Abstract
Background
The benefits of intravascular imaging-guided percutaneous coronary interventions (PCI) are well established. Intravascular imaging guidance improves short- and long-term outcomes, especially in complex PCI. Optical coherence tomography (OCT) has a higher resolution than intravascular ultrasound. However, the usage of OCT is mainly limited by the need to use contrast for flushing injections, which increases the risk of contrast-induced acute kidney injury, especially in patients with underlying chronic kidney disease. The aim of this study was to prove that flushing techniques with normal saline instead of contrast can be used in OCT imaging and can generate high-quality images.
Methods
This prospective single-center observational study included patients with indications for OCT-guided PCI. For OCT pullbacks, heparinized saline was injected by an automatic pump injector at different rates, and additional extension catheters for selective coronary artery engagement were used at the operator’s discretion. Recordings were made using the Ilumien Optis OCT system (Abbott) and the Dragonfly (Abbott) catheter and were analyzed at 1-mm intervals by two operators. Pullbacks were categorized as having optimal, acceptable, or unacceptable imaging quality. A clinically usable run was determined if >75% of the region of interest length was described as having optimal or acceptable imaging quality.
Results
A total of 32 patients were enrolled in the study; 47 different lesions were assessed before and after PCI. In total, 91.5% of runs were described as clinically suitable for use.
Conclusion
Heparinized saline injections for OCT imaging are effective in generating good-quality OCT images suitable for clinical use.
Keywords
Introduction
Approximately 7% of patients undergoing percutaneous coronary intervention (PCI) experience contrast-induced nephropathy (CIN). 1 However, in patients with advanced kidney disease, the incidence of CIN increases to around 30%.1,2 Contrast-induced nephropathy has been strongly associated with adverse clinical outcomes, including death, the need for hemodialysis, increased hospital costs, and lengths of stay.1–6 The ratio of the contrast media (CM) volume used to creatinine clearance is one of the strongest predictors of CIN development following PCI.7,8 Therefore, to reduce the incidence of CIN, it is important to focus on strategies to reduce the volume of CM used, particularly in patients with chronic kidney disease (CKD).
Multiple studies and meta-analyses have shown that compared to coronary angiography alone, the use of intravascular imaging techniques for PCI guidance reduces the risk of cardiovascular death and adverse events.9–17 Optical coherence tomography (OCT) has superior resolution compared to intravascular ultrasound (IVUS).18,19 Optical coherence tomography techniques have been shown to more accurately assess vessel morphology, sizing, and complications following stenting compared to angiography or IVUS.19,20 This makes OCT a powerful tool for optimizing the outcomes of percutaneous coronary intervention, particularly in complex lesions.17,19
Optical coherence tomography is a high-resolution (10 μm) catheter-based intracoronary imaging technique. Similar to IVUS, OCT provides cross-sectional images of the vessel. However, instead of sound waves, OCT uses near-infrared light waves for tissue analysis, enabling visualization of coronary lesions with microscopic precision.21,22 Furthermore, red blood cells interfere with the propagation of infrared light; therefore, OCT requires displacement of the red blood cells from the vessel lumen during image acquisition to avoid artifacts that may limit good visualization of the coronary artery wall. 23 This result was previously achieved by relatively complex techniques, such as occluding the coronary lumen with an inflated balloon and simultaneously flushing the vessel with saline through the balloon catheter. Currently, non-occlusive techniques that involve flushing the coronary artery with CM to displace the blood pool during the vessel’s scan allow full vessel visualization without occlusion. 21 CM is nephrotoxic, and its use during OCT significantly adds to the total volume of CM used during PCI. Depending on the size of the vessel, 11–17 mL of contrast media is used during one OCT pullback.23,24 Consequently, the usage of OCT is limited in patients with renal impairment. 25 Normal saline is an alternative agent that could potentially be used for the clearance of red blood cells during OCT. It is not nephrotoxic, is cheap, and easy to use. However, normal saline is less viscous compared to CM and will require an intracoronary injection of larger volumes at higher rates. No protocol for normal saline injections in OCT imaging has been published to date. The aim of our study was to assess if it is possible to get good-quality images by using normal saline for OCT imaging, to establish recommendations for injection rate, and to evaluate the safety of this procedure.
Methods
This was a prospective single-center observational study performed in the Clinic of Cardiology at the Hospital of Lithuanian University of Health Sciences Kaunas Clinics between September 2022 and September 2023. The study protocol was reviewed and approved by the regional bioethics committee. The study was conducted in accordance with the Declaration of Helsinki and approved by the Kaunas Regional Biomedical Research Ethics Committee (protocol code BE-2-7, dated February 22, 2022).
