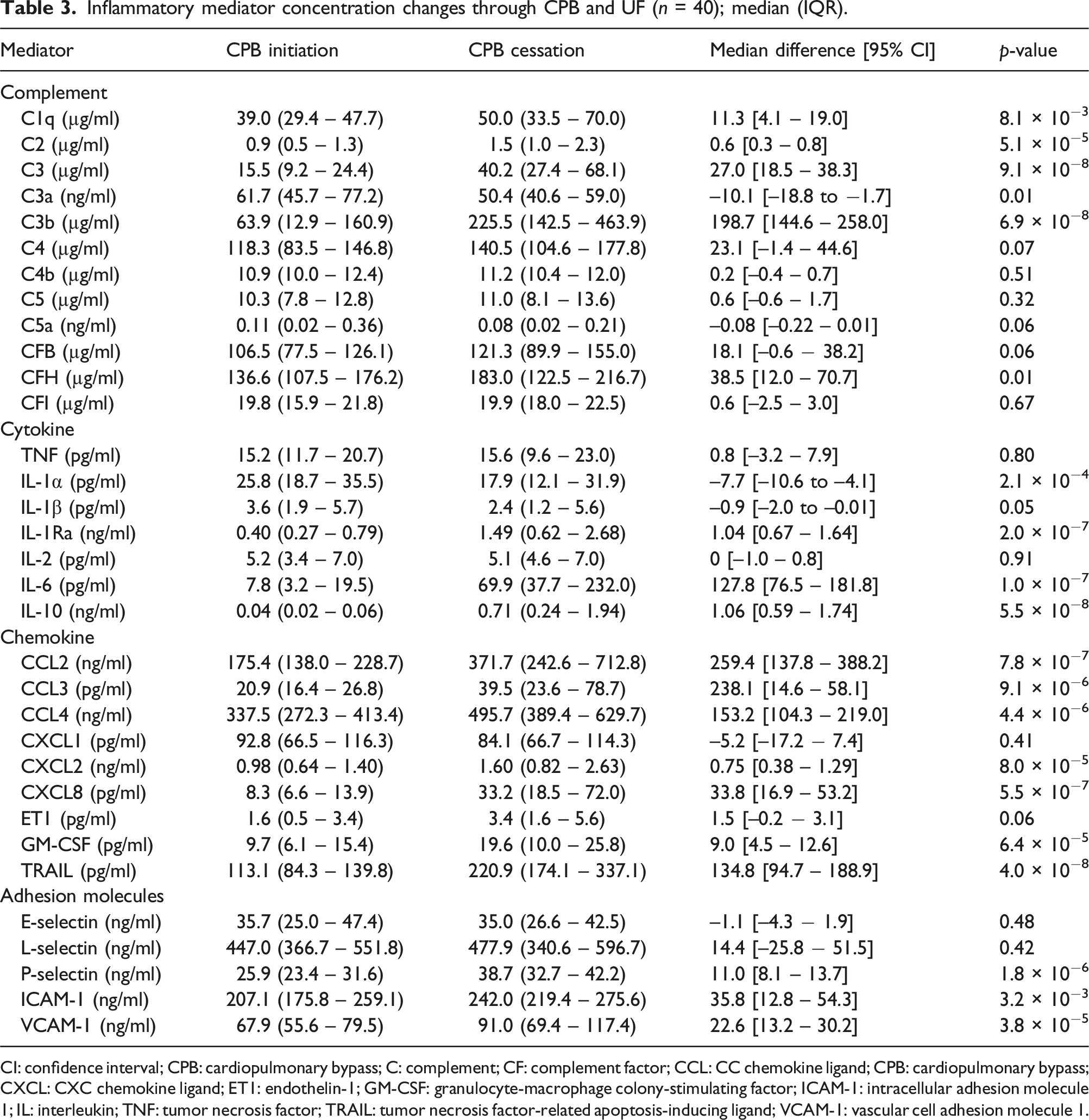
CI: confidence interval; CPB: cardiopulmonary bypass; C: complement; CF: complement factor; CCL: CC chemokine ligand; CPB: cardiopulmonary bypass; CXCL: CXC chemokine ligand; ET1: endothelin-1; GM-CSF: granulocyte-macrophage colony-stimulating factor; ICAM-1: intracellular adhesion molecule 1; IL: interleukin; TNF: tumor necrosis factor; TRAIL: tumor necrosis factor-related apoptosis-inducing ligand; VCAM-1: vascular cell adhesion molecule 1.
