Abstract
Introduction
To validate slaughterhouse hearts for ex-situ heart perfusion studies, we compared cold oxygenated machine perfusion in less expensive porcine slaughterhouse hearts (N = 7) to porcine hearts that are harvested following the golden standard in laboratory animals (N = 6).
Methods
All hearts received modified St Thomas 2 crystalloid cardioplegia prior to 4 hours of cold oxygenated machine perfusion. Hearts were perfused with homemade modified Steen heart solution with a perfusion pressure of 20–25 mmHg to achieve a coronary flow between 100–200 mL/min. Reperfusion and testing was performed for 4 hours on a normothermic, oxygenated diluted whole blood loaded heart model. Survival was defined by a cardiac output above 3 L with a mean aortic pressure above 60 mmHg.
Results
Both groups showed 100% functional survival, with laboratory hearts displaying superior cardiac function. Both groups showed similar decline in function over time.
Conclusion
We conclude that the slaughterhouse heart can be used as an alternative to laboratory hearts and provides a cost-effective method for future ex-situ heart perfusion studies.
Keywords
Introduction
With the reintroduction of donation after circulatory death (DCD) heart donation,1,2 both clinical and experimental research in ex-situ heart perfusion has intensified. A downside of experimental heart perfusion studies is the use of hearts that are harvested from laboratory (LAB) animals, of which the porcine heart is most commonly used. With the current focus on refinement, reduction and replacement in animal studies (the 3R’s), 3 alternatives to sacrifice laboratory animals are desired. The slaughterhouse (SLH) porcine heart can be used as an alternative source for ex-situ heart perfusion studies as their access is unlimited without sacrificing animals solely for research goals and thereby reducing experimental costs. The SLH-hearts have been used previously for similar studies. 4 Although an alternative, the use of slaughterhouse porcine hearts comes at a cost which is seen in terms of higher variability due to different farms of origin, stress due to transport and loading and a fatting diet.5,6 Furthermore, animals are 6 months old and around 120 kg as they are bred for human consumption, which also results in more hypertrophic hearts compared to experimental animals, and might lead to a larger variation in results obtained. Moreover, the harvesting of the SLH heart itself carries important implications. As regulations for the slaughter process require the sedation of animal prior to exsanguination, 7 the pig is electrically stunned and harvesting of the heart can only start after exsanguination of the animal. This results in an extended period of warm ischemia with potentially more severe ischemia reperfusion injury (IRI) upon reperfusion, which is more significant in hypertrophic hearts. 8 These factors together may impact myocardial function, but the exact effect in the slaughterhouse setting is unclear.
The harvesting procedure of the heart in LAB animals is in great contrast to SLH animals, since in LAB animals the heart can be harvested in the most optimal way. Animals undergo anesthesia prior to sternotomy followed by a controlled cardioplegic arrest of the heart. This procedure minimizes warm ischemia and prevents right ventricular distension and is known as the golden standard of heart harvesting. 9
In the current study, we compare cold oxygenated machine-perfused porcine slaughterhouse hearts to cold oxygenated machine-perfused porcine hearts that are harvested following the golden standard in laboratory animals in order to validate the slaughterhouse model for heart perfusion studies. The aim was to show that slaughterhouse hearts approach functional outcome and survival.
Material and methods
Slaughterhouse animals (SLH)
7 hearts were harvested from Dutch Landrace Hybrid pigs slaughtered for human consumption. The protocols were consistent with the EC regulations 1069/2009 regarding slaughterhouse animal material for diagnosis and research as supervised by the Dutch Government (Dutch Ministry of Agriculture, Nature and Food Quality) and were approved by the associated legal authorities of animal welfare (Food and Consumer Product Safety Authority).
Heart harvesting
Animals were electrically stunned, hung, exsanguinated, and declared dead prior to harvesting the heart. Death was determined if no reflex was seen with eye stimulus after draining at least 2 L of blood. No heparin was administered to the animal due to applicable regulations. Blood (3–3.5 L) was collected in a bag with 5000 IU/L heparin as anticoagulant. After parasternal incision, the heart and lungs were harvested en-bloc by transecting the trachea, the aorta distally from the arch vessels and the caval veins. Topical cooling was applied while the aorta was cannulated and modified St Thomas 2 crystalloid cardioplegia was administered at a pressure of 80–100 mmHg. 4 Warm ischemic time recorded ranged between the stunning of the animal to the delivery of cardioplegia.
Laboratory animals (LAB)
6 hearts were harvested from surplus laboratory specific pathogen free Topig Norsvin pigs, mean weight 86 kg ± 21.5. As the body weight of laboratory animals is normally lower than the weight of slaughterhouse animals, a weight difference between those groups was unavoidable. All personnel was trained according to the EU Directive 2010/63/EU on the protection of animals used for scientific purposes, with adequate knowledge on hygiene measures to prevent zoonosis.
Anesthesia
Animals received premedication (midazolam intramuscular 0.4 mg/kg, ketamine intramuscular 10 mg/kg and atropine intramuscular 0.05 mg/kg) followed by thiopenthal (intravenous 4 mg/kg) for induction of anesthesia after which the animals were intubated. Anesthesia was maintained by midazolam (intravenous 1 mg/kg/h) and nimbex (intravenous 0.6 mg/kg/h).