Informed consent was obtained from all patients before enrollment in the study. The primary endpoint was to assess the efficacy of saline OCT in generating high-quality images in anatomically selected coronary arteries. The secondary endpoint was to create a protocol for saline injection into the coronary artery depending on the visual angiographic diameters of the vessel and to assess angiographic features that would be useful in identifying anatomical angiographic parameters to predict the suitability of the vessel to obtain good-quality OCT images. We enrolled patients for whom intravascular imaging-guided PCI was indicated. To be enrolled in the study, patients had to be ≥ 18 years old and willing to participate by signing an informed consent form. Patients with acute ST-elevation myocardial infarction were not eligible. Patients with specific angiographic findings that could affect the quality of imaging were not included: an angiographically small vessel <2.0 mm in diameter, an angiographically large or ectatic vessel >5.0 mm in diameter, ostial and left main lesions, very distal lesions, long lesions >45 mm in length, tortuous and severely calcified lesions. During the study period, 3585 PCI procedures were performed in our center. During this period, heparinized saline was used for all OCT imaging injections. All 32 patients were enrolled in the study. Before enrollment, patients had to meet all angiographical study inclusion criteria. For patients who did not meet angiographical study inclusion criteria, intravascular ultrasound was performed instead of OCT.
For the OCT-guided PCI procedure, coronary arteries were planned to be imaged at least twice: before PCI for vessel and atherosclerosis morphological assessment, and after PCI to assess stent expansion, apposition, and stenting-related complications.
Heparinized saline was injected into the coronary system for blood clearance during OCT image acquisition. All OCT pullbacks were performed using an automatic syringe injector (Medrad, Germany). The flushing injections might be performed manually or by using the automatic pump injector, as per the operator’s preference. 25 Manual injections have been widely used between operators due to the fast and easy flushing, which does not require any additional preparation. However, automatic pump injections require additional preparation and precise de-airing of the system; the parameters of automatic pump injections are more controlled and all changes can be exactly registered and monitored. Because of the need to monitor all injection parameters carefully, all injections were performed using an automatic pump injector. Due to the lower viscosity of saline, it had to be injected at a higher rate and higher volume compared to CM injections. For selective engagement of the coronary artery, additional extension catheters were available for use under the judgment of the operator (guiding extension catheter or “mother in child/5 in 6 F catheters”). Injections to the right coronary artery (RCA) and selective injections to the left anterior descending (LAD) and the left circumflex (LCX) arteries were done at a rate of 4 mL/s, and nonselective injections to the left coronary artery (LCA) at a rate of 5 mL/s. The operator could perform as many runs as necessary. The speed and volume of injection could be increased further at the operator’s discretion if pull-back imaging quality was not sufficient. Due to the poor viscosity of normal saline, when imaging longer lesions, two runs might be taken to assess the proximal and distal lesion/stent segments. Optical coherence tomography pullbacks last <3.0 s, and no CM is used for flushing; multiple repeated runs are considered to be benign for patients.
Recordings were made using the Ilumien Optis intravascular OCT imaging system (Abbott, Santa Clara, California) using a Dragonfly (Abbott, Santa Clara, California) imaging catheter at a speed of 20 mm/s. Heparinized saline was injected into the coronary system for blood clearance during OCT image acquisition. The guiding catheter was properly engaged before image acquisition. Any remaining CM was withdrawn from the guiding catheter before the controlled injection of heparinized saline. Before every OCT run, 100 μg of intracoronary nitroglycerine (NTG) was given to avoid catheter- or saline-induced coronary artery spasm. Electrocardiographic and hemodynamic changes were observed during pullback.
Recordings were analyzed at 1-mm intervals by one or two experienced operators. The assessment of imaging quality was based on imaging clarity, external elastic membrane (EEL) or lumen contour visibility, the system’s ability to perform automatic calculations, and the absence of artifacts or squiring. Optimal imaging (Oi) was defined as a completely clear vessel lumen and visible lumen contour or EEL for 360° of vessel circumference; no blood swirl in the run; and detailed coronary lesion and stent characteristics. Acceptable imaging (Ai) was defined as a visible lumen contour or EEL >270°; visible blood in the run, but not hampering the coronary lesion characteristics and minimum lumen area; a visible proximal and distal reference landing zone; visualization of stent strut apposition, stent edge dissection, and plaque prolapse. Unacceptable imaging (Ui) was defined as a visible lumen contour and/or EEL <270°; no diagnostic information could be obtained, largely because of poor clearance of blood. If one OCT run was not enough to get good-quality imaging along the entire length of the lesion, then the lesion could be divided into halves or thirds, and multiple runs could be performed to assess each region. A clear imaging field (CIF) was defined as a visible lumen contour >270°.
