Abstract
Introduction
Acute type A aortic dissection (ATAAD) is one of the most critical emergencies in cardiovascular surgery. Additional complications such as organ malperfusion can significantly decrease the chances of survival. Despite promptly performed surgical treatment, impaired organ perfusion may persist, thus close postoperative monitoring is recommended. But, is there a surgical consequence due to the existence of a preoperatively known malperfusion and is there a correlation between pre-, peri- and postoperative levels of serum lactate and proven malperfusion?
Methods
Between 2011 and 2018, 200 patients (66% male; median age: 62.5 years; interquartile range: +/−12.4 years) that received surgical treatment at our institution for an acute dissection DeBakey type I were enrolled in this study. The cohort was divided into two groups according to the preoperative existence of malperfusion and non-malperfusion. At least one kind of malperfusion occurred in 74 patients (Group A: 37%), while 126 patients (Group B: 63%) showed no evidence of malperfusion. Furthermore, lactate levels of both cohorts were differentiated into four periods: preoperative, intraoperative, 24 hours after surgery, and 2–4 days after surgery.
Results
The patients’ status differed significantly prior to surgery. Group A (malperfusion) showed an elevated requirement for mechanical resuscitation (A: 10.8%; B: 5.6%;
Conclusions
Preexisting malperfusion due to ATAAD may significantly increase the chance of early mortality in patients with ATAAD. Serum lactate levels were a reliable marker for inadequate perfusion from admission until day 4 after surgery. Despite this, early intervention survival in this cohort remains limited.
Keywords
Introduction
Acute type A aortic dissection (ATAAD) is one of the most critical emergencies in cardiovascular surgery. The diagnosis of ATAAD requires clinical indications as well as radiographic evidence of an aortic dissection. An intimal tear in the ascending aorta extends into the aortic wall media to create a false lumen and a dissection flap. This can result in a severe decrease in organ perfusion (Figure 1). Even without peripheral malperfusion, ATAAD remains a life-threatening disease. Additional complications such as organ malperfusion can significantly decrease the chance of survival. Due to this fact, the rapid restoration of flow into the true lumen is necessary and surgical aortic repair remains the gold standard. However, clinical signs of the extent of a compromised perfusion are typically not well presented at later stages. Nevertheless, despite promptly performed surgical treatment, organ malperfusion may persist, thus postoperative monitoring should be closely performed. Therefore, reliable biomarkers such as serum lactate have been suggested as potential indicators for detecting persistent ischemia and increased early mortality.1–4 3D CTA reconstruction; ATAAD with malperfusion left renal artery and right common iliac artery.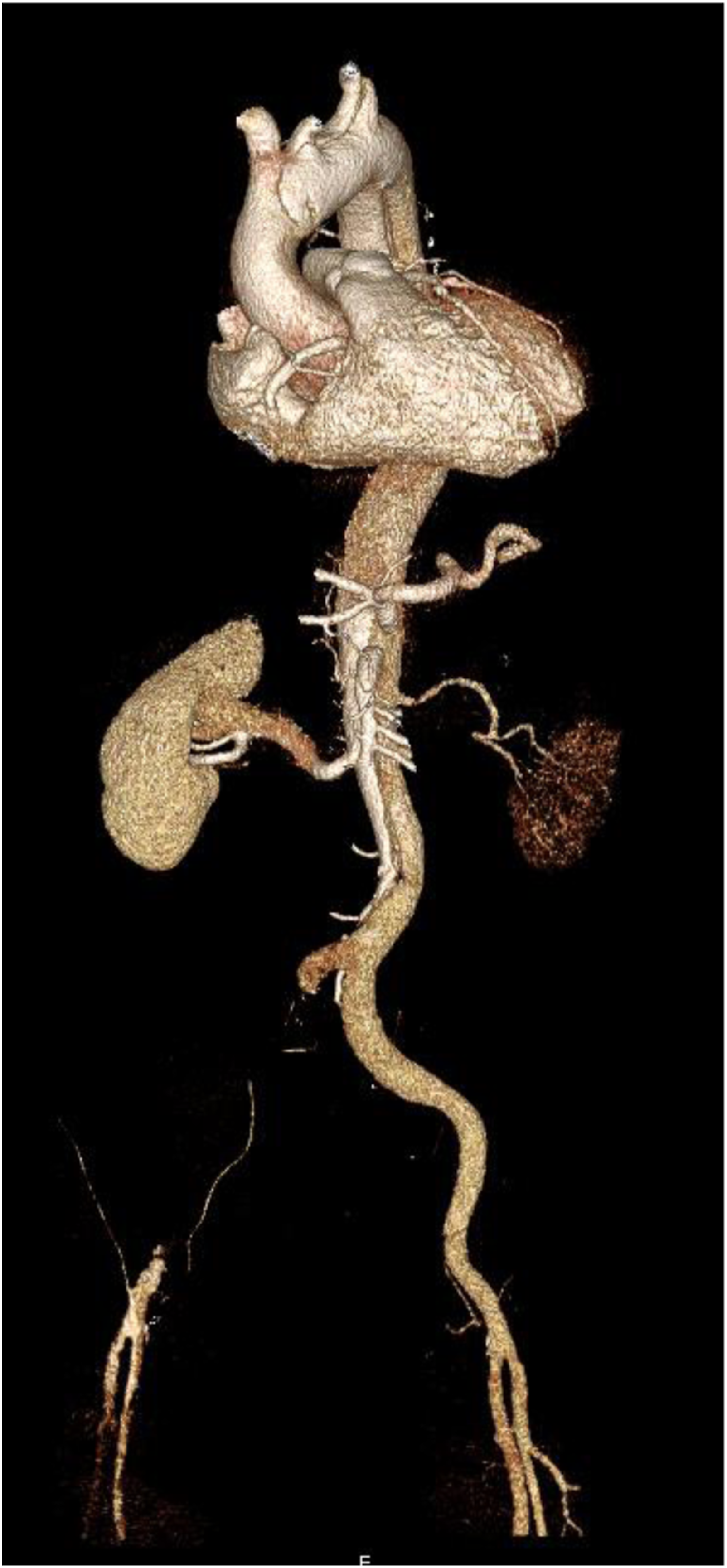
