Abstract
Introduction
The objective of this study was to describe the implementation and outcomes of a protocol outlining angiotensin-II utilization for vasoplegia following cardiac surgery.
Methods
This was a retrospective chart review at a single-center university hospital. Included patients received angiotensin-II for vasoplegia refractory to standard interventions, including norepinephrine 20 mcg/min and vasopressin 0.04 units/min, following cardiac surgery between April 2021 and April 2022.
Results
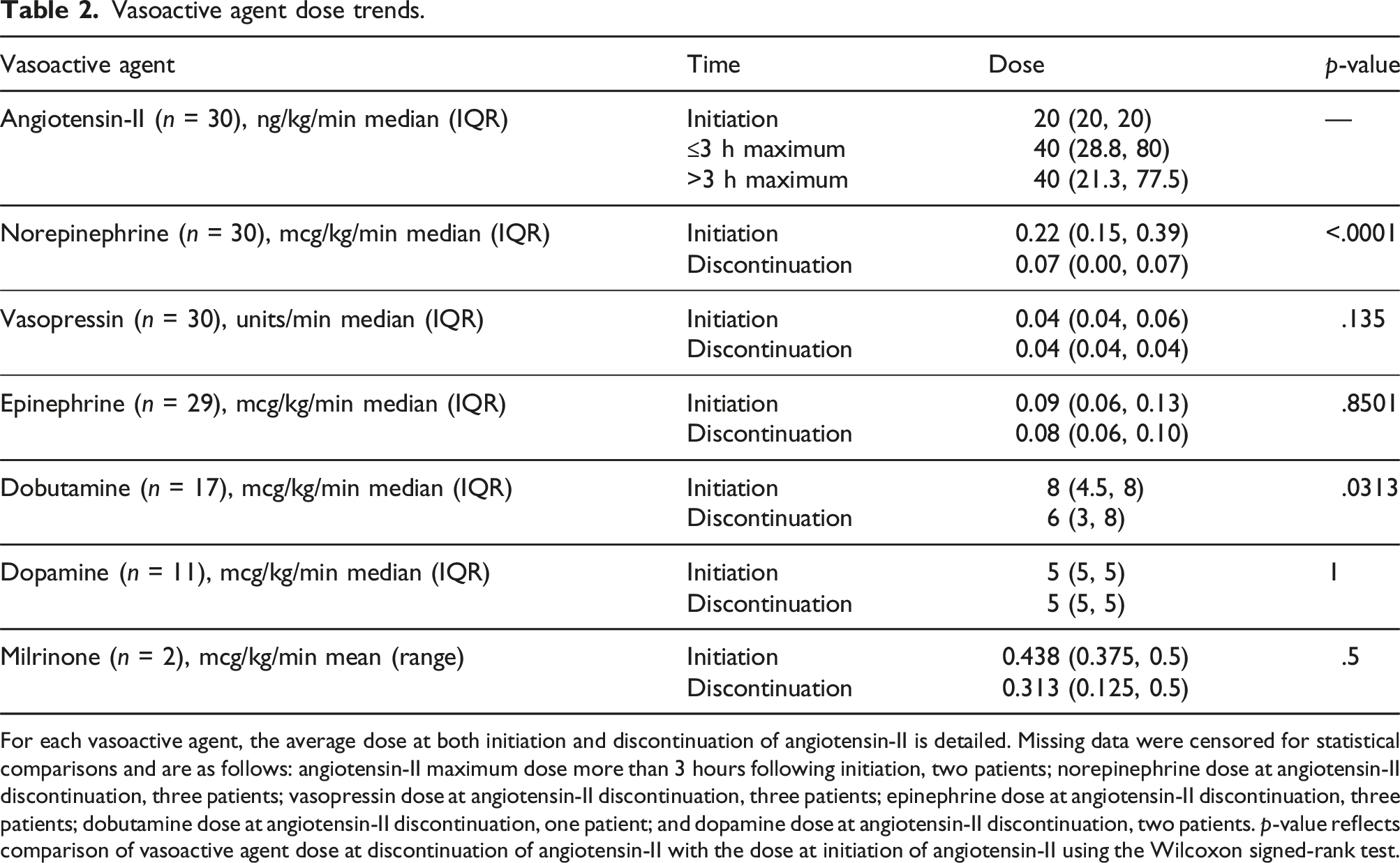
30 patients received angiotensin-II for refractory vasoplegia. Adjunctive agents at angiotensin-II initiation included corticosteroids (26 patients; 87%), epinephrine (26 patients; 87%), dobutamine (17 patients; 57%), dopamine (9 patients; 30%), milrinone (2 patients; 7%), and hydroxocobalamin (4 patients; 13%). At 3 hours, the median mean arterial pressure increased from baseline (70 vs 61.5 mmHg, p = .0006). Median norepinephrine doses at angiotensin-II initiation, 1 hour, 3 hours, and angiotensin-II discontinuation were 0.22, 0.16 (p = .0023), 0.10 (p < .0001), and 0.07 (p < .0001) mcg/kg/min. Median dobutamine doses decreased throughout angiotensin-II infusion from eight to six mcg/kg/min (p = .0313). Other vasoactive medication doses were unchanged. Three patients (10%) subsequently received hydroxocobalamin. Thirteen (43.3%) and five (16.7%) patients experienced mortality by day 28 and venous or arterial thrombosis events, respectively.
Conclusions
The administration of angiotensin-II to vasoplegic patients following cardiac surgery was associated with increased mean arterial pressure, reduced norepinephrine dosages, and reduced dobutamine dosages.
Introduction
Vasoplegia is a frequent complication associated with cardiopulmonary bypass (CPB).1,2 Vasoplegic shock is generally considered vasodilation (systemic vascular resistance [SVR] < 800 dynes-s/cm5) in the setting of physiologic or supraphysiologic cardiac output (cardiac index >2.2 L/min/m2), and end-organ hypoperfusion, occurring within 24 hours of separation from CPB.3,4 Pathophysiologic mechanisms of vasoplegia following CPB are incompletely understood and multifactorial but likely include augmented nitric oxide production, development of acidemia in smooth muscle, reduction of adenosine triphosphate availability, and depletion of circulating angiotensin-II and vasopressin. 4