24
Adequately imaged vessel length was determined as the segment of recording containing all frames with a CIF and further categorized as continuous length or proportional length. A clinically usable run (CUR) was considered suitable for clinical use if >75% of the region of interest length was described as imaging of Oi or Ai quality. Figure 1 shows examples of saline OCT imaging from this study. Examples of saline OCT images of different quality. (A and B) optimal imaging quality; (C) acceptable imaging quality; (D) unacceptable imaging quality.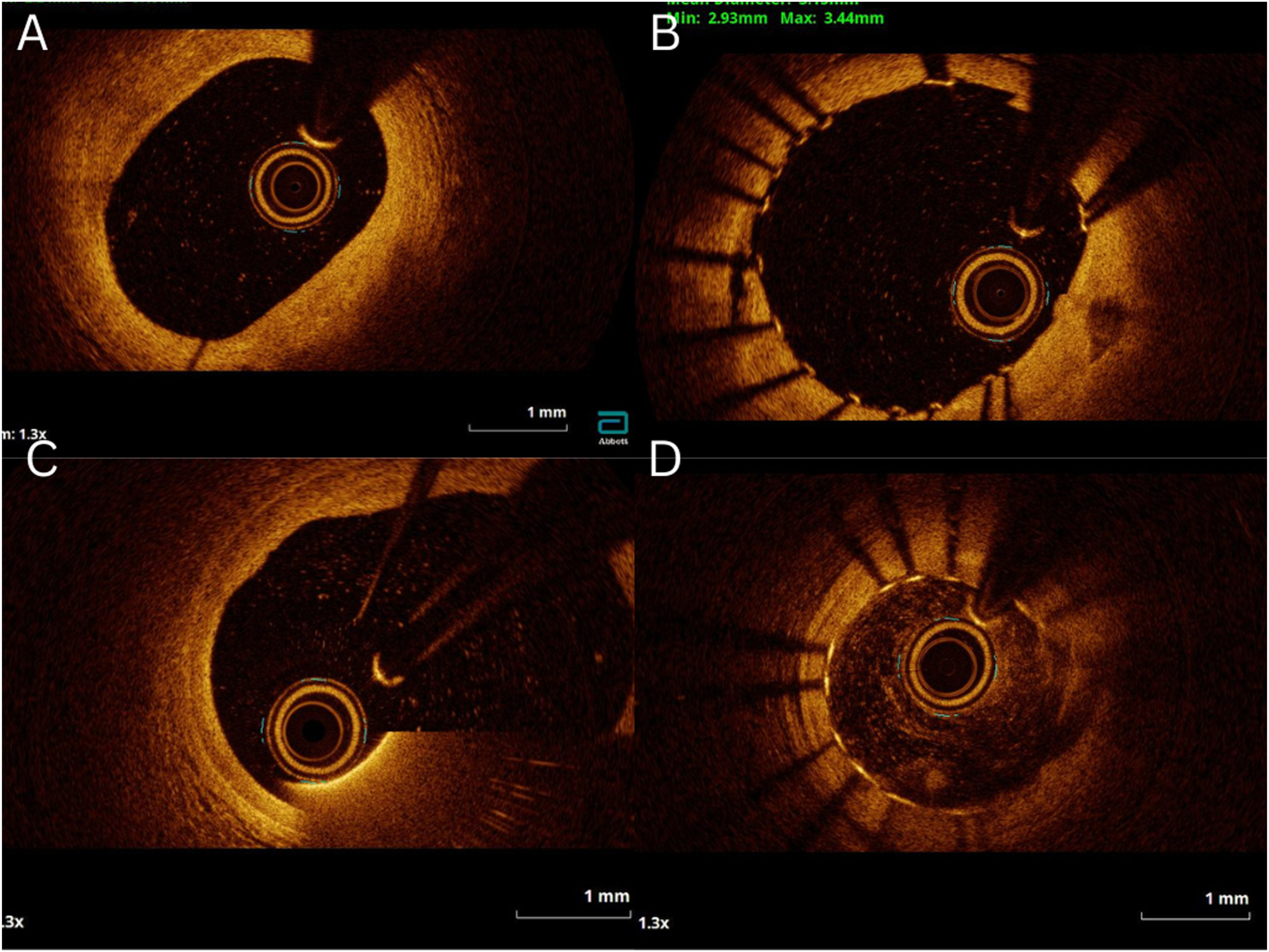
Periprocedural complications related to the usage of OCT imaging catheters and/or additional extension catheters were recorded. These included dynamic ECG changes (transient ST depression and transient ST elevation), transient chest pain, transient arrhythmias (bradycardia, ventricular tachycardia, and ventricular fibrillation), iatrogenic coronary artery dissection, no reflow after OCT imaging pullback, and coronary artery spasm. All mentioned complications were registered based on patients’ symptoms, ECG changes, and angiographic views.