Heart harvesting
The thorax and pericardium were opened. The pulmonary artery was dissected from the aorta and the root needle was inserted. 25.000 IU/L heparin was administered and after clamping the aorta, 1 L modified St Thomas cardioplegia crystalloid solution (added: Heparin 5000 IU/L) was administered with a target pressure of 100 mmHg and topical cooling was applied. The inferior caval vein was transected to unload the right atrium and the heart was excised.
Blood collection
As draining of 3.0–3.5 L of blood in the laboratory animals was impossible, the required amount of blood was collected from a SLH pig as mentioned before to assess the function of the heart on the PhysioHeart™ platform as described below.
Group difference
The difference in the animals per group were due to uncertainty of COMP settings in one heart. To secure statistical analysis another heart was added to the SLH group. After thorough analysis of the COMP perfusion data, we decided the heart could be used for analysis.
Cold oxygenated machine perfusion (COMP)
All hearts were stored with cold oxygenated machine perfusion (COMP) for 4 hours (Figure 1). The heart box system (XVIVO Perfusion, modified Kidney Assist transport box) consists of a reservoir, a pulsatile driven rotary pump (60BPM) and control unit. The heart box was assembled, primed with homemade modified Steen heart solution at 4°C (Supplementary Table 1), and de-aired carefully. The temperature was regulated and maintained around 8°C by placing ice around the reservoir. The machine was set to a pressure of 5 mmHg to maintain low flow before the heart was connected in the slaughterhouse. Schematic representation of the study design.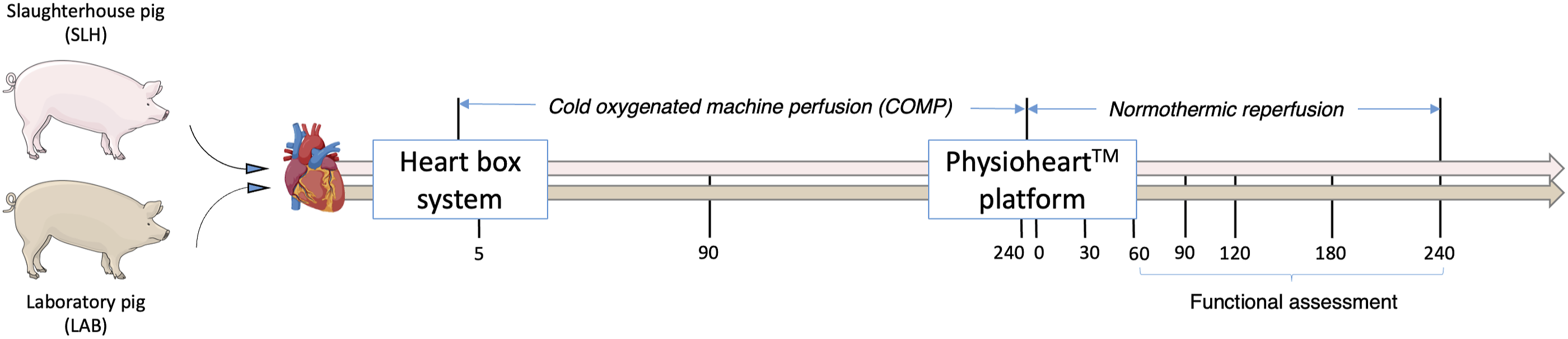
The aorta was cannulated and de-aired carefully while the heart was submerged in preservation solution. Perfusion pressure was set to 20–25 mmHg with a target flow of 100–200 mL/min; all hearts met these criteria and were transported to our laboratory. The hearts were perfused for 4 hours including preparations for the PhysioHeart™ model.
Working heart model - PhysioHeart™
The PhysioHeart™ platform enables loading of the left side of the heart, which allows for functional analysis of left ventricular function. The platform can thus be seen as a surrogate to transplantation, which is more expensive and complex to perform in an experimental setting. Reperfusion of the heart on the PhysioHeart™ platform has been previously described. 10 The system contains a pre- and afterload chamber and has the possibility to perfuse both in Langendorff and working mode. The system was primed with autologous heparinized diluted whole blood in the SLH group and with non-autologous heparinized diluted whole blood in the LAB group (diluted with modified Krebs Henseleit Buffer) to obtain 5 L final volume with 26% ± 2.41 hematocrit (Supplementary Table 2). The blood was kept at 38°C and physiological blood gas parameters were maintained using a gas mixture of oxygen and carbon dioxide with an FiO2 of 25%. The perfusate was supplemented with glucose and insulin to maintain physiological levels.
The left atrium, aorta and pulmonary artery were cannulated and connected to the system, while both caval veins and azygotic vein were closed with a suture. The hearts were then perfused in Langendorff mode with a target aortic pressure of 70–80 mmHg. Shortly after restoring blood flow to the myocardium, irregular contractions were observed, and the hearts were defibrillated with 30–50 J. If heartrate was less than 80 BPM, ventricular pacing using temporary pace leads with a target rate of 100 BMP (Medtronic Inc., Minneapolis, MN, USA) was initiated. A recovery perfusion interval of 60 minutes in Langendorff mode was performed prior to switching to working mode by applying preload to the heart by filling of the left atrium and left ventricle thereby enabling cardiac output (CO). The left atrial pressure (LAP) was kept between 10 and 20 mmHg and the afterload was adjusted to create a physiological setting of CO and mean aortic pressure (MAP). 4 Inclusion criteria for further analysis were a CO >3.0 L with a LAP <20 mmHg despite dobutamine administration. If these criteria were met, hearts were maintained in working mode for another 3 hours and function was assessed at specified time points (90, 120, 180, and 240 minutes).