We evaluated the surgical consequences resulting from the existence of a preoperatively known malperfusion. In addition, pre- and postoperative levels of serum lactate were verified for a possible correlation with proven malperfusion.
Methods
Definitions
Clinical signs, including abdominal or back pain, were observed, and signs of malperfusion or neurological symptoms may also occur as unspecific symptoms. The radiographic equivalent of an ATAAD was either the existence of a dissection membrane starting in the ascending aorta or an intramural hematoma inside the aortic wall. Malperfusion was defined as an occlusion or a false lumen perfusion of one relevant artery per organ. We characterize end-organ malperfusion according to imaging findings and clinical signs of malperfusion. Furthermore, the diagnosis of malperfusion was defined according to Sievers et al. 5 M2: Dissection of at least 1 supra-aortic vessel or aortic arch true lumen collapse with (M2+) or without (M2-) clinical symptoms of cerebral (stroke) or upper extremity (pulse deficit, pain, pallor, paraesthesia) malperfusion; M3: dissection or false lumen origin of at least 1 visceral, renal or 1 iliac artery or aortic true lumen collapse entailing functional closure of at least 1 visceral, renal or iliac artery offspring, with (M3+) or without (M3-) clinical symptoms of bowel (abdominal pain, ileus, bloody diarrhoea), kidney (anuria, renal failure signs) or lower extremity (pulse deficit, pain, pallor, paraesthesia) ischaemia. 5 Therefore, patients with stages M2 and M3
((−), (+)) were assigned to the malperfusion group. The presence of extension of the aortic dissection, the entry location and the dissection of the aorta, supra-aortic, visceral and iliac arteries were analysed according to a CTA. To differentiate lactate levels according to the anatomic location we further distinguished the cohort into cerebral, visceral, renal, and limb malperfusion. Furthermore, lactate levels of both cohorts were differentiated into four periods: preoperative, intraoperative, 24 hours after surgery, and 2–4 s after surgery. During these time periods, the highest value of serum lactate was recorded for each period.
Patients with severe neurologic symptoms at admission, like hemiplegia, dysarthria, or apraxia, without a performed cerebral CT scan and with postoperative evidence of stroke were assigned to the preoperative stroke cohort. Postoperative stroke had to be confirmed by magnetic resonance imaging or computed tomography. Accidently induced ATAAD during open heart surgery is referred to as iatrogenic dissection. Dissections of coronary arteries were either preoperatively discovered during coronary angiography or intraoperatively visible.
Study population and study design
Individual consent was obtained from all patients to allow for further follow-up examinations. In this study, we included 200 patients (66% male; median age: 62.5 years; interquartile range: +/−12.4 years) that received surgical treatment at our institution between 2011 and 2018 due to an ATAAD. DeBakey type II + III dissections as well as chronic dissections were not included.
Patients characteristics.