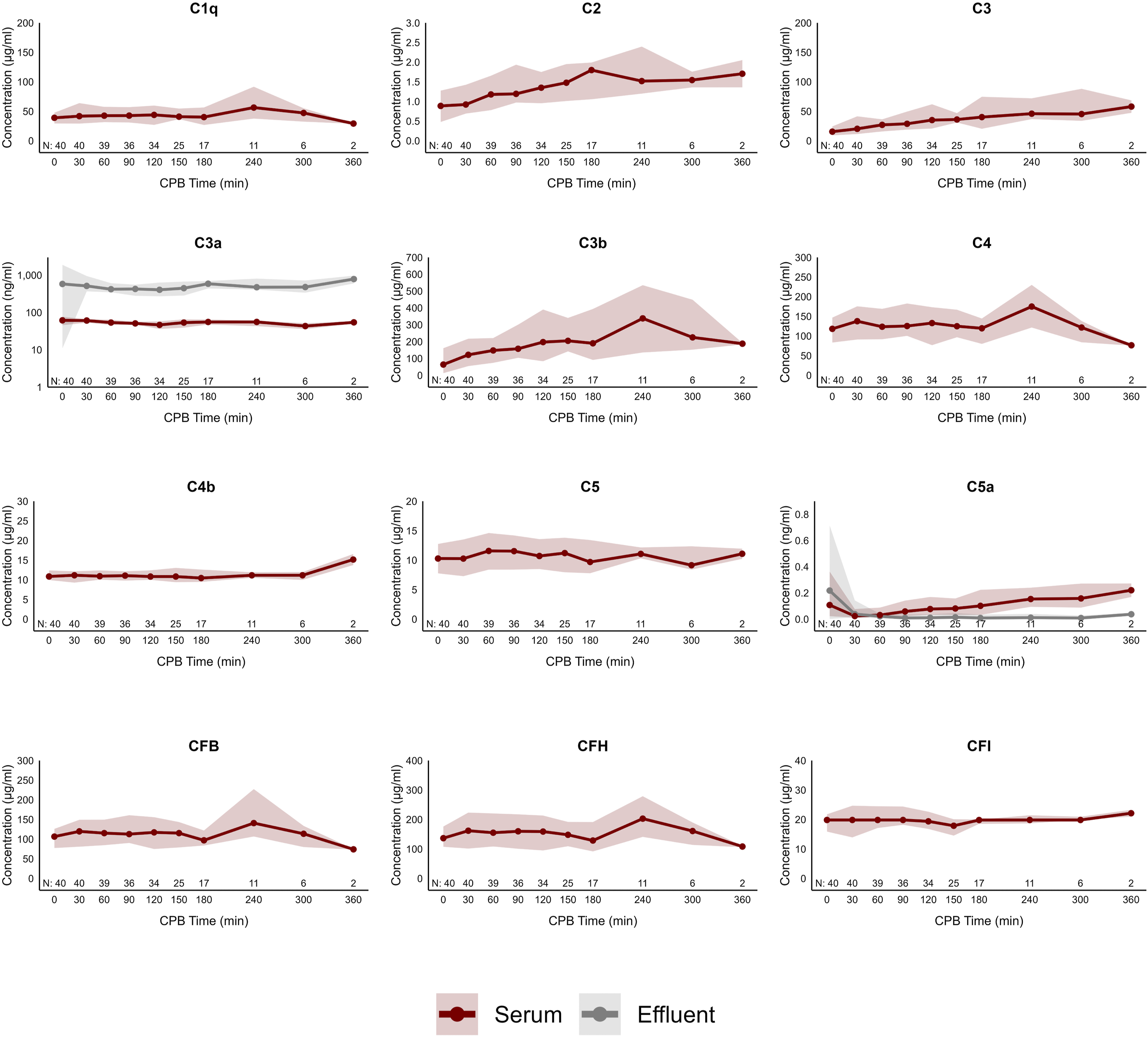
Complement mediator concentrations throughout CPB with UF. Values are displayed as median and IQR. Observations (N) through the time series are recorded. Only C3a and C5a concentrations were measured in the effluent while concentrations were not measured for the other complement factors with large molecular mass. C: complement; CF: complement factor; CPB: cardiopulmonary bypass.
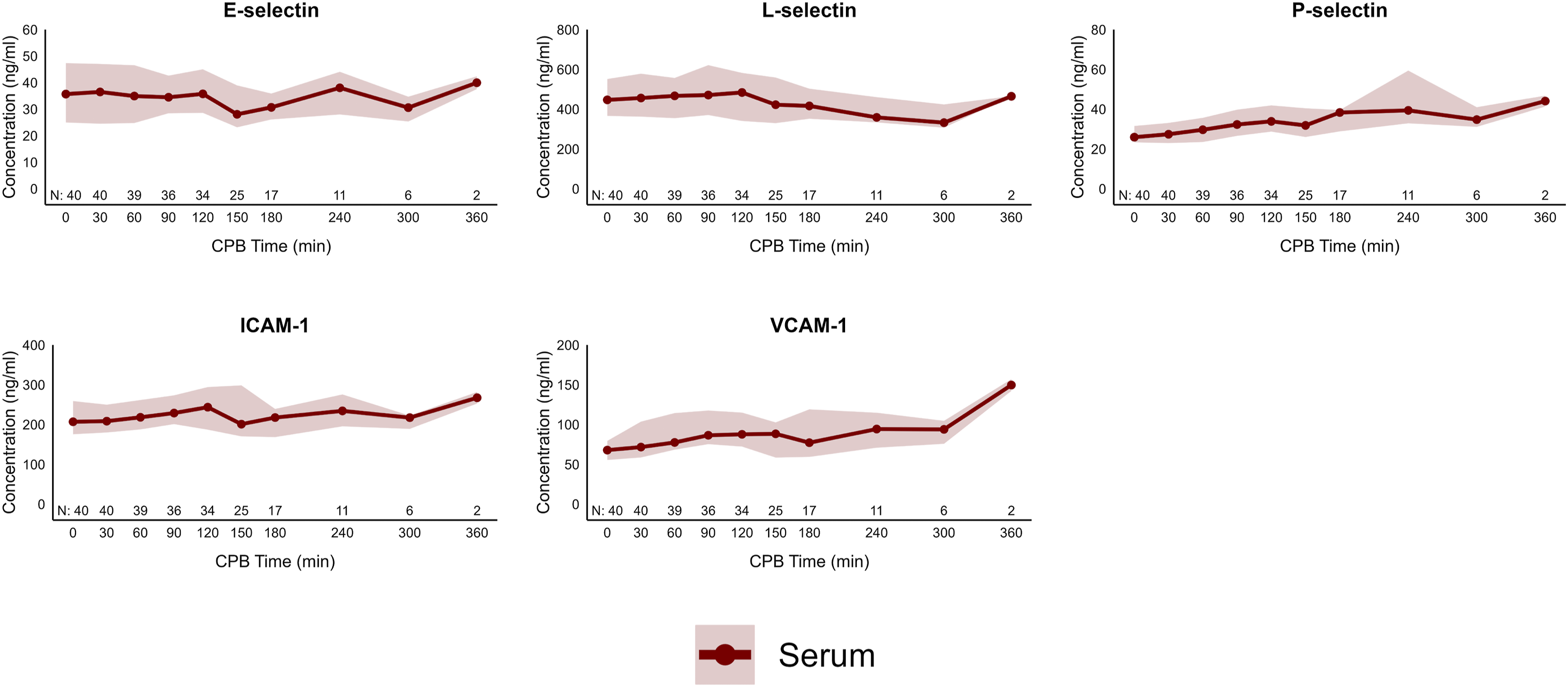
Cytokine and chemokine serum and effluent concentrations are found as a time series in Figure 2. IL-6, CXCL8, CCL2, CCL3, CCL4, CXCL2, CXCL8, GM-CSF and TRAIL had dramatic serum concentration increases during the CPB time with UF (p < .05) while ET1 trended towards a statistically significant increase (p = .06). IL-1α and IL-1β statistically decreased (p < .05) while IL-2 and CXCL1 did not change. Soluble adhesion molecule concentrations in the serum are displayed in Figure 3. P-selectin, ICAM-1 and VCAM-1 increased at the end of CPB with UF (p < .05) relative to initiation while E-selectin and L-selectin did not change. Cytokine and chemokine mediator concentrations throughout CPB with UF. Values are displayed as median and IQR. Observations (N) through the time series are recorded. CCL: CC chemokine ligand; CPB: cardiopulmonary bypass; CXCL: CXC chemokine