Abstract
Introduction
Bleeding and thrombotic complications are common in extracorporeal membrane oxygenation (ECMO) patients and are associated with increased mortality and morbidity. The optimal anticoagulation monitoring protocol in these patients is unknown. This study aims to compare the incidence of thrombotic and hemorrhagic complications before and after a protocol change. In addition, the association between hemostatic complications, coagulation tests and risk factors is evaluated.
Methods
This is a retrospective single center cohort study of adult ECMO patients. We collected demographics, ECMO parameters and coagulation test results. Outcomes of the aPTT guided and multimodal protocol, including aPTT, anti-Xa assay and rotational thromboelastometry were compared and the association between coagulation tests, risk factors and hemostatic complications was determined using a logistic regression analysis for repeated measurements.
Results
In total, 250 patients were included, 138 in the aPTT protocol and 112 in the multimodal protocol. The incidence of thrombosis (aPTT: 14%; multimodal: 12%) and bleeding (aPTT: 36%; multimodal: 40%), did not significantly differ between protocols. In the aPTT guided protocol, the aPTT was associated with thrombosis (Odds Ratio [OR] 1.015; 95% confidence interval [CI] 1.004-1.027). In both protocols, surgical interventions were risk factors for bleeding and thrombotic complications (aPTT: OR 93.2, CI 39.9-217.6; multimodal OR 17.5, CI 6.5-46.9).
Discussion
The incidence of hemostatic complications was similar between both protocols and surgical interventions were a risk factor for hemostatic complications. Results from this study help to elucidate the role of coagulation tests and risk factors in predicting hemostatic complications in patients undergoing ECMO support.
Keywords
Introduction
Extracorporeal membrane oxygenation (ECMO) is a vital treatment option for patients with severe cardiopulmonary failure. 1 Unfortunately, hemostatic complications, including thrombotic and hemorrhagic complications, occur in one-third of patients and are associated with mortality and morbidity. 2
The etiology of hemostatic complications is multifactorial and includes the hypercoagulable state of the critically ill patient, in combination with endothelial damage, an induced systemic inflammatory response, variable blood flow rates and the blood-biomaterial interface, leading to thrombosis.3–6 To prevent thrombosis, unfractionated heparin (UFH) is administered. Currently, UFH is recommended by the Extracorporeal Life Support Organization (ELSO) and most frequently used due to its short half-life and the availability of an antagonist. 7 Unfortunately, UFH increases the risk of bleeding complications. Prevention of both bleeding and thrombotic complications is challenging due to the delicate hemostatic balance. Therefore, a reliable diagnostic strategy to monitor this balance is imperative.
Generally used standard coagulation tests include activated partial thromboplastin time (aPTT), anti-factor Xa assays (aXa) and activated clotting time (ACT). 8 In contrast to these standard coagulation tests, alternative coagulation tests such as thromboelastography (TEG) or rotational thromboelastometry (ROTEM) provide a representation of the whole clotting process within 30 min, which might be an advantage in monitoring coagulation in ECMO patients. 9 Several studies have investigated the association between laboratory coagulation parameters and hemostatic state.10–12 Despite these efforts, the optimal method to monitor the hemostatic state during ECMO is unknown. As a consequence, heterogeneity exists in anticoagulation monitoring protocols among ECMO centers worldwide.8,13
In our Intensive Care Unit (ICU) in Rotterdam, The Netherlands, the coagulation monitoring strategy was changed from an aPTT guided to a multimodal coagulation monitoring protocol in 2013 after two cases of fatal pump thrombosis. In this study, we aim to compare the incidence of thrombotic and hemorrhagic complications between two coagulation management protocols in adult ECMO patients. Furthermore, we aim to investigate the association between coagulation test results, risk factors and hemostatic complications in both protocols.
Methods
Study design and patients
This is a retrospective single center cohort study of all adult ECMO patients admitted to the ICU and cardiac care unit (CCU) of the Erasmus Medical Centre (MC) in Rotterdam, The Netherlands between November 2004 and October 2020. This study has been approved by the Erasmus MC Medical Ethics Committee (MEC-2022-0581) and patient consent was waived.
All patients over 17 years old undergoing veno-venous (VV) or veno-arterial (VA) ECMO support with systemic UFH therapy and available coagulation test results were eligible for this study. Patients who received extracorporeal cardiopulmonary resuscitation (E-CPR), defined as initiation of ECMO during cardiac arrest or within 20 min after achieving spontaneous circulation were excluded due to the impact of circulatory arrest on hemostasis. 14 Patients with COVID-19 infection, ECMO runs shorter than 72 h, roller-pump ECMO runs and second ECMO runs were excluded. In addition, patients treated in the transition period from November 2013 to February 2015 were excluded due to non-coherence to the new diagnostic strategy.
