Abstract
Background: Monitoring the anticoagulant effect of unfractionated heparin (UFH) in extracorporeal membrane oxygenation (ECMO) patients is complex but critically important to balance the risks of treatment related bleeding and circuit thrombosis. While guidelines recommend using more than one method to monitor UFH activity, the use of thromboelastometry (ROTEM) to monitor UFH in ECMO patients has not been investigated in detail.
Methods: This is an observational, single-center retrospective study looking at adult ECMO patients on UFH that had ROTEM and thromboelastography (TEG) tests obtained concurrently. A total of 20 samples were obtained from nine patients during the study period, seven of which were on veno-arterial (VA) ECMO and two of which were on veno-venous (VV) ECMO.
Results: Under institutional standard operating practice, when TEG and/or activated partial thromboplastin time (aPTT) were considered therapeutic, intrinsic thromboelastometry clotting time (INTEM CT) was only 1.2 times higher than the normal range. TEG based monitoring compared to aPTT based monitoring tended to result in lower anti-Xa levels and less intensive anticoagulation. For the total cohort, bleeding events, driven by the need for blood transfusions, were more common compared to ischemic events (77% vs 11%; p = 0.02).
Conclusion: INTEM CT tended to be less sensitive to lower doses of UFH with a value of 1.2 times higher than the normal range when aPTT and/or TEG were considered therapeutic. Due to the relative insensitivity of ROTEM, our institution decided to continue to use TEG instead of ROTEM. Larger, multicenter trials may be helpful to validate these findings.
Keywords
Introduction
Extracorporeal membrane oxygenation (ECMO) is a circulatory support technique for providing respiratory and cardiac support. Patients on ECMO are at high risk of both bleeding and thrombosis. During ECMO, there is continuous contact between circulating blood and the foreign surface of the extracorporeal circuit resulting in an increased risk of thrombosis. Therefore, anticoagulation therapy, most commonly with unfractionated heparin (UFH), is necessary to preserve the integrity of the ECMO circuit. 1 However, monitoring UFH in ECMO patients is complex and the coagulation assays available are not well validated nor are anticoagulation thresholds standardized.1,2 The Extracorporeal Life Support Organization (ELSO) guidelines suggest using more than one method to monitor UFH activity but otherwise leaves it up to the individual institution to develop a monitoring and titration approach. 1 To date there is no consensus on the optimal monitoring strategy.
There has been a growing interest in using viscoelastic testing with thromboelastography (TEG) and rotational thromboelastometry (ROTEM) in monitoring coagulation status in patients on ECMO.2–5 Whole-blood viscoelastic point-of-care tests quantifies the properties of clot formation and the integrity of the coagulation cascade at multiple phases and may be more representative of in vivo coagulation than traditional tests like activated partial thromboplastin time (aPTT). This may be because all blood constituents participating in coagulation are available for interacting in whole blood viscoelastic testing as opposed to only coagulation proteins analyzed in platelet-poor aPTT testing. Its use in ECMO can be highly beneficial given that multiple derangements in critically ill patients on ECMO support (shock liver, disseminated intravascular coagulation, unfractionated heparin, azotemia) that can occur by itself, or in combination, may contribute to coagulation abnormalities. 1
Studies have shown that using a TEG-guided anticoagulation approach results in lower doses of UFH utilized without increasing thromboembolic events.3–5 Patients with significantly delayed clot initiation per TEG (TEG-R time 2-4X normal) frequently had aPTT values <1.5X baseline. 2 At our institution, we use a TEG and aPTT guided UFH monitoring protocol. Due to hospital plans of transitioning from TEG to ROTEM monitoring in the operating room, there was a need to develop a ROTEM UFH monitoring protocol for adult ECMO patients in the intensive care unit for simplification. However, very little guidance was found in literature looking at ROTEM monitoring in this patient population. The purpose of this study is to evaluate ROTEM for monitoring UFH anticoagulation by comparing it to TEG, aPTT, and anti-Xa.
Methods
A retrospective, single-center study was performed of ECMO-supported patients, at least 18 years old, from April 1, 2022 to May 31, 2022 that had both ROTEM and TEG lab orders obtained at the same time. The study was approved by the CommonSpirit Health Research Institute Institutional Review Board (IRB) (ID 191562-1) in which informed consent was waived.