ligand; ET1: endothelin-1; GM-CSF: granulocyte-macrophage colony-stimulating factor; IL: interleukin. Adhesion molecule concentrations throughout CPB with UF. Values are displayed as median and IQR. Observations (N) through the time series are recorded. Effluent concentrations were not measured for large molecular mass adhesion molecules. ICAM-1: intracellular adhesion molecule 1; VCAM-1: vascular cell adhesion molecule 1.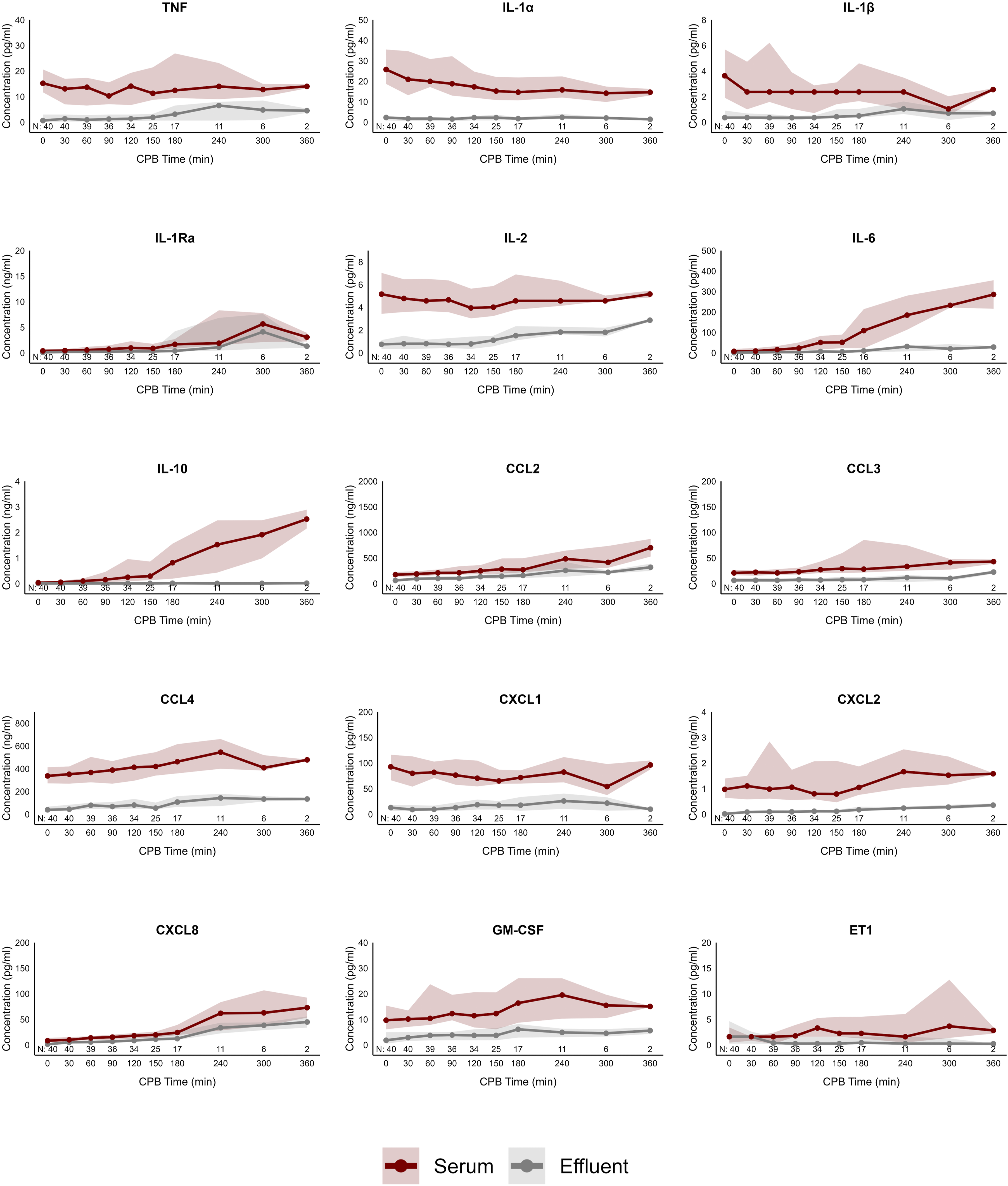
C3a, C5a, TNF, IL-1α, IL-1β, IL-1Ra, IL-2, IL-6, IL-10, CCL2, CCL3, CCL4, CXCL1, CXCL2, CXCL8, ET1, GM-CSF and TRAIL were detected in the UF effluent throughout CPB with varying concentrations (Figures 1–3). At CPB cessation, C3a was unique in that the effluent concentrations of 449 (337 – 672) ng/ml were consistently higher than those measured in the circulation at 56 (47 – 65) ng/ml. C5a effluent concentrations were 0.03 (0.01 – 0.07) ng/ml, compared to 0.10 (0.02 – 0.20) ng/ml in the plasma. CXCL8 concentrations were more similar in the effluent (24 (10 – 46) pg/ml) and plasma (32 (18 – 97) pg/ml) while IL-6 effluent concentrations were 117–20 pg/ml, far lower than the paired plasma at 90 (51 – 274) pg/ml. IL-10 effluent concentrations showed a larger divergence between effluent (0.02 (0.01 – 0.02) ng/ml) and paired plasma concentrations (0.8 (0.4 – 3.0) ng/ml).