Extracorporeal membrane oxygenation circuit and coagulation strategy
All patients were supported with the integrated Cardiohelp System (Maquet Cardiopulmonary GmbH, Rastatt Germany) or the iLA-activve ECMO system with a diagonal DP3 pump (Xenios AG, Heilbronn Germany). The Cardiohelp System was used in combination with HLS cannulae with Bioline (heparin-albumin) coating. The iLA-activve system kits are coated with a multi-layer coating made of heparin and albumin (x.ellence ® coating). Before ECMO cannulation, the circuits were primed with crystalloid solution and 1.000 IU of UFH. During ECMO cannulation, a single dose of 5.000 IU heparin was administered. Cannulation of VA-ECMO patients was performed by the thoracic surgeon or interventional cardiologist. In VA-ECMO, cannulation using the femoral vein and artery were the most common cannulation strategies. In all cases, the patient was provided with an antegrade femoral artery cannula to guarantee the perfusion of the leg distally to the arterial cannula. Cannulation of VV-ECMO was performed with the Seldinger technique. 15 In VV-ECMO, femoral vein - right internal jugular was the most common configuration. After cannulation, a starting dose of 20.000 IU/24 h UFH was administered.
aPTT only protocol
Between 2004 and 2013, the UFH dose was monitored using aPTT measurements every 6 hours, with an aPTT target between 45 and 65 s for all patients. When aPTT was below 45 s, the heparin dose would be raised with 100 IU/hr. In case of an aPTT value above 65 s, the heparin dose would be lowered with 100IU/hr. Additionally, fibrinogen, platelet count and the International Normalized Ratio (INR) were checked once daily. Plasma was administered in case of fibrinogen levels below 1 g/L and a platelet transfusion was considered in patients with an absolute platelet count below 50 × 109/L or with significant bleeding in conjunction with a platelet count below 100 × 109/L. Red Blood Cell (RBC) transfusion was indicated when hemoglobin levels dropped below 9.6 g/dL.
Multimodal protocol
In 2013, the coagulation monitoring protocol was changed to the multimodal protocol. This protocol incuded the aPTT, aXa and ROTEM. In every patient, the aPTT and ROTEM tests were performed daily during ECMO support. In case of clinical suspicion or proven thrombotic or bleeding event, aPTT, aXa and ROTEM were performed more intensively up to four times a day. At ECMO initiation, target ranges for aPTT were between 55 and 65 s. Coagulation target ranges were altered based on discretion of the attending physician and were increased to 65-80 s in case of high risk of thrombosis or ECMO flow under 2 L per minute and decreased to 40-55 s in patients prone to bleeding, for example after cardiac surgery. The target range of aXa was between 0.3 and 0.7 U/ml and the targeted difference of clotting time (CT) in the INTEM and HEPTEM (dCT INTEM-HEPTEM) was 80 s. In case of a bleeding complication, target ranges of aPTT were lowered to 40-55 s for all risk groups, aXa to 0.2-0.4 U/ml and dCT INTEM-HEPTEM between 40 and 60 s. In case of a thrombotic complication, the aPTT, aXa and dCT INTEM-HEPTEM target levels were increased to 65-80 s, 0.6-1.0 U/ml and 120-140 s, respectively. To reach this target in dCT INTEM-HEPTEM, the infused amount of heparine was raised or reduced by 20% from baseline when the measured dCT was too low or too high.
In both protocols, an ECMO circuit change was indicated in patients with a thrombus in the circuit larger than 1 cm, steep increase of trans oxygenator pressures, D-dimers above 30 g/L or free hemoglobin above 20 µmol/L for more than two consecutive days. Over the entire time period analysed, the ECMO tubing system was checked for thrombi three times a day optically by both the ICU nurse and doctor.
Data collection
Collected data included demographics (age, weight, gender) and ECMO parameters (diagnosis type, indication for ECMO support, number of support days, successful weaning from ECMO, survival to discharge) as well as hemostatic data (aPTT, INR, aXa levels, ROTEM results, fibrinogen levels and platelet count). The CT and the Maximum Clot Firmness (MCF) of intrinsic (INTEM) and extrinsic (EXTEM) coagulation pathways, INTEM with heparinase (HEPTEM) and the MCF of fibrin formation (FIBTEM) and dCT INTEM-HEPTEM were included. Only data from the first 14 days of ECMO support were collected as patients are commonly supported less than 14 days in our institution. In case of a bleeding or thrombotic complication, the hemostatic laboratory results of the 24 h period before the detection of the complication were analyzed and compared to the data of the non-complication days of the same subject. Surgical interventions following bleeding complications within 12 h were excluded.
Coagulation tests
All coagulation tests were performed in the hemostasis laboratory of the Erasmus MC (Rotterdam, The Netherlands). For all coagulation tests, blood was collected in 3.2% trisodium citrate tubes (citrate to blood ratio 1/9) and were centrifuged for 15 min at 1700 g. ROTEM results were obtained using the ROTEM delta (Werfen) with reagents from Werfen. aPTT (Actin FS, Siemens), PT-INR (Thromborel™ S Reagent, Siemens) and aXa (Liquid Heparin testkit; HemosIL™, (IL)) were performed using the Sysmex CS5100 coagulation analyzer (Siemens Healthcare Diagnostics B.V.).
