Abstract
Type A aortic dissection (TAAD) is a life-threatening disease with high mortality and poor prognosis, usually treated by surgery. There are many complications in its perioperative period, one of which is oxygenation impairment (OI). As a common complication of TAAD, OI usually occurs throughout the perioperative period of TAAD and requires prolonged mechanical ventilation (MV) and other supportive measures. The purpose of this article is to review the risk factors, mechanisms, and treatments of type A aortic dissection-related oxygenation impairment (TAAD-OI) so as to improve clinicians’ knowledge about it. Among risk factors, elevated body mass index (BMI), prolonged extracorporeal circulation (ECC) duration, higher inflammatory cells and stored blood transfusion stand out. A reduced occurrence of TAAD-OI can be achieved by controlling these risk factors such as suppressing inflammatory response by drugs. As for its mechanism, it is currently believed that inflammatory signaling pathways play a major role in this process, including the HMGB1/RAGE signaling pathway, gut-lung axis and macrophage, which have been gradually explored and are expected to provide evidences revealing the specific mechanism of TAAD-OI. Numerous treatments have been investigated for TAAD-OI, such as nitric oxide (NO), continuous pulmonary perfusion/inflation, ulinastatin and sivelestat sodium, immunomodulation intervention and mechanical support. However, these measures are all aimed at postoperative TAAD-OI, and not all of the therapies have shown satisfactory effects. Treatments for preoperative TAAD-OI are not currently available because it is difficult to correct OI without correcting the dissection. Therefore, the best solution for preoperative TAAD-OI is to operate as soon as possible. At present, there is no specific method for clinical application, and it relies more on the experience of clinicians or learns from treatments of other diseases related to oxygenation disorders. More efforts should be made to understand its pathogenesis to better improve its treatments in the future.
Introduction
Aortic dissection (AD) is a rupture in the intima of the aorta caused by various factors. Anatomically, AD can be classified into two types: Stanford type A aortic dissection (TAAD) is involved in the ascending aorta while Stanford type B aortic dissection (TBAD) refers to dissection appearing in the descending and distal aorta. 1 Presently, it is unclear how AD arises, but the cardiovascular diseases, inflammatory reaction and so on are believed to contribute to AD. 2 Recently, CT and ultrasound have become the most common techniques to diagnose AD, increasing the precision of diagnosis. In terms of treatment, surgery serves as the main method for TAAD, while TBAD is mostly treated with drugs or interventional therapy. 1 Efforts have been spent in recent years and in-hospital mortality has decreased significantly from 31% to 22% through medical progress, while several serious complications such as heart failure, and neurological impairment still remain associated with TAAD. Oxygenation impairment (OI) is one of the main complications that patients may experience. In this paper, we reviewed the risk factors of type A acute aortic dissection-related oxygenation impairment (TAAD-OI), as well as the mechanisms and treatments of this condition. To facilitate clinicians' understanding of the complication, the progress in this field is described in as much detail as possible.
Clinical features and epidemiology
The specific definition of TAAD-OI is still not settled. Some researchers defined it as TAAD-related hypoxemia, with an oxygenation index (PaO2/FiO2) ≤200 mmHg as the limit, 3 while others took PaO2/FiO2≤300 mmHg as the limit. 4 Other researchers defined it as type A aortic dissection-related acute lung injury (TAAD-ALI) or acute respiratory distress syndrome (TAAD-ARDS).5,6 Statistics indicate that preoperative TAAD-OI occurs in 30%–50% of patients, 7 while the incidence of postoperative TAAD-OI is between 20.4% and 56.2%.3,8–14 Once TAAD-OI appears, patients may develop shortness of breath, cyanosis, hypoxemia and other manifestations. Postoperative TAAD-OI often requires prolonged intubation and mechanical ventilation (MV). At present, the risk factors for TAAD-OI are numerous, the mechanisms are complicated, and there is no recognized specific treatment.
Risk factors
At present, there are lots of studies on the risk factors of TAAD-OI. Several typical risk factors that have been mentioned frequently were selected to be introduced, so as to raise clinicians’ vigilance and facilitate them to identify high-risk group.
Preoperative TAAD-OI is a hard nut to crack for clinicians. It increases the risk of ALI/ARDS and other complications after surgery, sometimes even causing the surgeon to delay surgery for better optimization (in the case of stable dissection).15,16 Therefore, the identification of patients at high risk of preoperative TAAD-OI can enable preventive adjustments (such as the adjustment of circulation arrest time, etc.) to reduce the incidence of postoperative complications.
5
Elevated inflammatory marker was referred as a strong risk factor. Interleukin-6 (IL-6) was higher in preoperative TAAD-OI patients according to Pan and Duan.6,17 Additionally, Duan discovered that preoperative TAAD-OI was linked to C-reactive protein (CRP) and inhibiting the inflammatory response using ulinastatin (a kind of human urine protease inhibitor, more details will follow) would improve patients’ oxygenation.
17
This finding was confirmed by Zhao et al who claimed that serum CRP ≥9.20 mg/L on admission was a reliable indicator for preoperative TAAD-OI.