Standard management of vasoplegia includes attainment of euvolemia, correction of electrolytes and normalization of acid-base status, and titration of vasoactive medications. 4 Scarce evidence informs pharmacologic interventions for vasoplegia refractory to standard interventions, although limited evidence generally supports the efficacy of either methylene blue (ProvayBlue®; Shirley, NY) or hydroxocobalamin (Cyanokit®; West Conshohocken, PA).5–7
Exogenous angiotensin-II (Giapreza®; San Diego, CA) is an emerging therapy for vasoplegia following CPB. Administration of exogenous angiotensin-II directly stimulates smooth muscle contraction to increase mean arterial pressure (MAP). 8 In 2017, the United States Food and Drug Administration approved angiotensin-II to increase blood pressure in adults with septic or other distributive shock on the basis of the Angiotensin II for the Treatment of High-Output Shock (ATHOS-3) trial.8,9 Vasoplegia following CPB occurred in only 16 of the 321 patients. The nine patients treated with angiotensin-II in this subgroup experienced improvement in MAP and reduction in vasopressor requirements consistent with the main results.9,10 Further clinical experience with angiotensin-II for the treatment of vasoplegia following CPB is limited to retrospective reports of heterogenous angiotensin-II utilization to treat a small number of patients.11–21
The purpose of this study is to describe the implementation and outcomes of a protocol outlining angiotensin-II utilization for vasoplegia following cardiac surgery and test the hypothesis that angiotensin-II is associated with MAP increases in real-world practice.
Methods
This was a single center, retrospective, observational study performed by reviewing the medical charts of patients admitted to a tertiary academic medical center. Inclusion criteria in the intervention cohort consisted of adult patients administered angiotensin-II between April 9, 2021 and April 30, 2022. This duration and resultant sample size was selected to permit internal review of angiotensin-II utilization by the local Pharmacy and Therapeutics committee. Patients administered angiotensin-II for an indication other than vasoplegia following cardiac surgery were excluded. The study was approved by the local Institutional Review Board (study number 211,443 on August 2, 2021).