Data collection
Functional data
Functional data was recorded for 10 seconds with 1000 Hz to create 10,000 data points per time point. Directly recorded data included: time in seconds, cardiac output (CO), coronary flow (CF), left ventricular pressure (LVP), left atrial pressure (LAP) and mean aortic pressure (MAP). With LVP and time, maximum developed pressure (dP/dT max) and minimum developed pressure (dP/dT min) were calculated. To correct for the difference in heart weights, CO and CF were divided by the heart weight, which resulted in cardiac index (CI) and coronary flow index (CFI). Survival was defined by a CO >3 L with a MAP >60 mmHg and a LAP <15 mmHg.
Bloodgas analysis
Bloodgas analysis was performed with a VetScan iSTAT 1 bloodgas analyzer (Abaxis, Union City, CA, USA) with CG8 + cartridges (Abbott, Chicago, IL, USA). Analysis was performed during hypothermic perfusion (5, 90, and 240 minutes), after priming of the PhysioHeart™ setup and during normothermic perfusion (10, 30, 60, 90, 120, 180, and 240 minutes)
Perfusate samples
Perfusate samples were collected during COMP and normothermic testing in Lithium-Heparin (BD Vacutainer plastic Lithium-Heparin tube) and Serum tubes (BD vacutainer plastic serum tube), centrifuged at 2300G (2 × 10 minutes), aliquoted and stored at −80°C. Samples were collected during hypothermic perfusion (5, 90, and 240 minutes) and normothermic perfusion (baseline, 30, 60, 90, 120, 180, and 240 minutes). As weight could impact the results, variables analyzed were normalized to the mean weight of 450 g of all hearts included (variable was divided by the weight of the heart and multiplied by the mean weight of 450 g).
Statistical analysis
Data analysis was performed using SPSS (IBM SPSS Statistics 26.0.0.1 (32bit)). Data is presented in mean ± standard deviation (SD) and median with interquartile range, as applicable. Differences between groups were calculated either with student t-test or Mann-Whitney U depending on the data distribution. A mixed ANOVA was used to analyze the impact of time and a Kaplan-Meyer curve with a log rank test was performed to detect differences in survival. A p-value below 0.05 was found significant. Correlations were determined by a Pearson correlation coefficient.
Results
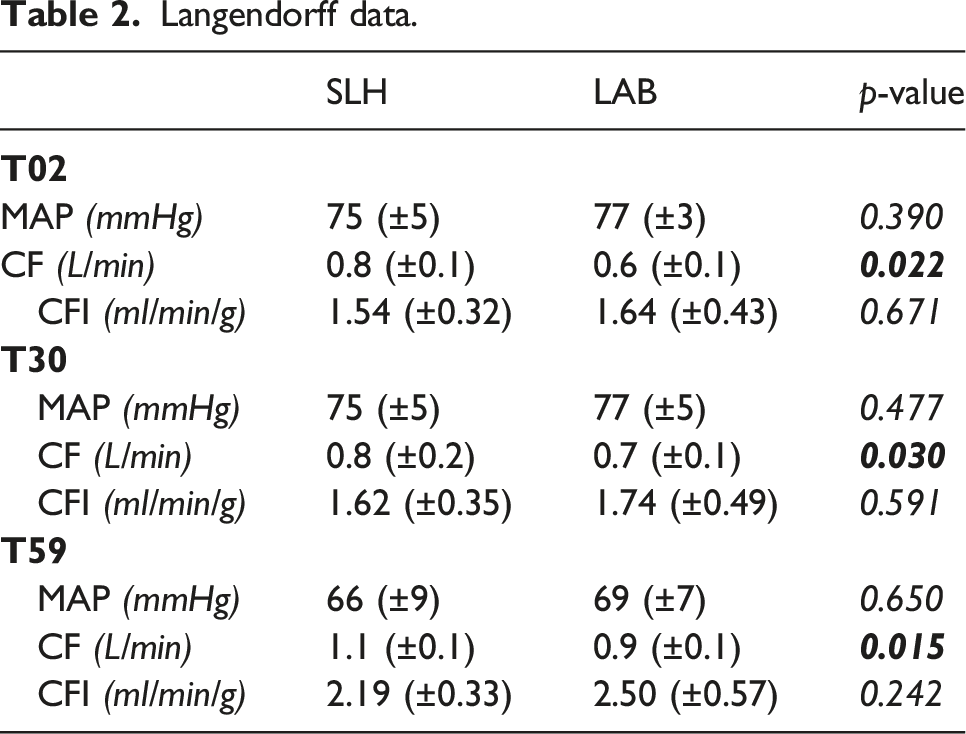
Warm ischemic time, baseline heart weight and Langendorff data
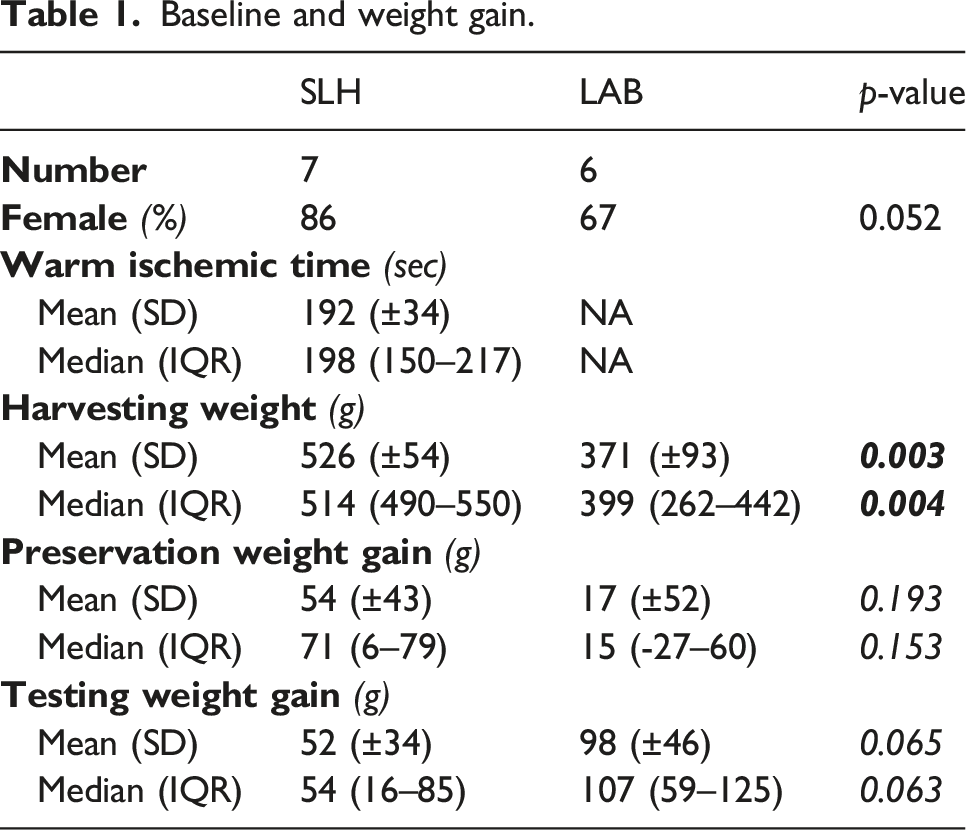
Baseline and weight gain.