18
Moreover, coagulation fibrinolytic factors also contribute to TAAD-OI. Gao et al mentioned that tissue factor (TF) and tissue factor pathway inhibitor (TFPI) in the blood and bronchoalveolar lavage fluid (BALF) of preoperative TAAD-OI patients continued to rise before surgery.
5
Guo et al mentioned that a low fibrinogen served as a marker for preoperative TAAD-OI
6
(Figure 1). These above findings can help identify high-risk groups before surgery. However, due to the rapid progression of TAAD, quite a few of patients will develop TAAD-OI before the test results come out. More efficient and early markers must therefore be sought. Risk factors for type A aortic dissection-related oxygenation impairment: TAAD-OI: type A aortic dissection-related oxygenation impairment; ECC: extracorporeal circulation; BMI: body mass index.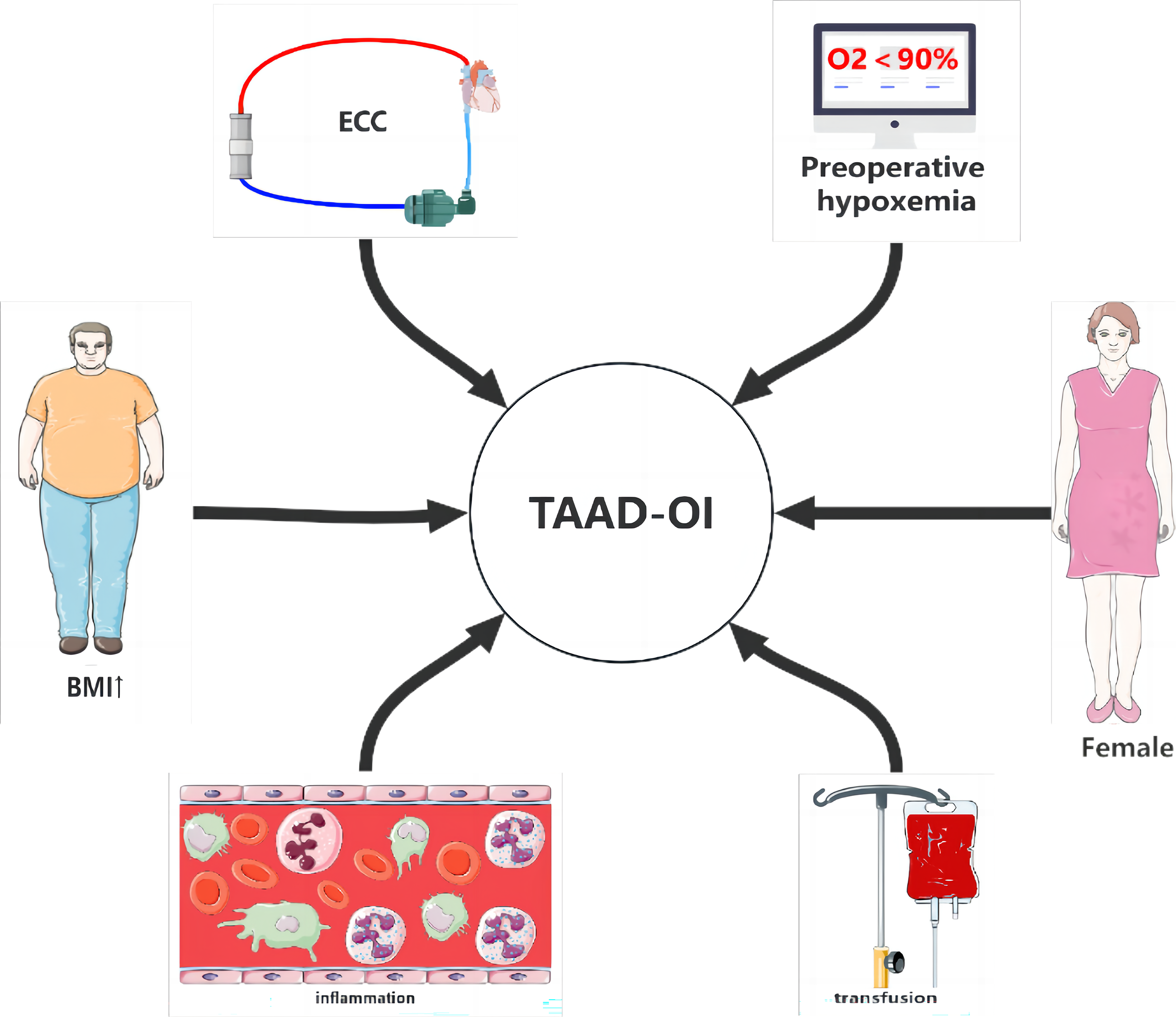
Although preoperative TAAD-OI is an undesirable condition, its occurrence can help to identify patients susceptible to postoperative TAAD-OI. Wang called attention to the fact that patients with preoperative PaO2/FiO2≤100 mmHg were more likely to develop postoperative TAAD-OI. 19 Analogous findings were published by Sheng, Zhao, and Liu et al, who pointed that preoperative PaO2/FiO2≤300 mmHg was a risk factor for postoperative TAAD-OI3,10,12 (Figure 1). Furthermore, for patients with preoperative TAAD-OI, the risk of postoperative TAAD-OI will increase by 7.830-14.611 times and make other complications more severe.3,10–12,14
Besides preoperative inflammatory markers, the amount of leukocyte also elevates before surgery, which will affect the degree of intraoperative inflammatory response in TAAD patients and hence affect postoperative oxygenation. 10 Sheng revealed that patients with preoperative elevated leukocyte developed more severe postoperative TAAD-OI, 3 so did Ge et al. 9 Liu confirmed that a leukocyte count>15,000/μL prior to surgery was a risk factor for postoperative TAAD-OI 10 (Figure 1). Such patients with a preoperative elevated leukocyte will develop more severe intraoperative inflammatory response, resulting in the release of numerous proinflammatory cytokines, the mass accumulation and activation of neutrophils and macrophages in the alveoli, and the release of toxic mediators and proteolytic enzymes, which increase the permeability of endothelial and epithelial cells, impair oxygenation, and cause postoperative TAAD-OI. 10
Female gender has been identified in quite a few studies as a risk factor for adverse outcomes of cardiac surgery,20–22 while its role in TAAD-OI remains controversial. Gong et al found that obesity and female gender were risk factors for postoperative TAAD-OI. 23 However, Wang et al pointed that although the incidence of postoperative TAAD-OI in women was higher than men, the effect did not show a statistical difference. 13 Fukui also noted in his study that there wasn’t statistic difference in postoperative mortality between women and men, but women were older and were operated more conservatively, and whether this was an artificial selection bias caused by surgeons was up for debate. 24 Therefore, it still needs more experiments characterized strict factors such as minimizing selection bias or meta-analyses reflecting the effect of gender on outcome.