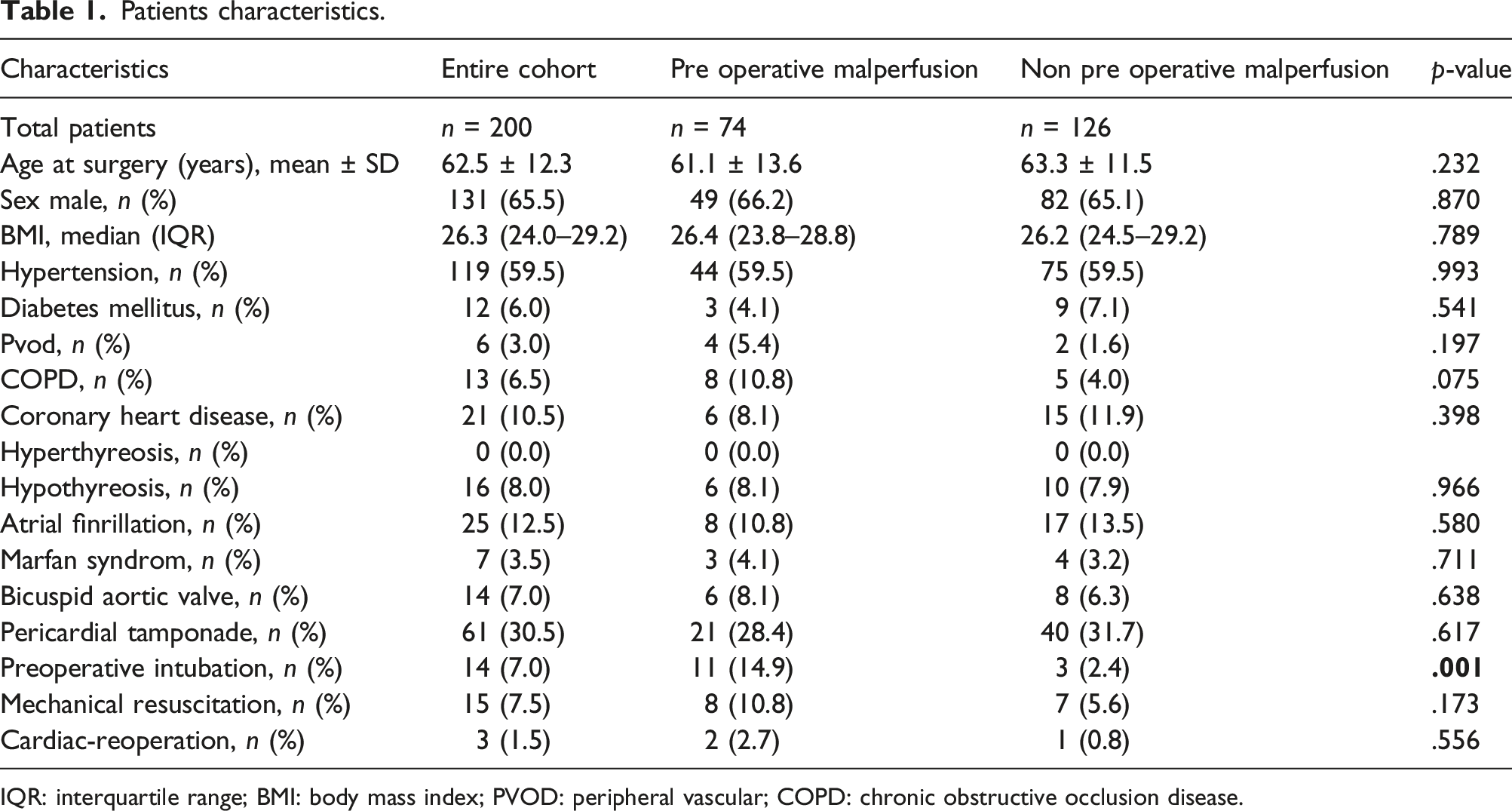
IQR: interquartile range; BMI: body mass index; PVOD: peripheral vascular; COPD: chronic obstructive occlusion disease.
Preoperative data.
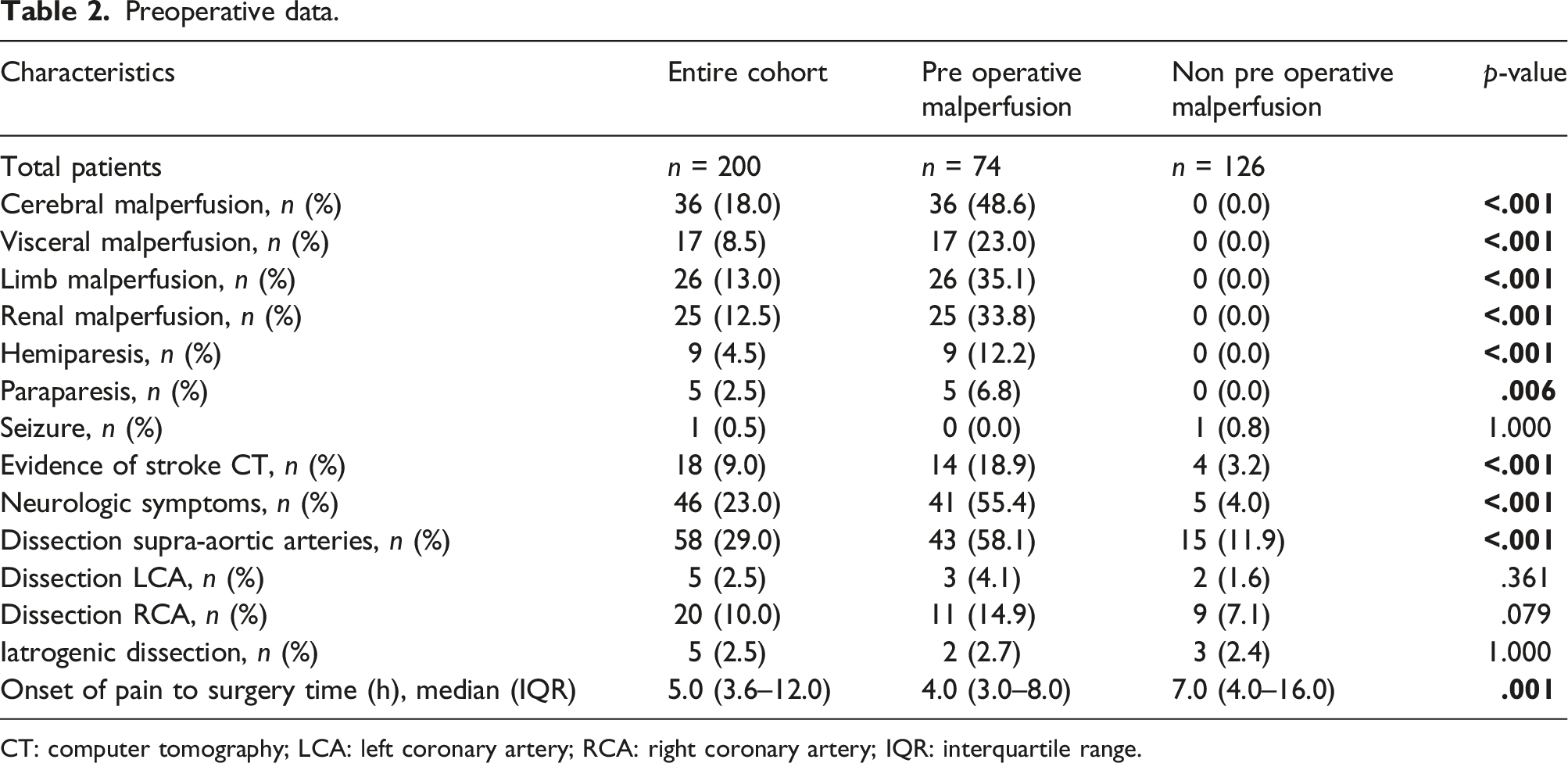
CT: computer tomography; LCA: left coronary artery; RCA: right coronary artery; IQR: interquartile range.
Follow-up
We obtained informed consent from patients to allow the collection of follow-up data. The clinical follow-up ended and was completed in November 2021. Patients were seen frequently in our aortic outpatient clinic. Imaging via magnetic resonance or computed tomography angiography was performed regularly after surgery and during follow-up.
Perioperative management and surgical technique
According to our standard protocol, all patients with acute ATAAD were transferred to the operation theater directly after first diagnosis. To avoid decompensation, intubation was not performed before anesthetic and surgical preparations were completed. After median sternotomy, extracorporeal circulation (ECC) was established. Our cannulation technique of the ascending aorta, also in ATAAD, was previously published by our group.6,7 We prefer a direct cannulation of the ascending aorta after identifying the true lumen using transesophageal echocardiography. The left side of the heart was vented through the right superior pulmonary vein. The aorta was clamped, also in ATAAD patients. In all dissections, cardioplegia was administered directly into the coronary ostia. Blood cardioplegia was our preferred method of myocardial protection. According our root-first policy we performed root/ascending aortic