Perfusate samples during COMP
Lactate remained significantly lower in LAB hearts at 240 minutes (1.0 (±0.2) vs 4.9 (±0.8) p = 0.000). This was also observed for lactate dehydrogenase and ammonia, whereas troponin levels did not differ. Oxygen levels decreased over time, with a steeper decline in the SLH hearts (Figure 2). Perfusate samples during cold oxygenated machine perfusion. SLH: slaughterhouse group; LAB: laboratory animal group; * ≤ 0.05; ** ≤ 0.001.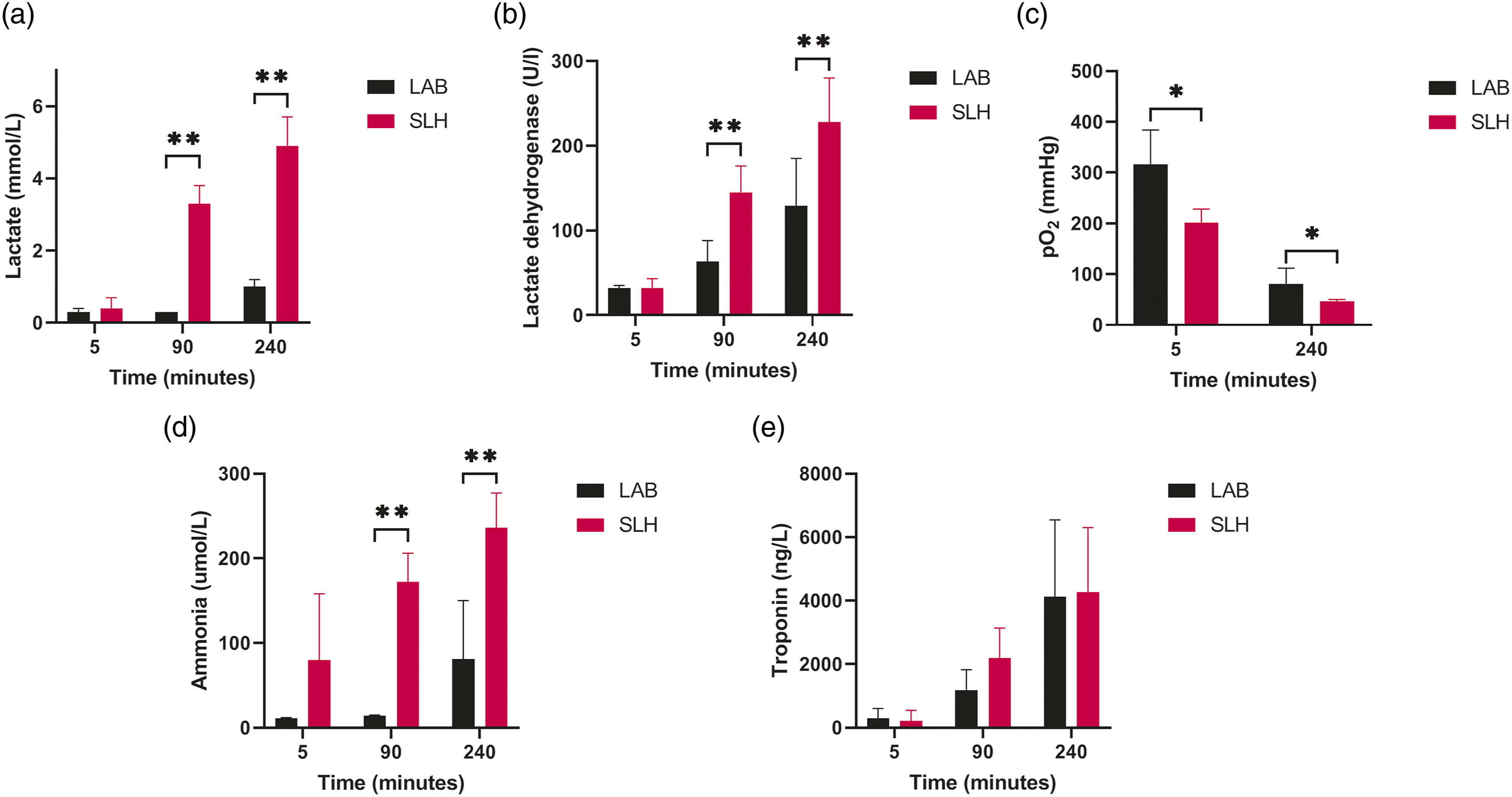
Lactate levels at 240 minutes of COMP correlated with the CI at 240 minutes of functional testing (r = −0.868, p = < 0.000) (Figure 3). Correlation plot for lactate at 240 minutes of cold oxygenated machine perfusion and cardiac index at 240 minutes of functional testing.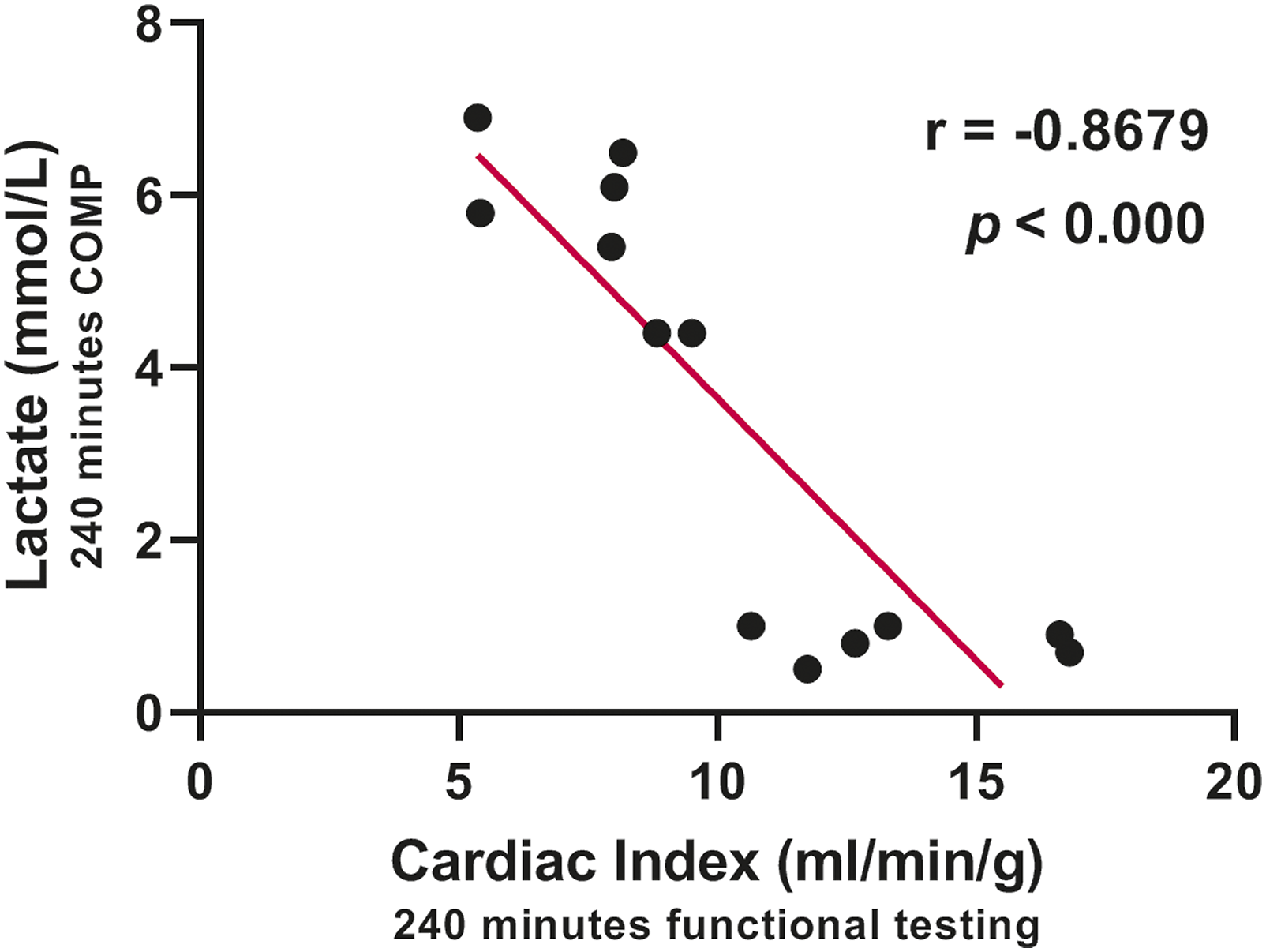
Baseline blood gas PhysioHeart™
No statistical differences were observed in most blood gas parameters at the start of normothermic perfusion on the PhysioHeart™ platform besides pO2 (LAB 139 (±31) vs SLH 176 (±25), p = 0.040) and potassium (LAB 4.4 (±0.3) vs SLH 4.0 (±0.3), p = 0.030) (Supplementary Table 2). One could argue whether these difference at the start of reperfusion are clinically relevant.