As a parameter that reflects one’s nutritional and weight status, body mass index (BMI) is another marker for postoperative TAAD-OI. Gong et al noted that obesity was a risk factor for TAAD-OI. 23 Zhao et al found that those whose BMI >25 kg/m2 would develop postoperative TAAD-OI more possibly, which was the same as Zhang’s findings12,25 (Figure 1). This phenomenon may be attributed to the decline in functional residual capacity (FRC) and other pulmonary function indicators in higher BMI groups whose respiratory work will increase at the same time. Oxidative stress may also play an important role in the process. 23
In recent years, improvements such as hemoperfusion technology have been made in extracorporeal circulation (ECC) methods to reduce complications,26,27 but 20% to 25% of patients still suffer from pulmonary dysfunction after ECC. 14 As Sheng pointed, the use of deep hypothermic circulatory arrest (DHCA) was associated with postoperative TAAD-OI. 14 Further research revealed that the risk of postoperative TAAD-OI increased with the duration of DHCA10,11 (Figure 1). The ongoing perspective is that the systemic inflammatory response syndrome (SIRS) instigated by blood contact with ECC pipes and ischemia reperfusion injury (IRI) during ECC are the primary drivers of postoperative TAAD-OI. 3 Despite the fact that hypothermia and antegrade cerebral perfusion might maintain satisfactory cerebral function during ECC, malperfusion of other organs might in any case prompt broad IRI. 11
Blood transfusion is required most of the time in cardiac surgery and is often associated with postoperative pulmonary morbidity.28,29 For instance, Sheng indicated that transfusion and its volume within postoperative 24 h were credible markers for TAAD-OI. 3 In addition, Wang discovered a link between postoperative TAAD-OI and blood transfusion volume greater than 3000 mL. 11 Moreover, Chen and Su found that the higher the rate of perioperative blood transfusion, the greater the rate of postoperative TAAD-OI30,31 (Figure 1). Due to the coagulopathy and nature of TAAD surgery, 31 transfusion is essential during and after surgery, which may lead to transfusion-related acute lung injury (TRALI). 30 It may be attributed to the fact that stored blood products are rich in leukocyte antibodies, cytokines and other bioactive substances, which will increase pulmonary microvascular permeability, leading to lung injury and capillary leakage. Likewise, microthrombi, cell flotsam and jetsam and inflammatory mediators in stored blood also count. 31 Therefore, improving surgical techniques and enhancing intraoperative hemostasis, thus reducing the use of blood products, are essential to reduce the incidence of postoperative TAAD-OI. Maybe in the future, an randomized controlled trial(RCT) that compared individualized transfusion strategies with conventional transfusion strategies on TAAD-OI patients is not a bad idea.
Besides the aforesaid factors, there are other markers reported to lead to TAAD-OI. Researchers from China pointed that patients with postoperative TAAD-OI were often with a homocysteine exceeding 1.95 mmol/L. 14 Other researchers found that a left atrial diameter ≥35.5 mm and a left ventricular posterior wall thickness ≥10.5 mm were risk factors for postoperative TAAD-OI. 19 Beyond these, researchers also suggested that an increased APACHE ⅱ score was a marker of postoperative TAAD-OI. 8 Also, there were other scholars mentioned that an elevated bladder temperature may be potentially related to postoperative TAAD-OI 32 (Figure 1).
It is apparently that countless factors account for TAAD-OI. Some can be controlled while others are uncontrollable because of the characteristics of the patient. Therefore, it reinforces the fact that reliable prediction model should be developed to identify patients of high-risk so that preventive measures can be taken as soon as possible. And correlative cohort or RCTs should be carried out to see whether the incidence of TAAD-OI will descend by controlling the above risk factors.