In April 2021, our institution implemented a protocol designating angiotensin-II as the preferred treatment modality for refractory vasoplegia associated with cardiopulmonary bypass. This protocol defines refractory vasoplegia as the presence of MAP <65 mmHg despite achievement of euvolemia, optimization of cardiac function, correction of electrolytes and acid-base status, empiric treatment of adrenal insufficiency, and titration of standard vasoactive medications (specifically, norepinephrine ≥20 mcg/min and vasopressin ≥0.04 units/min). Assessment of hypovolemia included comparison of current with baseline central venous and pulmonary arterial pressures, thermodilution cardiac output, mixed venous oxygen saturation, lactate, measurement of chest tube output every 15 min, and, when indicated, echocardiography measure to assess for obstructive vs hypovolemic shock. Confirmation of adequate cardiac function was attained by agreement between two of the following criteria: 1) a cardiac index of >2.2 L/min/m2, 2) a mixed venous oxygen saturation of >65%, and 3) transthoracic or transesophageal echocardiography imaging report describing the right (fractional area change) and left ventricular function (ejection fraction). Upon request of the primary service to initiate angiotensin-II, a protocol-designated anesthesiologist intensivist reviews the patient case and approves or denies the utilization of angiotensin-II (Figure 1). Angiotensin-II enrollment criteria.
At our institution, angiotensin-II dosage and titration is similar to ATHOS-3. 9 Angiotensin-II is initiated at 20 ng/kg/min and titrated by 10 ng/kg/min every 5 minutes to a maximum dose of 80 ng/kg/min for the first 3 hours and a maximum dose of 40 ng/kg/min thereafter. The continuous infusion is titrated by anesthesiologists in the operating room and nursing staff in the cardiovascular intensive care unit to achieve a MAP ≥65 mmHg. Our protocol requires the initiation of chemical thromboprophylaxis (i.e. subcutaneous heparin) as soon as clinically appropriate. Of note, our institution does not systematically insert pulmonary artery catheters for advanced hemodynamic monitoring.
The medical chart of each patient in the intervention cohort was reviewed to extract demographic information, procedural characteristics, and post-operative management data. The primary outcome was MAP 3 hours following initiation of angiotensin-II. Norepinephrine and vasopressin requirements were recorded at initiation, 1 hour following initiation, 3 hours following initiation, and at discontinuation of angiotensin-II. Other vasopressor and inotrope doses were recorded at initiation and discontinuation of angiotensin-II. Mortality was recorded as patient expiration by 28 days following angiotensin-II infusion. Death during the acute resuscitation was defined as death while angiotensin-II was infusing.
Trends in institutional utilization of methylene blue or hydroxocobalamin for CPB-associated vasoplegia were evaluated and compared during the intervention time period and a historical approximately 6-month control time period (September 28, 2020 through April 8, 2021). The control time period dates were selected to minimize confounding due to the coronavirus disease 2019 pandemic, allow one-half the number of days as compared to the intervention time period, and evaluate institutional practice immediately preceding protocol implementation. Inclusion in either time period consisted of adult patients administered methylene blue or hydroxocobalamin. Exclusion consisted of administration of methylene blue or hydroxocobalamin for indications other than vasoplegia following CPB. Evaluation of characteristics beyond methylene blue or hydroxocobalamin use in the control cohort was beyond the scope of this analysis.
Patient data were summarized utilizing descriptive statistics including count, percentage, mean, standard deviation, median, and interquartile range, as appropriate. MAP and vasoactive medication doses at various time intervals were compared to baseline using the Wilcoxon signed-rank test. The assumption of normality was determined by the Shapiro-Wilk test. Trends in institutional utilization of methylene blue or hydroxocobalamin for CPB-associated vasoplegia were compared using Fisher’s exact test. Statistical significance was achieved with a p-value of <.05. Statistical analyses were conducted in SAS version 9.4 (Cary, NC). Data may be available upon reasonable request to the primary investigator.