repair while cooling the patient to a nasopharyngeal temperature of 22–26°C. Other concomitant procedures (e.g., CABG) were performed if necessary. Cardioplegia was repeated approximately every 30 min. 7 Furthermore, in patients with extended arch repair we implemented a beating-heart technique in 2010. 8 In all cases, either a proximal, subtotal (involving replacement of the brachiocephalic trunk), or total arch replacement with ET or FET, hypothermic circulatory arrest (temperatures between 22°C and 26°C) and bilateral selective antegrade cerebral perfusion was performed. The application of SACP varied when a limited arch repair was performed.
Extended arch repair
From 2000–2010, the FET technique was performed using a custom-made Chavan-Haverich prosthesis followed by a prefabricated Chavan-Haverich hybrid graft9,10 (Curative GmbH, Dresden, Germany). From 2005 until 2010, the Jotec E-vita hybrid graft was used.
11
Supra-aortic vessels were reattached using the island technique (
Proximal arch repair
An isolated replacement of the proximal aortic arch was performed using established straight Dacron grafts.
Statistical analysis
SPSS 27 Statistics software (IBM Corp. released 2020; IBM SPSS Statistics for Windows, Version 27.0; Armonk, NY: IBM Corp.) was used for the data analysis. A normal distribution of variables was calculated using the Kolmogorov–Smirnov test. Categorical variables are stated as absolute numbers (
Results
Preoperative patient characteristics
Preoperative patient characteristics showed no significant difference regarding age (A: 61.1 y; B: 63.3 y;
Intraoperative data
Intraoperative data.
