Abstract
Purpose
To assess whether prone positioning (PP) increases bleeding risk compared to supine positioning in ARDS patients undergoing veno venous Extracorporeal Membrane Oxygenation (VV ECMO).
Materials and Methods
A single-center retrospective observational study was conducted between January 2012 and March 2023. Data were systematically collected from an institutional ECMO registry, including baseline characteristics, daily variables, bleeding events, and outcomes. We compared the relative risk (RR) and incidence rate ratio (IRR) of bleeding between ‘Prone' and ‘Supine' patients. Bleeding-free days were analyzed using Kaplan-Meier curves and the Log-Rank test.
Results
We included 136 consecutive severe ARDS patients undergoing VV ECMO (65% male, age 52 ± 11, 53% bacterial pneumonia), with 85 (62%) and 51 (38%) in the ‘Prone’ and ‘Supine’ group respectively. Bleeding occurred in 79 (58%) patients, with 43 of these being major bleeding events, including 14 intracranial hemorrhages. Fifty-two (61%) ‘Prone’ patients versus 27 (53%) ‘Supine’ patients had bleeding (RR 1.11 (95% CI: 0.81–1.52), p = .44; IRR 1.08 (95% CI: 0.68–1.75), p = .75), with a tendency towards lower major bleeding incidence in the ‘Prone’ versus ‘Supine’ patients (IRR 0.54 (95% CI: 0.29–1.03), p = .06). Kaplan-Meier survival analysis showed no significant difference in bleeding-free days between the ‘Prone’ and ‘Supine’ groups (p = .11).
Conclusions
PP during VV ECMO for ARDS was not associated with an increased incidence of bleeding. Further prospective studies are warranted to confirm these findings.
Introduction
Prone Positioning (PP) has been shown to improve outcomes in patients with acute respiratory distress syndrome (ARDS) 1 by enhancing overall ventilation/perfusion matching, reducing lung stress and strain, and potentially mitigating ventilator-induced lung injury.2–4 These benefits may theoretically be exploited also in patients supported with VV ECMO. During the COVID-19 pandemic, PP was widely used in many ECMO centers, with limited supporting evidence.5,6 Earlier studies suggested that PP during ECMO could accelerate weaning and improve survival, 7 but more recent evidence has produced conflicting results.8,9 In particular, concerns regarding the safety of PP during ECMO persist.5,9 Among the possible ECMO complications, bleeding may be particularly difficult to diagnose and manage while the patients are in PP. Several previous reports5,7,9–11 described the occurrence of bleeding during PP on ECMO, with an overall incidence rate of 98 episodes in 254 patients. 12 However, whether PP increases the risk of bleeding during VV ECMO remains unclear and has not been specifically evaluated. One possible, yet unproven, hypothesis is that frequent position changes (prone-supine) may contribute to catheter or cannula displacement, as well as pressure-induced soft tissue damage, particularly in vulnerable areas such as the face, which may potentially lead to bleeding. This mechanical stress could represent an indirect risk factor for hemorrhagic complications in prone patients.
The present study aims to evaluate whether the incidence of bleeding is higher among ARDS patients undergoing PP during ECMO than among those managed without PP.
Materials & methods
Study design
This retrospective observational study was conducted at the General Intensive Care Unit of the Foundation IRCCS Ca’ Granda - Ospedale Maggiore Policlinico (Milan, Italy), an Italian tertiary referral center for respiratory failure and ECMO. The study was conducted following the World Medical Association Declaration of Helsinki ethical principles for medical research involving human subjects 13 and the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for reporting observational studies. 14 The study received approval from the Territorial Ethics Committee Lombardia 3 (Reference number 4203_S_P). Written consent was waived due to the retrospective design of the study.
Patient population
All consecutive severe ARDS patients undergoing ECMO for more than 24 h between January 2012 and March 2023 were considered for inclusion. Inclusion criteria comprised: - diagnosis of ARDS (i.e., Berlin criteria 15 ), - use of VV ECMO support. Exclusion criteria were: - age below 18 years, - veno-arterial ECMO (VA-ECMO) support, - extracorporeal CO2 removal (ECCO2R), - pregnancy, - ECMO support in the immediate postoperative period after lung transplantation or as a bridge to lung transplant; - survival <24 h.
Clinical management of extracorporeal membrane oxygenation
Patients were treated following standardized protocols for sedation, anticoagulation and ECMO circuit management.16–18 Most patients received VV ECMO via femoro-femoral access. Alternative cannulation (femoro-jugular or jugulo-femoral) was used when femoro-femoral access was not feasible or did not provide adequate blood flow (BF). Cannulation was performed percutaneously, with cannula sizes based on ultrasound assessment of vessel dimensions. Getingue HLS® cannulas (21–25 Fr, 55 cm) were used for drainage, while Bio-Medicus Medtronic® cannulas (21–23 Fr, 50 cm) were used for reinfusion. In some cases, a bi-caval dual lumen 31 r cannula (Avalon Elite® (Getinge, Sweden)) was used. The extracorporeal systems utilized were the Cardiohelp® device with HLS Set Advanced or the PLS® membrane oxygenator with Rotaflow pump and console (Getinge, Sweden).
For detailed information on ECMO management and ventilation strategies, please refer to the Supplemental Digital Content (S1. ECMO Management and Ventilation Strategies).