At our institution, UFH is the anticoagulant of choice for ECMO anticoagulation. The ECMO circuit is heparin coated. For non-surgical patients, patients are bolused with 50 units/kg at time of cannulation. Per protocol, patients are started on 7 units/kg/hr of UFH with aPTT checked every 6 h and TEG twice daily. 2 Therapeutic goals were defined as an aPTT of 60–80 s (1.5-2X baseline) for VA ECMO and an aPTT of 50–70 s (1.25–1.75X baseline) for VV ECMO, a TEG reaction (TEG-R) time ratio 2–4X patient-specific baseline with no fibrinolysis, an anti-Xa level of 0.2–0.4 units/mL, and an anti-thrombin III level >50%. If the aPTT or TEG was supratherapeutic, the heparin infusion would be reduced by 100 units/hr. If both lab values were subtherapeutic, the heparin infusion would be increased by 100 units/hr. If one value was subtherapeutic and the other therapeutic, the patient’s anti-Xa level would be measured to help resolve the discordance. The intensivist team would then make a decision on dosing titration based on all available lab values; consultation with a clinical pathologist was available as needed if there were difficulties with interpretation of conflicting results. ROTEM was drawn on patients during the months of April – May 2022 at the same time as morning TEG draws in ECMO patients on UFH as the institution worked on transitioning from TEG to ROTEM. Treatment decisions were not based solely upon ROTEM results due to its investigational nature in our institution.
The TEG 5000 (Haemonetics Corporation, Braintree, MA) and ROTEM delta (Werfen, Barcelona, Spain) were used. Blood was sampled from the arterial line after an appropriate aliquot was wasted per protocol. Both ROTEM and TEG samples were collected at the same time in different tubes utilizing 2.7 mL of blood each in 3.2% sodium citrate tubes. The samples were analyzed immediately with commercially available reagents.
The TEG analyzer has a rotating cup in which a patient’s blood sample is placed. A stationary pin attached to a torsion wire is immersed in the blood. The cup oscillates as kaolin is added to activate primarily the intrinsic pathway of the coagulation cascade leading to thrombin and clot formation. The degree of rotational movement by the pin is converted to an electrical signal which produces a graphical tracing that reflects a hemostasis profile of clot formation. TEG variables include reaction time (R time, mins) which measures clot initiation, angle (degrees) which measures amplification, maximum amplitude (MA, mm) which measures stability of the clot, and clot lysis at 30 min after MA (LY 30). Heparinase can also be added to the sample to allow for evaluation of heparin effect by comparing the tracings. 5
ROTEM on the other hand uses a rotating pin instead and thus is less susceptible to vibration and mechanical shocks. Between pin and cuvette remains a gap of 1 mm which is bridged by the blood. The pin is rotated by a spring and as the blood clots, the rotation of the pin becomes more restricted which is detected optically. An integrated computer calculates the ROTEM curve. 6 ROTEM variables include clotting time (CT, sec) which measures clot initiation, clot formation time (CFT, sec) which measures speed of clot formation, maximum clot firmness (MCF, mm) which measures clot stabilization, and maximum lysis (ML; % decrease in amplitude 60 min after MCF) which measures fibrinolysis. With ROTEM, various activators or inhibitors are added to the sample in order to represent different processes of hemostasis. In measuring heparin activity, the INTEM assay may be most useful where the intrinsic pathway is activated. The HEPTEM assay contains heparinase and is coupled with the INTEM to determine coagulation status without heparin effect.6,7
For the purposes of this study, we collected TEG R-time, TEG R-time with heparinase (KH-TEG-R), EXTEM CT, INTEM CT, and HEPTEM CT. For conventional coagulation tests, we collected aPTT and anti-Xa. Additional data collected include demographic data (age, weight), comorbidities, ECMO type and indication, UFH dose, baseline complete blood count, INR, ATIII level, bleeding and thrombotic events, and mortality.
Bleeding was classified as major or minor according to the ELSO guidelines. 1 Major bleeding included a hemoglobin (Hgb) drop ≥4 g/dL in 24 h, use of more than 2 units of packed red blood cells (pRBC) in 24 h, bleeding at a critical site (gastrointestinal, pulmonary, intracranial), or bleeding requiring surgical intervention. One change was made to the ELSO definition of bleeding to account for Hgb drops that were simply due to critical illness and multiple blood draws. We used a threshold Hgb drop of ≥4 g/dL, instead of ≥2 g/dL as recommended in the ELSO guidelines, because this threshold was consistent with the anticoagulation policy that was already in effect at our institution. The second primary endpoint, thrombosis, was defined as thrombus formation anywhere in the circuit (whether or not it led to circuit replacement), disseminated intravascular coagulation (DIC), myocardial infarction, ischemic stroke, pulmonary embolism, deep vein thrombosis (DVT), or any arterial thromboembolic event.1,4 Based on institutional practice, circuit thrombosis was checked visually daily by perfusionists and reported to the medical team. DVTs and PEs were only checked based on clinical suspicion.
Continuous data were reported as median and interquartile range [IQR] and categorical data as absolute and relative frequency. Categorical data were compared by using the χ2 test; continuous data were compared by using a two-tailed t test or its nonparametric analogue, the Wilcoxon rank-sum test, as appropriate. An alpha level of 0.05 was used to determine statistical significance. Correlation was performed using the Spearman test. Data analysis was performed in Microsoft Excel and GraphPad Software (Boston, MA).
Results
Baseline characteristics (n = 9) at the time of ECMO cannulation.