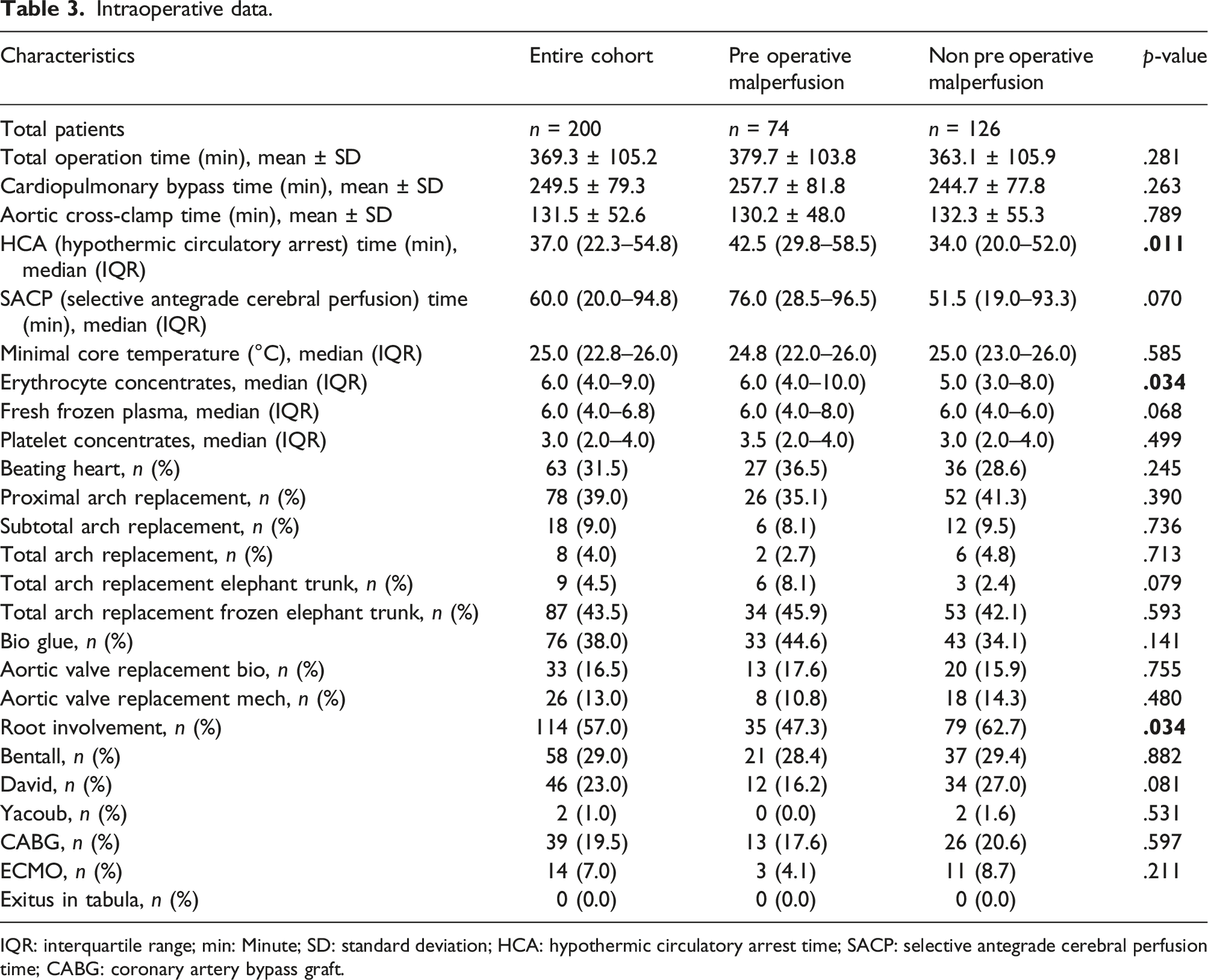
IQR: interquartile range; min: Minute; SD: standard deviation; HCA: hypothermic circulatory arrest time; SACP: selective antegrade cerebral perfusion time; CABG: coronary artery bypass graft.
Postoperative data
Postoperative data.