Statistical analysis was carried out using SPSS version 24.0. Categorical variables were expressed as percentages, and continuous variables with a normal distribution were expressed as mean ± standard deviation. Continuous variables that did not meet normal distribution criteria were expressed as mean (median). The Shapiro-Wilk and Kolmogorov-Smirnov tests were used to assess data normality. The chi-square test was used to compare categorical variables. Numerical data were assessed using Pearson’s, χ2, and Fisher exact tests. To assess normally distributed data between two groups, the independent-sample t-test was employed; to assess non-normally distributed data, the Mann-Whitney test was used. For comparisons among more than two groups, one-way analysis of variance (ANOVA) and Kruskal-Wallis tests were used accordingly.
Results
A total of 32 patients, with a mean age of 70.38 ± 8.78 years, were enrolled in the study, of whom 62.5% were male, and 25.0% had a diagnosis of CKD. Among these patients, 47 different lesions were assessed both before and after PCI. The distribution of these lesions by location was as follows: 18 (38.3%) in the LAD, 14 (29.8%) in the LCX artery, and 15 (31.9%) in the RCA.
Detailed characteristics of patients’ demographics and assessed lesions.

Ai - acceptable imaging; AH - arterial hypertension; BMI - body mass index; CA - coronary artery; CABG - coronary artery bypass grafting; CKD - chronic kidney disease; CrCl - creatinine clearance; CUR – clinically usable run; DM - diabetes mellitus; LAD - left anterior descending; LCX - left circumflex; MI - myocardial infarction; NSTEMI - non-ST-segment MI; OCT - optical coherence tomography; Oi – optimal imaging; PCI - percutaneous coronary intervention; RCA - right coronary artery; UA - unstable angina.
Data of detailed analysis on pre-PCI and post-PCI OCT technique and imaging.
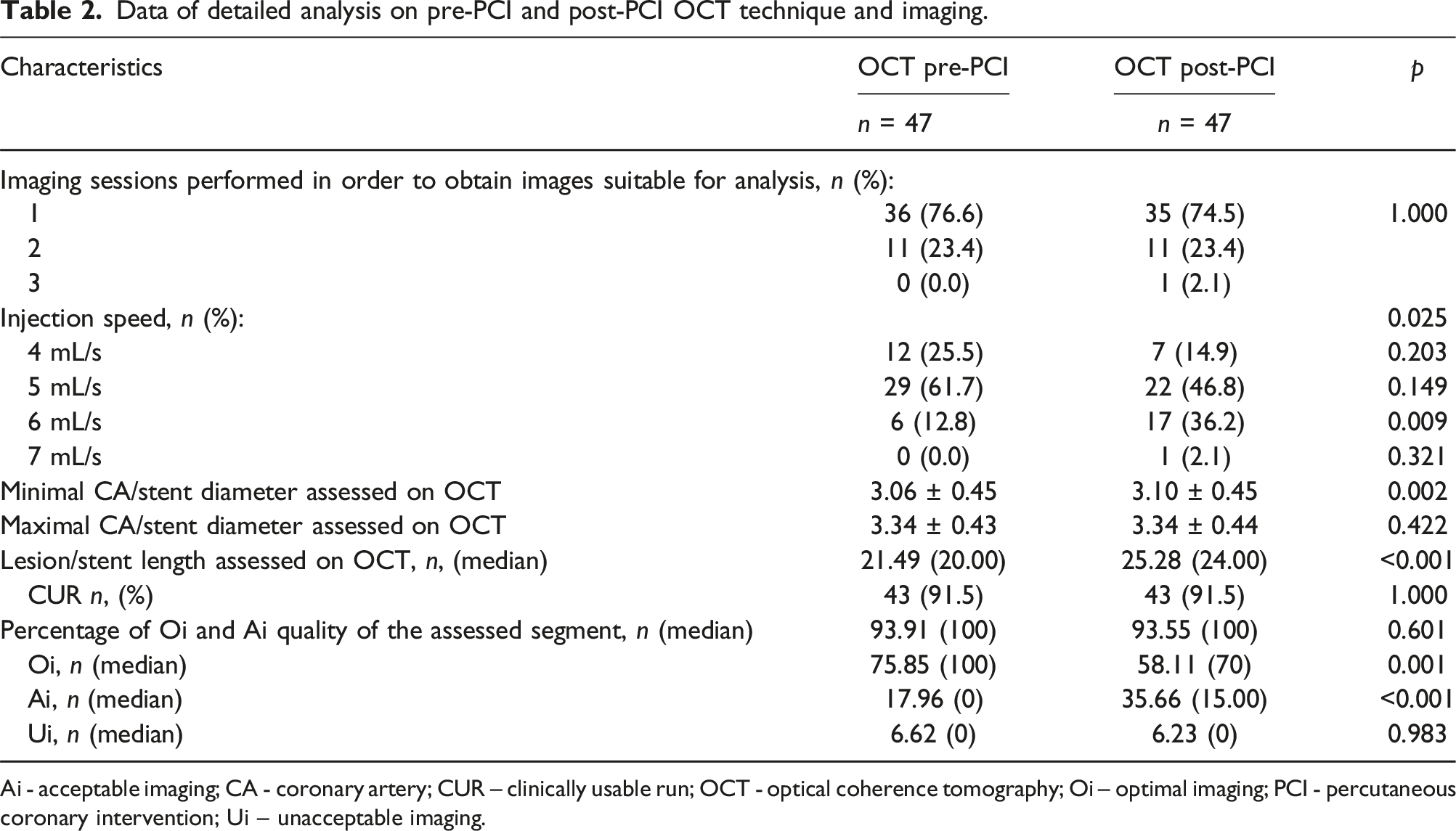
Ai - acceptable imaging; CA - coronary artery; CUR – clinically usable run; OCT - optical coherence tomography; Oi – optimal imaging; PCI - percutaneous coronary intervention; Ui – unacceptable imaging.
Upon analyzing the OCT assessment across different coronary arteries, there was no statistical significance in the number of runs required to obtain imaging suitable for inclusion in the analysis group. However, in the LCX artery, the percentage of two out of two suitable and included runs was lower compared to the LAD and RCA groups (6 (42.9%) versus 11 (61.1%) and 9 (60.0%), p > .05). Extension catheters to improve imaging quality were not needed in RCA arteries. The injection rate of 4 mL/s was used exclusively for selective engagement with extension and mother-in-child catheters; therefore, this rate was never used for imaging the