Abstract
Introduction
cardiac surgery associated acute kidney injury (CSA-AKI) has a number of preoperative and intraoperative risk factors. Cardiopulmonary bypass (CPB) factors have not yet been elucidated in a single multivariate model. The aim of this study is to develop a dynamic predictive model for CSA-AKI.
Methods
retrospective study on 910 consecutive adult cardiac surgery patients. Baseline data were used to settle a preoperative CSA-AKI risk model (static risk model, SRM); CPB related data were assessed for association with CSA-AKI. CPB duration, nadir oxygen delivery, time of exposure to a low oxygen delivery, nadir mean arterial pressure, peak lactates and red blood cell transfusion were included in a multivariate dynamic perfusion risk (DPR). SRM and DPR were merged into a final logistic regression model (multifactorial dynamic perfusion index, MDPI). The three risk models were assessed for discrimination and calibration.
Results
the SRM model had an AUC of 0.696 (95% CI 0.663–0.727), the DPR model of 0.723 (95% CI 0.691–0.753), and the MDPI model an AUC of 0.769 (95% CI 0.739–0.798). The difference in AUC between SRM and DPR was not significant (p = 0.495) whereas the AUC of MDPI was significantly larger than that of SRM (p = 0.004) and DPR (p = 0.015).
Conclusions
inclusion of dynamic indices of the quality of CPB improves the discrimination and calibration of the preoperative risk scores. The MDPI has better predictive ability than the existing static risk models and is a promising tool to integrate different factors into an advanced concept of goal-directed perfusion.
Introduction
Cardiac surgery associated acute kidney injury (CSA-AKI) is one of the most common postoperative complications, associated with an increased mortality risk.1–4 It is usually defined based on the changes in serum creatinine in the early postoperative period, and on the need for renal replacement therapy, and since a specific definition of CSA-AKI is still lacking, its grading usually follows the Acute Kidney Injury Network (AKIN) 5 or the Kidney Disease Improving Global Outcomes (KDIGO) 6 definition. According to these criteria, an increase of the serum creatinine by at least 50% is used to adjudicate an AKI (of any stage).
Several risk scores for CSA-AKI exist.7–11 They are based on preoperative risk factors and severity of the procedure. Additionally, they have been designed to predict the risk of most serious degrees of acute kidney injury, namely renal replacement therapy. The only model considering lower degrees of CSA-AKI cannot be considered a real preoperative risk model, since it includes postoperative variables. 12
The existing risk models do not take into consideration the cardiopulmonary bypass (CPB) associated factors, that in many studies have been linked to CSA-AKI.
The purpose of the present study is to develop a new model for prediction of CSA-AKI based on a “static” preoperative model, integrated with a number of CPB-related factors, ending up with a “dynamic” model inclusive of the preoperative risk factors, the procedure-related risk, and the CPB-related factors.
Methods
Study design and patient population
Retrospective cohort study on 910 consecutive adult patients undergoing cardiac surgery with CPB in the year 2019 at our Institution. Congenital heart patients were excluded from the study population. Patients treated at a bladder temperature <32°C were excluded. The final study population was therefore represented by 830 subjects. The study was approved by the Ethics Committee of San Raffaele Hospital. Given the retrospective nature of the study, a written informed consent was retrieved whenever feasible; all the patients however provided a written informed consent for the use of their data for scientific research, in an anonymous form.
All the patients were treated with a low-hemodilution CPB strategy, consisting of a priming volume of 800 mL of gelatins and sodium bicarbonate as a buffer.
Data collection and definitions
Data for evaluation of the static risk model (SRM) for CSA-AKI were retrieved from our Institutional database. These included demographics, co-morbidities, baseline serum creatinine and bilirubin, hematocrit (HCT, %), hemoglobin (Hb, mg/dL), type of surgical intervention, and other intervention-related factors (elective/urgent/emergent; redo surgery). From the preoperative data, the Cleveland Risk Score (CRS) 7 was calculated and chosen as the reference “static” CSA-AKI risk score. This score includes the following risk factors: gender; congestive heart failure; type 1 diabetes; chronic obstructive pulmonary disease; left ventricular ejection fraction; preoperative use of an intra-aortic balloon pump; history of previous cardiac surgery; type of current cardiac surgery; and preoperative creatinine. In agreement with the existing literature that considers age8–10 and a low preoperative HCT13–15 as risk factors for CSA-AKI, age and HCT were merged into the CRS, and the resulting logistic regression equation produced the static risk model (SRM).
Data from the CPB files were retrieved and manually analyzed for CPB-related factors. The files contained data hemodynamic and ventilatory parameters collected every 10 min during CPB: pump flow (L/min), mean arterial pressure (MAP, mmHg), total gas flow (L/min) and FiO2, plus central temperature (°C). Blood gas analyses were performed every 20 min and included acid-base balance, oxygen tension (mmHg) and saturation (%), HCT, Hb, electrolytes, and lactates (LAC, mmol/L). Oxygen delivery indexed for body surface area (DO2, mL.min−1.m−2) was calculated based on pump flow, Hb, and oxygen saturation. Venous blood gas analyses were not routinely performed and not at specific intervals of time. On-line measure of venous and arterial oxygen saturation and exhaled CO2 were available in a limited amount of patients.
