Abstract
Introduction: Comprehensive genetic testing with whole-exome (WES) or whole-genome (WGS) sequencing facilitates diagnosis, can optimize treatment, and may improve outcomes in critically ill neonates, including those requiring extracorporeal membrane oxygenation (ECMO) for respiratory failure. Our objective was to describe practice variation and barriers to the utilization of comprehensive genetic testing for neonates on ECMO.
Methods: We performed a cross-sectional survey of Level IV neonatal intensive care units in the United States across the Children’s Hospitals Neonatal Consortium (CHNC).
Results: Common indications for WES and WGS included concerning phenotype, severity of disease, unexpected postnatal clinical course, and inability to wean from ECMO support. Unexpected severity of disease on ECMO was the most common indication for rapid genetic testing. Cost of utilization was the primary barrier to testing. If rapid WES or WGS were readily available, 63% of centers would consider incorporating universal screening for neonates upon ECMO cannulation.
Conclusion: Despite variation in the use of WES and WGS, universal testing may offer earlier diagnosis and influence the treatment course among neonates on ECMO. Cost is the primary barrier to utilization and most centers would consider incorporating universal screening on ECMO if readily available.
Introduction
Extracorporeal membrane oxygenation (ECMO) is a life-saving therapy offered to the most critically ill neonates with severe respiratory and/or cardiac failure refractory to maximal medical management.1–5 Despite its success as a life-sustaining procedure, ECMO is invasive and costly, and carries a mortality of 10–40%.1,4,6
Whole exome sequencing (WES) sequences all ∼20,000 protein-coding genes or exons, however these account for only ∼2% of the entire genome. Alternatively, whole genome sequencing (WGS) sequences the entire genome, both the protein-coding genes or exons, as well as the non-protein-coding genes, or introns, which may include important regulatory regions.
Historically, for neonates with respiratory failure or pulmonary hypertension of unclear etiology, invasive diagnostic testing such as a lung biopsy was pursued while on ECMO. 7 However, lung biopsy is not without risk including air leak and bleeding. Recognition of underlying genetic defects, such as alveolar capillary dysplasia or surfactant protein deficiency disorders, has led to a shift from lung biopsy to genetic testing as the primary modality for diagnosis. Advances in technology have led to availability of increasingly sophisticated diagnostic tests. While more comprehensive genetic testing can allow a diagnosis to be made non-invasively, these tests have limitations. For example, chromosomal microarrays are only able to detect deletions and duplications while multigene panel testing only sequences a few genes of interest and does not have parental samples to interpret variants. Whole exome sequencing and WGS have the potential to provide rapid diagnosis, optimize treatments, and improve outcomes in critically ill neonates, including those requiring ECMO.8–20 Information obtained through comprehensive genetic testing with WES and WGS may alter the course of ECMO through changes in initiation or management, or in some situations, discontinuation of ECMO.
Costs and turnaround time for comprehensive tests such as WES and WGS vary, with some centers waiting several weeks for results. With newer technology, select centers offer “ultra-rapid” protocols with a turnaround time of 1–3 days. Multiple studies have shown benefits of universal screening with rapid WGS in neonatal intensive care units (NICUs), including changes in management and improvements in diagnostic efficacy.8-12 In 2021, Dimmock et al. found that 40% of infants received a diagnosis by rapid WGS and 32% of infants had a subsequent change in medical management. Rapid WGS also led to $2.2–2.9 million in cost savings. 9 Similarly, in a large multi-center randomized controlled trial, infants with WGS performed in 15 days were twice as likely to have a change in medical management compared to infants who had WGS with a turnaround time of 60 days. 10 While rapid WES and WGS are promising tools to aid in the management and prognostication of neonates on ECMO, little is known about their systematic use. 3 Thus, we sought to evaluate the current practices, variation in, and barriers to the utilization of comprehensive genetic testing among neonatal ECMO patients in Level IV NICUs.
Materials and methods
Data source
The Children’s Hospitals Neonatal Consortium (CHNC) is a multicenter collaborative of 41 Level IV NICUs in Children’s Hospitals in the United States and Canada dedicated to developing quality and research initiatives across participating institutions. Participating NICUs have > 400 annual admissions or > 25 NICU beds, and > 50% outborn neonates. 21
Study design
Cross-sectional survey of practice for neonatal respiratory ECMO among 41 Level IV neonatal intensive care units across the CHNC from June to September 2021. The survey was created, and study data were collected using Research Electronic Data Capture (REDCap) hosted at the Children’s Hospital of Philadelphia (CHOP).