Ultrafiltration immunomodulatory effects
UF immunologic effect throughout CPB (n = 40); median (IQR).
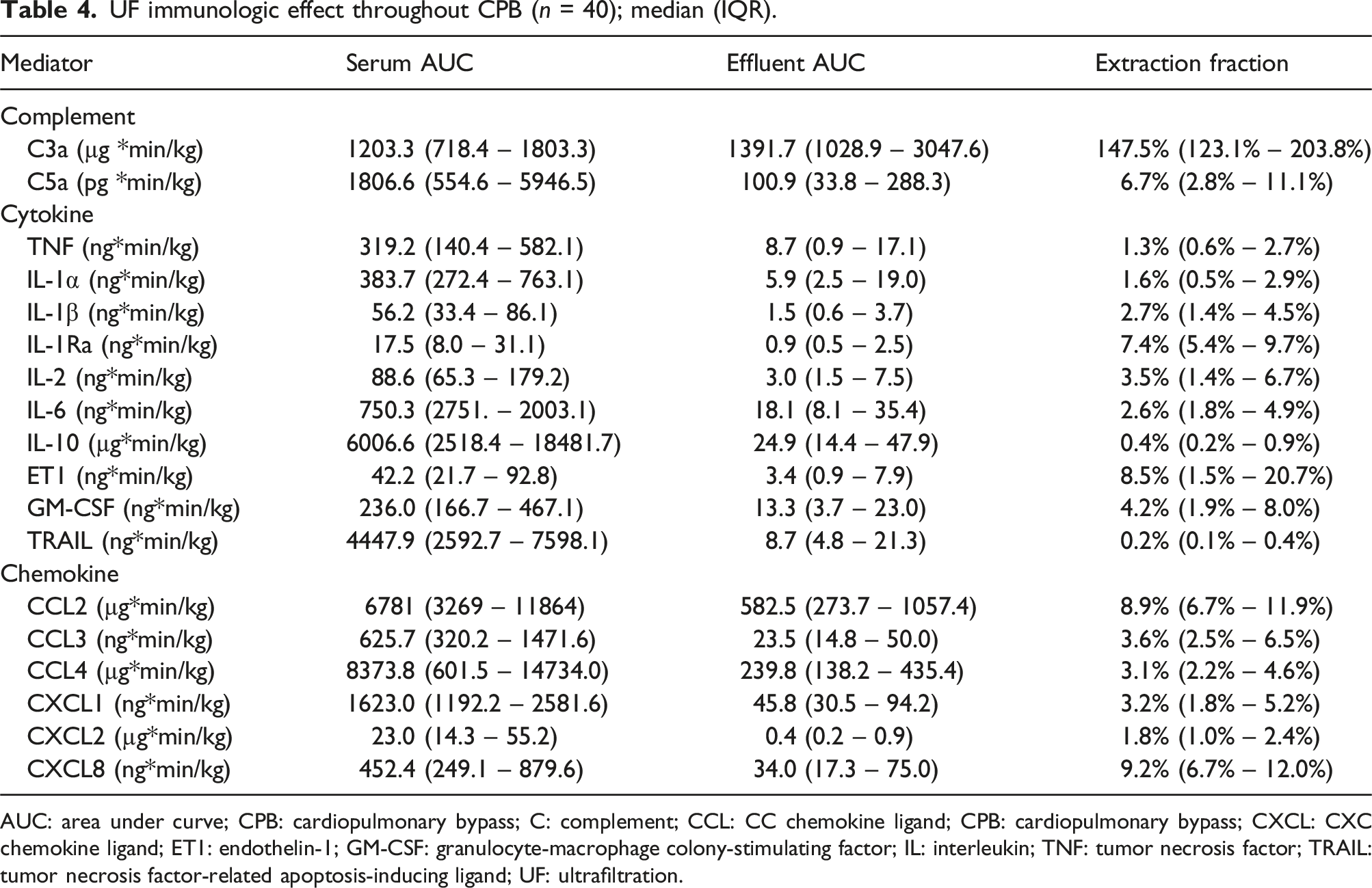
AUC: area under curve; CPB: cardiopulmonary bypass; C: complement; CCL: CC chemokine ligand; CPB: cardiopulmonary bypass; CXCL: CXC chemokine ligand; ET1: endothelin-1; GM-CSF: granulocyte-macrophage colony-stimulating factor; IL: interleukin; TNF: tumor necrosis factor; TRAIL: tumor necrosis factor-related apoptosis-inducing ligand; UF: ultrafiltration.