The use of allogeneic blood products (red blood cells, RBC) and of vasoconstrictors (norepinephrine) while on CPB were recorded.
Based on the above items, the following parameters were derived: • Nadir pump flow indexed for body surface area (L.min−1.m−2) • Nadir HCT, maintained for at least 10 min • Nadir DO2, maintained for at least 10 min • Time of exposure to a DO2 below the critical value (min) • Nadir MAP, maintained for at least 10 min • Use of vasoconstrictors (norepinephrine) • Peak blood lactates • RBC transfusion (yes/no) • CPB duration (min)
The outcome parameter (CSA-AKI) was defined as a serum creatinine increase of at least 50% of the baseline value, occurring within the first 48 h from surgery. This definition includes the stages I, II, and III of the KDIGO classification 6 and can therefore be labeled as CSA-AKI “of any kind.”
Models development and statistics
The sample size of 830 patients was considered adequate to develop multivariable models inclusive of more than 10 independent variables. Considering a CSA-AKI rate of 15%, and a ratio of 1:10 between variables and events, up to 12 independent variables can be admitted to the model. This number was considered adequate for the purpose of the present study.
Variables are defined as mean and standard deviation (SD) or number (%). Differences between patients with or without CSA-AKI were addressed with a Student’s t-test for unpaired data and a Pearson’s chi-square for differences in frequency.
Each CPB-related variable was tested for association with CSA-AKI using linear of polynomial regression equations. In case of non-linearity, adequate cut-off values were assessed based on the Youden index of receiver operating characteristics (ROC) curves.