Anticoagulation during ECMO was managed with unfractionated heparin according to the institutional protocol previously published by our center.18,19 In cases of heparin-induced thrombocytopenia (HIT), argatroban was administered instead of heparin. Further details are provided in the Supplemental Digital Content (S2. Anticoagulation and ECMO Circuit Management Protocol).
Bleeding events were classified as major or minor according to the ELSO criteria. Major bleeding was defined as visible bleeding associated with a hemoglobin drop of at least 2 g/dL within 24 h, blood loss greater than 20 mL/kg in the same period, or the need for one or more transfusions exceeding 10 mL/kg of packed red blood cells (PRBC). Retroperitoneal, pulmonary, or intracranial bleeding, as well as any bleeding requiring surgical or interventional procedures, were also considered major bleeding. Minor bleeding was defined as blood loss of less than 20 mL/kg per day, requiring no more than a single transfusion of 10 mL/kg of PRBC. 20
Management of prone position during ECMO
The proning maneuver was performed under direct supervision of the attending physician, following international guidelines and previously published protocols.1,17,18 Prone positioning was indicated in the presence of severe hypoxemia despite FiO2 and FSO2 set at 100%, ventilatory parameters suggestive of lung injury with plateau pressure greater than 32 cm H2O despite adjustments, or a lack of improvement in gas exchange and respiratory system compliance after 48 hours of ECMO support.
Before pronation, patients were sedated and paralyzed. The procedure required eight caregivers to safely position the patient while a perfusionist managed the ECMO tubing. Ventilator and ECMO settings (sweep gas flow, ECMO pump speed, respiratory rate, and tidal volume) were kept constant during and after pronation. The planned duration of prone positioning was at least 12 consecutive hours. The patient was returned to the supine position before the end of the pronation cycle in case of clinical deterioration, difficulty in maintaining adequate ECMO blood flow, or further worsening of oxygenation. PP was repeated if improvement in gas exchange and lung mechanics were observed and no complications occurred during the procedure.
Data collection
We systematically collected prospective data from an institutional ECMO registry, including baseline demographic and clinical characteristics. Variables collected comprised age, sex, predicted body weight, SAPS II score at ICU admission, ARDS etiology, and the dates of hospital admission, ICU admission, initiation of invasive mechanical ventilation, and ECMO cannulation and decannulation.
Clinical and physiological variables were recorded daily, including ventilation parameters, ECMO settings, hemodynamics, laboratory variables, and bleeding events (for more details, see Supplemental Digital Content S3. Data Collection).
We also collected data related to proning sessions, including the number, duration, and any instances of early interruption. The duration of mechanical ventilation, ICU length of stay, and hospital mortality were also recorded.
Statistical analysis
Categorical variables were reported as absolute and relative frequencies, while continuous variables were summarized as median values [interquartile range]. Missing data were not imputed, as they accounted for less than 10% of the observations for the variables of interest.
Patients were divided into two groups: those who underwent at least one PP maneuver during ECMO (‘Prone’ group) and those who received ECMO support without prone positioning (‘Supine’ group). PP occurring prior to ECMO connection or after ECMO decannulation was not a determinant of the stratification.
Continuous variables were compared between groups using Student’s t-test or Mann-Whitney U test, depending on the nomality of data distribution. The Shapiro-Francia test was used to assess the normality of continuous variables. For the comparison of categorical variables, the Chi-square test was used.
The cumulative incidence of bleeding and major bleeding was calculated as the ratio of the number of first events to the number of patients at risk at the beginning of ECMO.
The incidence rates of bleeding and major bleeding, expressed per 100 ECMO-patient days under treatment, were calculated by dividing the number of first bleeding events by the total number of days at risk.
For ‘Supine’ patients, time at risk corresponds to days from the start of ECMO until either bleeding occurs or ECMO end. For ‘Prone’ patients, time at risk corresponds to days from the start of first pronation in ECMO until either bleeding occurs or ECMO end. The corresponding confidence intervals were estimated using the Normal approximation method. Both cumulative incidence and incidence rate were estimated for the total population and stratified by groups. To compare cumulative incidence of bleeding and major bleeding between groups, we estimated the Relative risk (RR), as ratio between the two values, while the Incidence Rate Ratio (IRR) was used to compare incidence rates.
To assess the effect of prone positioning compared to supine positioning on bleeding events during ECMO, we used Poisson regression models with robust standard errors. Separate models were fitted for the total duration of ECMO exposure and for the days during which patients were in the prone position.
The models were also adjusted for potential confounders, including age, sex, BMI, SAPS-II score, and ARDS etiology (bacterial pneumonia, viral pneumonia, other causes). Incidence Rate Ratios (IRR) with 95% confidence intervals were estimated to assess the association between treatment position and bleeding outcomes.
Bleeding-free days were compared between groups using Kaplan-Meier survival analysis, with differences assessed using the Log-Rank test.
Finally, Cox proportional hazards models with time-dependent covariates were employed to investigate the impact of the duration of prone positioning on bleeding outcomes. In detail, prone positioning during ECMO was modeled as a time-varying covariate, transitioning from ‘prone’ to ‘supine’ either at the cessation of prone positioning or before the start of the next prone period. Hazard ratio (HR and 95% CI) was estimated to assess the association between prone positioning duration and both bleeding events (all and major), adjusting also for potential confounders (age, sex, BMI, SAPS II score, and ARDS etiology). The time at risk for bleeding events was defined from the start of ECMO until the event, censoring at the earliest of death, ECMO decannulation, or the end of the study period.