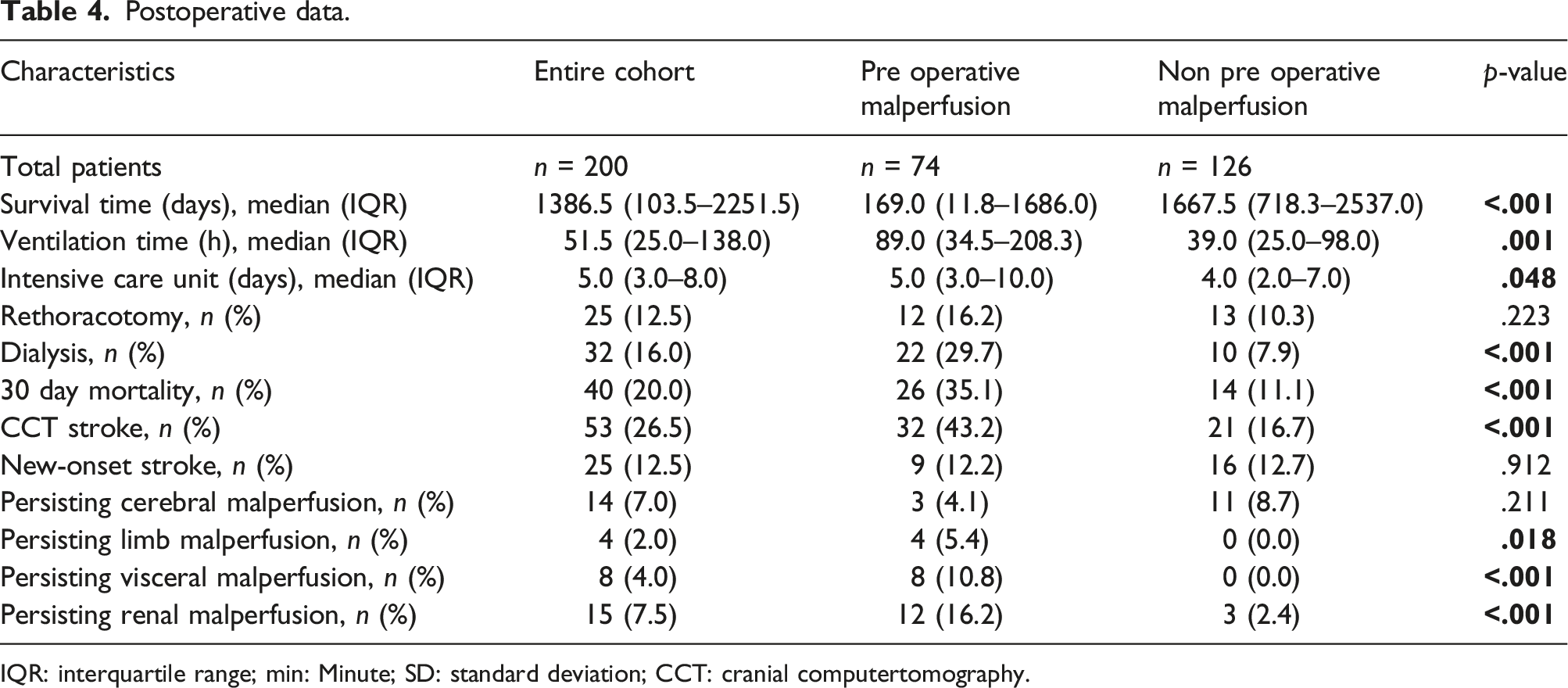
IQR: interquartile range; min: Minute; SD: standard deviation; CCT: cranial computertomography.
Long-term follow-up data
Follow up data.
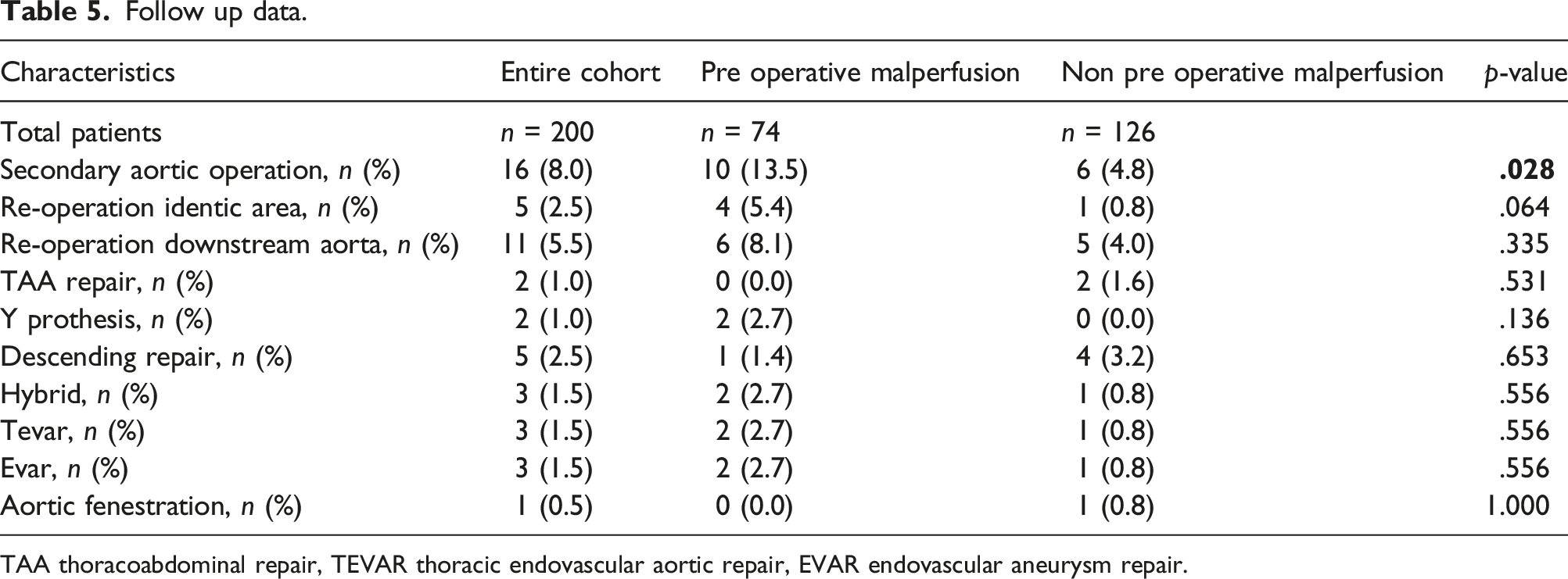
TAA thoracoabdominal repair, TEVAR thoracic endovascular aortic repair, EVAR endovascular aneurysm repair.