Results
Thirty-one patients received angiotensin-II during the study period. One patient received angiotensin-II for septic shock and was excluded. 30 patients were included for further review.
Baseline characteristics.

DVT: deep vein thrombosis; PE: pulmonary embolism; IABP: intra-aortic balloon pump; VA-ECMO: venoarterial extracorporeal membranous oxygenation; LVAD: left ventricular assist device; RVAD: right ventricular assist device; CVP: central venous pressure; PA: pulmonary artery; SVR: systemic vascular resistance; CI: cardiac index.
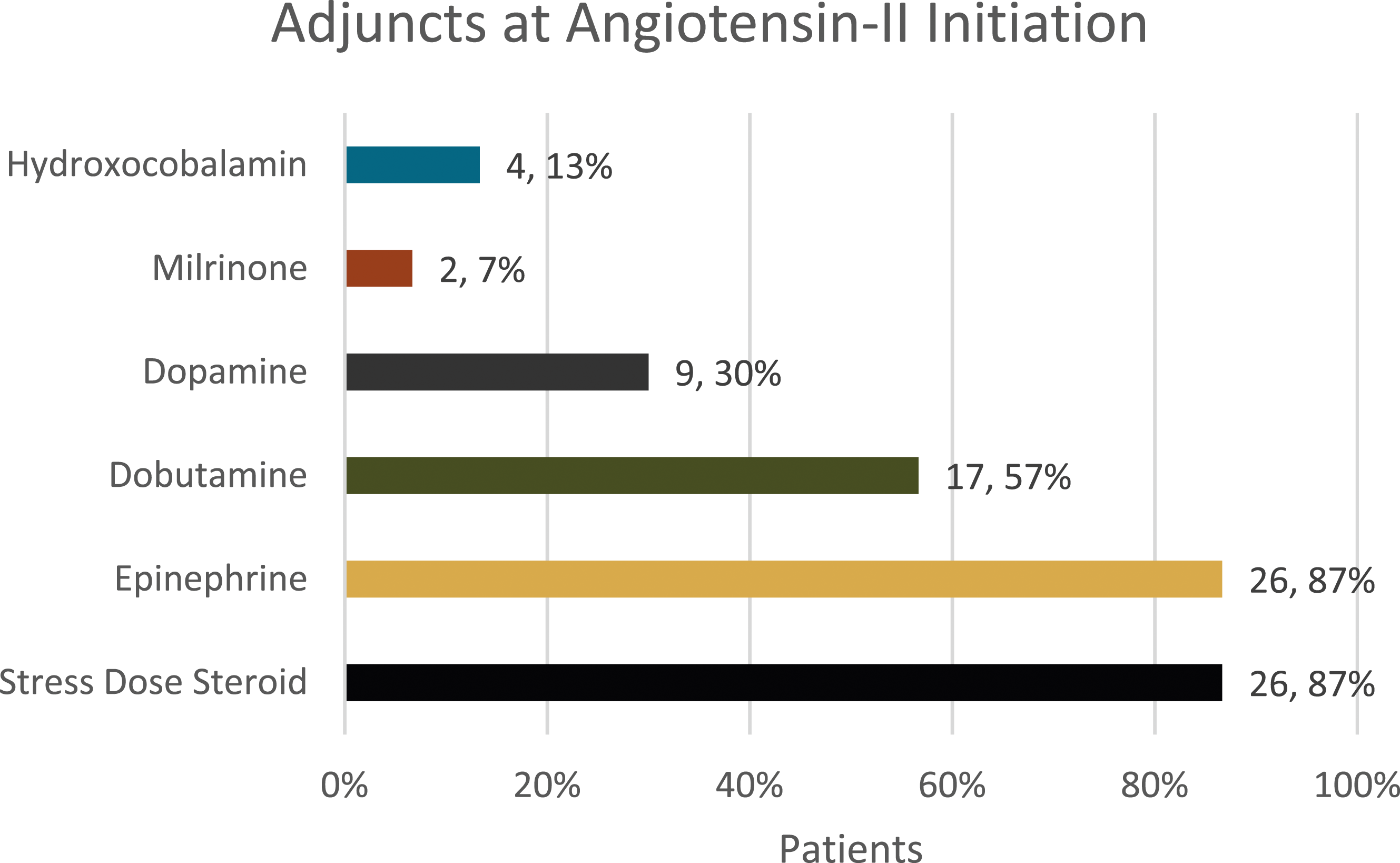
Adjuncts to norepinephrine and vasopressin at initiation.