Mechanism
TAAD-OI, whether occurring before or after medical procedures, has always been a troublesome issue for clinicians. Knowing relevant risk factors can identify high-risk groups. However, to fundamentally prevent it and its development, it is necessary to understand the mechanism of the disease. The exact mechanism of TAAD-OI remains ambiguous, but correlative pathways, including inflammation, coagulation, and fibrinolysis, have been suggested as possible mechanisms.
Predisposition
We have described a number of risk factors associated with TAAD-OI before, of which elevated BMI is thought to be closely related to it. There are three main mechanisms of higher BMI leading to TAAD-OI. Firstly, the vascular system in adipose tissue of obese patients is underdeveloped, which easily leads to hypoxia of adipose cells, thus stimulating the release of pro-inflammatory cytokines such as TNF-α, IL-6, IL-8, leptin, and IL-1 ß and activating pro-inflammatory signaling pathways such as c-Jun N-terminal kinase (JNK), IĸB kinase (IĸK)-ß, nuclear factor-ĸB (NF-ĸB), and redox-sensitive transcription factor, leading to lung dysfunction. In addition, fat accumulation stimulates the release of NADPH oxidase 4 (Nox4), a key factor in enzymatic cellular reactive oxygen species (ROS) production. ROS stimulates macrophages to infiltrate adipose tissue by inducing the production of monocyte chemotactic protein (MCP), further promoting the expression of local Nox4 and the production of ROS, causing oxidative stress and impairing lung function. Moreover, elevated free fatty acids in obese patients will bind to immune receptors such as Toll-like receptor 4 (TLR4), stimulate inflammation development, and lead to activation of the IĸK-ß/NF-ĸB system and the release of pro-inflammatory cytokines, triggering OI. 33
HMGB1/RAGE pathway
An inflammatory cascade is thought to be involved in the development of TAAD-OI. Zhang et al called attention to the fact that the number of high mobility group protein B1 (HMGB1) and receptor for advanced glycation end products (RAGE) in patients with TAAD-OI altogether arisen after admission and continued to increase in the ensuing time frame, while PaO2/FiO2 diminished at the same time. Once reaching their highest values, the number of HMGB1 and RAGE began to decrease while PaO2/FiO2 gradually changed in the opposite manner.
34
As a nuclear nonhistone protein, HMGB1 plays a key role in the initiation and maintenance of inflammatory cascade reaction. It interacts with RAGE to activate the mitogen-activated protein kinase (MAPK) and NF-κB pathway, thereby increasing the expression of cytokines such as IL and TNF, leading to an inflammatory cascade reaction, which eventually activates inflammatory cells in the lung, releasing inflammatory mediators that damage lung endothelial cells, leading to TAAD-OI (Figure 2). Mechanism of type A aortic dissection-related oxygenation impairment: TAAD: type A aortic dissection; TAAD-OI: type A aortic dissection-related oxygenation impairment; DAO: diamine oxidase; iFABP: intestinal fatty acid binding protein; IL: interleukin; TNF: tumor necrosis factor; Ang II: angiotensin II; AT1-R: angiotensin II type 1 receptor; P-IKB-α: phosphorylated protein IKBα; P-p65: phosphorylated p65; NF-κB: nuclear factor κ- B; MCP-1: monocyte chemoattractant protein one; MMP-9: matrix metalloproteinase nine; HMGB1: high mobility group protein B1; RAGE: receptor for advanced glycation end products; MAPK: mitogen-activated protein kinase; IRI: ischemia-reperfusion injury; GALT: gut-associated lymphoid tissue; BALT: bronchial associated lymphoid tissue.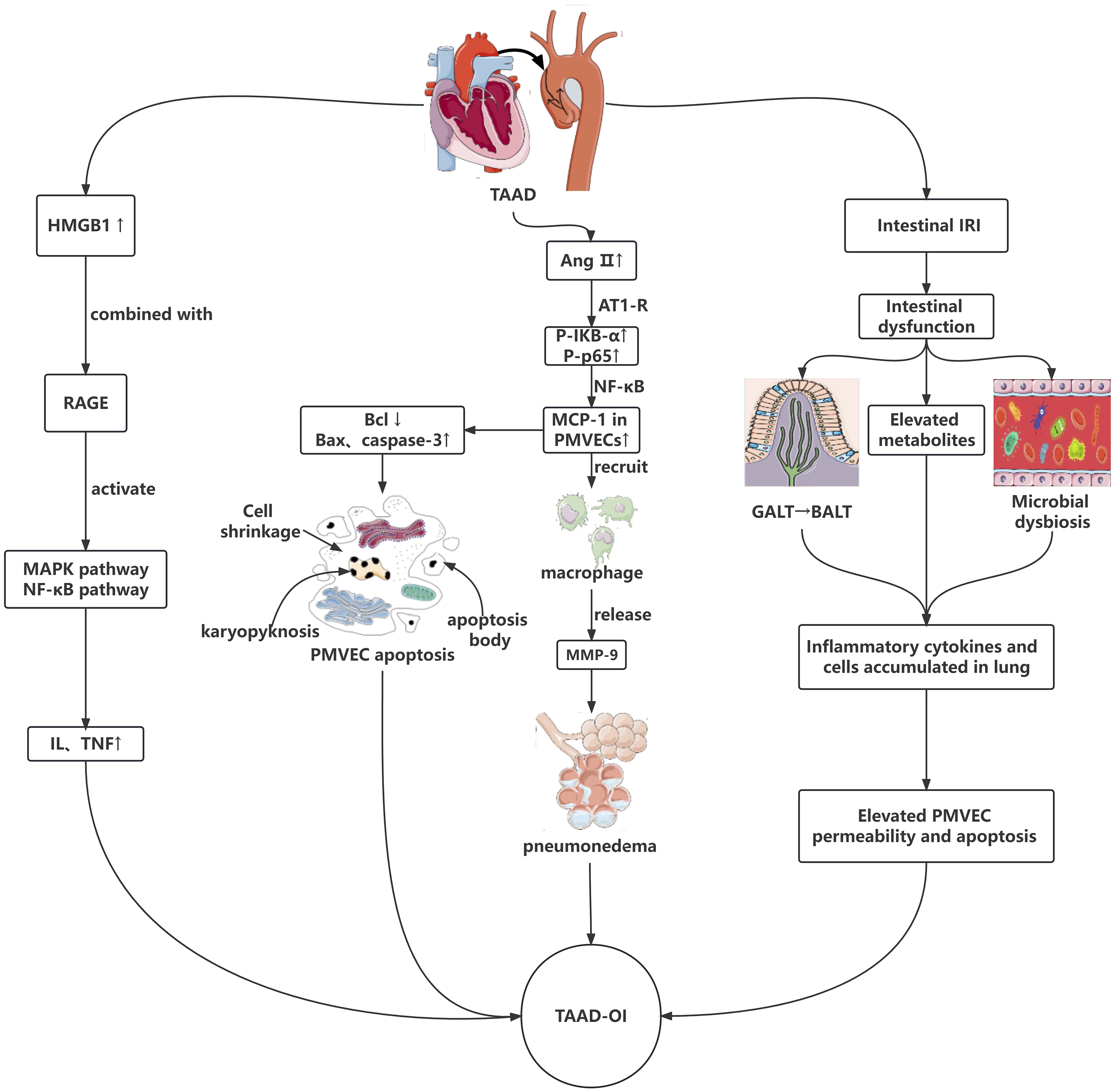