The statistical analysis used a 2-tailed significance level, with statistical significance defined as p ≤ .05. Statistical analysis was performed using STATA software version 16 (Stata Corp., College Station, TX, USA) and R version 4.3.2 (The R Project for Statistical Computing; https://www.r-project.org/).
Results
Study Population
Between January 2012 and March 2023, 311 patients were screened. Among them, 166 were excluded: 51 patients received VA ECMO, 39 underwent ECMO as a bridge to lung transplantation, 15 were treated with ECCO2R, and 61 received VV ECMO for conditions other than ARDS (54 for post-lung transplant support, 6 for COPD exacerbation, and 1 for hypothermia). In addition, 8 patients were excluded because ECMO support lasted less than 24 h (see Figure 1). Population flowchart. ECLS: extracorporeal life Support; VA-ECMO: veno-arterial extracorporeal membrane oxygenation; VV ECMO: veno-venous extracorporeal membrane oxygenation; ECCO2R: extracorporeal CO2 removal; DLTx: dual-lung transplantation.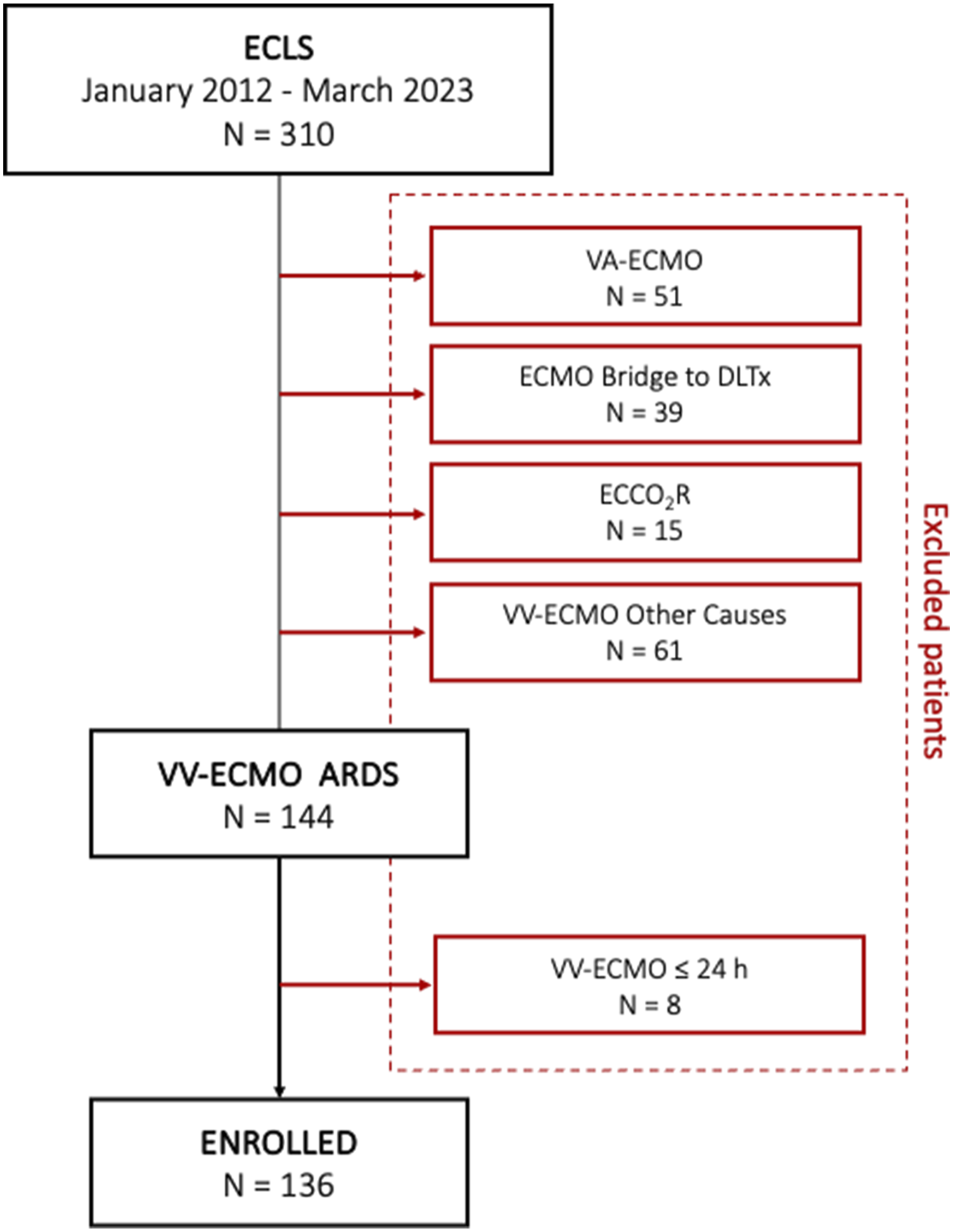
A total of 136 patients undergoing ECMO for severe ARDS were enrolled. Of these, 91 patients (67%) were referred from peripheral hospitals, and ECMO was initiated prior to transport in 73 cases.