RCA. A higher infusion rate, specifically 6 mL/s, was significantly more often used in the RCA (p = .033). While CUR imaging slightly varied across different types of arteries, no statistical significance was observed. Percentage of CUR imaging between different coronary arteries provided in Figure 2. Table 2 displays the percentage of CUR imaging across different coronary arteries, along with injection technique analysis and imaging analysis data in different coronary arteries. Further analysis and comparison of runs across different vessels are provided in Table 3. CUR imaging between different coronary arteries. LAD – left anterior descending, LCX – left circumflex, RCA – right coronary artery. Comparative analysis of LAD, LCX, and RCA imaging technique and imaging analysis data. Ai - acceptable imaging; CUR – clinically usable run; LAD - left anterior descending; LCX - left circumflex; OCT - optical coherence tomography; Oi – optimal imaging; PCI - percutaneous coronary intervention; RCA - right coronary artery; Ui – unacceptable imaging.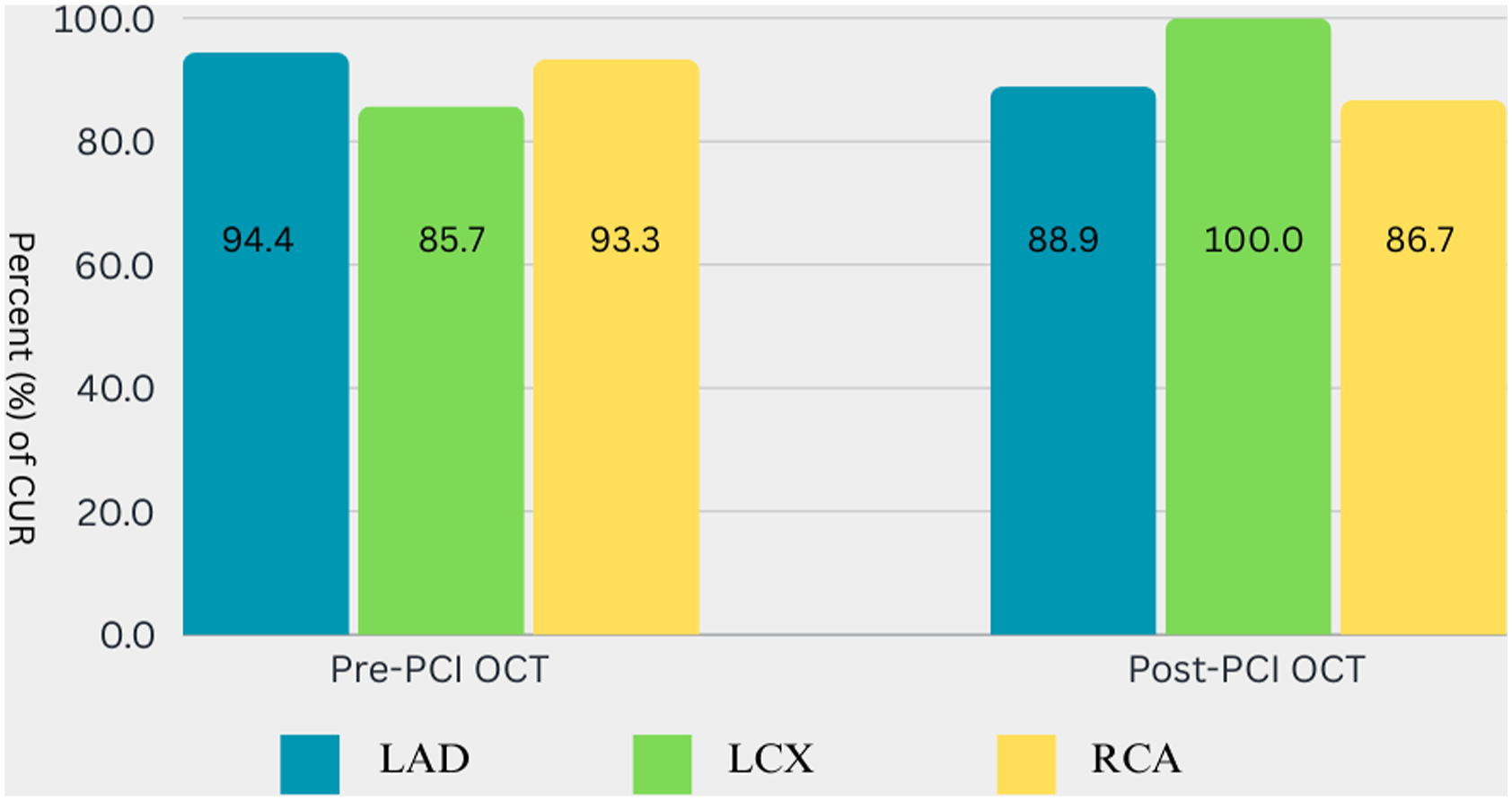

The speed of saline injection to the RCA pre-PCI varied from 5 to 6 mL/s, while to the LCA, it ranged from 4 to 6 mL/s; the speed of 4 mL/s was used as the starting speed for selective injections with extension catheters. We found that post-PCI imaging runs had to be performed with the same or higher injection volume compared to pre-PCI runs, with speeds varying between 4 and 7 mL/s. Changes of injections; speeds are provided in Figure 3. Alterations of post-PCI OCT injections speed increasement in comparison of pre-PCI OCT injection speed. OCT – optical coherence tomography; PCI - percutaneous coronary intervention.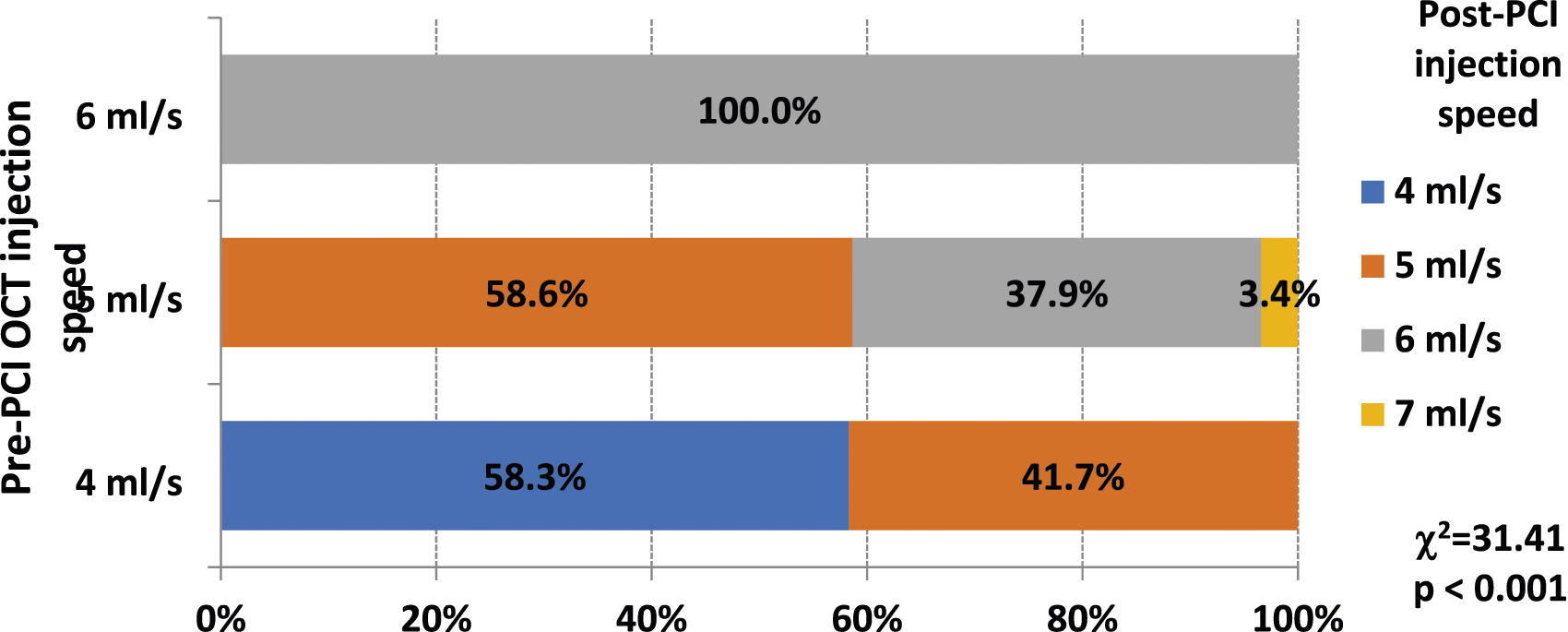
During all 117 saline OCT pullbacks, we observed two complications (1.7%): one complication (0.85%) was injection-related ventricular fibrillation, which was discontinued by electrical cardioversion, and the procedure was successfully completed; another (0.85%) was extension catheter-related artery dissection in the diseased segment, which was successfully stented. We did not register any transient ECG changes or episodes of chest pain.
Discussion
Despite the numerous benefits of OCT, its utilization in patients with impaired kidney function is limited due to the heightened risk of contrast-induced nephropathy (CIN).26–28 The objective of our study was to demonstrate that flushing techniques with normal saline can produce high-quality images, thereby reducing the need for contrast during OCT-guided procedures and broadening the application of this imaging modality. Our findings revealed that normal saline injections using an automatic pump at controlled volume and speed can yield adequate-quality images. Due to differences in viscosity between normal saline and contrast media (CM), normal saline must be injected at a higher speed and in a greater total volume. We provide recommendations for normal saline injection volumes and speeds that have not been previously published.