Definitions
Major bleeding were defined according to the ELSO definition. 16 In short, any clinically overt bleeding with an associated hemoglobin fall of at least 2 g/dl in a 24 h period, bleeding more than 20 mL/kg over a period of 24 h or bleeding with a transfusion requirement of one or more 10 mL/kg packed RBCs over that same period of time was defined as major bleeding. Furthermore, intracranial bleeding and any blood loss requiring surgical intervention were considered major bleeding complications. Bleeding events that did not meet the ELSO definition were defined as minor bleeding. Any thrombotic complication leading to ECMO circuit change, cardiac thrombosis, venous thromboembolism, pulmonary embolism, bowel ischemia or cerebral infarction was considered a thrombotic event. Surgical interventions were defined as any invasive procedure with penetration of the skin, excluding all percutaneous procedures such as placement of intravenous lines or endovascular procedures. Sepsis was defined as a life-threatening organ dysfunction caused by a dysregulated host response to infection. 17
Statistical analysis
Data are represented as mean ± standard deviation or as median with interquartile range (IQR). A univariate logistic regression analysis was used to assess the association between coagulation test results and bleeding and thrombotic events. Separate GEE models have been generated for each ROTEM variable (INTEM, EXTEM and HEPTEM: CT and MCF and FIBTEM MCF) with major bleeding as the dependent variable. As the subjects in the aPTT protocol were not treated with a ROTEM guided strategy, the data regarding the ROTEM analysis only applied to the subjects included in the multimodal protocol. Separate analyses were performed for bleeding and thrombosis. All parameters with a significant