Characteristics of the study population and Outcomes stratified by treatment allocation.
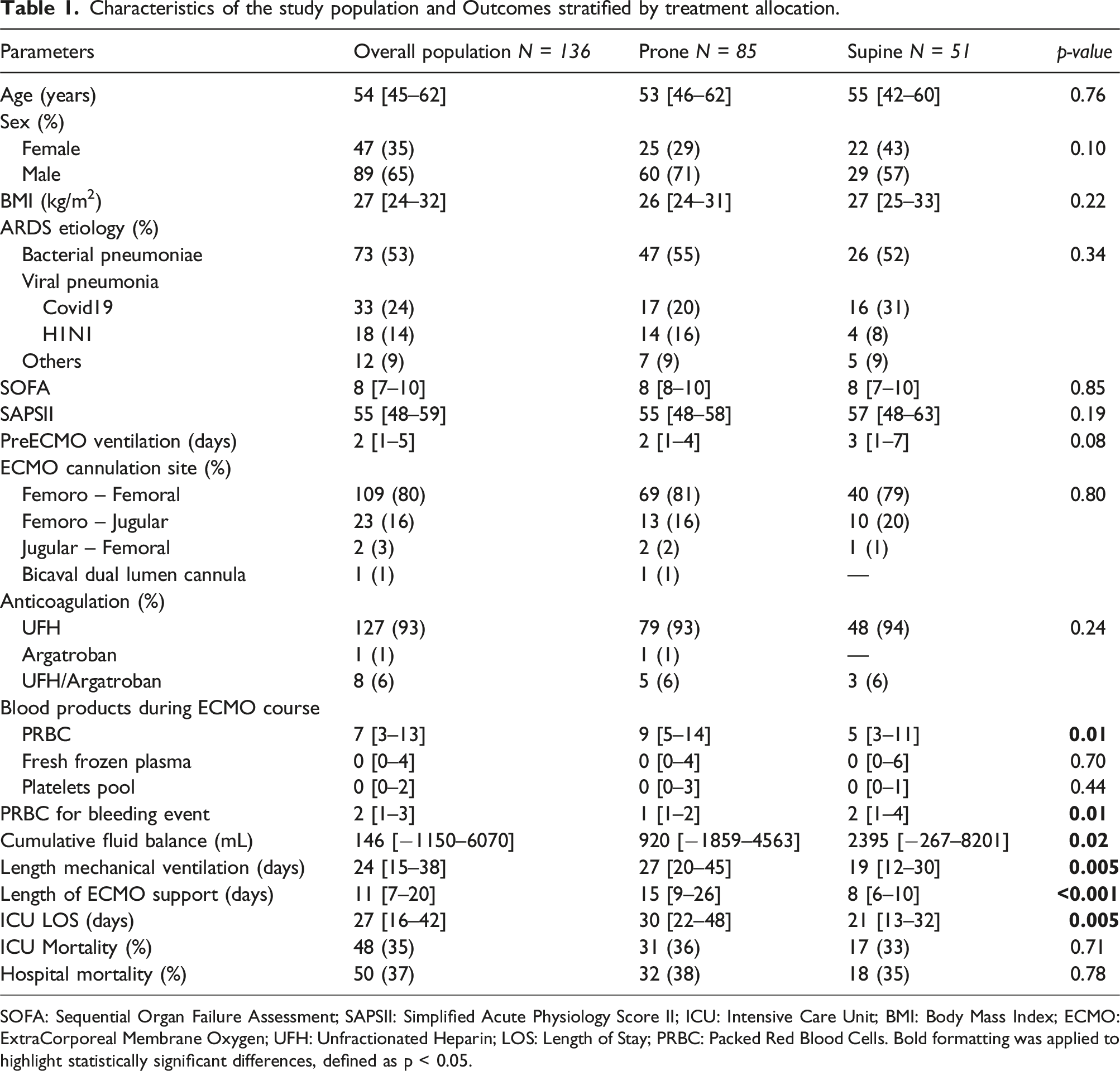
SOFA: Sequential Organ Failure Assessment; SAPSII: Simplified Acute Physiology Score II; ICU: Intensive Care Unit; BMI: Body Mass Index; ECMO: ExtraCorporeal Membrane Oxygen; UFH: Unfractionated Heparin; LOS: Length of Stay; PRBC: Packed Red Blood Cells. Bold formatting was applied to highlight statistically significant differences, defined as p < 0.05.
Pre-ECMO characteristics of the study population stratified by treatment allocation.