Optical coherence tomography provides visualization of the coronary artery microstructure at a resolution 10 times better than IVUS, allowing for microscopic assessment of artery structures such as edge dissections, tissue coverage of stent struts, and malposition, which cannot be adequately assessed by IVUS. 29 Consequently, OCT has become the second most commonly used intracoronary imaging technique, with increasing utilization over the years. However, despite its advantages, OCT imaging requires flush media for blood clearance to achieve visualization with near-infrared light, typically utilizing contrast media (CM).29–32 This reliance on contrast media poses a significant limitation to OCT usage, particularly for patients with underlying CKD and a higher risk of contrast-induced nephropathy (CIN).25,29,32
Comparing different studies, a trend of decreasing contrast media usage for OCT procedures over time can be observed. For instance, in the iSIGHT study, the total contrast volume used in the OCT group was significantly higher than in the angiography group (94.10 ± 40.54 mL [27.63 ± 7.39 mL due to OCT] vs 72.3 ± 35.8 mL, p = .018), but not significantly different from the contrast volume used in the IVUS group (82.1 ± 41.3 mL, p = .277). An average of 12.85 ± 3.05 mL of contrast was used per OCT run, with no significant differences observed between pre-PCI and post-PCI runs (12.44 ± 2.81 vs 13.0 ± 3.11 mL, p = .218). Furthermore, OCT runs in the left coronary system required more contrast than imaging of the right coronary artery (14.45 ± 2.52 vs 10.62 ± 2.21 mL, p < .001). 33 In comparison with other studies, the iSIGHT study demonstrated a notably smaller average contrast volume of 94 mL for OCT-guided PCI, significantly less than that used in the ILUMIEN III (222 mL) 34 and OPINION (164 mL) 35 studies. Moreover, the mean contrast volume (94 mL) used in the iSIGHT study for OCT-guided PCI cases was nearly two times smaller than that used in the angiography-guided PCI of ILUMIEN III (183 mL) 34 and much smaller than that used in the IVUS arm of OPINION (138 mL) , 35 even considering the higher complexity of lesions treated in the iSIGHT study. 33