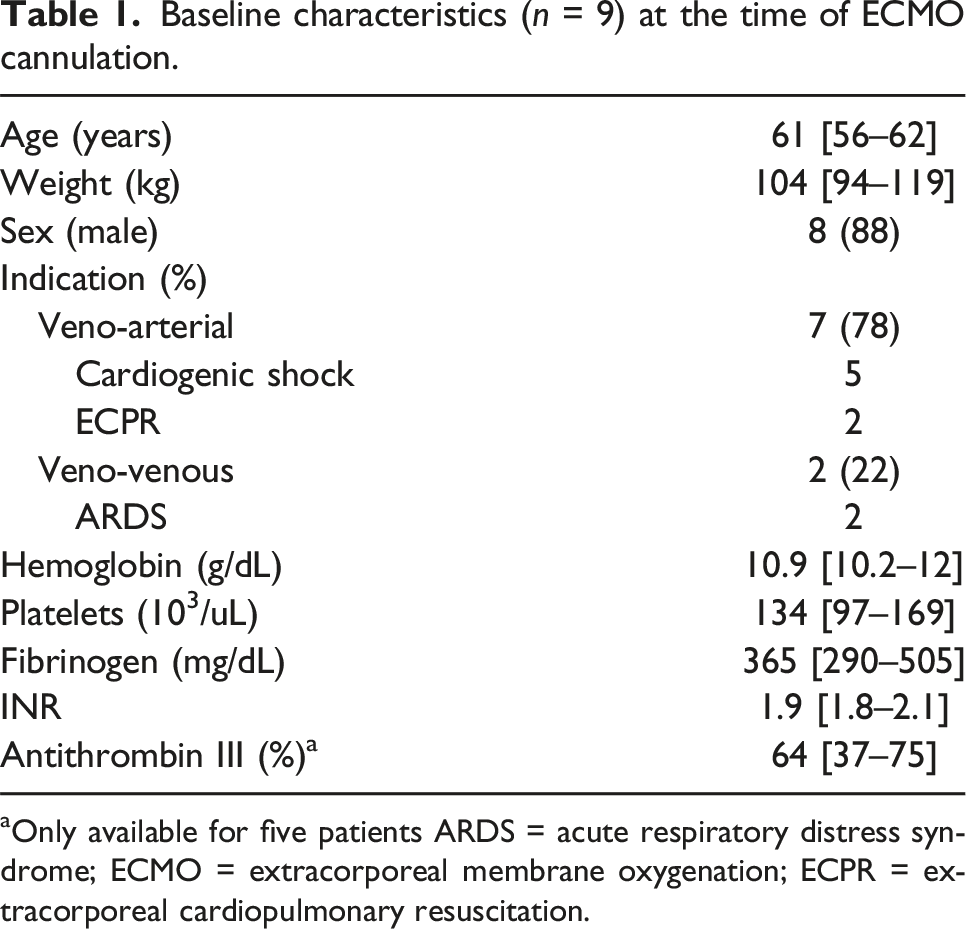
aOnly available for five patients ARDS = acute respiratory distress syndrome; ECMO = extracorporeal membrane oxygenation; ECPR = extracorporeal cardiopulmonary resuscitation.
Coagulation parameters based on therapeutic TEG (n = 18). a
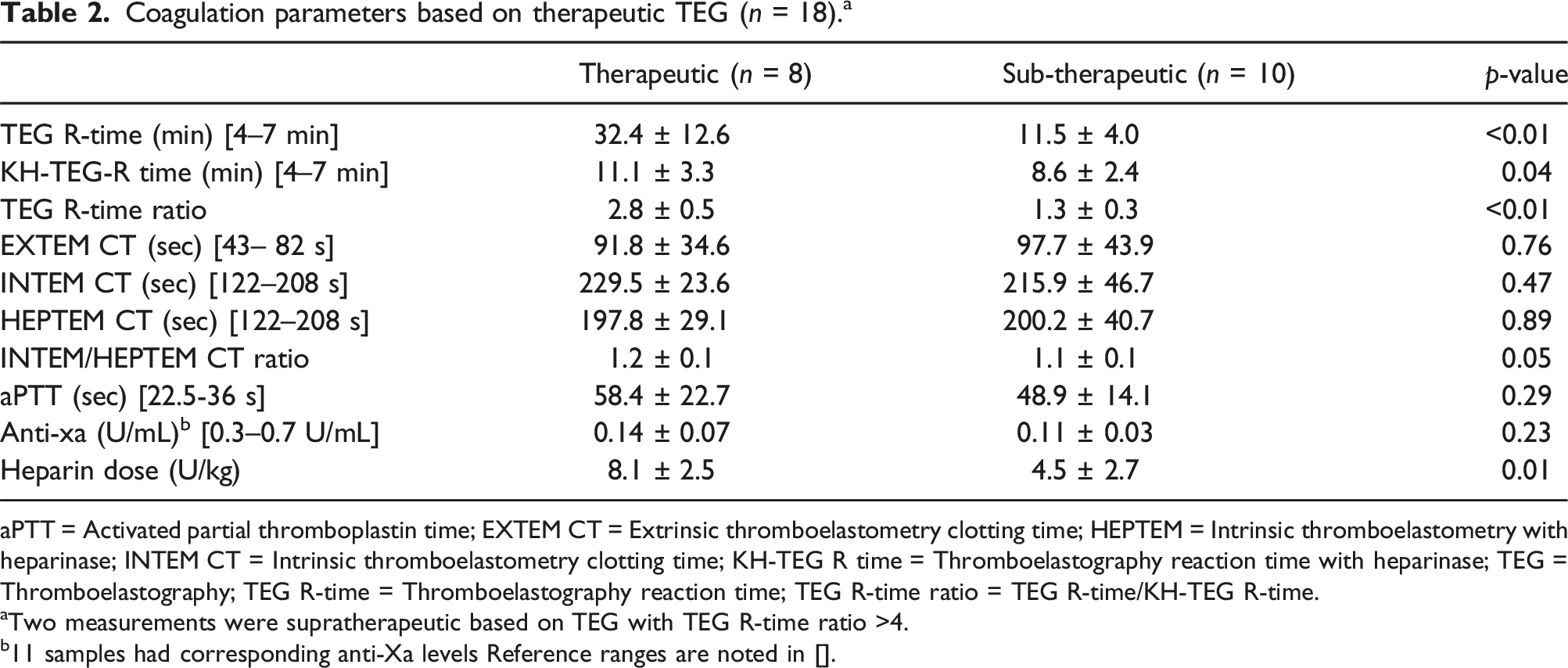
aPTT = Activated partial thromboplastin time; EXTEM CT = Extrinsic thromboelastometry clotting time; HEPTEM = Intrinsic thromboelastometry with heparinase; INTEM CT = Intrinsic thromboelastometry clotting time; KH-TEG R time = Thromboelastography reaction time with heparinase; TEG = Thromboelastography; TEG R-time = Thromboelastography reaction time; TEG R-time ratio = TEG R-time/KH-TEG R-time.