Multivariable analyses were based on logistic regressions, producing odds ratios (OR) and 95% confidence intervals (CI). Based on the logistic regression equation, three separate risk models were developed: the SRM (based on preoperative factors), the dynamic perfusion risk (DPR) based on the perfusion-related variables, and the multifactorial dynamic perfusion index (MDPI) which is the combination of SRM and DPR.
The MDPI was tested for discrimination using a ROC analysis producing areas under the curve (AUC), and it was compared to other three existing risk factors for CSA-AKI (CRS, 7 Simplified Predictive Index, 8 and Bedside Index 9 ) Differences between AUCs were investigated with the DeLong method. Calibration properties were investigated using calibration plots that are considered the gold standard for this assessment. 16
An internal validation of the final model was performed using a bootstrap technique with 1000 iterations, producing the values of bias and 95% confidence interval.
All the statistical analyses were performed with computerized packages (SPSS 20.0, IBM, Chicago, IL, GraphPad, GraphPad Software, Inc, San Diego, CA, and MedCalc, MedCalc Software, Ostend, Belgium). A p value <0.05 was considered significant for all the statistical tests.
Results
The static risk model
Preoperative and operative details of the patient population (N = 830).
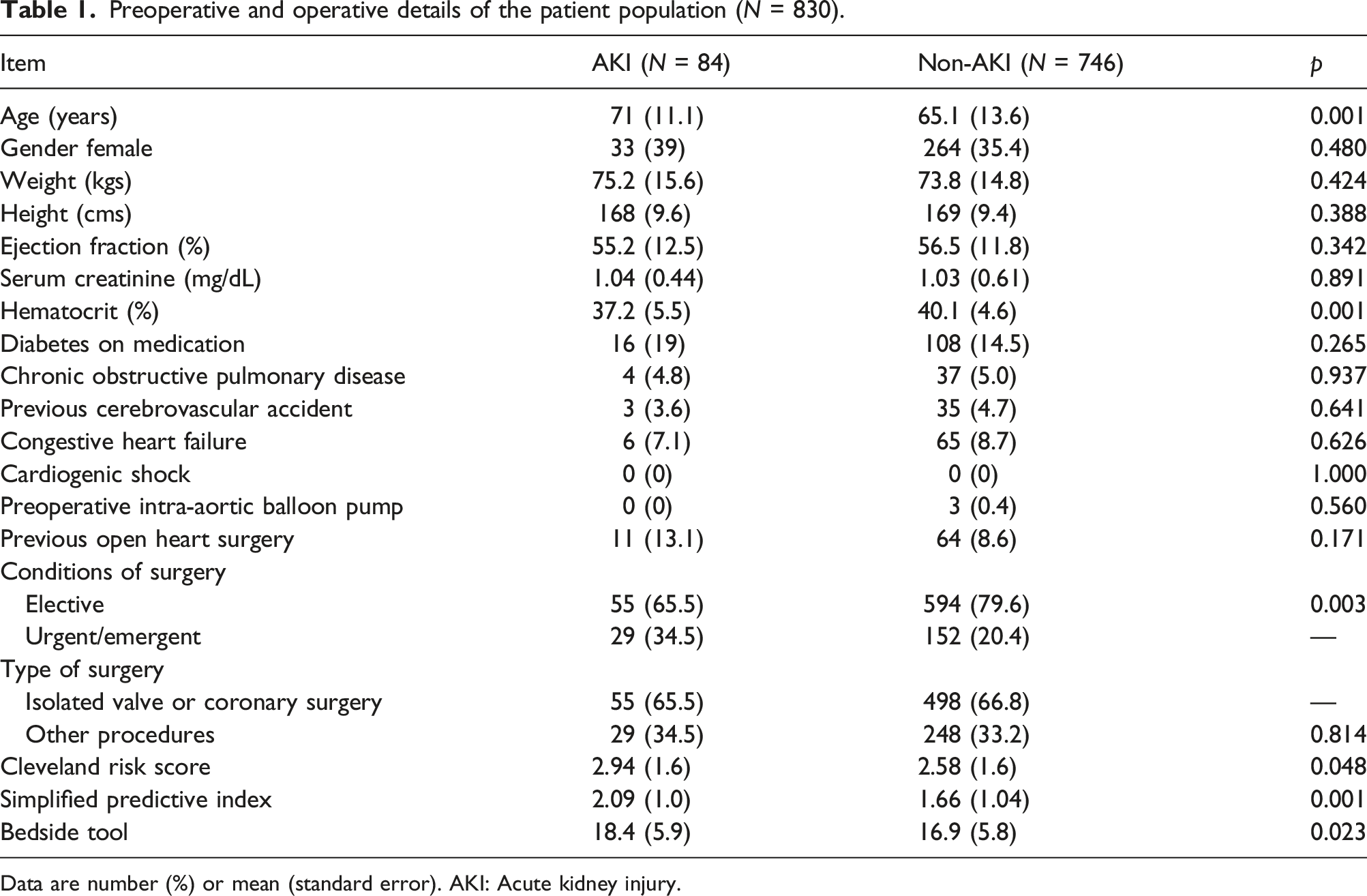
Data are number (%) or mean (standard error). AKI: Acute kidney injury.
All the existing risk scores were significantly higher in AKI patients. Other factors being significantly different were age and preoperative hematocrit. The SRM for AKI is defined by the following logistic regression equation: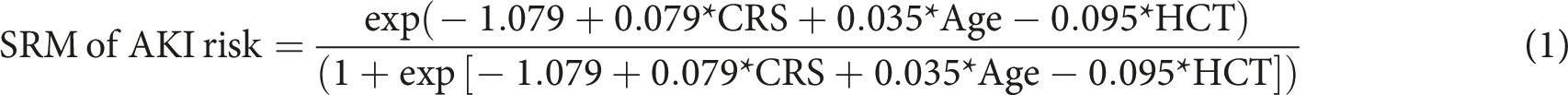
The dynamic perfusion risk
Cardiopulmonary bypass-related factors in AKI and non-AKI patients.
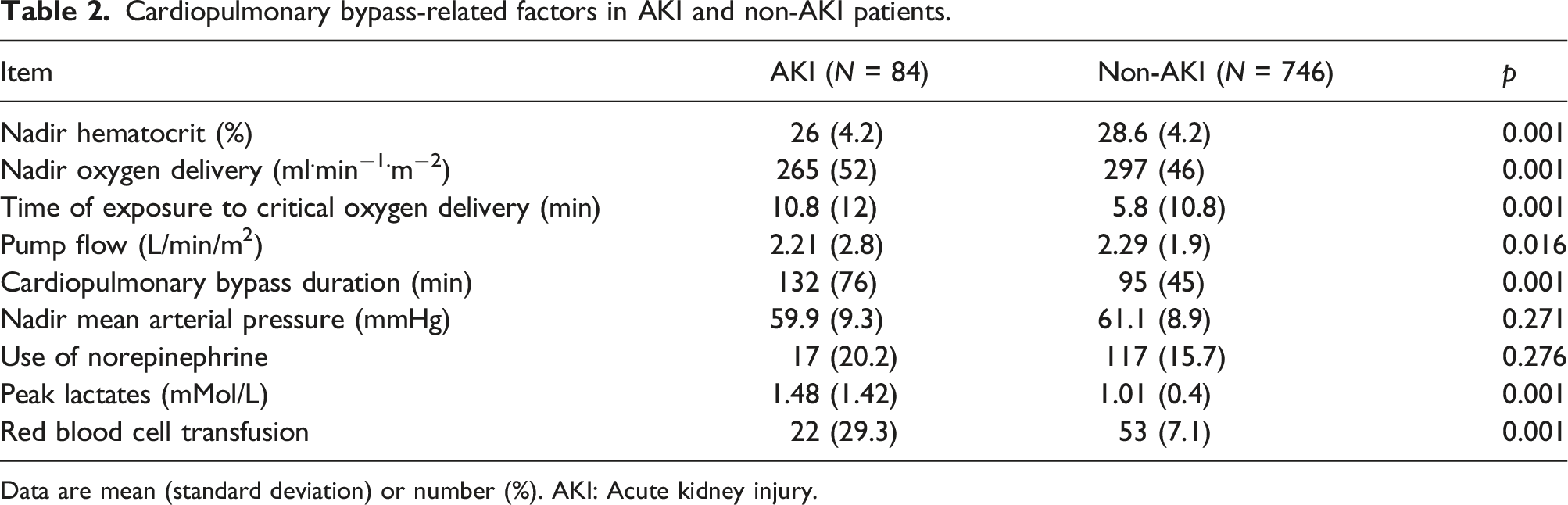
Data are mean (standard deviation) or number (%). AKI: Acute kidney injury.