Angiotensin-II was associated with a reduction in concurrent vasoactive medication requirements. Median norepinephrine requirements at initiation, 1 hour, 3 hours, and angiotensin-II discontinuation were 0.22, 0.16 (p = .0023), 0.10 (p < .0001), and 0.07 (p < .0001) mcg/kg/min, respectively (Figure 3). Median dobutamine requirements significantly decreased from eight to six mcg/kg/min (p = .0313) from initiation to discontinuation of angiotensin-II, respectively (Table 2). Other vasoactive medication requirements were unchanged. Three (10%) patients were subsequently administered hydroxocobalamin for persistent vasoplegia (Table 3). There was no methylene blue use. Norepinephrine continuous infusion dosages charted at various time points relative to angiotensin-II continuous infusion initiation. The lower and upper borders of each box represent the 25th and 75th percentile of norepinephrine dosage, respectively, at that time point. The median norepinephrine dosage at each time point is represented by the middle horizontal line. The lower and upper whiskers represent the minimum and maximum values, respectively, of non-outliers. Outlier norepinephrine dosages are represented by extra dots. At the 3 hour time point, data were missing for two patients (28 measurements represented). At discontinuation of angiotensin-II, data were missing for three patients (27 measurements represented). These data were censored during individual Wilcoxon signed-rank tests. **p < .01 ***p < .001 Hr: hour. Vasoactive agent dose trends. For each vasoactive agent, the average dose at both initiation and discontinuation of angiotensin-II is detailed. Missing data were censored for statistical comparisons and are as follows: angiotensin-II maximum dose more than 3 hours following initiation, two patients; norepinephrine dose at angiotensin-II discontinuation, three patients; vasopressin dose at angiotensin-II discontinuation, three patients; epinephrine dose at angiotensin-II discontinuation, three patients; dobutamine dose at angiotensin-II discontinuation, one