p value were used in a multivariate logistic regression analysis for repeated measurements based on generalized estimating equations (GEE). The independent variables in these analyses were the results of ROTEM (CT and MCF of INTEM, EXTEM and HEPTEM and the MCF of FIBTEM and delta INTEM-HEPTEM CT and MCF), day of ECMO support (coded as a continuous variable), age, sepsis, heparin dose, ECMO duration and surgical interventions. A similar approach was used for thrombosis, using thrombosis as the dependent variable in the uni- and multivariate analysis. An independent working correlation matrix was used in the GEE models to account for the within-subject correlations. The association between aPTT and aXa and hemostatic complications was investigated in the same manner as ROTEM components. The Chi-square test was used to calculate the difference in survival, sex, pre-ECMO surgery, ECMO configuration and ECMO indication between protocols. The Mann-Whitney test was used to calculate the difference in age, body mass index (BMI), duration of ECMO support, Survival After Veno-Arterial ECMO (SAVE) and Respiratory ECMO Survival Predication (RESP) scores between protocols and between patients with and without hemostatic complications.
A p-value of 0.05 was considered the limit of significance in all tests. A Bonferroni correction for multiple testing was applied for all analysis with coagulation tests, resulting in a significance level of αaltered = 0.05/9 = 0.006. All statistical analysis were performed using SPSS V. 24.0 (IBM, Chicago, Illinois, USA).
Results
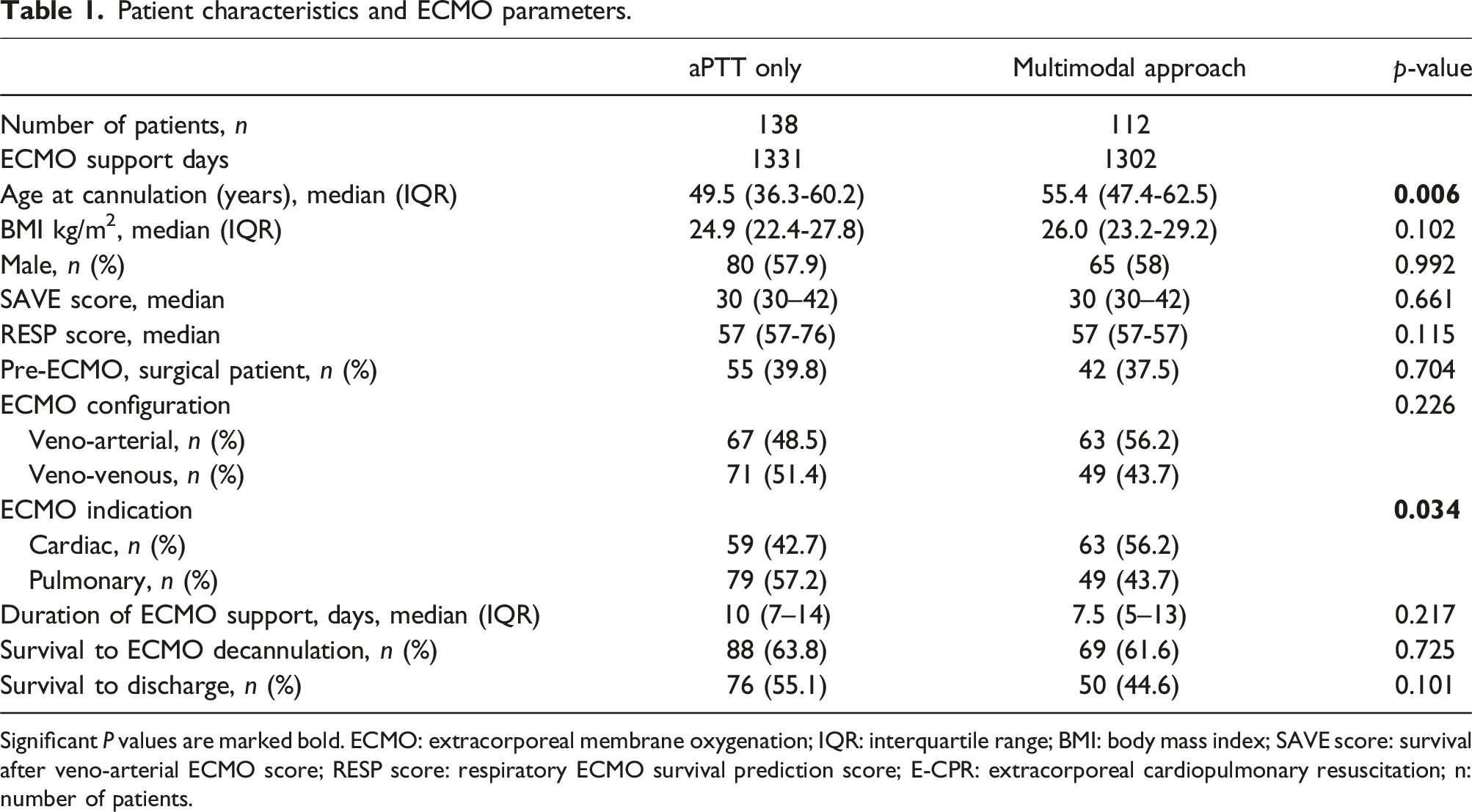
Patient characteristics
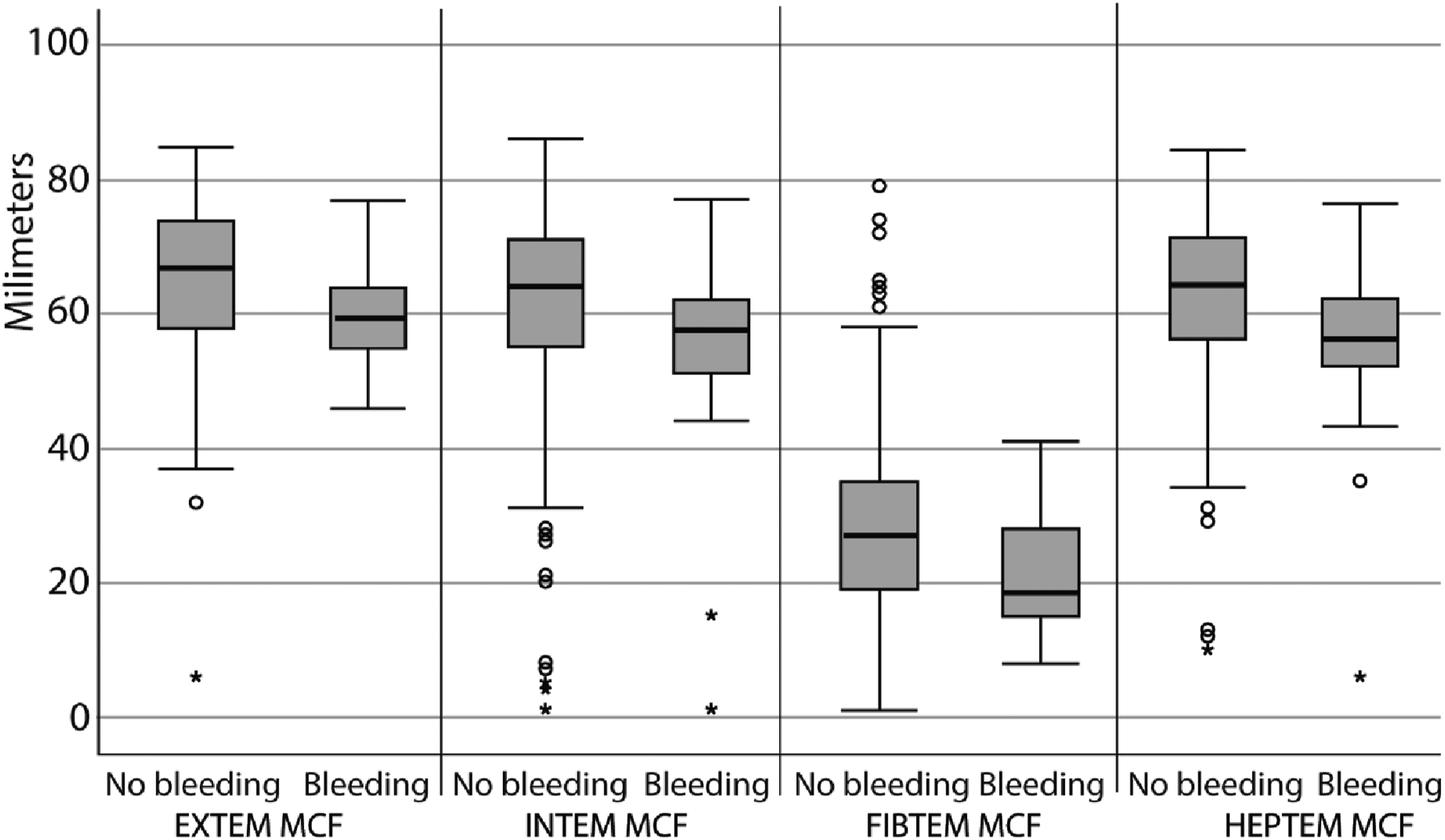