The analyses of association between continuous CPB-related factors and CSA-AKI are reported in Figure 1. The relationship is linear for CPB duration (relative risk 0.1% per each CPB minute) and peak LAC (relative risk increase 11.2% per each mMol/L); quadratic for MAP and cubic for the time of exposure to the critical DO2. Nadir HCT and DO2 levels behave linearly below a cut-off value that was identified at 26% and 289 mL.min−1.m−2 respectively; for values above these points, the risk of CSA-AKI is constant at 6.4% and 5.3% respectively. Association between cardiopulmonary bypass (CPB) factors and risk of acute kidney injury (AKI). DO2: Oxygen delivery; HCT: Hematocrit; MAP: Mean arterial pressure.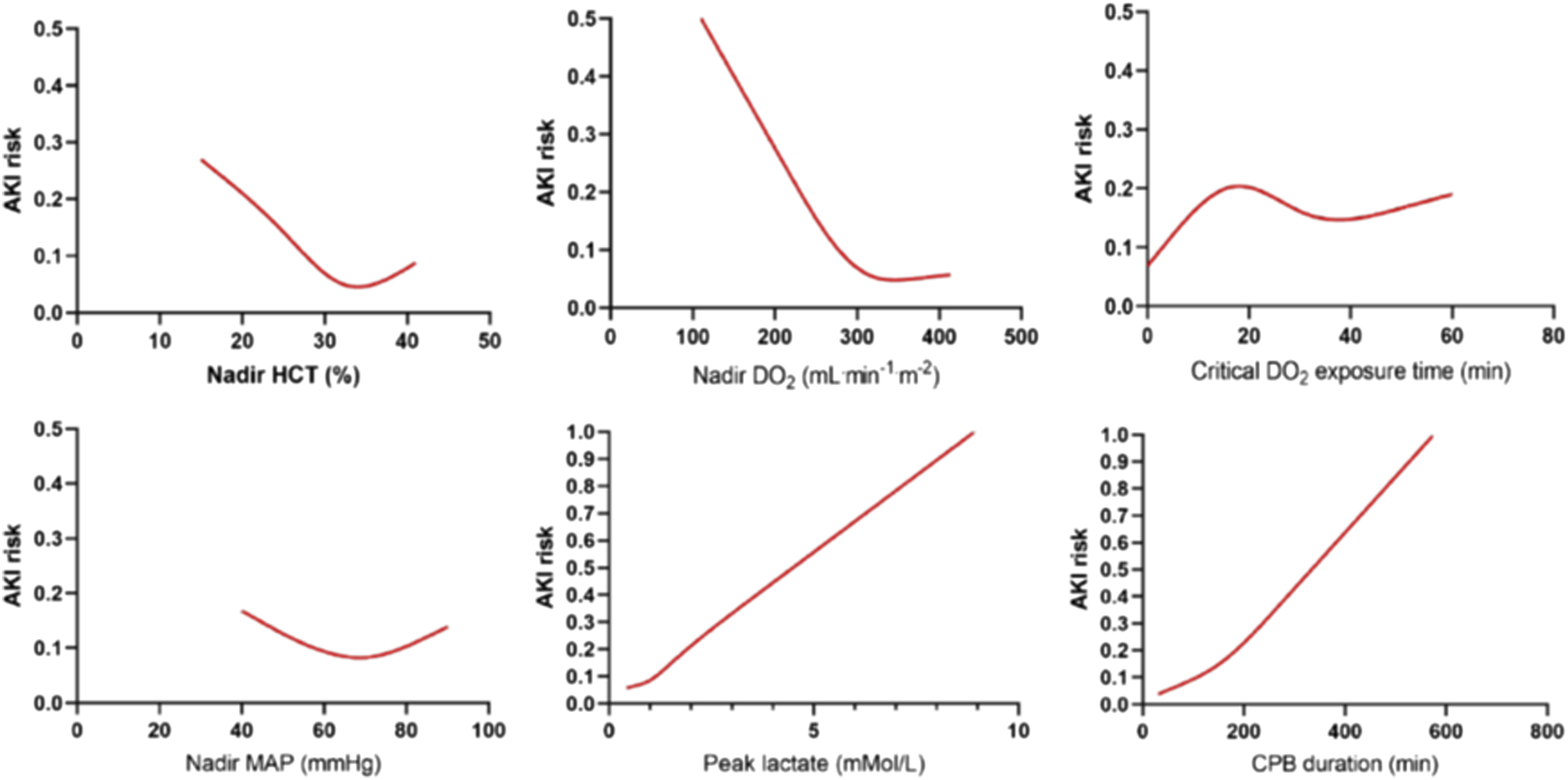
Logistic regression parameters for the dynamic perfusion risk.