patient; and dopamine dose at angiotensin-II discontinuation, two patients. p-value reflects comparison of vasoactive agent dose at discontinuation of angiotensin-II with the dose at initiation of angiotensin-II using the Wilcoxon signed-rank test. Surrogate endpoints and clinical events. Data not available or not applicable for all patients for the cardiac index and oxygen saturation, urine output, and lactate endpoints. CI = cardiac index; SvO2 = venous oxygen saturation; ScvO2 = central venous oxygen saturation.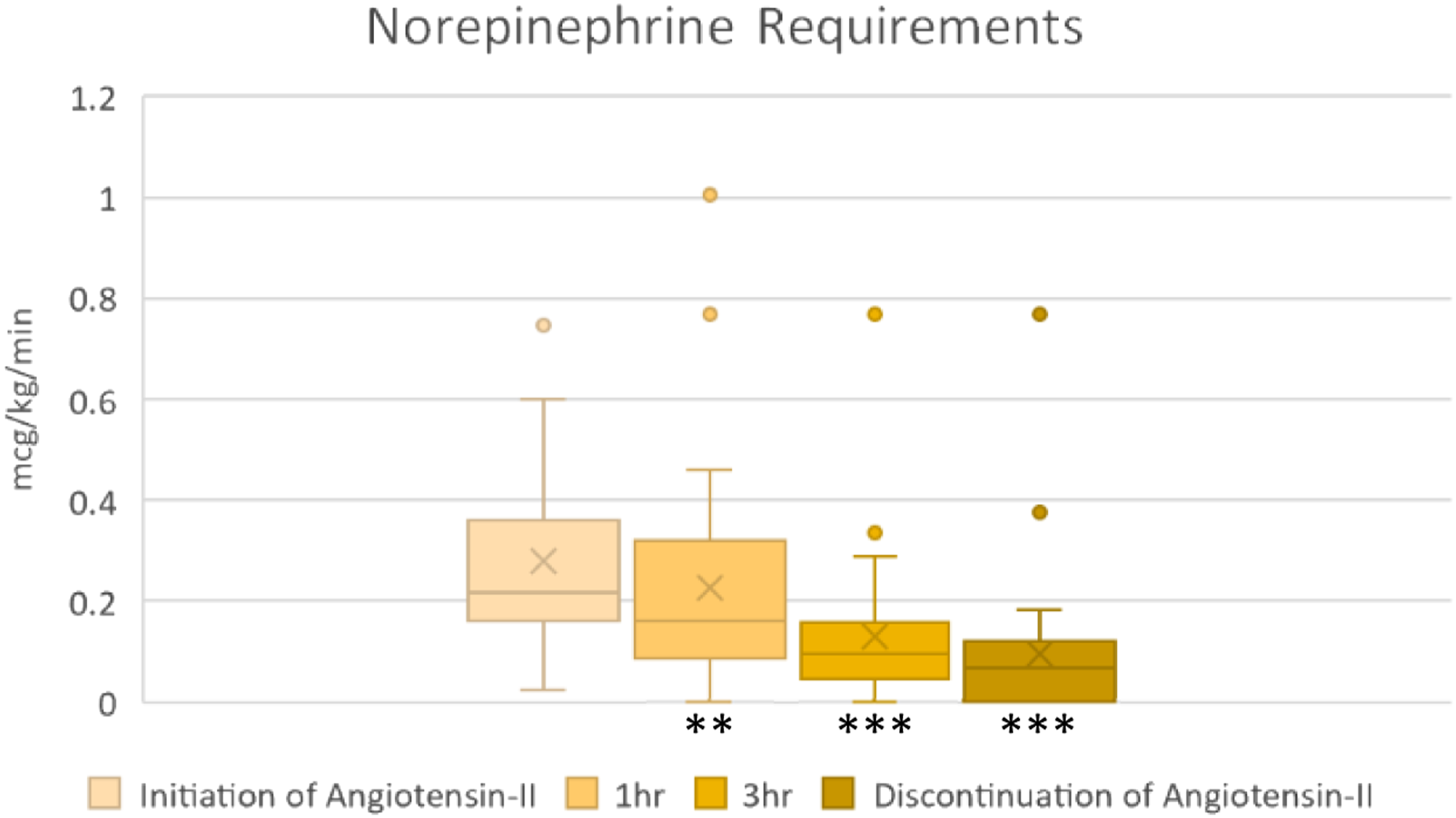

Furthermore, there was an association with improvement in hemodynamic parameters. Three hours following angiotensin-II initiation, the median MAP significantly increased from 61.5 to 70 mmHg (p = .0006). Twenty-seven (90%) patients achieved MAP >65 mmHg while angiotensin-II was infusing. Twenty-two (88%) patients achieved a cardiac index ≥2.0 L/min/m2, venous oxygen saturation >60%, or central venous oxygen saturation >70% during angiotensin-II infusion.
Thirteen (43.3%) patients expired by day 28. All three patients who did not achieve a cardiac index ≥2.0 L/min/m2, venous oxygen saturation >60%, or central venous oxygen saturation >70% during angiotensin-II infusion expired by day 28. Four (13.3%) patients expired during the acute resuscitation period. The cause of death was heterogenous across the cohort. Venous or arterial thrombosis during the hospitalization following angiotensin-II occurred in five (16.7%) patients, and chemical thromboprophylaxis was initiated in 26 (86.7%) patients a median of 0.5 days following angiotensin-II initiation. Thromboprophylaxis was initiated prior to the identification of thrombosis in all patients who developed thrombosis. None of the thrombotic events were considered life-threatening. All patients who experienced thrombotic events received prothrombin complex concentrate. While angiotensin-II was infusing, urine output >0.5 mL/kg/hour was achieved in 11 (52.4%) patients and lactate normalized in five (20.8%) patients (Table 3).