Gut-Lung axis
In recent years, many studies have indicated that intestinal barrier dysfunction is an important factor in the occurrence and development of ALI. The same results were also published in the TAAD-OI field. In 2016, findings demonstrated that elevated intestinal barrier damage-related factors intestinal fatty acid binding protein (iFABP), diamine oxidase (DAO), and peptidoglycan (PGN)—a bacteremia marker--were observed in TAAD-OI patients. 35 It may be due to the inference that IRI caused by TAAD can induce intestinal injury, leading to the translocation of intestinal microbiota, bacteremia and excessive expression of inflammatory factors, and finally TAAD-OI (Figure 2). In fact, current studies involved in asthma, chronic obstructive pulmonary disease (COPD), respiratory infection and so on have pointed to a close link between the lung and gut, from which the concept of the gut-lung ax evolves. 36 Shock, vascular surgeries and ECC often cause intestinal IRI, destroy the homeostasis of the gut and its environment, lead to intestinal dysfunction,37,38 and then induce distal organ dysfunction, in which ALI is the most characteristic manifestation. 39 At present, it is believed that the relevant factors may include the gut microbiota and its metabolites, microbial dysbiosis, and common mucosal immune system. 40 Studies indicate that succinate elevates during intestinal IRI and that ALI is exacerbated by succinate receptor 1 (SUCNR1) dependent alveolar macrophage polarization. 39 In addition, intestinal IRI can cause intestinal barrier dysfunction, result in microbial imbalance and induce bacteria to produce a large amount of lipopolysaccharide (LPS), which will enter the circulation to activate the lung TLR4 signaling pathway. TLR4 is an important pattern recognition receptor, which can recognize pathogenic bacteria that have translocated into the blood circulation, initiate the downstream signal transduction pathway, activate the NF-κB pathway, up-regulate the expression of various inflammatory factors, and trigger the inflammatory cascade in the lung, thus resulting in ALI by direct injury of endothelial cells or activation of neutrophils and so on. 41 Moreover, it is found that the gut and lung are part of a common mucosal immune system. This system mainly consists of gut-associated lymphoid tissue (GALT) and bronchial associated lymphoid tissue (BALT). Blood and lymph vessels can transport immune cells and cytokines from GALT to BALT, enhancing the immune resistance of the respiratory system. 40 However, when intestinal IRI appears, exosomes, TLR4, inflammatory cytokines and cells are excessively transported to the lung, which leads to the accumulation of a large number of inflammatory cells and cytokines in the lung, the production of harmful substances such as oxygen free radicals, the increase of endothelial permeability, the induction of apoptosis of endothelial cells, and the triggering of ALI. 42 However, the exact mechanism between the gut–lung axis and TAAD-OI is yet to be defined, and further basic researches are still needed to illuminate the role of the gut–lung axis in TAAD-OI.