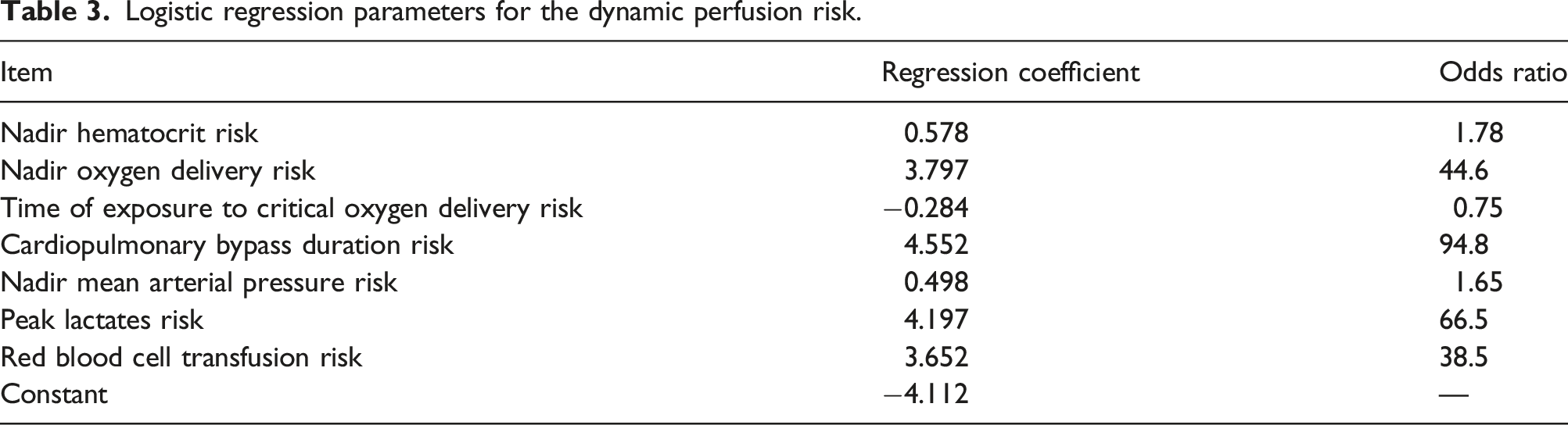
The multifactorial dynamic perfusion index
The CSA-AKI risk as determined by the SRM and the DPR were merged into a single logistic regression equation (equation (2)) defining the MDPI-CSA-AKI risk. The equation is the following: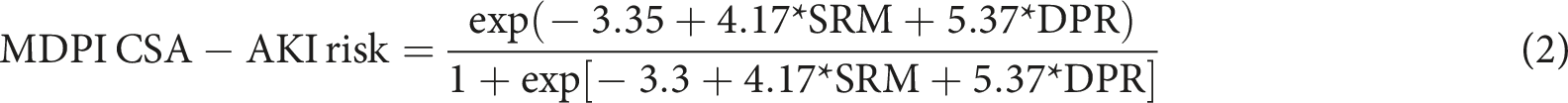
Internal validation
The equation (2) was internally validated with a bootstrapping of 1000 iterations. The bias of the three coefficients was negligible and the p values remained highly significant: • Constant: Bias −0.033, standard error 0.229, 95% CI –3.90 to −2.96, p = 0.001 • SRM: Bias −0.058, standard error 1.51, 95% CI 1.26–7.30, p = 0.004 • DPR: Bias 0.238, standard error 1.55, 95% CI 2,89–8.96, p = 0.001
The MDPI was tested for discrimination using an ROC analysis (Figure 2), in comparison with the other three existing CSA-AKI predictive models. The CRS had an AUC of 0.580 (95% CI 0.545–0.614), the Simplified Predictive Score an AUC of 0.631 (95% CI 0.597–0.664), the Bedside Tool an AUC of 0.587 (95% CI 0.552–0.621) and the MDPI-based equation reached an AUC of 0.769 (95% CI 0.739–0.798). The difference in AUC between the MDPI and the other scores yielded a p < 0.001. Receiver operating characteristics curves for the four risk models. MDPI: Multifactorial dynamic perfusion index.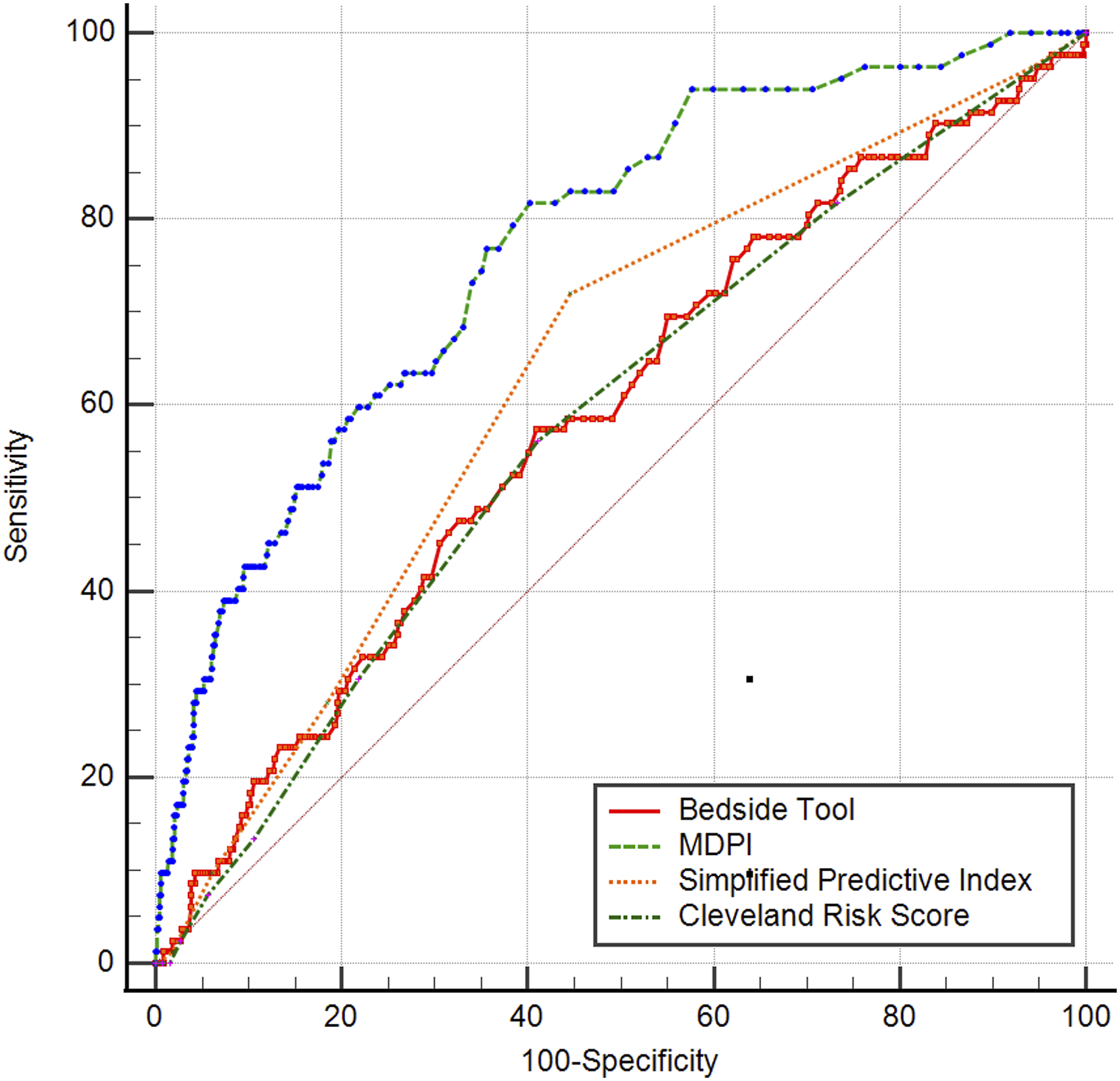