Finally, introduction of the angiotensin-II protocol was associated with similar institutional utilization of methylene blue or hydroxocobalamin for CPB-associated vasoplegia. During the intervention time period, 17 patients of the 1440 (1.2%) CPB cases were administered either methylene blue or hydroxocobalamin, whereas, during the control time period, 16 patients of the 655 (2.4%) CPB cases were administered either methylene blue or hydroxocobalamin (p = .0573, Supplementary Table 1). The proportion of patients receiving treatment for vasoplegia with methylene blue, hydroxocobalamin, or angiotensin-II between the intervention and control time periods was unchanged (3.0% vs 2.4%, p = .4810).
Discussion
This report describes implementation of a vasoplegia protocol outlining angiotensin-II administration following cardiac surgery and the associated hemodynamic changes, vasoactive medication dose adjustments, clinical outcomes, and changes in institutional use of methylene blue or hydroxocobalamin following CPB. Angiotensin-II for post-operative vasoplegia was associated with a significant reduction in norepinephrine and dobutamine requirements and an increase in MAP. Despite this, the rate of 28-days mortality was 43.3%, corroborating the high mortality rate associated with vasoplegia. 22
Administration of angiotensin-II for post-CPB vasoplegia is attractive, given its mechanism of action. As angiotensin converting enzyme is expressed predominantly in the lung capillaries, exclusion of blood flow through the pulmonary circuit during CPB contributes to decreased circulating angiotensin-II and development of post-operative vasoplegia.23,24 Pre-operative renin-angiotensin-aldosterone system (RAAS) inhibition is a known risk factor of post-operative vasoplegia. 25 Subsequent increases in renin concentrations to upregulate RAAS further supports this pathophysiologic process.11,26 Angiotensin-II binds to receptors on vascular smooth muscle cells to stimulate contraction, clinically translating to an increased blood pressure. 8 Administration of angiotensin-II to vasoplegic patients following CPB has been demonstrated to reduce plasma renin concentrations, suggesting reversal of the dysfunctional RAAS in the pathophysiology of post-operative vasoplegia.11,26
Similarly to the ATHOS-3 substudy evaluating angiotensin-II for vasoplegia following CPB, our study demonstrated angiotensin-II was associated with both a significant MAP increase from 61.5 to 70 mmHg and a significant norepinephrine dose decrease from 0.22 to 0.10 mcg/kg/min at hour three. This study provides value in addition to previous reports in that it consists of a real-world population, includes a relatively large number of patients treated with angiotensin-II, and describes the outcomes of a specific protocol for angiotensin-II utilization. Therefore, this study provides a framework for angiotensin-II implementation at other cardiac surgery centers.
While the cumulative data suggest angiotensin-II increases MAP while decreasing vasopressor requirements, a known risk of angiotensin-II is thrombosis. The rate of arterial and venous thrombotic events in the ATHOS-3 study in patients treated with angiotensin-II was 13% compared with 5% of patients treated with placebo. 8 To address the risk of thrombosis, our local protocol requires the implementation of chemical thromboprophylaxis as soon as clinically appropriate. Despite utilization of chemical thromboprophylaxis in 86.7% of patients an average of 0.5 days following initiation of angiotensin-II, the incidence of venous or arterial thrombosis in the present study was similar to the thrombosis rate in ATHOS-3 at 16.7%. All patients who experienced thrombosis in the present study received prothrombin complex concentrate, which is a known risk factor for thrombosis.27,28 While the initiation of chemical thromboprophylaxis in angiotensin-II-treated patients may address the risk of thrombosis, consideration must be given to the individualized bleeding risk given the potential of worsened outcomes associated with major bleeding. 29 The patient population in the present study was at an increased risk for both thrombosis and bleeding given the presence of mechanical circulatory support in one-half of patients at the time of angiotensin-II initiation. This study adds to the limited data that exist regarding angiotensin-II utilization in a mechanical circulatory support population.12,15,16,19,20,30,31
The presence of vasoplegia is a risk factor for mortality.22,32 In the present study, despite implementation of the protocol for angiotensin-II use, 43.3% of patients expired by day 28. In the ATHOS-3 study, the rate of 28-days mortality in angiotensin-II treated patients was 46%, which was not different from placebo. 9 Of note, in the present study, only four patients expired during the resuscitation period while angiotensin-II was infusing. The median survival time of the remaining 10 expired patients was 195 h (interquartile range 105 to 295 h). The impact of angiotensin-II on mortality in a cardiac surgical population warrants further investigation.