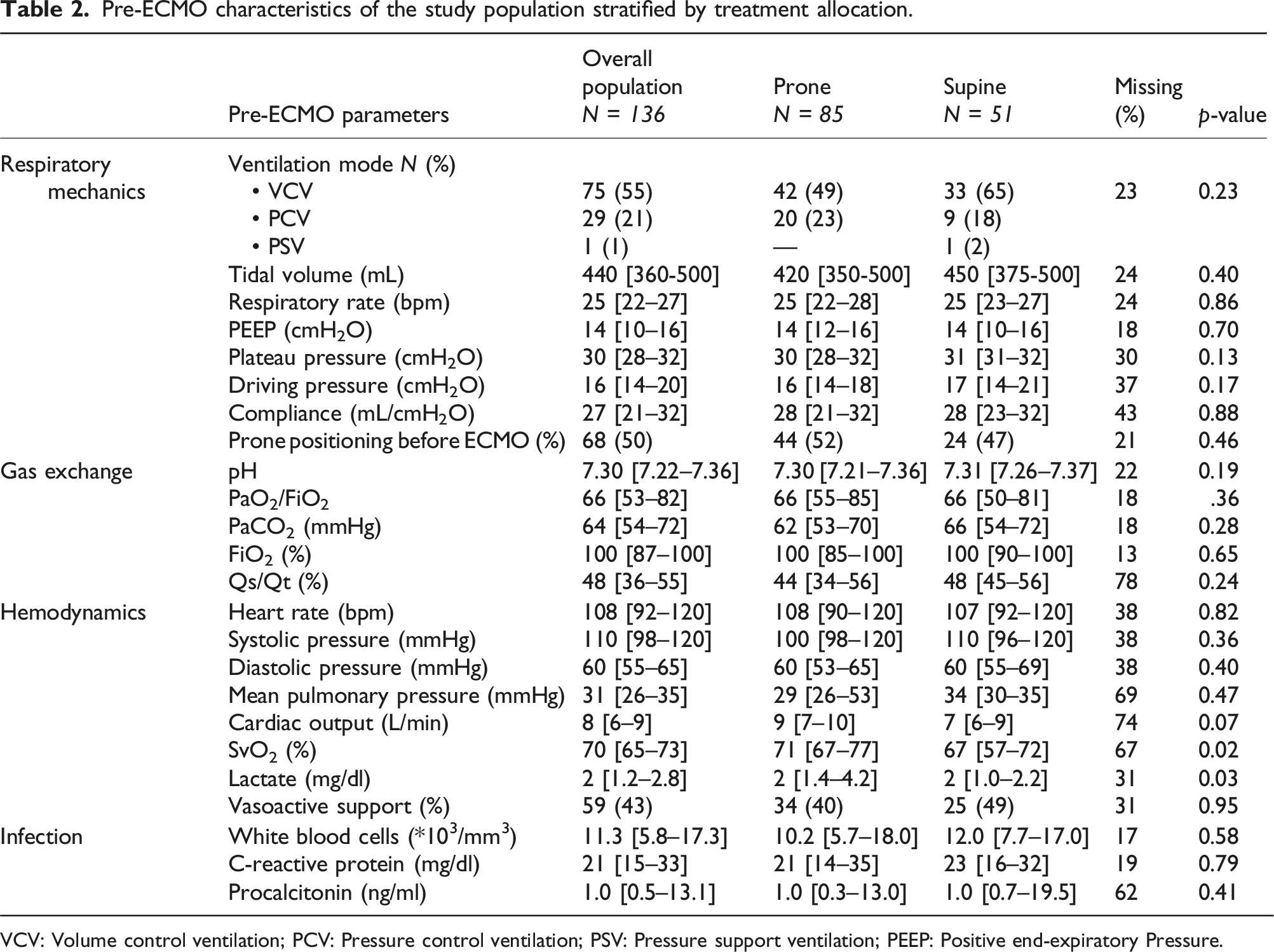
VCV: Volume control ventilation; PCV: Pressure control ventilation; PSV: Pressure support ventilation; PEEP: Positive end-expiratory Pressure.
The median duration of mechanical ventilation was 24 days [15–38], and the median length of ECMO support was 11 days [7–20]. Tracheostomy was performed in 67 patients (49%) on day 14 [10–21]. Hospital mortality was 37% (50 patients).
Prone versus supine
Among the enrolled patients, 85 (63%) underwent at least one prone positioning session during VV ECMO. Baseline characteristics were similar between the ‘Prone’ and ‘Supine’ groups (Table 1). On average, patients in the ‘Prone’ group were proned 4 [2–6] times. Prone positioning sessions were always completed without interruptions due to safety concerns or clinical deterioration. No accidental decannulation, endotracheal tube dislodgment, or technical malfunctions were observed. In one patient (1%) of the ‘Prone’ group, tracheostomy was performed before the first proning session.
Patients in the ‘Prone’ group had a significantly longer duration of ECMO support (15 [9–26] days vs 8 [6–10] days; p < .0001), mechanical ventilation (27 [20–45] days vs 19 [12–30] days; p = .005) and length of ICU stay (30 [22–48] days vs 21 [13–32] days; p = .005).
Additionally, the cumulative fluid balance was significantly lower in the ‘Prone’ group compared to the ‘Supine’ group (920 [−267 to −8201] mL vs 2395 [−267 to −8210] mL; p = .02). ICU mortality was similar between the two groups (31 [36%] in the ‘Prone’ group vs 17 [33%] in the ‘Supine’ group; p = .71), as was hospital mortality (32 [38%] in the ‘Prone’ group vs 18 [35%] in the ‘Supine’ group; p = .78). No other significant differences were observed between the two groups.
Incidence of bleeding
Bleeding characteristics and bleeding risk stratified by treatment allocation.