In an effort to reduce the volume of contrast media (CM) used for OCT-guided procedures, several alternative flushing agents have been investigated in vivo. 36 Low-molecular-weight dextran, with a viscosity very similar to contrast, has been considered as a potential flushing agent for achieving high-quality imaging.37,38 However, dextran poses a risk of renal injury as it is excreted from the kidneys unmetabolized, similar to contrast.38,39 Carbon dioxide, previously utilized for peripheral angiography, 40 is not employed for intracoronary imaging due to concerns about inducing myocardial ischemia. Clear crystalloid solutions, such as Ringer’s solution, have been used in older occlusive OCT pullback techniques, demonstrating effective imaging quality.20,41 More recently, three small studies have investigated normal saline as a suitable flushing medium for OCT imaging.42–44 However, these studies utilized manual saline injections for imaging, lacking a clear protocol for achieving high-quality images. Nevertheless, no significant complications were documented with this imaging technique. Nalin et al. conducted a study with 27 patients undergoing intracoronary OCT imaging using manual injections of normal saline, but only 61% of the images were deemed suitable for analysis. 42 Two other studies compared vessel measurements between OCT imaging with contrast media and normal saline, finding no significant differences in vessel diameter measurements between the two techniques.43,44
Our study demonstrated that normal saline (NS) might serve as an effective, non-nephrotoxic alternative as a flushing material for OCT imaging. However, the lower rate of high-quality Oi images after PCI may be attributed to the increased lumen area and imaging lesion length post-procedure. This challenge was easily overcome by adjusting the injection speed in post-PCI imaging pullbacks. For OCT imaging with contrast media (CM), the contrast flush rate is typically set at 4 mL/s for a total volume of 14–16 mL for the left coronary artery (LCA) and 3 mL/s for a total volume of 12–14 mL for the right coronary artery (RCA). 25 NS, having a lower viscosity compared to contrast, allows for higher injection speed limits for flushing injections. The NS flushing injection speed depends on the length of the imaging segment and the size of the vessel; however, even repeated runs did not have any significant impact on the patient.
Repeated injections of NS may have a negative cumulative effect on patients with reduced left ventricle ejection fraction (LVEF) and/or heart failure (HF) symptoms. The maximal flushing speed of 7 mL/s was used only once in our study, with 6 mL/s deemed a safe and effective speed limit. During standard OCT imaging involving two pullbacks (pre and post PCI), an average of up to 40 mL may be injected. However, if multiple imaging pullback runs are repeated, the amount of NS can exceed 100 mL or more. This increase in fluid volume may have a negative effect on patients with reduced LVEF and HF symptoms. Although our study did not specifically analyze patients with LV EF as a subgroup, the potential negative impact on their HF status should be considered if multiple repeated pullbacks with NS are performed.