aTwo measurements were supratherapeutic based on TEG with TEG R-time ratio >4.
b11 samples had corresponding anti-Xa levels Reference ranges are noted in [].
Coagulation parameters based on therapeutic aPTT (n = 19). a
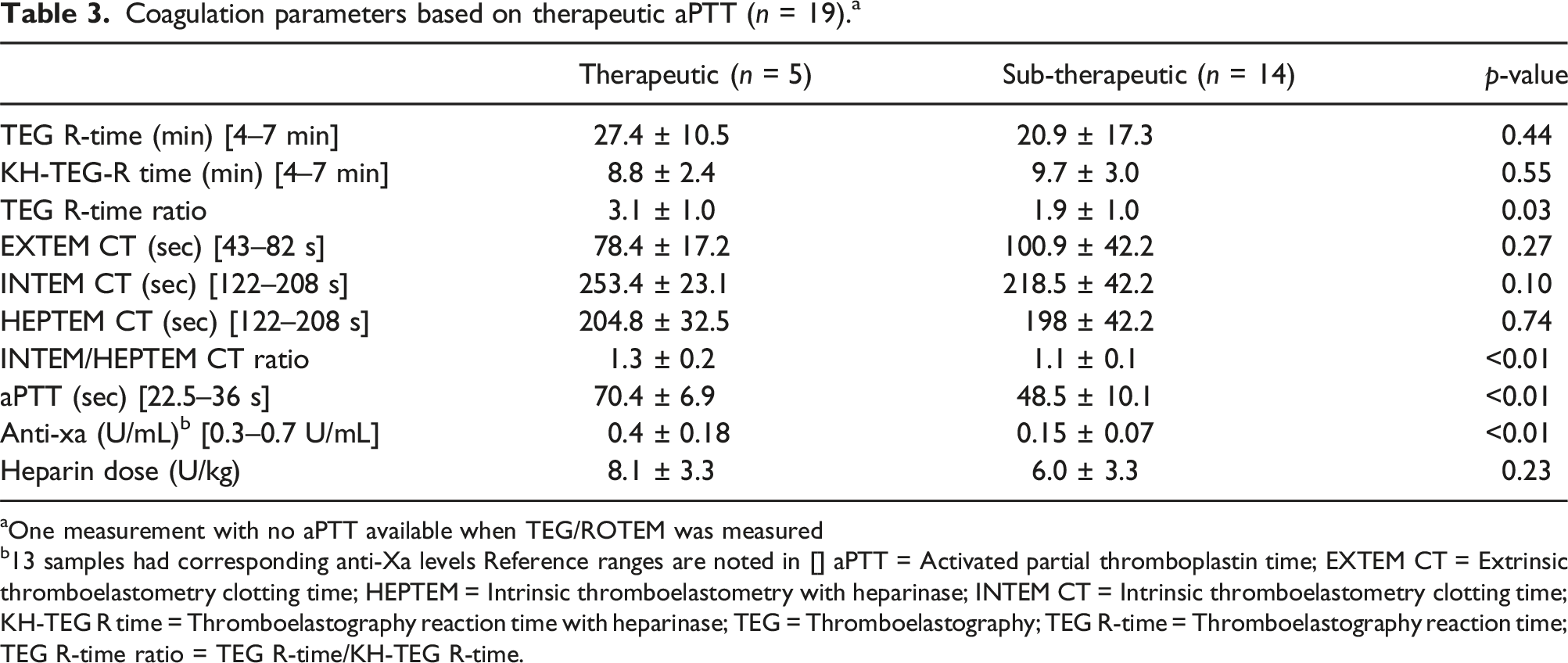
aOne measurement with no aPTT available when TEG/ROTEM was measured
b13 samples had corresponding anti-Xa levels Reference ranges are noted in [] aPTT = Activated partial thromboplastin time; EXTEM CT = Extrinsic thromboelastometry clotting time; HEPTEM = Intrinsic thromboelastometry with heparinase; INTEM CT = Intrinsic thromboelastometry clotting time; KH-TEG R time = Thromboelastography reaction