Calibration of the MDPI was tested with a calibration plot (Figure 3). The MDPI showed a good calibration until a predicted CSA-AKI risk of 50%; for higher risk values, it was overestimating the risk. However, this is due to the fact that only 14 patients (1.6%) had an expected CSA-AKI risk >50%, and therefore in this very high risk patient population the sample size was insufficient to guarantee an adequate calibration. Calibration plot for the multifactorial dynamic perfusion index. AKI: Acute kidney injury.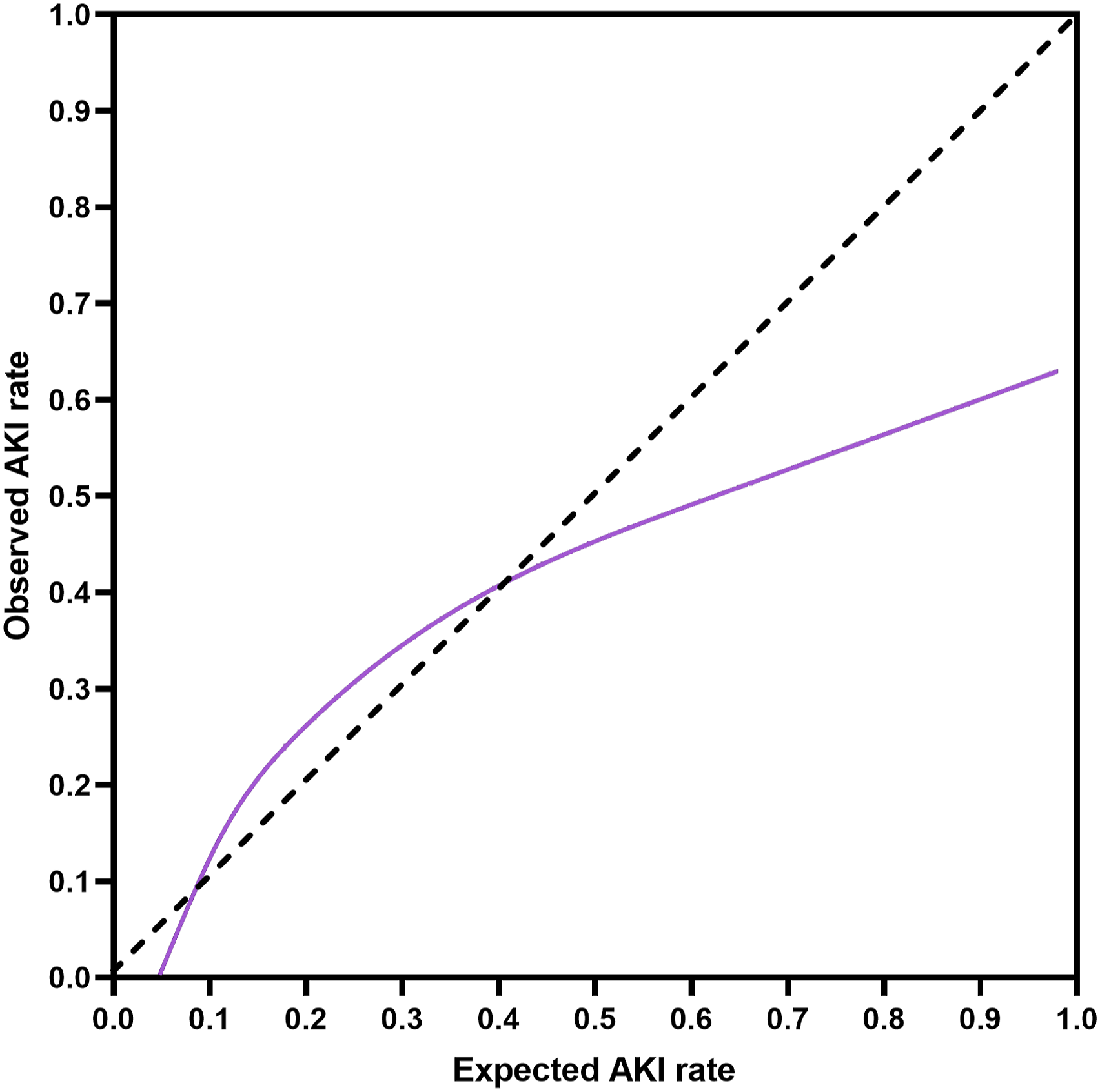
Discussion
Our study proposes a new approach to CSA-AKI risk assessment, based on the combination of a pre-procedural, static and non-modifiable risk score with a dynamic and largely modifiable combination of CPB-related hemodynamic and metabolic indices. The MDPI demonstrated a good discrimination ability, superior to the existing static scores, and a good calibration in the range of CSA-AKI risk between 0% and 50%, and an optimal internal validation.
The existing risk models are generally inappropriate to predict the risk of minor degrees of AKI (namely stage I and II in the KDIGO classification). 17 The main reason is that they include the baseline serum creatinine, that is an excellent predictor of AKI requiring renal replacement therapy but not of serum creatinine increase. 17 Additionally, they do not take into consideration the cardiopulmonary bypass (CPB) associated factors, that in many studies have been associated with CSA-AKI. Given these points, it is not surprising that the MDPI has a higher discrimination power, being titrated on any stage of CSA-AKI and incorporating CPB-derived parameters.