Between 2004 and 2020, 501 patients were treated with ECMO therapy in the Erasmus MC and 250 patients were included (Figure 1). Demographics and ECMO parameters are described in Table 1. Patients in the multimodal protocol had a significantly higher age at ECMO initiation (aPTT: 49.5 years; multimodal 55.4 years, p = .006) and had significantly more frequently a cardiac ECMO indication (aPTT 43%, multimodal 56%, p = .034). (Table 1) The iLA-active ECMO system was used in three patients from the aPTT guided protocol and in two patients from the multimodal protocol Figure 2. Flowchart of patient inclusion. ECMO: extracorporeal membrane oxygenation; E-CPR: extracorporeal cardiopulmonary resuscitation; n: number of patients. Patient characteristics and ECMO parameters. Significant P values are marked bold. ECMO: extracorporeal membrane oxygenation; IQR: interquartile range; BMI: body mass index; SAVE score: survival after veno-arterial ECMO score; RESP score: respiratory ECMO survival prediction score; E-CPR: extracorporeal cardiopulmonary resuscitation; n: number of patients. Mean ROTEM clotting times before bleeding events against mean ROTEM clotting times in non-bleeding patients. MCF: maximum clot firmness; EXTEM: Extrinsically activated thromboelastometry; INTEM: Intrinsically activated thromboelastometry; FIBTEM: Fibrin-based extrinsically activated thromboelastometry; HEPTEM: Intrinsically activated thromboelastometry with heparinase. The univariate analysis shows a decrease of EXTEM MCF, INTEM MCF, FIBTEM MCF and HEPTEM MCF in the patients with a bleeding complication. This significantly increased risk of hemorrhage was no longer seen in the multivariate analysis.
Incidence of hemostatic complications
Overview of hemostatic complications.
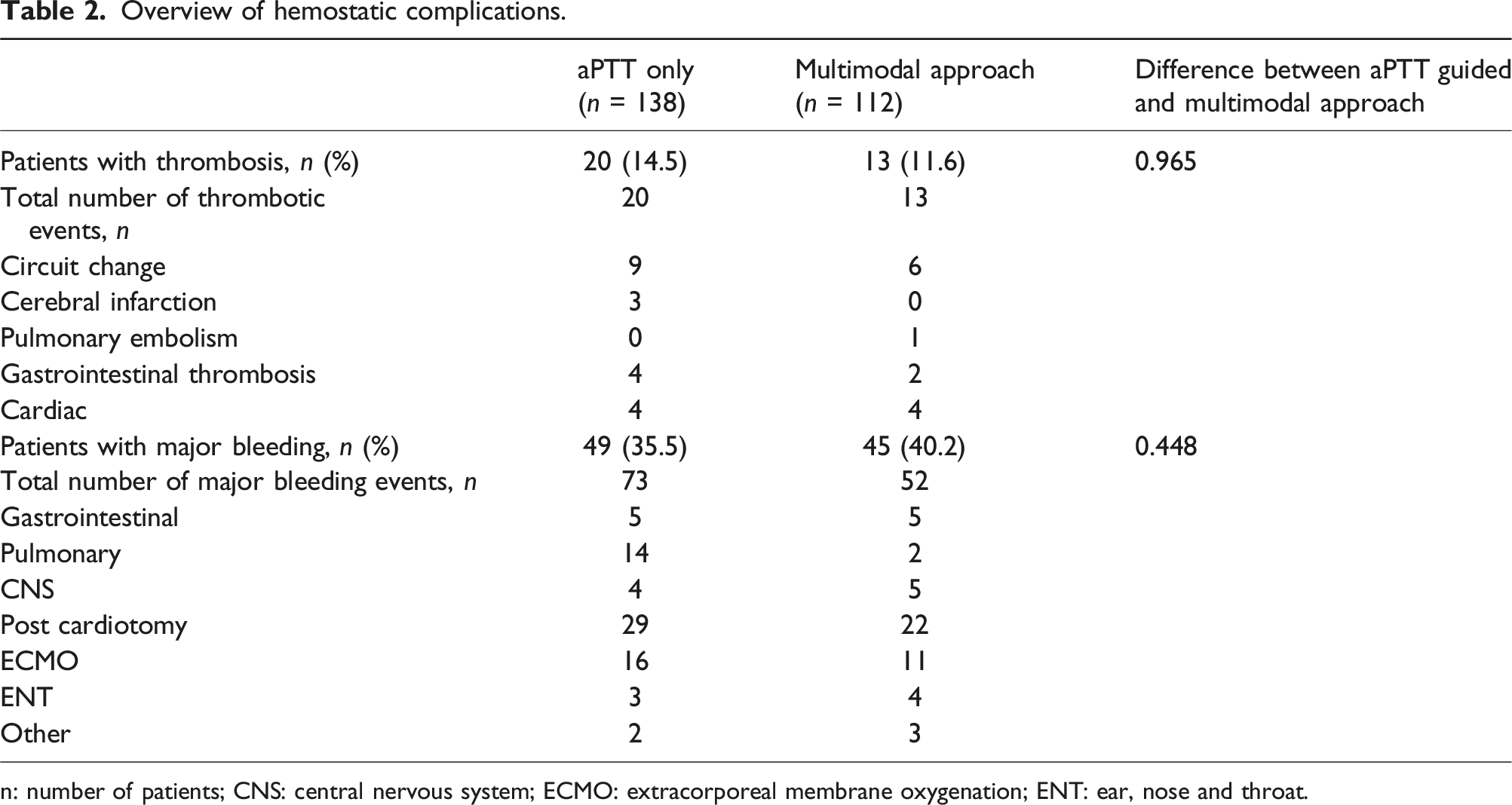
n: number of patients; CNS: central nervous system; ECMO: extracorporeal membrane oxygenation; ENT: ear, nose and throat.