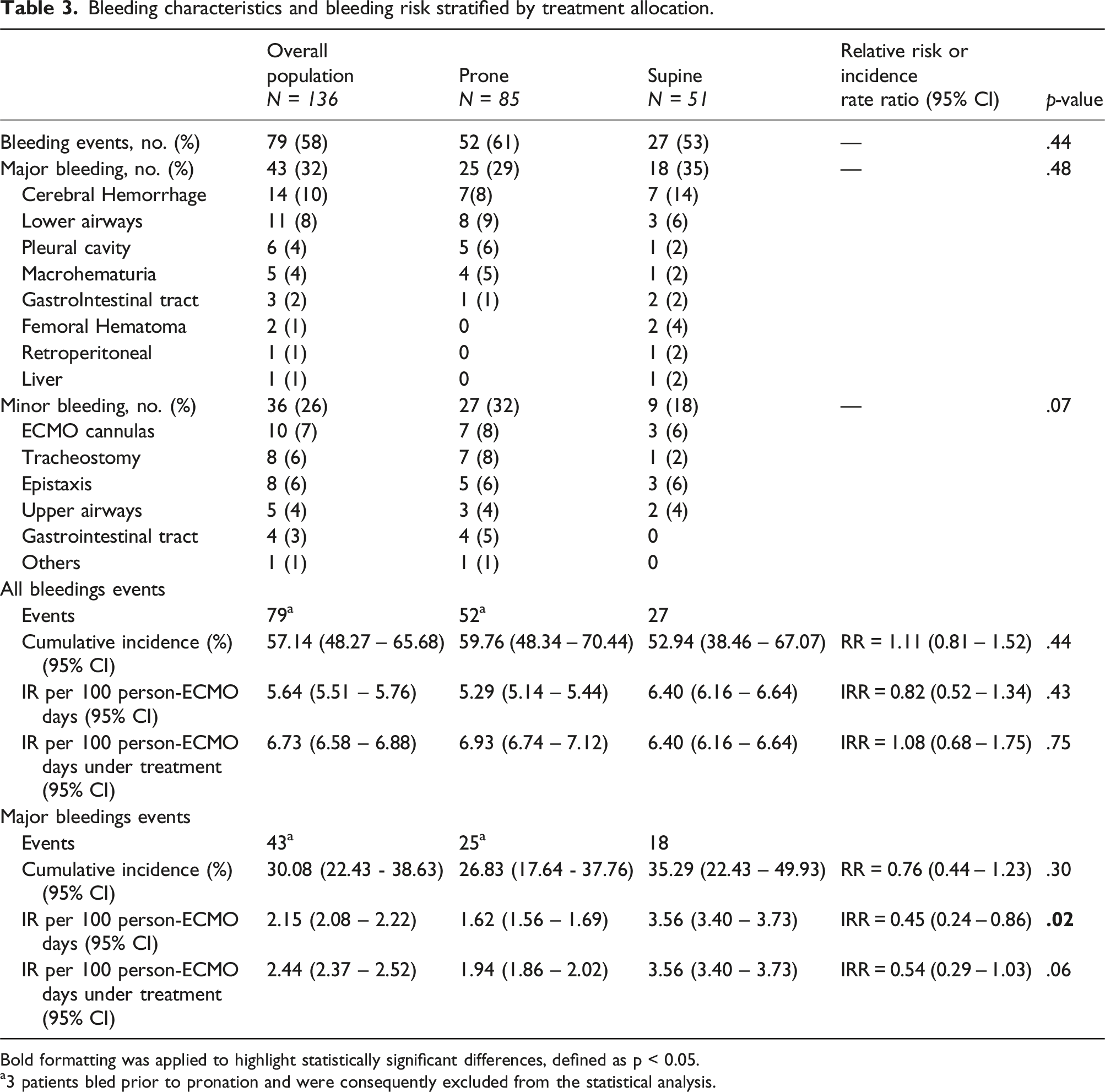
Bold formatting was applied to highlight statistically significant differences, defined as p < 0.05.
a3 patients bled prior to pronation and were consequently excluded from the statistical analysis.
A higher absolute number of bleeding events occurred in the ‘Prone' group compared to the ‘Supine’ group (52 patients [61%] vs 27 patients [53%]); however, the difference was not statistically significant (p = .44).
During ECMO support, patients received 7 [3–13] units of PRBC. The median number of transfused units was higher in the ‘Prone’ group (9 [5–14]) compared to the ‘Supine’ group (5 [3–11]; p = .01). For each bleeding event, the median number of PRBC was 2 [1–3], with 1 [1–2] unit in the ‘Prone’ group and 2 [1–4] units in the ‘Supine’ group (p = .01). In the ‘Prone’ group, the rate of units of PRBC transfusions was 59.33 per 100 ECMO-days (95% CI: 55.79–62.94), while in the ‘Supine’ group, it was 71.83 units per 100 ECMO-days (95% CI: 64.39–79.28), with an IRR of 0.83 (95% CI: 0.73–0.93; p = .003).
No significant differences were found between groups in the use of fresh frozen plasma and platelet transfusions. In 14 patients, surgical or interventional procedures were required to control bleeding. Specifically, thoracotomy was performed in 3 cases, exploratory laparotomy in 1, gastroscopy in 3, colonoscopy in 4, pericardiocentesis in 1, tracheostomy revision in 3, and interventional radiology procedures in 2 cases. In 3 patients, more than one procedure was necessary.
No significant difference was observed in the median dose of heparin administered between the two groups, with a dose of 16 [11–20] U/kg/h in the ‘Prone’ group and 14 [11–19] U/kg/h in the ‘Supine’ group (p = .38).