Macrophage
It was reported that angiotensin II (Ang II), matrix metalloproteinase 9 (MMP-9) and monocyte chemoattractant protein 1 (MCP-1) significantly increased in TAAD-OI patients. At the same time, angiotensin II type 1 receptor (AT1-R) overexpressed, and macrophages expressing MMP-9 accumulated extensively in lung tissue. Given that the gene promoter of MCP-1 contained a binding sequence of the NF-ĸB pathway, researchers from China created a rat TAAD-OI model by Ang II to verify the hypothesis that MCP-1 may participate in the pathogenesis of TAAD-OI by regulating macrophage recruitment through NF-ĸB pathway.43,44 As was expected, the aforementioned phenomenon was observed, the expression of phosphorylated protein IKBɑ (P-IKB-ɑ) and phosphorylated p65 (P-p65) in pulmonary microvascular endothelial cells (PMVECs) upregulated, so did MCP-1. 43 It reminds us that TAAD can cause a higher level of Ang II, which then binds to AT1-R and increases PMVECs’ expression of P-IKB-ɑ and P-p65, thus activating the NF-ĸB pathway and induces MCP-1 to release. Then, MCP-1 recruits macrophages into lung tissue to increase the expression of MMP-9, which can cause the denaturation of collagen and elastin-the main components of the extracellular matrix and vascular basement membrane, as well as the degradation of type III-V collagen. As a result, the alveolar capillary barrier is breached, capillary permeability increases, inflammatory cells invade, pulmonary edema develops, and ultimately TAAD-OI occurs 44 (Figure 2).
In another study, researchers discovered that Ang II increased the expression of MCP-1, Bax and caspase-3 and decreased the expression of Bcl-2 in PMVECs, which were all related to apoptosis while Bcl-2 restrained apoptosis, Bax induced apoptosis and the activation of caspase played an important role in the executive phase of apoptosis45,46 (Figure 2). This suggests that MCP-1 can also induce apoptosis in PMVECs by disrupting the alveolar capillary barrier and regulating the expression of proteins related to apoptosis thus contributing to TAAD-OI.
Treatment
A variety of pharmacological and non-pharmacological approaches are currently being explored or already applied for ALI such as corticosteroids, exosomes, stem cells, prone positioning and so on. 47 It still remains unknown whether traditional methods and other novel treatments will have deeply impressive results for TAAD-OI. With great interest, several cut-and-try therapeutic methods are listed as follow.
Nitric oxide (NO)
NO is a selective vasodilator, which is currently used for pulmonary hypertension, respiratory failure and other diseases. Several studies have found that postoperative application of NO could be a potential way for TAAD-OI. Ma et al noted that statistically, PaO2/FiO2 ratio was higher and MV duration was shorter in patients treated with NO,
48
so was Zhang’s finding.
49
Zheng and his teammates also announced through a prospective observational study that PaO2/FiO2 increased after NO treatment
50
(Figure 3). It seems that NO can work on the oxygenation of TAAD-OI patients, without clear secondary effect. Inspiring as it may be, the current conclusions are based on a series of observational studies. Besides, previous RCTs and meta-analyses have concluded that NO has no benefit for decreasing mortality or duration of MV in patients with ARDS.51,52 So, its exact efficacy needs to be validated by RCTs. Based on following the principles of randomization, control and blind experimental design, the main population of these trials should be TAAD population, with NO treatment as the experimental group. It isn’t a bad idea to set the primary outcome as postoperative PaO2/FiO2 or something like a composite of oxygenation measurements. Then whether NO is applied intraoperatively or postoperatively remains to be discussed. The safe and effective dosage and time of application should be explored. In order to improve the level of evidence, the number of participating centers should be appropriately increased while ensuring the quality of the study. Treatments for type A aortic dissection-related oxygenation impairment: TAAD-OI: type A aortic dissection-related oxygenation impairment; NO: Nitric oxide; IL-22: interleukin-22; ECMO: extracorporeal membrane oxygenation.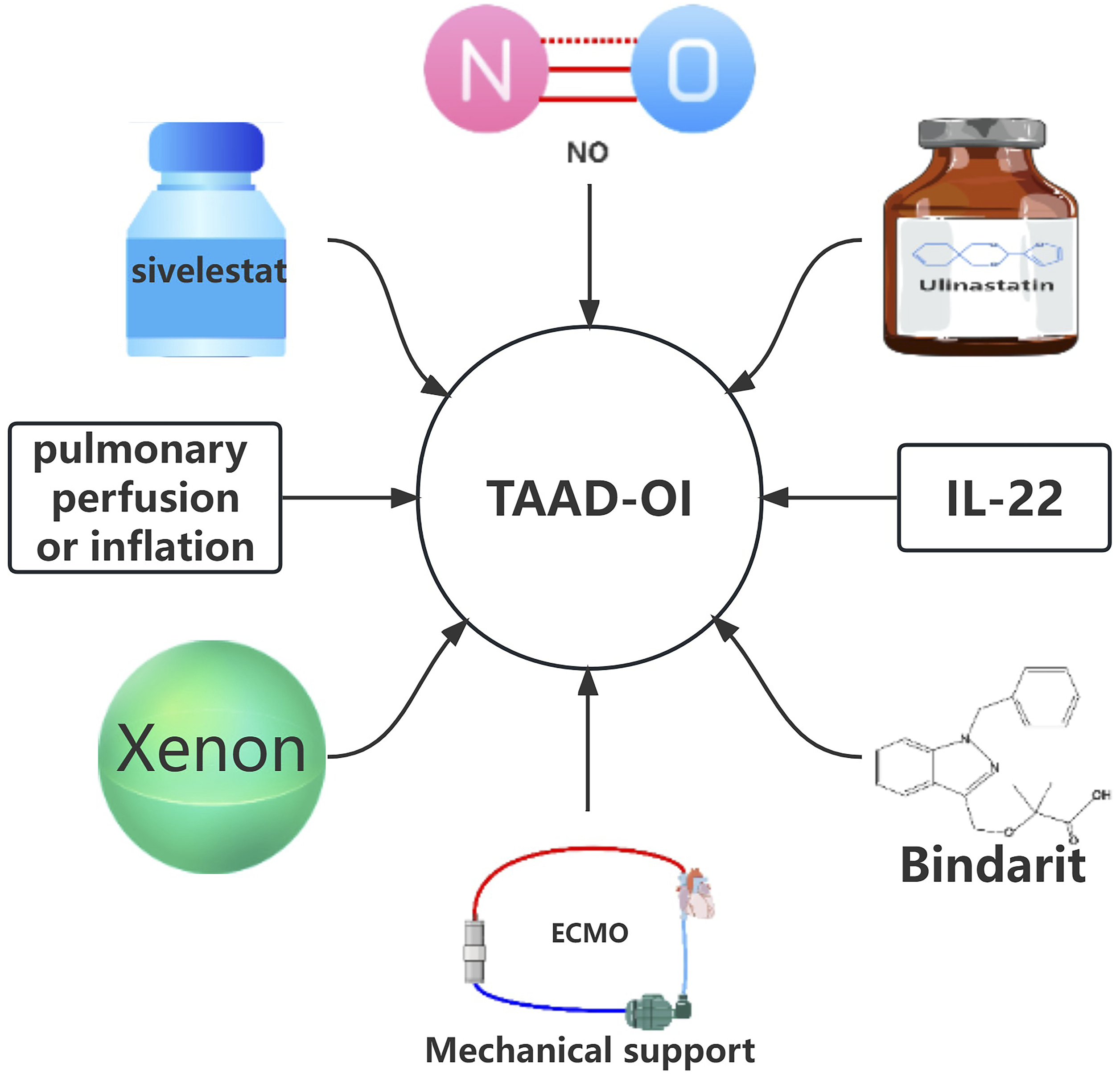