Langendorff mode
Langendorff data.
Perfusate samples during physioHeart™
No differences were seen in lactate levels during normothermic testing, although lactate dehydrogenase was higher in the SLH group. Ammonia showed a trend towards higher levels in the LAB group, whereas no difference was found in troponin levels between groups (Figure 4). Perfusate samples during normothermic perfusion and testing. SLH: slaughterhouse group; LAB: laboratory animal group; * ≤ 0.05.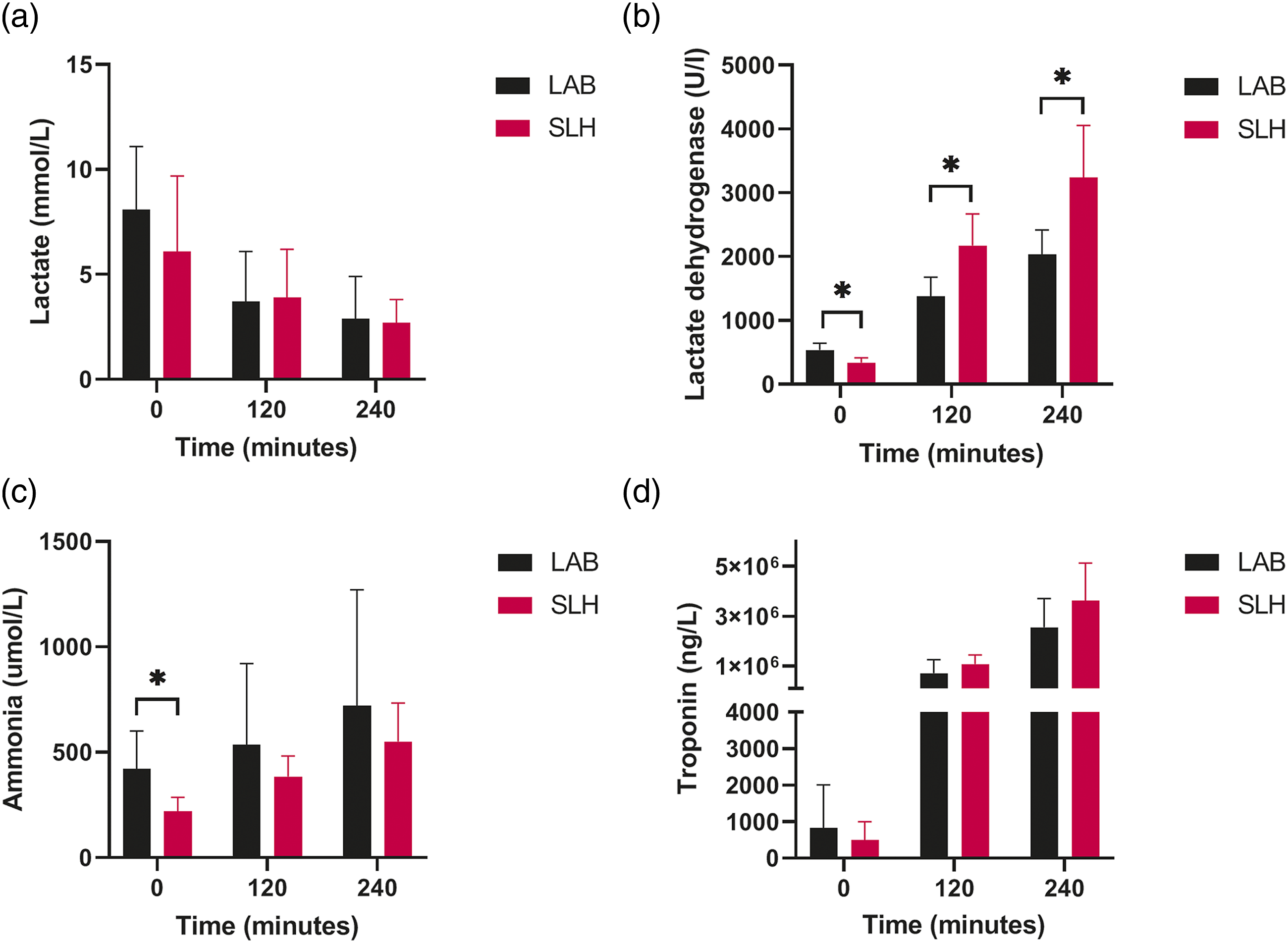
Functional survival
Functional survival was similar in both groups, but LAB hearts showed significant better CI, CFI, LAP and developed pressures (Figure 5). Mixed ANOVA showed a significant time effect in functional decline and significant differences between groups for CO, CI, developed pressures, LAP, CFI and MAP (Supplementary Table 3). Functional outcome. SLH: slaughterhouse group; LAB: laboratory animal group; dP/dTmax: maximum developed pressure; dP/dTmin: minimum developed pressure; * ≤ 0.05; ** ≤ 0.001.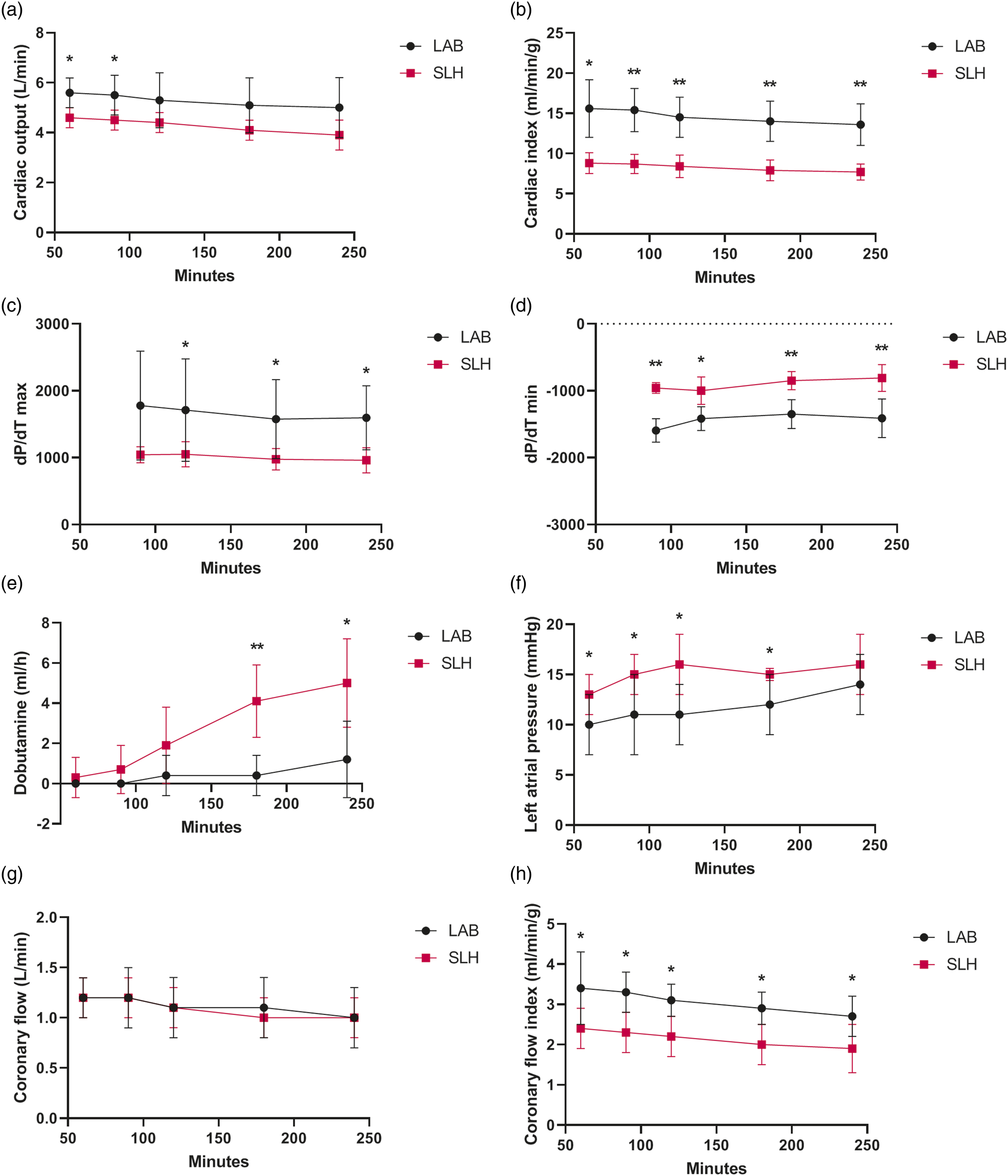
Discussion
In this study, we compared 4 hours of COMP of SLH and LAB hearts to validate the use of SLH hearts for ex-situ machine perfusion studies. Functional survival in both groups was 100% during normothermic testing, but although LAB hearts had a superior function and required less dobutamine, both groups displayed a similar decline in functional parameters over time. The SLH hearts can therefore be used as an alternative to LAB hearts for ex-situ heart perfusion studies, as they respond in a similar way to the exposure of the perfusion setup with comparable survival and trends of function over time. The difference in function and differential requirement of dobutamine should be considered when analyzing data from SLH heart experiments.
Earlier studies have shown successful preservation and survival of porcine hearts from experimental animals for up to 24 hours using normothermic perfusion 11 and COMP.12–14 However, this paper is the first that describes the survival of SLH hearts for 4 hours. SLH hearts accessibility is unlimited, and they concomitantly reduce the cost of experiments. SLH hearts are harvested without buying and housing laboratory animals, the use of operating room, medication and extra personnel needed during the harvesting procedure.