Counterfactual analysis
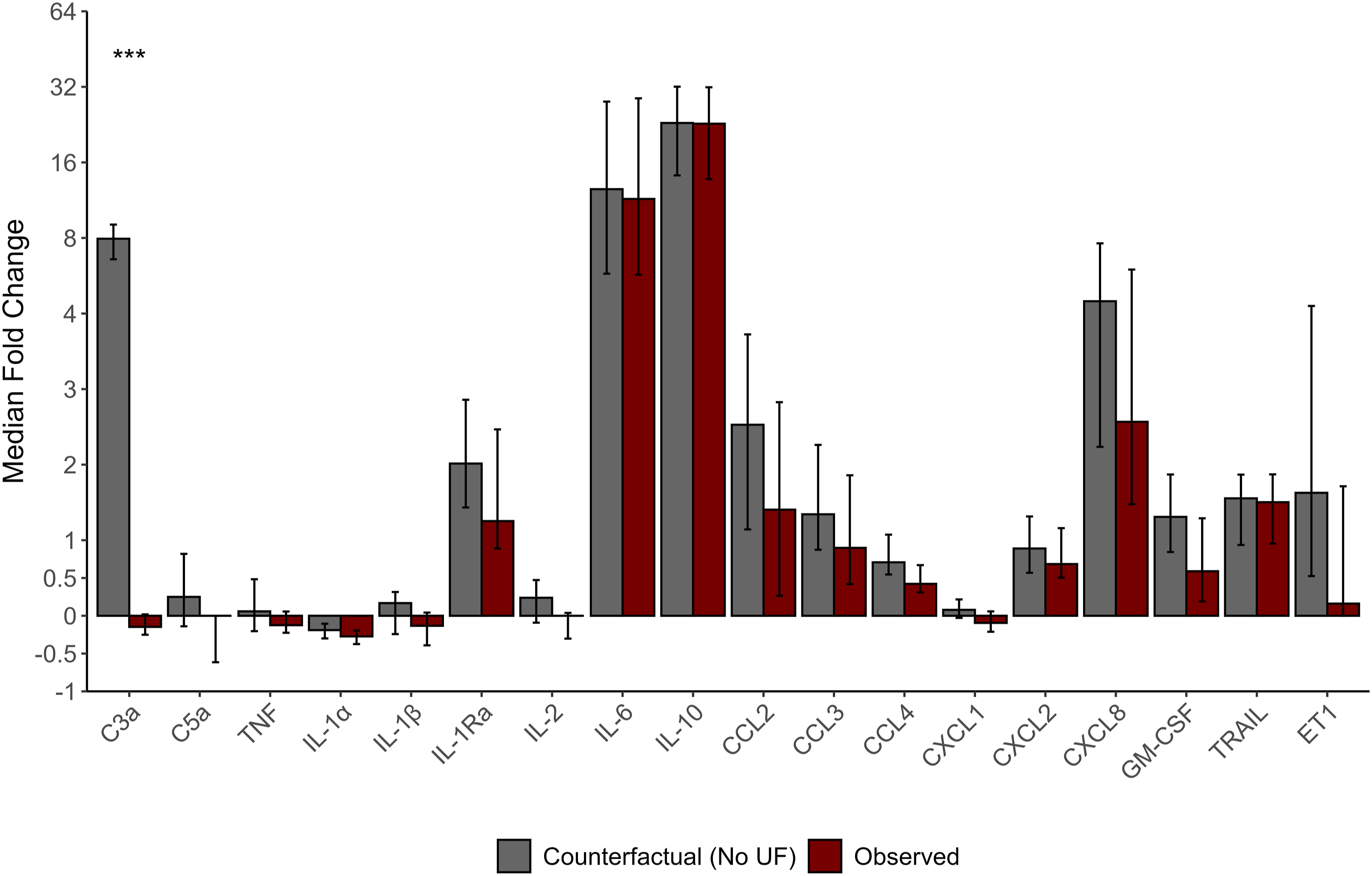
Mediators extracted in the effluent of UF were included in the counterfactual analysis to estimate the immunomodulatory effect of the therapy. The observed (UF) and counterfactual (no UF) times series along with the corresponding differences are plotted in Figure 4. Assessed by AUC, only C3a had statistically different time series between counterfactual and observed curves (p = 3.5 × 10−12). Similarly, the mediator fold change over CPB in the observed and counterfactual scenarios (Figure 5) was only different for C3a (p = 1.4 × 10−14) where the observed C3a concentration had a median fold decrease [95% CI] of −0.1 [−0.2 to 0] times compared to a counterfactual (no UF) which was 7.9 [6.6 – 9.0] times higher. GLMEM revealed differences between counterfactual and observed datasets in C3a (p = 2.3 × 10−87), C5a (p = .02), IL-1β (p = .03), IL-1Ra (p = .04), IL-2 (p = .02), CCL2 (p = 4.9 × 10−3), CXCL1 (p = .01), GM-CSF (p = .05) and ET1 (p = .05) but other cytokine and chemokine masses were not statistically significantly different (p > .05). The C3a mediator mass at CPB cessation was higher in the counterfactual scenario (no UF) compared to the observed (UF) data (63.0 vs 7.3 ug/kg; p = 1.5 × 10−14) alongside IL-2 (0.88 vs 0.76 ng/kg; p = .04) and CXCL1 (13.5 vs 11.9 ng/kg; p = .03). Other mediators were not statistically differentiable (p > .05) between counterfactual and observed mediator masses at the end of CPB (Table 5). Comparison of counterfactual (No UF) and observed (UF) mediator mass. The difference between counterfactual and observed concentrations is displayed. Statistical comparison between the counterfactual and observed mediator mass over the time series is by AUC and GLMEM with the direct comparison and model interaction term p-value displayed. Values are displayed as median and IQR. Observations (N) through the time series, equivalent between the counterfactual and observed groups, are recorded. AUC: area under curve; C: complement; CCL: CC chemokine ligand; CPB: cardiopulmonary bypass; CXCL: CXC chemokine ligand; ET1: endothelin-1; GLMEM: generalized linear mixed-effects model; GM-CSF: granulocyte-macrophage colony-stimulating factor; IL: interleukin. Comparison of counterfactual and observed mediator mass expressed as median fold change [95% CI] mass (n=40 for each group). Fold change calculated by ((mediator mass CPB cessation – mediator mass CPB initiation ) / mediator mass CPB initiation) and [95% CI] estimated by 1000 nonparametric bootstrap samples. *** indicates statistical significance with p < 0.001. C: complement; CCL: CC chemokine ligand; CXCL: CXC chemokine ligand; ET1: endothelin-1; GM-CSF: granulocyte-macrophage colony-stimulating factor; IL: interleukin. Comparison of observed and counterfactual mediator mass (n = 40 for each group); median (IQR). CPB: cardiopulmonary bypass; C: complement; CF: complement factor; CCL: CC chemokine ligand; CPB: cardiopulmonary bypass; CXCL: CXC chemokine ligand; ET1: endothelin-1; GM-CSF: granulocyte-macrophage colony-stimulating factor; IL: interleukin; TNF: tumor necrosis factor; TRAIL: tumor necrosis factor-related apoptosis-inducing ligand.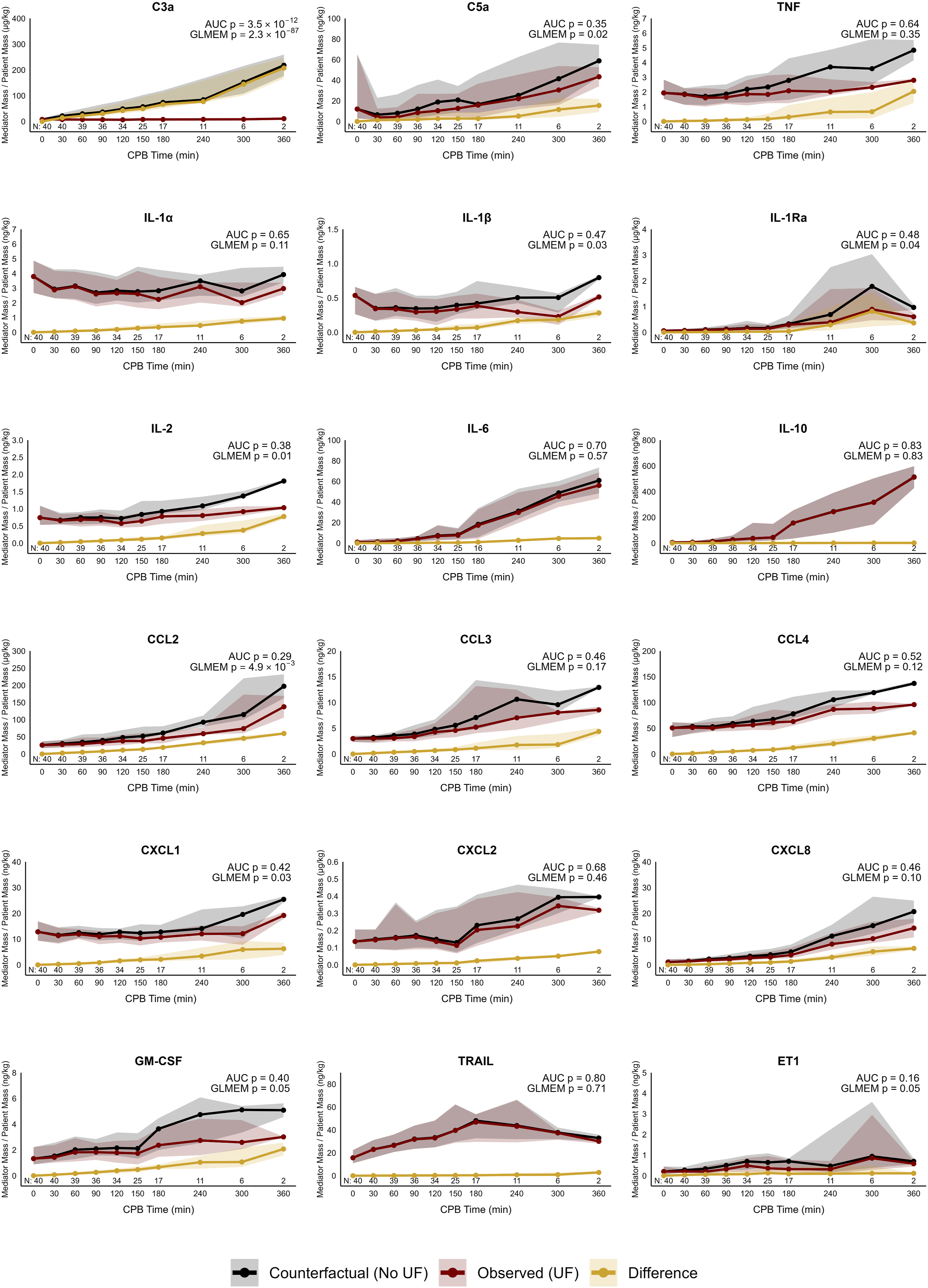
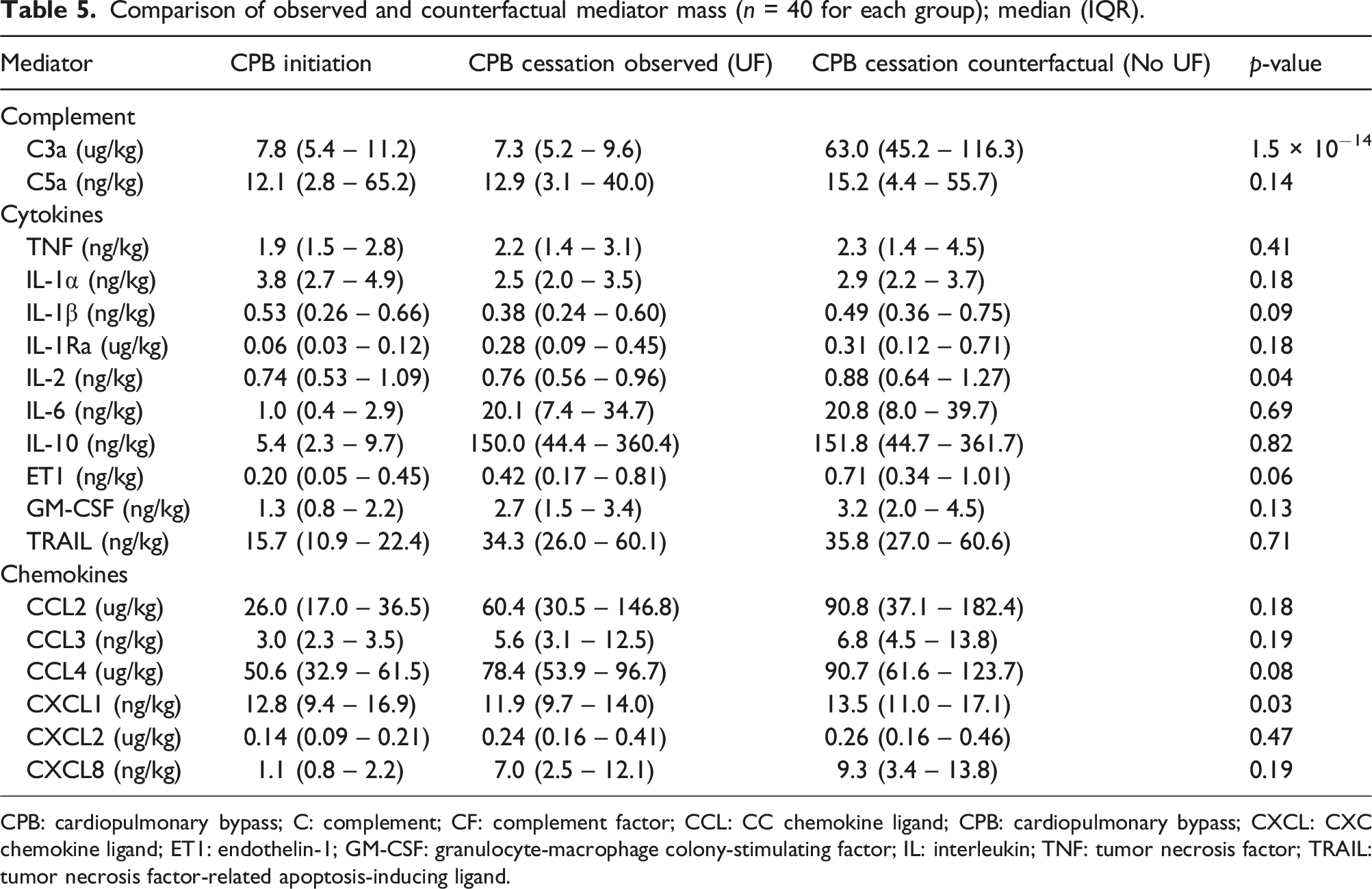
Discussion
This study examined 33 inflammatory mediators in time-series during pediatric CPB and continuous UF. C3a along with seventeen cytokines and chemokines were detected in the ultrafiltration effluent throughout UF. In general, the effluent concentrations of these mediators, with the exception of C3a, were far lower than their paired plasma samples. Prior research has come to a variety of conclusions on the effective sieving coefficient, probably related to different types of filters and immunoassay techniques.15,25–27 Because sieving coefficient, alone, is insufficient to describe the efficacy of UF on these mediators, extraction fraction was used to contextualize the sieving coefficient to the volume of effluent removed relative to the patient circulating volume, thereby more accurately describing the anti-inflammatory effect of UF during CPB. Other than C3a, the inflammatory mediators had a generally low median extraction fraction ranging from 0.2% to 9.2%. In a counterfactual analysis that considers weight-standardized mediator masses in the circulation, UF effectively extracted C3a with statistically significant difference between observed (UF) and counterfactual (no UF) data assessed by time series AUC, GLMEM and fold change methods. UF extraction of IL-1β, IL-1Ra, IL-2, CCL2, CXCL1, GM-CSF and ET1 appeared to be borderline as only the GLMEM method sensed a difference between observed and counterfactual scenarios. Therefore, the anti-inflammatory impacts do appear limited to C3a as the therapy extracts significant amounts C3a from the patient’s circulation leading to a stable concentration of this anaphylatoxin throughout CPB. Thereby, any potential beneficial immunomodulatory clinical effects are most likely related to C3a extraction as other pro-inflammatory mediators were not significantly impacted.