Pulmonary perfusion/inflation
Continuous cerebral perfusion has become a common practice during TAAD surgery, which has lowered the risk of postoperative brain complications. Actually, IRI is considered to play a crucial role in TAAD-OI. Refer to this method, researchers tried continuous perfusion for lung protection. De Santo found that patients receiving continuous lung perfusion during surgery obsessed higher postoperative PaO2/FiO2 than patients who did not. Additionally, the MV duration of the perfusion group was shorter. 53 Long before that, researchers discovered that the use of ECC with continuous perfusion in children could preserve pulmonary function. 54 However, De Santo didn’t get the same results when he changed the perfusate from oxygenated blood to celsior solution-a kind of organ preservation solution 55 (Figure 3). Jin et al used 50% xenon gas to continuously inflate the lungs during TAAD surgery. At the end of the surgery, the decrease in PaO2/FiO2 was reduced. 56 Analytically, the arrest of pulmonary blood flow during ECC plays an important role in pulmonary dysfunction, 57 which can be attributed to IRI. Animal and human studies have shown that the less pulmonary blood flow deprivation, the less pulmonary function damage. This is because it relieves the degree of IRI, reduces neutrophil isolation, and alleviates endothelial cells’ functional and structural damage. 55 As for xenon, it may be the possibility that xenon inflation prevents alveolar collapse better than oxygen due to its solubility, and therefore, absorption is very limited as opposed to oxygen. Besides, xenon treatment attenuates oxidative stress, indicated from the results that compared to baseline, MDA and MPO levels decreased in the xenon group postoperatively. 56 Although promising, questions are so many to be settled: what is the best perfusate/gas? In what range of perfusion/inflation pressure should be maintained? What is the recommended perfusion rate? Maybe in the future, studies should be devoted to solving the above problems.
Neutrophil/macrophage inhibitors
Ulinastatin is a human urine protease inhibitor that can inhibit the infiltration of neutrophil and prevent it from releasing elastase and other chemical mediators. Xu et al showed that the application of ulinastatin reduced the concentration of inflammatory cytokines and patients’ postoperative respiratory support parameters such as peak inspiratory pressure were improved. 58 Duan et al also confirmed that the large doses of ulinastatin caused a decline in the incidence of TAAD-OI. 17 According to the current results, ulinastatin seems to have a promising therapeutic effect on TAAD-OI, but due to the small sample size of this clinical trial, it may cause the increase of sampling error, thus reducing the efficiency of the test. So, whether the results are really believable needs to be further verified. Another neutrophil elastase inhibitor, sivelestat sodium was reported to improve PaO2/FiO2 in TAAD-OI patients who have undergone MV for more than 96 h and lower white blood cell counts (Figure 3), although sivelestat sodium did not improve postoperative oxygenation index overall. 59 Ulinastatin and sivelestat sodium are both neutrophil inhibitors. However, macrophages also win a place in the course of TAAD-OI and the application of macrophage inhibitors, bindarit, reduced the aggregation of macrophages in lungs and the incidence of TAAD-OI. 60 Presently, neutrophil and macrophage inhibitors alone have shown barely satisfactory results. Perhaps in future, a drug that can inhibit both neutrophils and macrophages or a combination of neutrophil and macrophage inhibitors should be more effective in patients with TAAD-OI.