Association between thrombotic complications, coagulation tests and risk factors
Multivariate analyses of thrombotic complications in aPTT guided versus multimodal approach of anticoagulation.
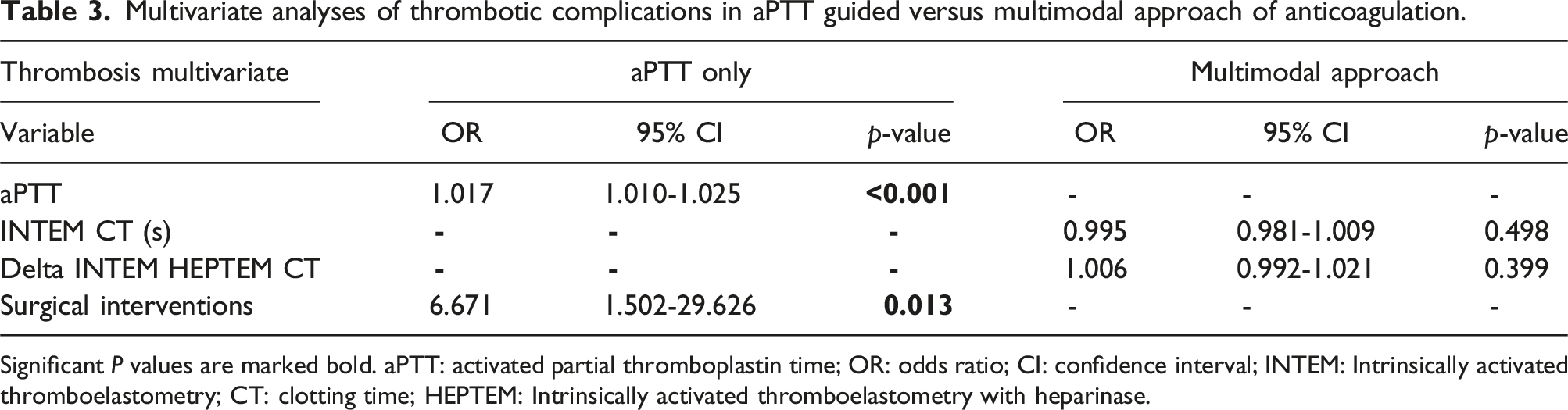
Significant P values are marked bold. aPTT: activated partial thromboplastin time; OR: odds ratio; CI: confidence interval; INTEM: Intrinsically activated thromboelastometry; CT: clotting time; HEPTEM: Intrinsically activated thromboelastometry with heparinase.
Association between bleeding complications, coagulation tests and risk factors
Multivariate analyses of major bleeding complications in aPTT guided versus multimodal approach of anticoagulation.M clot firmness, aPTT activated partical thromboplastin time, CI confidence interval, OR odds ratio, mm mm.