time with heparinase; TEG = Thromboelastography; TEG R-time = Thromboelastography reaction time; TEG R-time ratio = TEG R-time/KH-TEG R-time.
Spearman correlation (n = 20).
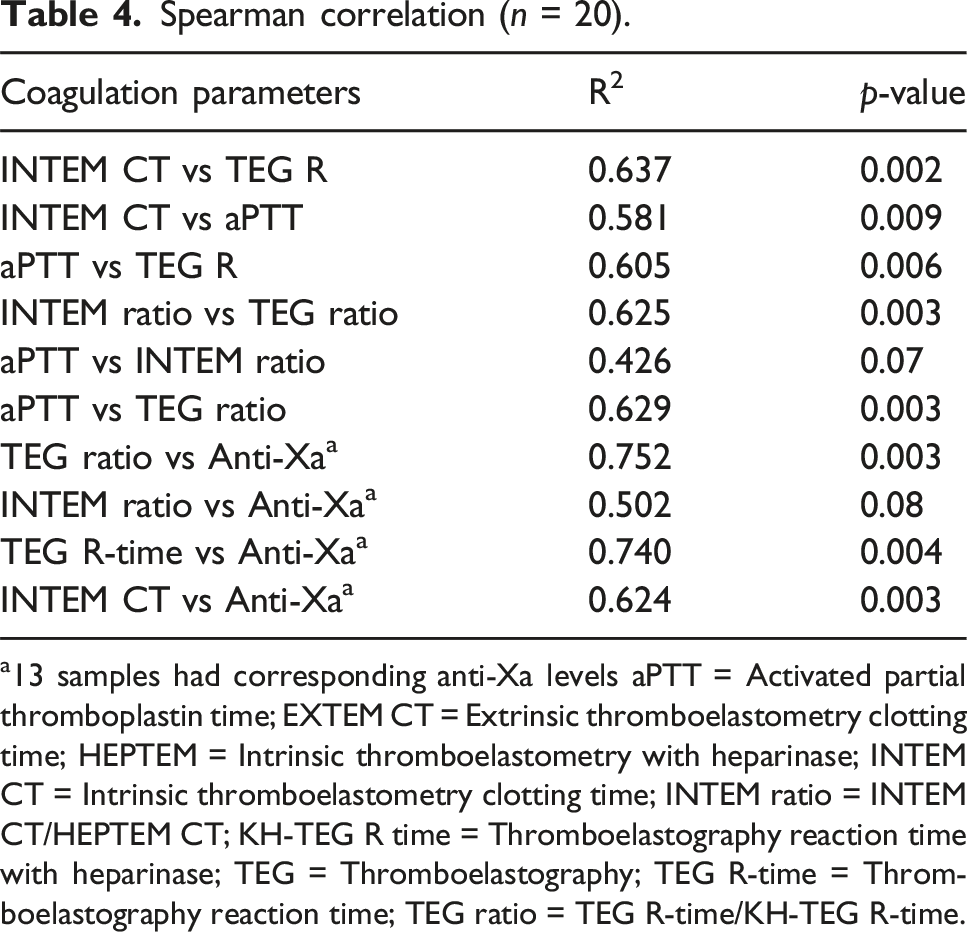
a13 samples had corresponding anti-Xa levels aPTT = Activated partial thromboplastin time; EXTEM CT = Extrinsic thromboelastometry clotting time; HEPTEM = Intrinsic thromboelastometry with heparinase; INTEM CT = Intrinsic thromboelastometry clotting time; INTEM ratio = INTEM CT/HEPTEM CT; KH-TEG R time = Thromboelastography reaction time with heparinase; TEG = Thromboelastography; TEG R-time = Thromboelastography reaction time; TEG ratio = TEG R-time/KH-TEG R-time.