Three patients in the ‘Prone’ group were excluded from the bleeding incidence analysis because they experienced bleeding before their first session of PP. Among the 133 patients included, the cumulative incidence of bleeding was 59.76% (95% CI 48.34–70.44) in the ‘Prone’ group and 52.94% (95% CI 38.46–67.07) in the ‘Supine’ group, and no significant difference in the occurrence of bleeding was observed between the groups (RR 1.11, 95% CI: 0.81–1.52, p = .44).
The overall bleeding incidence rate was 6.73 episodes per 100 ECMO-days (95% CI 6.58–6.88). In the ‘Prone’ group, the bleeding incidence rate was 6.93 episodes per 100 ECMO-days (95% CI 6.74–7.12), whereas in the ‘Supine’ group, it was 6.40 episodes per 100 ECMO-days (95% CI 6.16–6.64), with an IRR of 1.08 (95% CI 0.68–1.75; p = .74).
The incidence of major bleeding was 2.44 episodes per 100 ECMO-days (95% CI 2.37–2.52) overall, 1.94 episodes per 100 ECMO-days (95% CI 1.86–2.02) in the ‘Prone’ group, and 3.56 episodes per 100 ECMO-days (95% CI 3.40–3.73) in the ‘Supine’ group. The IRR was 0.54 (95% CI 0.29–1.03; p = .06).
The multivariate regression analysis adjusting for potential confounders, including age, SAPS II scores, and ARDS etiology, is presented in Table S1 (see Supplemental Digital Content). The adjusted analyses confirmed that PP was not associated with an increased risk of overall bleeding events (adjusted IRR 0.88, 95% CI 0.55–1.45, p = .61). A post hoc power analysis confirmed that the study had a high power (95%) to detect a significant difference in major bleeding incidence.
No difference in bleeding-free days in ‘Supine’ and ‘Prone’ patients was observed according to survival analysis (p = .11), refer to Supplemental Digital Content, Figure S1, for the Kaplan-Meier analysis.
This finding was further supported by time-to-event analyses adjusting for multiple potential confounders, which showed no significant association between prone positioning duration and the incidence of either all bleeding events or major bleeding events (refer to Supplemental Digital Content, Table S2).
Discussion
This study investigated the incidence of bleeding among patients with severe ARDS undergoing VV ECMO and aimed to compare patients managed with PP to those in the supine position. Study findings do not support our initial hypothesis that PP might increase the bleeding risk.
Bleeding remains one of the most frequent complications in patients undergoing ECMO, despite advances in coagulation management,18,19 the widespread adoption of percutaneous cannulation techniques,21,22 and the use of biocompatible extracorporeal circuits. 23
Overall, the prevalence of hemorrhagic complications is still up to 50% of the patients 24 with possible catastrophic complications such as intracranial hemorrhage (ICH) and associated increased mortality.9,25
The incidence of bleeding complications observed in our cohort is consistent with previous reports. In a similar population of ARDS patients with mixed etiologies, Martucci et al. 26 reported a bleeding incidence of 52.5%, with an incidence rate of 11.2 episodes per 100 ECMO-days. Moreover, we observed a prevalence of ICH around 10%, consistent with several recent studies.8,9 All our cases of ICH were diagnosed ex-post through CT scan analyses carried out following an abrupt occurrence of signs of intracranial hypertension or uncal herniation (i.e., fixed dilated pupils or anisocoria) associated with hypertension and bradycardia.
In such a fragile cohort of patients, the benefits of any new therapy, diagnostic procedure, or management approach have thus to be carefully weighed against the increased risk of hemorrhage. In patients with ARDS, PP remains one of the few therapeutic strategies with a proven beneficial effect on survival.1,3 Therefore, it is reasonable that PP has been increasingly applied in the most severe ARDS patients,6,8,27 including those receiving ECMO. However, PP could theoretically increase the risk of bleeding through cannula displacement, mechanical trauma, or pressure sores, as described in some case reports.25,28 A multicenter retrospective analysis 9 reported that 1.5% of the procedures caused minor bleeding, with a similar incidence of fatal hemorrhage between patients undergoing PP and not (i.e., around 15%). A recent randomized controlled trial showed a similar prevalence of bleeding (defined as the need for >2 packed red blood cell transfusion) between ECMO patients allocated to PP or supine position (i.e., 27% vs 32%, respectively), 8 with an incidence of hemorrhagic stroke of 2%, regardless of treatment allocation. Given the absence of measures of incidence rate (i.e., the number of events occurring during exposure time) in these previous reports, a formal comparison with our results cannot be made. Nevertheless, our work is consistent with the cited studies in that it does not show an increased incidence of bleeding in patients undergoing PP during ECMO. On the contrary, we observed a clinically relevant but non-statistically significant lower incidence rate of major bleeding in the prone group.
This finding could be explained by selection bias and lack of equipoise in the application of PP. Prone positioning may have been preferentially offered to patients with a perceived lower risk of bleeding or greater hemodynamic stability early after ECMO initiation. Another consideration is that in patients who experience bleeding, we might avoid PP and expedite ECMO weaning compared to our standard criteria. This approach is driven by the challenges associated with proning a patient who is actively bleeding. In such cases, prone positioning not only complicates the clinical monitoring and visual inspection of the bleeding but also could make its management significantly more difficult. Nevertheless, we did not observe any significant baseline differences between groups, including demographic characteristics and severity scores at cannulation, such as SOFA and SAPS II.