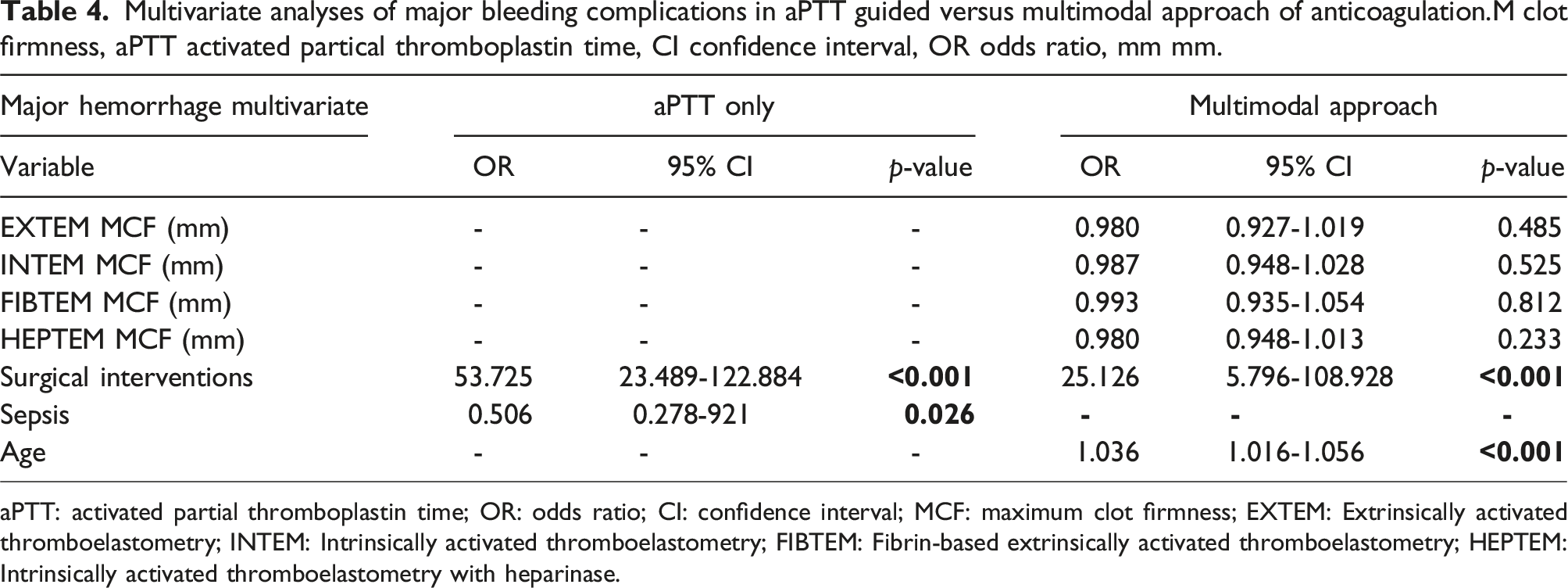
aPTT: activated partial thromboplastin time; OR: odds ratio; CI: confidence interval; MCF: maximum clot firmness; EXTEM: Extrinsically activated thromboelastometry; INTEM: Intrinsically activated thromboelastometry; FIBTEM: Fibrin-based extrinsically activated thromboelastometry; HEPTEM: Intrinsically activated thromboelastometry with heparinase.
Discussion
In this cohort, hemostatic complications developed in half of patients undergoing ECMO support. The incidence of these complications did not significantly differ between protocols. In the aPTT only protocol, the aPTT was associated with thrombotic complications. None of the coagulation parameters was significantly associated with increased bleeding risk in either protocol. Surgical interventions were a risk factor for hemostatic complications in both protocols.
Incidence of hemostatic complications
Half of patients suffered from a hemostatic complication in this cohort. This incidence falls within the incidence range described in literature.2,18–23 The incidence of hemostatic complications did not differ between protocols. Two similar studies in 100 and 123 ECMO patients also found no difference in the incidence of hemostatic complications after a protocol change.24,25 The majority of thrombotic complications were ECMO circuit clots leading to an ECMO circuit change, similar to previous studies.20,26 In addition, in accordance with the present results, previous studies have demonstrated that the majority of bleeding events developed on surgical and ECMO cannulation sites.2,21
Association between hemostatic complications and coagulation tests
The decrease in aPTT was associated with thrombosis in the aPTT protocol. Contradictory results about the association between the aPTT and thrombosis have been described in literature. Two retrospective studies in 63 and 172 ECMO patients revealed that the mean aPTT was an independent risk factor for thrombosis.27,28 However, another retrospective study found no correlation between daily aPTT measures and thrombotic events in 265 ECMO patients. 29 Contrary to our expectations, this study did not find an association between the aPTT and bleeding complications. In literature, several retrospective cohort studies reported an association between a prolonged aPTT and the risk of hemorrhagic events.18,21–23,30,31 Strikingly, a switch from an ACT based to an aPTT based protocol did not affect the incidence of hemostatic complications in two retrospective cohort studies.32,33 In the Erasmus medical center, an elevated aPTT was treated by decreasing the UFH dose immediately to prevent bleeding complications. The absence of an association between aPTT and bleeding complications might reflect the beneficial effect of this policy. However, the optimal aPTT target range and the correlation between aPTT and bleeding risk in ECMO patients remain unclear. 33
In the current study, no ROTEM parameter was associated with bleeding or thrombotic complications in the multivariate analyses. Conflicting findings regarding the association between viscoelastic tests and hemostatic complications have been described. First of all, Laine et al. described an association between a decreased MCF and bleeding complications in 24 ECMO patients. No thrombotic complications occurred. 34 In addition, Hellmann et al. showed in a cohort of 57 ECMO patients that patients with bleeding complications had a significantly prolonged INTEM CT35,36 Furthermore, two other studies described no significant difference in the incidence of bleeding and thrombotic complications between an aPTT guided and TEG based protocol.24,26 These conflicting results might be explained by the timing of the ROTEM analysis in relation to the dose change of heparin. Because of the retrospective nature of our study, we have only analysed the ROTEM parameters known in the 24 h before a hemostatic event. The adjustment of heparin dose in IU/hour and extra intravenous pushes in response to these parameters could not be taken in consideration. Futhermore, correction of out of range ROTEM coagulation times with coagulation factors or transfusion of fresh frozen plasma and thrombocytes was not looked into.