Survey development
The survey was developed by the study investigators who are members of the CHNC ECMO Focus Group, a collaborative group of neonatologists and pediatric surgeons from CHNC participating NICUs with interest and expertise in ECMO. Survey questions underwent several iterations for readability, clarity, and understandability based on feedback from members of the focus group. The final survey contained 19 questions (Supplemental Table 1).
Survey distribution
The survey was distributed to CHNC site sponsors by email via the organizational listserv in June 2021. The email contained a link to the online survey which collected data in REDCap. A survey reminder was sent twice between June and September 2021. Surveys were completed collaboratively between neonatologists and geneticists for accuracy.
Statistical analysis
Data analysis was conducted using descriptive statistics. Simple reporting of survey results are reflected in results. No secondary analysis was performed due to sample size.
Results
Survey responses - Likelihood of ordering genetic testing for critically ill neonates, neonates on ECMO with identified and unidentified causes of pulmonary hypertension.
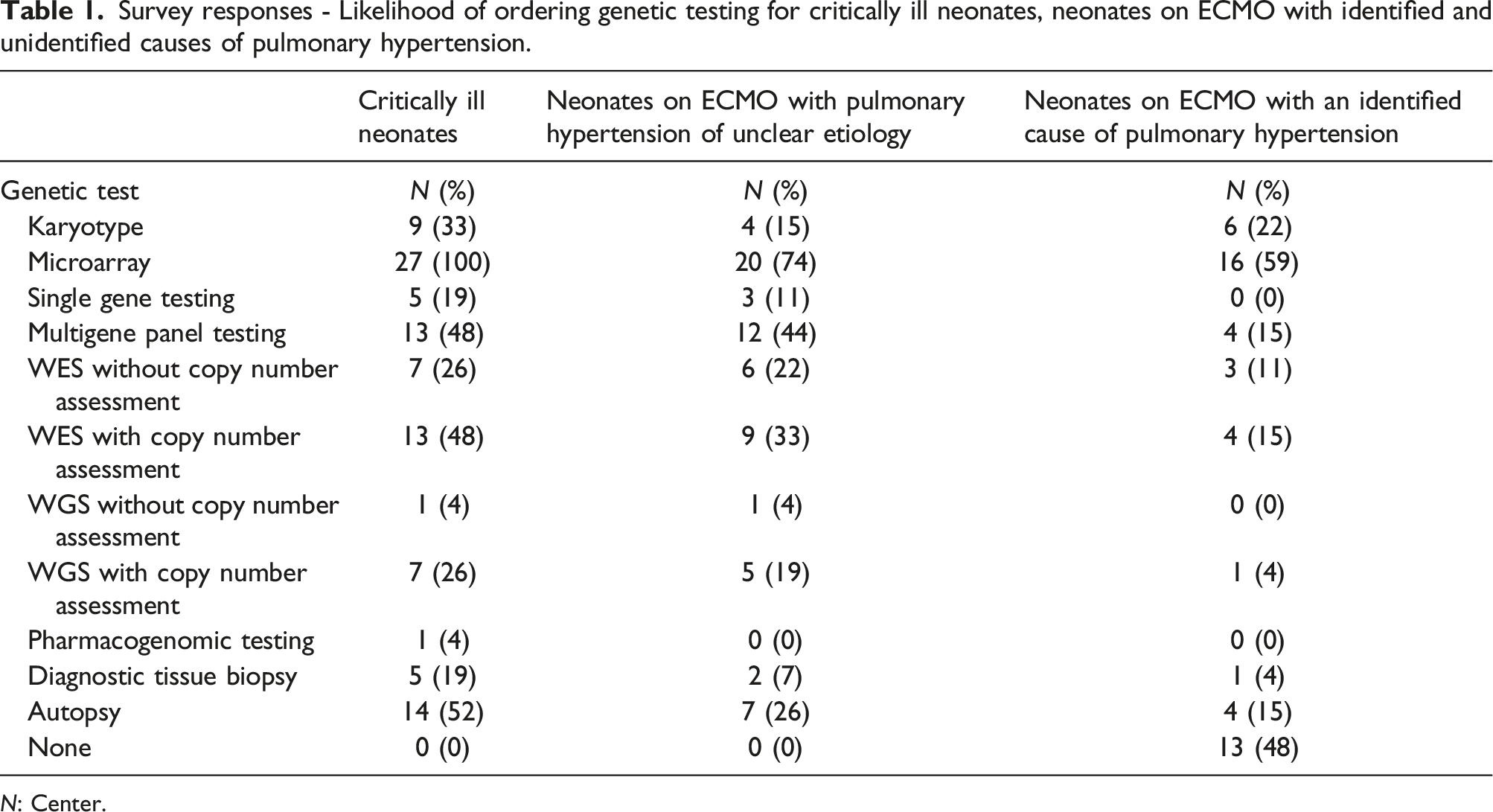
N: Center.