Our study also showed that patients in the ‘Prone’ group experienced longer ECMO runs and ICU stays 21 but had similar survival rates. These associations, or lack thereof, must be interpreted cautiously: the study’s retrospective design and the limited sample size do not allow any causal relationship between PP and such outcomes to be ascertained. Immortal time bias (i.e., patients that die early cannot be proned) can further skew the results of such a study design, limiting the interpretation and external validity of our results. Still, such a difference in ECMO length must be carefully considered when assessing the risk of bleeding. For this reason, in contrast to most previous studies, we assessed bleeding risk considering the duration of exposure. Although the prevalence of hemorrhage was higher among prone patients, their bleeding incidence rate (IR) was lower.
When studying prolonged therapies such as ECMO, the IR, rather than the prevalence, more appropriately measures actual risk. Moreover, while a significant difference was observed in the overall number of packed red blood cells transfused between the two groups, PP patients had fewer transfusions per 100 ECMO-days. In other words, while the overall transfusion needs were higher in PP patients, the IR of transfusion was lower.
We also analyzed whether bleeding episodes during PP might be more serious than during supine positioning. On the contrary, we observed a lesser need for bleeding-associated transfusion in the PP cohort and a similar need for interventional hemorrhage control.
Overall, these findings suggest that an extended exposure to ECMO, rather than PP, may be associated with an increased risk of bleeding complications. Thus, our results contribute to the growing evidence suggesting that PP may be a safe and feasible maneuver during VV ECMO.
We observed lower cumulative fluid balances in the prone group. On the one hand, this may suggest that PP positively impacts hemodynamics by possibly improving right ventricle function.1,29–31 On the other hand, it may reveal a selection bias when applying PP. Indeed, despite similar hemodynamics at ECMO connection, it is not uncommon to refrain from PP and maintain supine position in patients with shock or right ventricle failure. Further prospective observational studies are necessary to evaluate such association.31,32
Limitations of the study
This study has several limitations. The study’s retrospective nature has inherent biases, such as the previously mentioned selection biases in applying PP. A randomized controlled trial design is necessary to overcome these limitations. Nevertheless, given the established benefits of PP in ARDS patients not undergoing ECMO, lack of therapeutic equipoise and perceived life-saving beneficial effects of PP may still hamper the successful conduct of such RCT. In addition, the anticoagulation protocol adopted in our center (described in the Supplemental Digital Content), although consistently applied to all patients in both groups and aligned with current international guidelines, may nonetheless have influenced the bleeding outcomes and limited our ability to detect potential between-group differences. Furthermore, we acknowledge that other confounding factors, such as circuit thrombosis, type of anticoagulation, platelet count, pre-cannulation antiplatelet or anticoagulant use, and PTT modulation, may have influenced the observed results. While our dataset did not allow for an exhaustive adjustment for all these variables, their potential effects should be explored in future research.
Moreover, our study was performed at a tertiary referral center with decennial experience in PP and ECMO application, with specific protocols, expertise, and dedicated personnel. Thus, the applicability of our findings to different clinical scenarios may be limited. Finally, although a post hoc power analysis confirmed adequate power to detect major differences, our study may still be underpowered to identify small but clinically relevant differences in bleeding rates between prone and supine patients. This highlights the need for adequately powered studies to validate our findings.
Conclusion
In conclusion, our findings support the safe inclusion of prone positioning in VV ECMO management protocols for patients with severe ARDS. While prone positioning did not significantly increase the incidence of bleeding, its impact on patient outcomes remains uncertain due to potential unmeasured confounders and limitations in statistical power. Future prospective studies with adequate sample sizes and more detailed data on anticoagulation strategies and coagulation parameters are fundamental to validate our findings.
Supplemental Material
Supplemental Material - Prone positioning and bleeding risk during extracorporeal membrane oxygenation in severe ARDS patients
Supplemental Material for Prone positioning and bleeding risk during extracorporeal membrane oxygenation in severe ARDS patients by Alessio Caccioppola, Francesco Padovano, Vittorio Scaravilli, Fabiana Madotto, Cristina Dulama, Alfredo Lissoni, Mauro Panigada and Giacomo Grasselli in Perfusion
Footnotes
Acknowledgements
We extend our heartfelt gratitude to the dedicated team of medical professionals, including nurses, residents, and students at the Adult Intensive Care Unit of Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico in Milan.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministero della Salute.
CRediT authorship contribution statement
Alessio Caccioppola: Conceptualization, Methodology, Validation, Formal analysis, Writing original draft, Writing review & editing. Francesco Padovano: Conceptualization, Methodology, Validation, Formal analysis, Writing original draft, Writing review & editing. Vittorio Scaravilli: Conceptualization, Methodology, Supervision, Writing original draft, Writing review & editing. Fabiana Madotto: Methodology, Validation & Formal analysis. Cristina Dulama: Validation, Formal analysis & Writing original draft. Alfredo Lissoni: Conceptualization, Methodology, Supervision, Writing original draft, Writing review & editing. Mauro Panigada Investigation, Methodology, Supervision, Writing review & editing. Giacomo Grasselli: Investigation, Methodology, Supervision, Writing review & editing.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