The novelty of our approach is the inclusion of different parameters into a single algorithm. As a matter of fact, taken one by one, the seven predictors included in the DPR have already been identified as independent risk factors for CSA-AKI.
Nadir HCT
The deleterious effects of excessive hemodilution on CPB, as represented by a low HCT, have been discovered since the mid 90s’.18–22 These effects have been largely demonstrated as determinants of CSA-AKI, but they appear to affect other organs dysfunction and even mortality. 22 It is still unclear which is the cut-off value for entering the critical level of hemodilution; however, it seems to be around 26%.21,22
In our series, we could confirm this critical level of HCT, below which the CSA-AKI risk linearly increases. Severe hemodilution is per se a risk factor for bad outcomes. In particular, it has been demonstrated that severe hemodilution is deleterious for the microcirculation. 23
Nadir DO2
A low DO2 has been associated with CSA-AKI in many studies. In the first study addressing this topic, we could find a critical DO2 at a value of 272 mL.min−1.m−2. 24 In our series, the critical DO2 was found at 289 mL.min−1.m−2, in substantial agreement with the previous findings. A low DO2 is certainly deleterious for any organ function, but visceral organs and namely the kidney are the more susceptible to a mismatch between oxygen needs and supply. The nadir DO2 depends of course on the HCT and the pump flow. Pump flow was excluded from our model due to the minimal difference between patients with or without CSA-AKI, and to avoid collinearity.