Preserving hearts using COMP instead of SCS, which is still the golden standard in heart transplantation, is based on our hypothesis that COMP is a better preservation method. Key factors that support this hypothesis are a reduction in metabolism due to hypothermia, the washout or dilution of waste products and supplying oxygen, which has also been demonstrated in other hypothermically perfused organs.15–17 The washout of lactate, lactate dehydrogenase and ammonia were higher in the SLH hearts, whereas rise in troponin level was similar. These findings could be a result of the harvesting procedure in the SLH hearts with a period of warm ischemia, resulting in suboptimal preservation of energy stores prior to COMP. Although a higher level of damage markers was seen during COMP, all hearts showed clearance of lactate during normothermic perfusion and acceptable functional survival for 4 hours.
Interestingly, lactate levels at 240 minutes of COMP correlated with CI after 240 minutes of functional testing (Figure 3). At the start of clinical normothermic ex-situ heart perfusion, lactate was seen as a possible predictor of outcome after transplantation 18 and was used as a marker to accept or deny hearts during normothermic perfusion. 19 More recent research has shown that lactate during normothermic perfusion is not very strongly correlated with functional outcomes.20,21 As a marker of adequate perfusion, the decrease in lactate provides sufficient information, for that reason no venous lactate was measured. Lactate as predictor for cardiac function during COMP is not well documented and needs further research.
Although SLH hearts showed functional survival for 4 hours, their function was inferior to LAB hearts. The superior cardiac function of LAB hearts can be attributed to the fact that these hearts were harvested in an optimal setting, resulting in the absence of warm ischemia. The length of the warm ischemic interval followed by reperfusion is inversely associated with cardiac function. 22 Other important factors contributing to the functional difference between LAB and SLH animals are the electrical stunning of SLH animals that potentially contributes to more myocardial injury, the more extensive stress of these animals prior to harvesting and the finding that the SLH hearts are more hypertrophic compared to hearts from LAB animals. Hypertrophy was not precisely measured in this setup but was based on the inspection of axial coupes at the end of the experiment. The main factor that influences the hypertrophy is the fact that SLH animals are bred for human consumption. Farmers aim to reach a weight of around 120 kg by the age of 6 months with specific feeding strategies to optimize growth, unfortunately the weight of the SLH animals could not be measured due to logistic reasons. This is in contrast with LAB animals, which are not fed to gain weight in a short timeframe. The difference in harvesting weight was suboptimal but unavoidable. Laboratory animals are normally used at a younger age and are not bred or fed to gain weight in a short timeframe, which make animals lighter at similar age. Besides, the housing and handling of 120 kg pigs is uncommon in the laboratory setting. For this reason, we decided to use hearts from animals that are commonly used in our animal facility and normalized perfusate samples to the mean weight of the group as described in our methods.
Despite the inferior function of SLH hearts, the trend of cardiac function over time showed to be comparable for both groups. This reflects a similar response to reperfusion and normothermic testing. On the one hand this is a positive outcome, as this highlights again that SLH hearts can be used for ex-situ perfusion studies to optimize machine perfusion or test therapeutics. On the other hand, it highlights that potentially the reperfusion and normothermic testing for all hearts can be optimized. Literature describes this decline in function over time, 23 with metabolic pathways as potential target.
Another limiting factor in this study is the use of non-autologous blood during normothermic testing in the LAB hearts. Although we attempted to harvest the required amount of blood, we were not able to collect enough blood from the LAB animals. Since a minimal amount of full blood is needed for the normothermic testing phase, we have chosen to use non-autologous blood. The use of non-autologous blood in prior non-transfused pigs normally does not lead to hemolytic transfusion, but can have an impact on endothelial function, coagulation and inflammation. 24 Together this can impact cardiac function, although the extent of which is unknown. It could be that the decrease in cardiac function over time is partly due to the use of non-autologous blood, although similar declining trends were observed in the SLH hearts that were run with autologous blood. Although the use of non-autologous blood could have had an impact in the LAB group, cardiac function in the LAB group remained superior to the SLH hearts.
In conclusion, this study demonstrates that the SLH heart can be used as an alternative to LAB hearts for ex-situ perfusion studies. Although LAB hearts showed superior function at a lower dobutamine concentration, both LAB and SLH hearts showed similar functional survival and had a similar trend in functional decline during normothermic testing. The use of SLH hearts can reduce the need for laboratory animals and enables high volume access to porcine hearts for ex-situ perfusion studies in a cost-effective manner.
Supplemental Material
Supplemental Material - Validation of the slaughterhouse porcine heart model for ex-situ heart perfusion studies
Supplemental Material for Validation of the slaughterhouse porcine heart model for ex-situ heart perfusion studies by Selma E Kaffka genaamd Dengler, Mudit Mishra, Sjoerd van Tuijl, Saskia CA de Jager, Joost PG Sluijter, Pieter A Doevendans, Niels P van der Kaaij in Perfusion.
Footnotes
Declaration of conflictintg interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Regenerative Medicine crossing borders (RegMed XB). This work was supported by European Research Council (ERC) under the EVICARE grant (number 725,229) to JS.
Supplemental Material
Supplemental Material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