Utilization of comprehensive genetic testing (WES/WGS) for neonates on ECMO.

N: Center.
Reported barriers to testing and time to result for comprehensive genetic testing (WES/WGS) for neonates on ECMO.
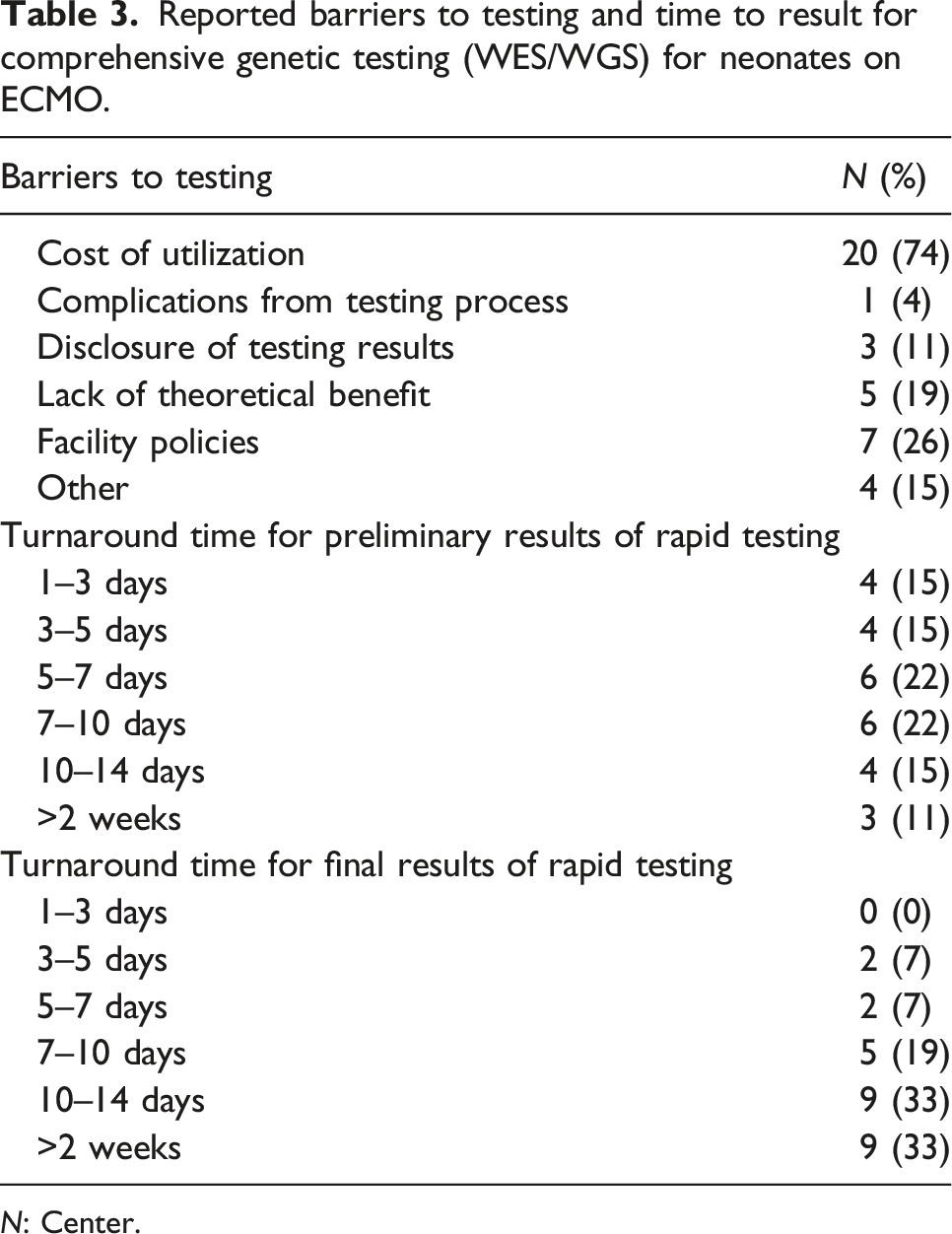
N: Center.