Time of exposure to the critical DO2
It is reasonable to hypothesize that the longer the patient stays below the critical DO2, the greater will be the risk of CSA-AKI. There are few studies addressing this topic. Rasmussen et al. 25 demonstrated that actually the duration of exposure to a low DO2 is associated with an increased risk of CSA-AKI. In our series, this finding was confirmed; however, the risk increases until 20 min of exposure; then it stabilizes at around 20%, without further increase for longer exposure to the critical DO2.
Peak lactates
Even if other mechanisms are possible, the main mechanism leading to hyperlactatemia is the transition from the aerobic to the anaerobic metabolism. In the setting of CPB, this is again to be ascribed to a mismatch between oxygen needs and oxygen supply.
Other studies have highlighted that hyperlactatemia on CPB is associated with bad outcomes, including mortality.26,27 In our series, lactate concentration has a linear relationship with CSA-AKI, with a relative risk of 11.2% per each mMol/L of lactate, thus confirming the important role of this marker.
Nadir MAP
The role of MAP during CPB as a determinant of worse outcomes and namely CSA-AKI is widely debated. The existing guidelines simply suggest to maintain the MAP between 50 and 80 mmHg; 28 however, a retrospective study found an association between a MAP <65 mmHg and a composite outcome of stroke, AKI or mortality. 29
From our data, it seems that both hypotension (nadir MAP <40 mmHg) and hypertension (nadir MAP >90 mmHg) increase the CSA-AKI risk to a value of about 15%. Therefore, the MAP-related risk was defined by a quadratic equation, with the lowest risk placed around 70 mmHg.
CPB duration
The notion that the longer the CPB, the worse the outcome, is well established. Although some authors30,31 recently proposed cut-off values of CPB duration (between 70 and 110 min), our data suggest that the relationship is linear, with an incremental relative risk of 0.1% per minute of CPB.
RBC transfusion
Allogeneic blood product transfusions have been associated with CSA-AKI in a number of studies,32-34 although the timing of transfusion (on CPB or after CPB) has not been elucidated yet. In our series, the CSA-AKI risk was triple (at the univariate analysis) in patients receiving RBC transfusion on CPB. However, it should be considered that RBC transfusion has a potentially favourable impact on other parameters included in the MDPI such as nadir HCT, nadir DO2, and time to exposure to the critical DO2. The combined effects of RBC transfusion (deleterious) and the consequent increase in HCT and DO2 (beneficial) can be calculated from the multivariable logistic equation from which the MDPI is derived.
MDPI in clinical practice
The MDPI is intended to offer a guide to the perfusionists and the anesthesiologists in order to limit the perfusion-related risk of CSA-AKI, possibly decreasing the preoperative, static risk. Preoperative risk models are basically composed by non-modifiable risk factors; conversely, the MDPI includes a number of modifiable risk factors, like the nadir HCT, the nadir DO2, the time of exposure to the critical DO2, the mean arterial pressure, and the use of RBC transfusion. The perfusionist has a number of tools to correct the DPR; some of them are proactive (i.e. limiting the hemodilution in order to preserve the HCT), others are reactive (increasing the pump flow to preserve the DO2). Basically, the concept is that the perfusionist should consider the SRM as the starting point, follow the DPR and the MDPI, and apply corrective measure in order to decrease the SRM or, at least, not to increase it. In this sense, the MDPI should be considered as a quality marker of the CPB, probably associated with outcomes other than the CSA-AKI. The incorporation of the various equations is theoretically possible in some of the data monitoring systems integrated in the CPB equipment, with the possibility to follow in real-time the changes in DPR and MDPI.
The level of discrimination of the MDPI is acceptable, considering that it includes preoperative and CPB-related factors, but not post-CPB factors that certainly influence the incidence of CSA-AKI: among these, hypotension, low cardiac output, need for inotropic agents and vasoconstrictors.
Limitations
The main limitations are (i) the possible lack of some predictors (i.e. the mixed venous oxygen saturation) that were not routinely collected in our files; (ii) the single-Institution nature of this study and (iii) the absence of an external validation series. Although the internal validation is satisfying, an external series obtained in multiple Institutions is certainly recommended, and will be the endpoint of future studies. Actually, the incidence of CSA-AKI of any stage greatly differs among Institutions. A multicenter study 35 conducted in 10 Institutions in Europe, USA, Australia and New Zealand showed a CSA-AKI incidence ranging from 9.1% to 27.3%. In our series, the CSA-AKI rate was 10.1%, so on the low-side of the distribution. The inclusion of Institutions with a larger rate of CSA-AKI could be useful to adjust the coefficients of the MDPI.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Marco Ranucci is a Consultant for Livanova and Medtronic; Mauro Cotza is a Consultant for Livanova, Medtronic, and Qura. The algorhitms composing the Multifactorial Dynamic Perfusion Index are protected by a patent owned by IRCCS Policlinico San Donato.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was partially funded by the Italian Ministry of Health, within the funding of Clinical Research Hospitals network, which includes our Institution.