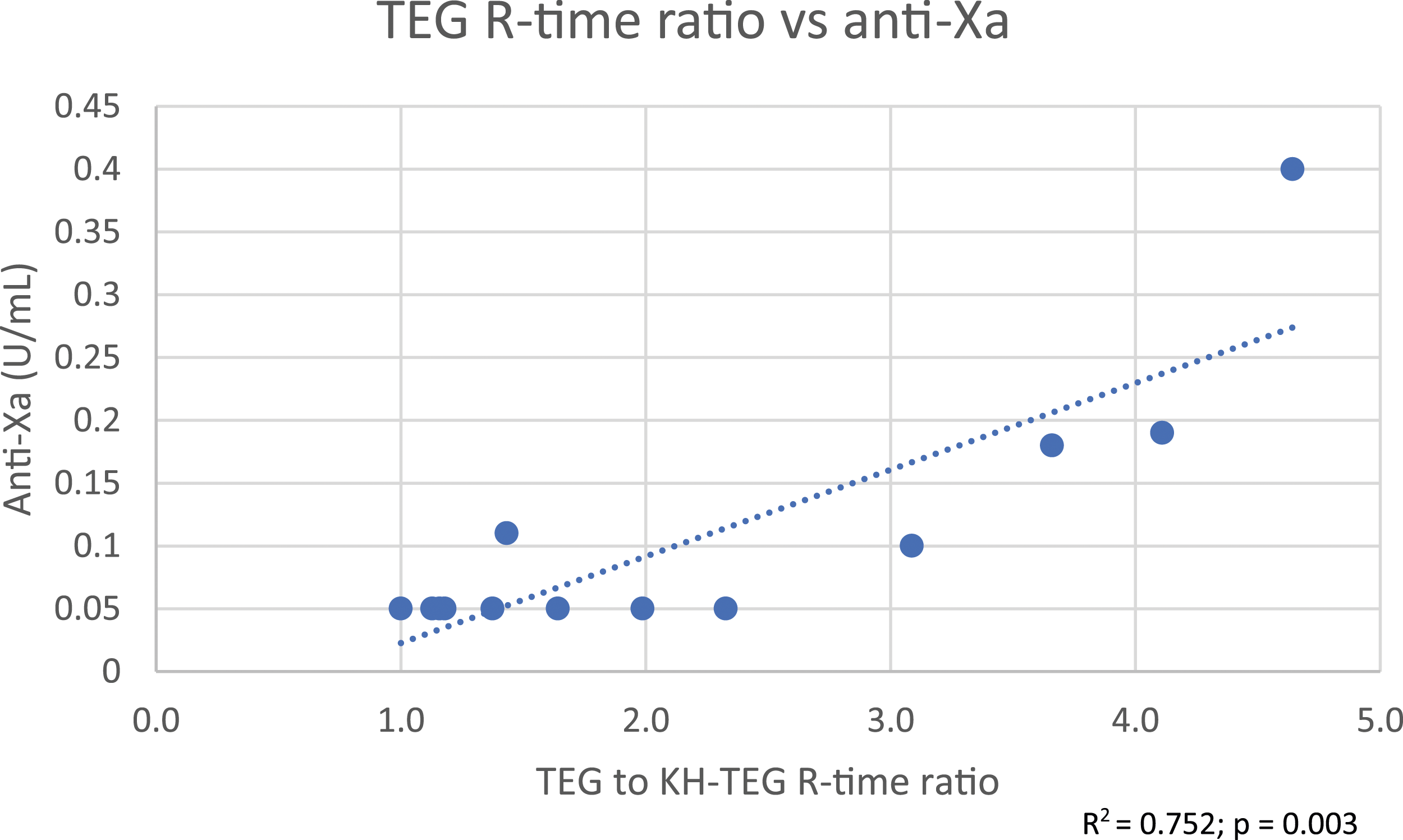
TEG R-time ratio versus anti-Xa.