For neonates on ECMO specifically, WES and WGS were more likely to be utilized in neonates with pulmonary hypertension of unclear etiology, compared to those with a known etiology such as congenital diaphragmatic hernia (CDH). For neonates undergoing ECMO support with pulmonary hypertension of unclear etiology, 74% (N = 20) send chromosomal microarray, 44% (N = 12) send multigene panel testing such as an interstitial lung disease gene panel or a pulmonary hypertension gene panel, 56% (N = 15) send WES, and 22% (N = 6) send WGS. For neonates undergoing ECMO support with an identified cause of pulmonary hypertension, such as CDH, 59% (N = 16) send microarray, 15% (N = 4) send multigene panel testing, 26% (N = 7) send WES, and 4% (N = 1) send WGS. Autopsy was recommended for critically ill neonates in 52% of centers; in 26% of centers autopsy was recommended for neonates with pulmonary hypertension of unclear etiology and in 15% of centers for neonates with an identified cause of pulmonary hypertension. These results are summarized in Table 1.
As shown in Table 2, the most common indications for WES and WGS included concern for an inheritable condition based on family history (78%, N = 21), phenotype on exam concerning for a genetic disorder (93%, N = 25), severity of disease inconsistent with primary ECMO indication (74%, N = 20), inability to wean from ECMO support (70%, N = 19), and severe unexpected complications related to ECMO (74%, N = 20). While inability to wean from ECMO support was the most common indication for sending comprehensive testing, there are centers that send testing prior to and upon ECMO cannulation. For comprehensive genetic testing, 59% (N = 16) use WES and 19% (N = 5) use WGS. Genetic testing is most often done sequentially with microarray followed by WES or WGS if microarray returns non-diagnostic in 63% (N = 17) compared to comprehensive testing with WES or WGS that includes copy number evaluation as first line testing in 33% (N = 9). “Rapid” WES or WGS are most often offered for patients with severity of disease inconsistent with primary ECMO indication in 63% (N = 17). In 78% (N = 21), testing is recommended collaboratively between geneticists and neonatologists and in 74% (N = 20), results are disclosed collaboratively between geneticists and neonatologists. These results are shown in Table 2.