The unique dynamics of C3a during CPB and SBUF are a major finding of this investigation. Initiating the alternative compliment pathway activation, C3 is cleaved via the ‘roll-over tick’ mechanism during contact with the non-endothelialized CPB circuit, producing the split products C3a (a potent pro-inflammatory anaphylatoxin) and C3b (which goes on to form amplifying C3- and C5-convertase machinery , and itself, is a pro-inflammatory stimulus).2,6,7,28 Although C3a and C3b are known to have different degradation pathways, the extreme divergence observed in our study of C3a versus C3b concentrations, is notable.29,30 Relative to CPB initiation, C3a showed a median decrease of 10.1 ng/mL while C3b showed a median increase of 198.7 ug/mL. Opposite to observations in this report, prior investigations without continuous ultrafiltration showed a linked response of concentration increase during pediatric CPB. 31 Journois et al. identified that C3a is extracted well by zero-balance ultrafiltration with a correspondingly lower plasma concentration than a control group, similar to our results here reported. 9 Although causality cannot be proven in this observational data set, the link between C3a extraction and decreased concentration throughout CPB is difficult to ignore. This result suggests that continuous ultrafiltration might be considered an “anti-C3a therapy” given the magnitude of effect observed and associated decrease of C3a concentrations during CPB. It is yet to be determined if the lowering of C3a concentration by continuous UF, leads to a lower concentration of all downstream inflammatory mediators.
One striking observation is that the effluent concentrations of C3a were often higher than the paired serum concentrations The unexpected relationship could potential be explained by the sampling method of serum and effluent, which are paired in time but not sampling location as the plasma sample was taken from the post-filter CPB arterial line, remote from the hemoconcentration device which the effluent sample was taken. A previous dedicated analysis on C3a concentrations sampled pre- and post-filter as well as the effluent, also revealed a similar pattern of effective C3a extraction where pre-filter inflow concentration was higher than the post-filter outflow with the difference proportional to the measured effluent concentration. 32 Similar to our results, this study also exhibited effluent C3a concentrations (2465 ± 780 ng/mL) more than the paired post-filter samples (2022 ± 407 ng/mL) but less than the pre-filter sample (4592 ± 1246 ng/mL). 32 The analysis presented in this report specifically focuses on the mediator mass circulating and extracted in the effluent, as measured, which likely overcomes any measurement bias described above.