Complications related to OCT are rare (0.6%) and typically self-limiting. They include transient chest pain and QRS widening/ST-depression/elevation, ventricular fibrillation due to catheter occlusion and/or deep guide catheter intubation, air embolism, and vessel dissection.36,45–47 Ventricular fibrillation is the most common arrhythmia, with an incidence as high as 1.1%. 47 A recent study by Terada et al. revealed that contrast volume was the only independent predictor of ventricular fibrillation, with a cut-off value of 19.2 mL for predicting arrhythmia (area under the curve, 0.713, p < .001; diagnostic accuracy, 87.1%). 48 In our study, we demonstrated that our proposed protocol for normal saline injection during the OCT pullback is safe. We did not register any procedure-related major complications, even with high volume and saline injection rates.
Obtaining good-quality images often necessitates elective target vessel engagement with an extension guiding catheter or a 5 to 6 French “mother-in-child” catheter to achieve adequate clearance, particularly in lesions with a larger diameter. While our suggested protocol has shown effectiveness and safety, larger-scale randomized clinical trials are needed to confirm the findings of our study.
Study limitations
The major limitation of this study is the absence of a control group, in which patients undergoing OCT would have received traditional flushing with contrast media (CM). This comparison would have allowed for demonstrating the potential benefits of the method, such as reduced CM volume used for the procedure and a possible decrease in the development of contrast-induced nephropathy (CIN). The lack of a control group diminishes the efficacy of assessing the safety of the saline OCT method. However, we compared the complication rate of OCT with saline to that of traditional OCT methods using CM, as reported in published clinical trials. For further evaluation of the safety and efficacy of our proposed method, additional randomized studies with a larger sample size are necessary.
Conclusions
This study demonstrated that heparinized saline injections for OCT imaging are effective in generating good-quality OCT images suitable for clinical use and assessment. Heparinized saline flushing injections at rates of 4 and 7 mL/s were found to be safe and well tolerated by patients, with no significant complications observed. However, the results of this pilot study need to be confirmed in multicenter randomized controlled trials.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