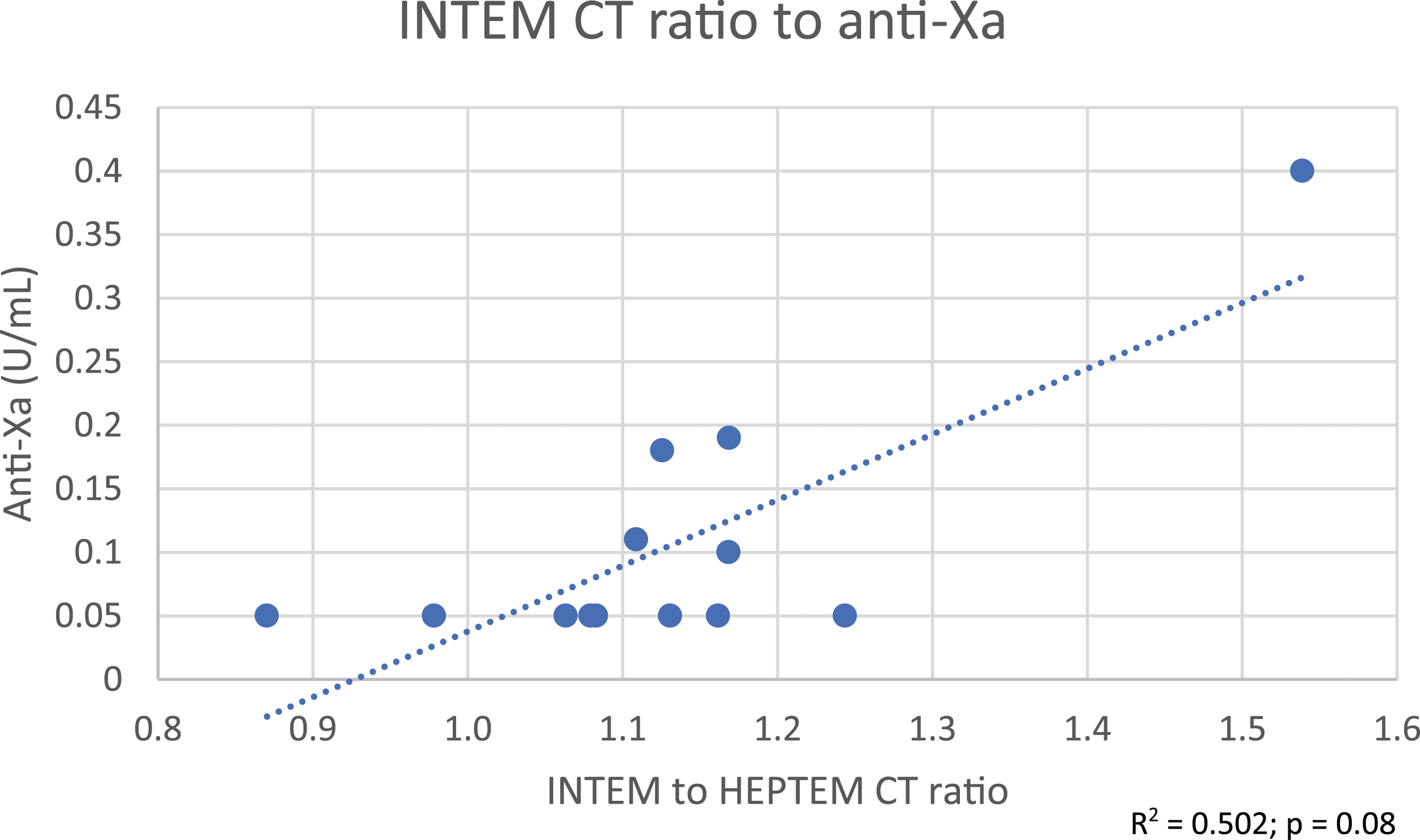
INTEM CT ratio versus anti-Xa.
Clinical outcomes (n = 9).

ECMO = Extracorporeal membrane oxygenation.
Discussion
Evaluation of the coagulation system in patients on ECMO support is challenging. These patients have many modifications from the normal hemostasis background including acute-phase reaction, organ compromise, consumption of factors, and hemodilution. 8 While UFH is commonly used for anticoagulation – its typical measure through aPTT methodology is challenged by the marked increase in factor VIII and fibrinogen that can contribute to “heparin refractoriness” by shortening the aPTT masking the true UFH effect. 9 Whether UFH is playing a significant role in coagulation or whether the global impact of UFH on coagulation is underappreciated based upon assay design is uncertain.
Alternative measures of anticoagulant activity include the activated clotting time (ACT) and anti-Xa. However, the ACT is affected by multiple clinical factors including thrombocytopenia, elevated D-dimers, low fibrinogen, hypothermia, and hemodilution or anemia. All of these factors are frequently present in ECMO patients; therefore, expertise in understanding the result is required as UFH titration based off a specific number or range may not be appropriate. 9 Anti-Xa is better associated with UFH dose and results in less variability than aPTT; however, it does not take into account the patient’s baseline coagulopathy and is affected by antithrombin levels. Additionally, the assay may be affected by hyperlipidemia, hyperbilirubinemia, and hemolysis, all of which may be present in ECMO patients, and ultimately falsely lower the anti-Xa level.1,9
Global hemostasis – taking into account all constituents that participate in clot formation (circulating proteins, platelets, red blood cells) is an attractive alternative approach. The degree of anticoagulation required for ECMO is not well established. Previously levels required in a post-thrombotic state as with deep vein thrombosis were recommended. 1 Evolving data suggests that a less intense course of anticoagulation, with anti-Xa levels of 0.1–0.2 U/mL preferred to 0.3–0.7 U/mL used in post-thromboembolism care, may be preferred given the fact that overall bleeding complications seem to be more frequent than thrombotic complications.2,9–12 This is particularly of interest in VV-ECMO where multiple retrospective studies suggest that either lower (or no) anticoagulation is safe and feasible.10,13,14. Several studies have shown that a TEG-guided anticoagulation approach leads to lower doses of anticoagulation being utilized with similar rates of thrombosis but less bleeding events.3–5 In this study, the same anticoagulation protocol was used as the Colman study. 2 Although patients were considered to be ‘sub-therapeutic’ 14 of 19 readings in the aPTT group versus 10 of 18 readings in the TEG group, bleeding complications still tended to be more frequent (77%) compared to thrombotic events (11%). Using a lower aPTT goal or targeting a TEG R-time ratio of 2-4X baseline may be preferred to provide adequate ECMO-related thrombosis protection and reduce major bleeding rates.