Survival: Kaplan-Meier curves showing survival with and without preoperative cardiac tamponade. The
Lactate levels
Serum lactate levels in mmol/l (millimole per liter) over time. Preoperative, intraoperative, 24 hours after surgery, day 2–4 after surge.

Discussion
Acute type A aortic dissection is a manifold and life-threating disease. In the present study we distinguished patients with ATAAD regarding the existence (Group A) or absence (Group B) of preoperative malperfusion. Furthermore, the relevance of serum lactate levels as a marker for malperfusion was evaluated. The opinion that malperfusion is an independent risk factor is well established and evidence-based.14–16 Furthermore, Bennet et al. 17 assume a correlation between increased serum lactate levels and one-year mortality. However, a simple assimilation of elevated lactate levels with malperfusion would be fatal. Hyperlactemia may also be caused by hypoperfusion during cardiogenic shock in a compromised cohort. 18 However, is there an adjustment in perioperative management or surgical treatment due to proven malperfusion? And is serum lactate really a reliable marker for malperfusion during the decision-making process?
The patients’ conditions at admission to our hospital differed significantly between the groups. The malperfusion cohort fulfilled further independent risk factors,
19
such as an increased incidence of preoperative intubation (A: 14.9%; B: 2.4%;
In keeping with the higher number of dissected supra-aortic arteries with proven malperfusion, perioperative data show a significant longer median SACP time in Group A (A: 76 min (28.5–96.5 min); B: 51.5 min (19.0–93.3 min);
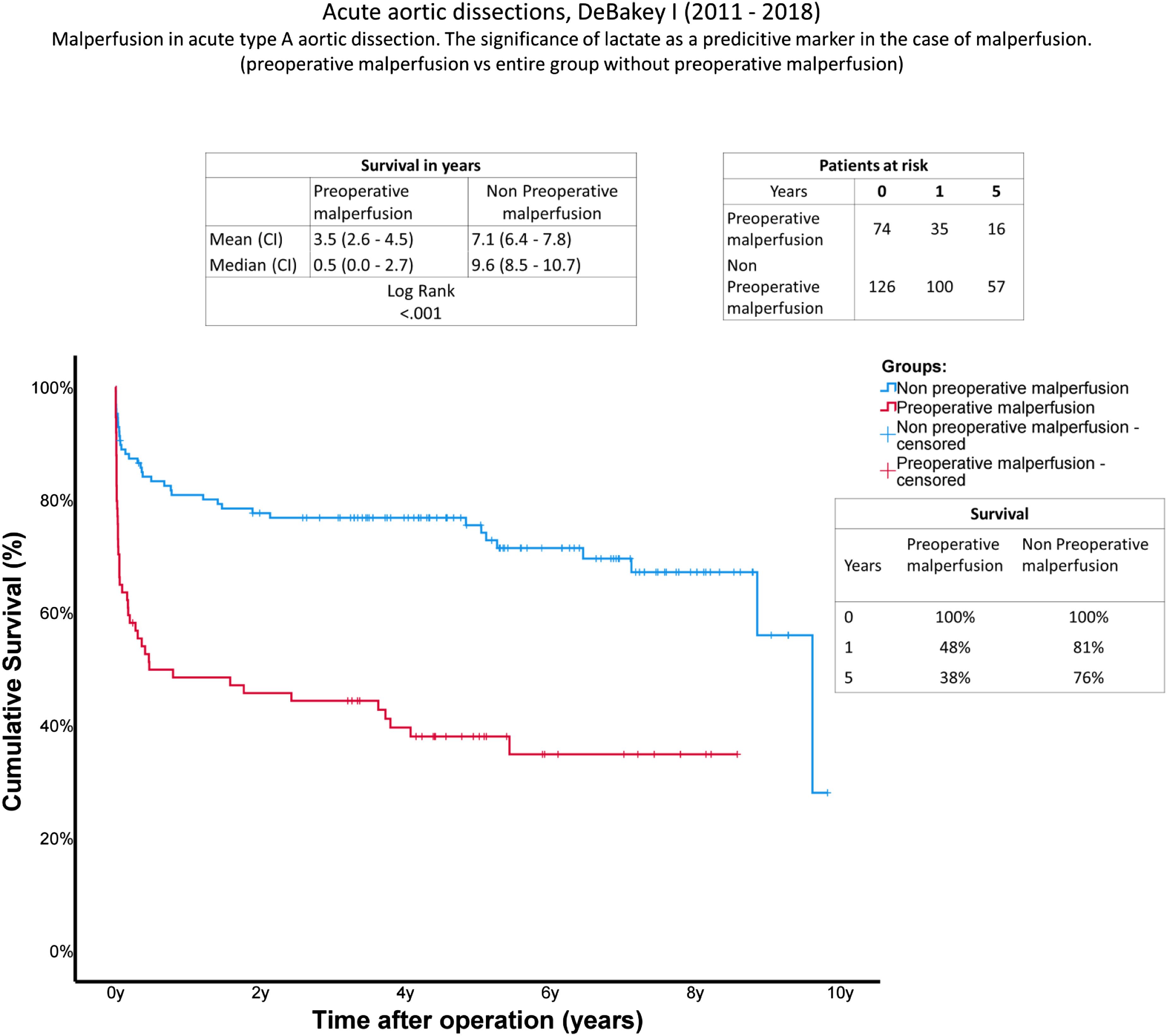
Patients with preoperative existing malperfusion fulfilled all the criteria for a limited postoperative outcome compared to the control group without malperfusion. Median survival time (A: 169.0 d; B: 1667.5 d;
Postoperative visible (remaining) dissections also occurred in Group B. A new-onset cerebral malperfusion showed in 11 patients, and a new impairment of the renal perfusion in three patients. Ether dissection membrane proceeded between the first CT scan and surgery, or even preoperatively. All patients with remaining cerebral malperfusion received further interventions for the re-establishment of perfusion.
Despite the fact that both groups received a balanced extent of surgical procedures, the malperfusion cohort showed a significantly higher requirement for secondary aortic surgery (A: 13.5%; B: 4.8%;