Similar to prior investigations, these results showed that IL-6, CXCL8, IL-1Ra and IL-10 were dynamically produced in response to multiple pro-inflammatory stimuli including the artificial CPB circuit and myocardial ischemia-reperfusion.1–3 Interestingly, and in contrast to previous reports, TNF concentrations were largely static during CPB while IL-1α and IL-1β showed modest decreases in concentration.1–3 These three pro-inflammatory mediators are usually upregulated during CPB as part of the systemic pro-inflammatory response.4,9,31,33 Our observational results, however, are not able to definitively conclude if ultrafiltration is the direct causal reason for the suppression of these important mediators. Indeed, it is unlikely, as the extraction fractions of TNF, IL-1α and IL-1β are less than 5%. Several chemokines including CCL2, CCL3, CCL4 and CXCL2 along with GM-CSF and TRAIL were also significantly elevated by the end of CPB with SBUF, while ET-1 trended towards an increase. The soluble forms of adhesion molecules ICAM-1, VCAM-1 and P-selectin were also found at higher concentrations after CPB exposure.
IL-10 is a potent immunosuppressive mediator in both the innate and adaptive systems that control pro-inflammatory responses and reduce tissue damage.34,35 IL-10 is produced by most subtypes of leukocytes and functions as a homodimer, with molecular mass of roughly 30 kDa. It exerts most of its suppressive function on neutrophils, macrophage by inhibiting pro-inflammatory NF-κβ cytokine production and also inhibits antigen-presenting cell function.34,35 Similar to previous reports, IL-10 is produced in substantial quantities during pediatric cardiac surgery with CPB.1,2,5,36 Furthermore, we calculated the IL-10 extraction fraction to be only 0.2%. Despite its small molecular mass, IL-10 is consistently found in trace concentrations in ultrafiltration effluent as demonstrated here and in previous work, with sieving coefficients between 0.1% and 2.5%.15,37 Although many cytokines have sieving coefficients less than 50%, IL-10 falls at the low end of the spectrum, perhaps because of other quaternary interactions in the circulation. 15 In any case, there is substantial evidence to suggest that ultrafiltration during CPB spares this important anti-inflammatory mediator. Focused research on IL-10 is required to identify clinically relevant anti-inflammatory impacts during pediatric cardiac surgery and CPB.
CPB-associated inflammation is a well-described syndrome during and after pediatric cardiac surgery, however, effective immunomodulatory remain elusive.1,4,38 There is little consensus on which mediator or collection of mediators is most important to the post-operative clinical outcomes. Prior work utilizing principal component analyses and linear correlations have suggested that complement activation and anaphylatoxins could be the most relevant to inotrope and ventilator use as well as intensive care unit length of stay.5,39 Furthermore, there is conflicting evidence that IL-6 and CXCL8 influence post-operative recovery.5,40 Based on these results here presented, it appears that UF during CPB can only meaningfully influence C3a concentrations and further comparative research is required to assess if C3a extraction, and any cascading immunologic sequalae, corresponds to clinically relevant benefits after children’s heart surgery.
The results of this study should be considered relative to several limitations. First, the data rises from a single-arm observational series of patients without a comparator group, thus the findings are largely hypothesis-generating, requiring validation by prospective comparative evidence. Second, the patient population is heterogenous in several variables relevant to systemic inflammation including age, type of congenital heart disease, use of prophylactic steroids, sanguineous CPB prime, myocardial ischemia time and CPB duration. Heterogeneity influences the recorded variance in immunologic biomarker measurements throughout CPB but also facilitates generalizability of the results across a diverse congenital heart disease patient population. Third, the results are largely limited to Terumo Capiox® Hemoconcentrator HCO5 as other filters might have different sieving effects on the mediators of interest. Hemoconcentrators from various manufacturers might differ in material, pore size, hydrophobicity and biochemical charge that influence the movement of mediators across the membrane into the effluent. 26 Finally, the counterfactual approach of analysis can only consider immediate first order impacts of not utilizing UF (each mediator’s concentration) and is not able to determine critical second and third order dynamics of mediator interactions in the systemic inflammatory cascade. The ongoing ULTRA study (NCT04920643) is a randomized trial that was designed to understand the immunologic and clinical impact of continuous ultrafiltration rate to enhance recovery after children’s heart surgery by comparing a high- and low-exchange SBUF protocols, which should overcome this limitation in the counterfactual approach. 41
In conclusion, continuous UF during pediatric CPB with median effluent extraction of 52 mL/kg/hr extracted eighteen inflammatory cytokines, chemokines and complement anaphylatoxin C5a with minimal effectiveness. Only C3a was significantly extracted by UF in observed data and exploratory counterfactual analysis, which is consistent with a limited number of studies that specifically investigated this mediator’s characteristics of UF extraction. The predominant anti-inflammatory mediator IL-10 was preserved in circulation with virtually no extraction by SBUF. For the first time, our findings suggest that ultrafiltration during pediatric cardiac surgery and CPB could be considered an anti-C3a therapy, subject to confirmation by prospective comparative trial data.
Footnotes
Acknowledgements
The authors thank the clinical departments of Cardiac Surgery, Cardiology, Critical Care, Clinical Perfusion and Peri-Operative Care at the IWK Health Centre. Specific recognition to Suvro Sett MD and Amy Lockhart RFNA for clinical support of the project as well as Nong Xu and Dr Ian Haidl for Luminex immunoanalysis support. Above all, we would like to acknowledge the patients and families who participated in this study.
Author contributions
All authors were responsible for translational study design, interpretation of results and editing of this manuscript. JB, RS, MH, and DH collected clinical data. JB and JM completed acquisition of immune mediator data. JB and PA conducted statistical analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from Dalhousie University’s Department of Surgery, the Dalhousie Medical Research Foundation, Research Nova Scotia, and the Nova Scotia Department of Health and Wellness through the Clinician Investigator Program.
Ethical approval
IWK Health Centre Research Ethics Board approved (#1024869) this study on November 21, 2019, and was maintained for the duration of the investigation. Written informed consent was obtained from all participant’s substitute decision maker.
Data Availability Statement
The datasets generated and/or analyzed during the study are not publicly available due to patient data and information confidentiality but are available from the corresponding author on reasonable request.