Among available global hemostasis systems, the current configuration of TEG may be a better fit for this patient population. TEG R-times and ratios were able to respond to the lower desired concentrations of heparin compared to ROTEM. In fact, Prakash et al. found that INTEM CT results were >50% of the time in normal range when aPTT was considered therapeutic. They concluded that due to the relative insensitivity to ROTEM, INTEM CT guided UFH titration could lead to excessive UFH overexposure. 15 In our study, INTEM CT values were only in the normal range when TEG R-time ratios and aPTT were considered sub-therapeutic. When TEG and/or activated partial thromboplastin time (aPTT) were considered therapeutic, INTEM CT averaged 243 ± 27 s. However, this elevation is only around 1.2X the normal range compared to 2–4X times the normal range for TEG. Additionally, when comparing INTEM CT to HEPTEM CT, the ratio was also only 1.2. Considering real world practice, when considering UFH titration, evaluating a 1.2X change in value from baseline compared to a 2–4X change in value from baseline would be harder to assess. In other words, the use of the ROTEM by comparing INTEM to HEPTEM CT provides a very narrow therapeutic window that would be clinically difficult to sustain.
We also found that coagulation parameters tended to correlate with each other with the exception of the INTEM CT ratio when compared to aPTT or anti-Xa. This, however, differs from the conclusions described by Giani et al. They had found a poor correlation between TEG R-time and aPTT but a modest correlation between INTEM CT and PTT and no correlation between TEG and ROTEM. 5 This shows the necessity and importance of larger studies needing to be done in this area.
A modified system with a greater response to lower concentration so UFH may render ROTEM equivalent to TEG for the purpose of assessing comparative blood clotting. While ratios of clotting time seem crude in the modern context of measuring very specific aspects of the coagulation system (ex. anti-xa), one must remember that aPTT values are also based upon a ratio consideration. As systems adopt whole blood viscoelastic testing more widely, we hope that the niche – but important ECMO market will be considered so that institutions opting for a viscoelastic platform will be able to use a single system for all patients. At this time we elected to continue to use TEG-based testing for ECMO despite overall conversion to ROTEM in our intra- and post-operative cardiovascular patient population.
Limitations of this study include the small sample size; however, this was meant to be an exploratory laboratory assay validation study. This also limited the ability to correlate coagulation parameters with clinical outcomes.
Conclusion
In our exploratory, single-center study, the major finding was that INTEM CT tended to be less sensitive to lower doses of UFH with a value of 1.2 times higher than the normal range when aPTT and/or TEG were considered therapeutic. Coagulation parameters used to assess for heparin effect in ECMO tended to correlate with each other with the exception of the INTEM CT ratio with aPTT and anti-Xa. Following a TEG based approach tended to result in lower anti-Xa levels and less intensive anticoagulation. Whether staying on sub-therapeutic anti-Xa levels as measured by TEG is safe enough is still unclear due to the limited sample size in this study. When transitioning from a TEG to ROTEM monitoring strategy, an INTEM CT value of 210 – 260 s or an INTEM CT ratio of 1.1 – 1.3 correlated with a TEG R-time ratio of 2–4X baseline; clinical decision on dosing should be made in conjunction with a standard coagulation parameters (aPTT or anti-Xa). However, at this time, we decided to continue TEG and aPTT guided UFH titration. Bleeding events were more common compared to ischemic events even though coagulation parameters were considered sub-therapeutic more than 50% of the time. Ongoing study of the optimal set point for heparin-based anticoagulation in ECMO is needed.
Supplemental Material
Supplemental Material - Thromboelastography versus thromboelastometry for unfractionated heparin monitoring in adult patients on extracorporeal membrane oxygenation
Supplemental Material for Thromboelastography versus thromboelastometry for unfractionated heparin monitoring in adult patients on extracorporeal membrane oxygenation by Ellen B Yin, Arthur W Bracey and Subhasis Chatterjee in Perfusion.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