The aXa was not significantly associated with the risk of thrombotic or bleeding complications. A possible explanation for this might be that, although aXa reflects the heparin effect, it does not mirror the overall hemostatic balance in the patient. In contrast to our study, two retrospectice cohort studies including 22 and 24 ECMO patients found an association between low aXa levels and the risk of thrombotic complications.23,36 The aXa only might be a too specific test to monitor the complex hemostatic balance in ECMO patients and a combination of aXa and other coagulation tests may be preferable to a single test protocol.24,37 In addition, the aXa was only performed on discretion of the attending physician. As a consequence, selection bias may have played a role as well.
In the current study, multimodal monitoring did not decrease the number of hemostatic complications. In addition, none of the coagulation tests used in the multimodal approach were associated individually with hemostatic complications. This demonstrates the ongoing challenge in predicting hemostatic complications using coagulation tests. The optimal heparin monitoring strategy in ECMO patients remains to be elucidated. Large prospective studies are needed to determine the role of coagulation tests in the prediction of hemostatic complications in ECMO patients.
Risk factors for bleeding and thrombotic complications
In this cohort, surgical interventions were associated with increased risk of hemostatic complications. This finding underlines the hemostatic challenges in post surgical ECMO patients. In general, UFH treatment is often decreased or temporarily stopped around surgical interventions to decrease the post surgical bleeding risk. However, this increases the risk of thrombotic complications. In literature, a retrospective study in 149 ECMO runs described that surgical interventions increased the risk of bleeding complications. 22 In another retrospective study including 243 ECMO patients, postcardiotomy VA-ECMO was a risk factor for bleeding complications, as well. However, surgical interventions were not specifically identified as a risk factor. 38
Increasing age at ECMO initiation was another risk factor for bleeding complications in the multimodal protocol. This was surprising since several other retrospective studies in ECMO patients did not identify increasing age as a risk factor for bleeding complications.38–42 Age might be a risk factor for bleeding complications in the multimodal protocol because the population in this protocol was significantly older. In addition, older patients have a more fragile cardiovascular system and use anticoagulation more frequently. In the current study, correction for anticoagulant use before ECMO was not performed.
In our center, bleeding complications are often treated with surgical interventions. A strength of this study is that surgical interventions due to bleeding were excluded from analysis to prevent bias. Nevertheless, surgical intervention remaines an important risk factor for hemostatic complications. This study has several limitations. Firstly, the data was retrospectively collected, making it susceptible to time interval bias and missing data. Secondly, protocol adherence to the multimodal protocol was suboptimal. In an effort to prevent bias as result of non adherence, patients in the transition period between after introduction of the multimodal approach were excluded. However, ROTEM parameters were still not available in one third of bleeding events and half of thrombotic complications. In addition, the absence of heparin adjustment data rendered us incapable of ascertaining the extent of heparin dose modifications in both protocols, which could have been a valuable advantage of the multimodal monitoring strategy. Lastly, E-CPR patients were excluded due to the effect of cardiac arrest on the hemostatic balance and the risk of development of disseminated intravascular coagulation. 43 Thus, results in this paper are not applicable to E-CPR patients. Maintaining the hemostatic balance in E-CPR patients comes with a specific set of challenges which need to be addressed in separate studies. However, results from this study are generalizable to the general -non ECPR-adult ECMO population.
Conclusion
In this study, we conclude that hemostatic complications occur frequently in ECMO patients. The incidence of hemostatic complications did not differ between an aPTT guided and a multimodal monitoring protocol. The decreasing aPTT was associated with thrombotic events in the aPTT guided protocol. In this study, no clear benefit of the multimodal monitoring protocol was found. Surgical interventions were an important risk factor for hemostatic complications during ECMO support. The role of coagulation tests and surgical interventions in the development and prevention of hemostatic complications needs to be elucidated in future studies to improve care for this critically ill patient population.
Supplemental Material
Supplemental Material - Evaluation of an aPTT guided versus a multimodal heparin monitoring approach in patients on extra corporeal membrane oxygenation: A retrospective cohort study
Supplemental Material for Evaluation of an aPTT guided versus a multimodal heparin monitoring approach in patients on extra corporeal membrane oxygenation: A retrospective cohort study by Diman Taha, Joppe G Drop, Enno D Wildschut, Matthijs De Hoog, C. Heleen van Ommen and Dinis Dos Reis Miranda in Perfusion.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