There is evidence of associated increased risk with the administration of methylene blue and hydroxocobalamin.33–36 A relevant question is whether implementation of angiotensin-II as the preferred treatment of vasoplegia following cardiac surgery may reduce institutional utilization of methylene blue or hydroxocobalamin. In the present study, the implementation of this protocol was associated with a reduction in utilization of methylene blue or hydroxocobalamin for CPB-associated vasoplegia by one-half, though statistical significance was not achieved. To our knowledge, this study is the first to suggest a replacement of methylene blue or hydroxocobalamin with angiotensin-II for vasoplegia following CPB in real-world practice. This finding is of clinical significance as it reflects the possible substitution of therapies supported by low-quality evidence, or with notable safety concerns, for a therapy known to increase MAP relatively safely as demonstrated in a randomized controlled trial. While promising, prospective studies evaluating the relative efficacy and safety of angiotensin-II compared to methylene blue or hydroxocobalamin are lacking.
This study has several important limitations. The retrospective nature of the study relies on medical record documentation, which could have implications in outcomes such as vasoactive medication dosages and MAP changes. The lack of a comparator group precludes comparative evaluation of the outcomes of angiotensin-II administration with alternative interventions for refractory vasoplegia, such as methylene blue and hydroxocobalamin. Comparison of the outcomes of vasoplegic patients treated during the intervention and control time periods is beyond the scope of this analysis. We cannot exclude a component of selection bias, as methylene blue and hydroxocobalamin were available to clinicians as therapeutic modalities for refractory vasoplegia during the study period. As some patients received interventions such as hydroxocobalamin, there is the potential for confounding. Additionally, overlap may exist between post-cardiotomy cardiogenic shock and vasoplegia in the subset of patients on mechanical circulatory support due to 15 (50%) patients receiving mechanical circulatory support at the initiation of angiotensin-II. While the majority of patients had a mean cardiac index of 2.7 L/min/m2 and mean SVR 669.8 dynes-s/cm5 which would favor a vasoplegic state, due to the retrospective nature of the study, this overlap cannot be fully excluded. As a single-center study including 30 patients, results may be of limited generalizability to centers with differences in baseline patient characteristics, operative procedures, or post-operative management strategies. Despite the implemented local protocol, there was likely heterogeneity in individualized patient management, such as titration of concomitant vasoactive agents. Although average baseline hemodynamic parameters were consistent with vasoplegia, 17 (57%) patients were receiving vasodilating inotropes dobutamine or milrinone at the time of angiotensin-II initiation. While this was a consequence of routine institutional use of inotropic medications following cardiac surgery, this confounds the results. Finally, we did not evaluate the impact of pre-operative RAAS inhibitor medication use on response to angiotensin-II, which remains an area for further research.
In conclusion, the administration of angiotensin-II to vasoplegic patients following cardiac surgery was associated with both an increase in MAP and a reduction in norepinephrine and dobutamine dosages, which validates randomized controlled trial findings in real-world practice. Furthermore, implementation of a protocol outlining angiotensin-II as the preferred treatment modality for refractory vasoplegia was associated with a clinically relevant reduction in institutional utilization of methylene blue or hydroxocobalamin. Opportunities for further research regarding angiotensin-II include mortality impact in a larger sample, quantifying thrombotic risk in a cardiac surgery population, and comparative effectiveness with methylene blue and hydroxocobalamin.
Supplemental Material
Supplemental Material - Angiotensin-II for vasoplegia following cardiac surgery
Supplemental Material for Angiotensin-II for vasoplegia following cardiac surgery by Andrew J Johnson, William Tidwell, Andrew McRae, C Patrick Henson, Antonio Hernandez in Perfusion.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