It is known that malperfusion is an evidence-based risk factor for an increased mortality rate 23 and that lactate levels are a reliable predictor for mortality after the repair of ATAAD. 1 Furthermore, in comparison to existing publications, our study monitored lactate levels from the preoperative period until 2–4 days after surgery. Elevated serum lactate levels were significant at all time points in the malperfusion group. According our data, even early-stage surgical intervention in patients with malperfusion results in increased serum lactate levels from the preoperative period until day 4 after surgery.
Results of long-term survival using Kaplan-Meier curves confirmed an increased mortality rate during the early postoperative phase. If malperfusion occurs, the chance of survival is significantly reduced during the first year after surgery. Mortality rate is comparable with the control group one year after surgery (Figure 2). These are resonable data due to the fact that either acute or remaining malperfusion results mainly in acute organ failure.
Despite the proven existence of malperfusion an adjustment of the surgical therapy has not been taken place. Accordingly, further evaluations of the surgical procedure is mandatory. Goldberg et al. 24 evaluated the interesting option of a “Reperfusion First” - Concept using surgical approaches like central repair, fenestration and direct revascularization. Nevertheless, the prompt aortic repair remains the gold standard.
Limitations
This is a retrospective study and thus it carries all the potential risks and biases linked to studies of this nature. Furthermore, the individual decisions regarding the surgical procedures were based on the surgeon’s experience. Between the years 2008 and 2018, a total of 19 surgeons performed the operative treatment of these patients. Surgical skill levels may vary in this cohort. Furthermore, the diagnosis of a low cardiac output syndrome was not really evaluated in patients with ATAAD. Preoperative echocardiography data are not available for this population with a high urgent requirement for surgery. Only a minority of the patients presented malperfusion of one isolated perfusion area. For the further assignment of elevated lactate levels, multicenter studies are necessary. Furthermore, a relevant number of patients that died prior to admission can be expected.
Conclusion
In this study, we identified patients with preexisting malperfusion due to ATAAD as a severely compromised cohort with significantly increased chances of early mortality. This cohort prematurely presents strong clinical symptoms with the consequence of a reduced onset of pain to surgery time. In all cases of malperfusion, serum lactate levels were a reliable marker for inadequate perfusion from admission until day 4 after surgery. Despite this, early intervention survival in this cohort is limited. It can be assumed that survival is comparable in cases of malperfusion after surviving the vulnerable perioperative phase and the first year after surgery.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