IL-22
Risk factors and relevant references for type A aortic dissection-related oxygenation impairment; CRP: C-reactive protein; IL: interleukin; TAAD-OI: type A aortic dissection-related oxygenation impairment; BMI: body mass index; ECC: extracorporeal circulation.

Mechanical support
Mechanical life support systems such as MV, extracorporeal membrane oxygenation (ECMO) and continuous renal replacement therapy (CRRT) have helped countless patients since their invention. In this article, we review two systems currently used in TAAD-OI: high frequency jet ventilation (HFJV) and ECMO. MV is an important treatment for severe OI, among which HFJV has been found to be helpful for TAAD-OI. Japanese scientists reported two cases of TAAD-OI treated with HFJV. After treatment, patients’ pulmonary function was improved, pulmonary edema was alleviated and hemodynamics became stable.62,63 Although MV saves the lives of many patients with severe OI, it can lead to complications such as ventilator-related lung injury and increase mortality, where the role of ECMO is particularly important. 64 ECMO is a technique that mainly provides support to patients with severe respiratory or cardiac failure. 65 Initially, the evidence of ECMO for severe OI was not clear, but with the emergence of large RCTs and the practice in 2009 influenza A virus subtype H1N1 and COVID-19,66–68 the importance of ECMO for severe OI was gradually recognized. Two cases of TAAD-OI treated with venous-venous extracorporeal membrane oxygenation (VV-ECMO) were reported by Nanjing Drum Hospital, China. One of the patients reached an improvement of the disease and was successfully removed from ECMO. 69 Tokai University in Japan reported that a patient with TBAD progressed to TAAD. The patient showed no obvious effect after conventional and NO treatment after surgery. VV-ECMO combined with NO treatment was then started on the postoperative day eight, and ECMO was withdrawn 10 days later. After rehabilitation treatment, the patient could finally walk to the outpatient clinic for a follow-up visit. 70 Although there has been reports of successful application of ECMO in TBAD-OI, 71 ECMO for TAAD is rarely reported and controversial because the complexity of surgery and postoperative coagulation dysfunction limit the beneficial effects of ECMO support. In addition, there is a risk of retearing and ECMO will be futile if the underlying malperfusion syndrome is not corrected.72,73 The majority of existing literatures reporting the use of ECMO to support circulatory failure or circulatory failure accompanied with respiratory failure after TAAD are discouraging, especially before surgery.69,72–74 Anyway, VV-ECMO may be a feasible way for simple respiratory failure after TAAD surgery. 75 However, whether the contact with ECMO tubes aggravates the inflammatory response of TAAD needs to be carefully weighed. There may be significant progress in the treatment of TAAD-OI with ECMO in the future, but at present, it calls for more information.
Conclusions
At the moment, TAAD-OI is still a Gordian knot for clinicians no matter before or after surgery and our understanding of this complication is still insufficient, as is research in this field. Current studies have found many risk factors, but they are all based on retrospective studies, and there may be selection bias. At present, the research on the mechanism of inflammation, especially the macrophage-induced inflammatory pathway, has been deepened to a certain extent. Other directions are still in the preliminary speculation or exploration stage and need to be further explored in the future. As for treatment, in our center, it is recommended that patients with TAAD-OI should undergo surgery as soon as possible and clinicians should reduce the time of ECC and operation through skilled techniques. The application of moderate hypothermia technology, combined with related drugs for anti-inflammatory and anti-infection treatment is not a bad idea, as well as inhalation of NO in patients with pulmonary hypertension. Promoting blood protection thus reducing blood products transfusion should also be used; In terms of ventilation strategy, lung protective ventilation strategy should be implemented. If the above methods are still unable to maintain oxygenation, ECMO should be considered as the last way. In other centers, there are drugs and intraoperative organ protection therapies, but to apply the relevant results to the clinic, the safety and reasonable dosage of relevant drugs and technologies need to be verified again, as well as more RCTs or meta-analyses are required, but due to its high lethality, as well as the requirements for surgical equipment and techniques, RCTs may only be performed in top hospitals. So increasing the collection and quality of data may require cross-border cooperation. In the short run, it is an ideal research direction to find and verify risk factors and then identify high-risk patients. On the other hand, in the long term, exploring the pathophysiology of the disease in order to understand it from the perspective of molecular biology and find therapeutic targets is a problem that needs to be solved. However, until this becomes a reality, finding an effective treatment will still be a long way off. Accordingly, it is important to reinforce research interest in this field, investigate the particular components and hazard variables of the illness, and go to early lengths to forestall this entanglement.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