As shown in Table 3, the turnaround time for the results of rapid testing is highly variable. Preliminary results are most often available within 7 days and final results within 14 days. Cost of utilization is the primary barrier to testing in 74% (N = 20), but prolonged time to results may also represent a barrier to clinical utility. As shown in Figure 1, if cost were not an issue, most centers responded that they would be more likely to send WES and WGS in ECMO patients. Hospitals absorb the cost with institutional billing in 44% (N = 12) compared to insurance billing in 15% (N = 4). Twenty-six percent (N = 7) were unsure of how costs were paid. If rapid WES or WGS were readily available, 63% (N=17) would consider incorporating universal screening on ECMO patients upon cannulation. Removing cost of utilization, ECMO site likelihood to order comprehensive genetic testing (WES/WGS) versus diagnostic biopsy or autopsy.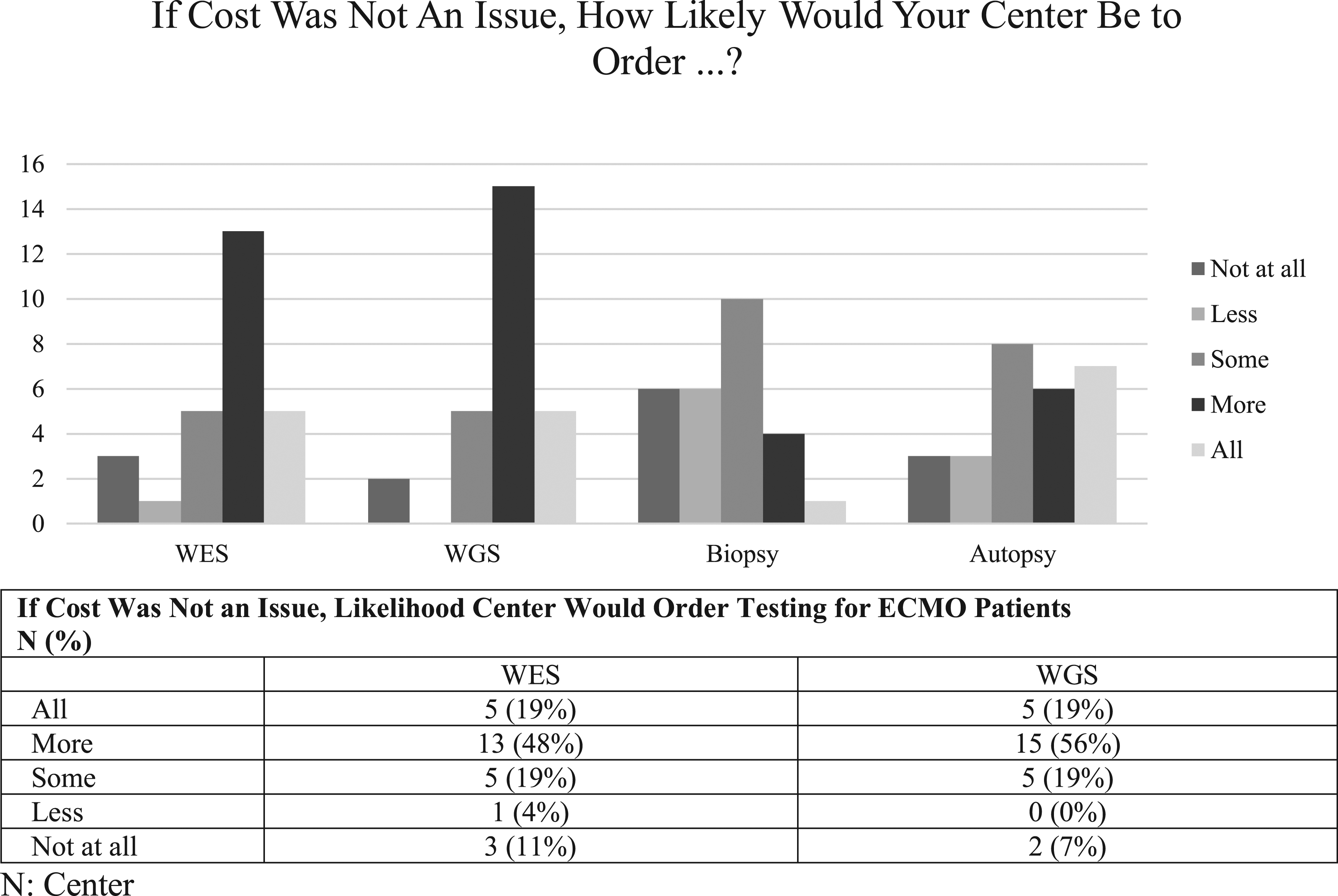
Discussion
Postnatal genetic testing utilization varies widely among ECMO centers. While all centers currently send microarray testing in the evaluation of critically ill neonates, use of comprehensive genetic testing is variable. Despite these variations, most centers would consider sending WES or WGS on neonatal ECMO patients if the cost of testing was not prohibitive.
Not surprisingly, WES and WGS were more likely to be sent in neonates on ECMO with pulmonary hypertension of unclear etiology compared to those with an identified cause of pulmonary hypertension, such as CDH. Similarly, rapid WES or WGS were offered most often for patients with severity of disease inconsistent with primary ECMO indication. In neonates with pulmonary hypertension of unclear etiology or unexpected severity of disease, a genetic diagnosis may explain the need for ECMO and guide decisions about the remainder of the ECMO course.
Multigene panel testing, often done as an interstitial lung disease panel or surfactant dysfunction panel, is sent by 48% of centers for critically ill neonates compared to 44% of centers for neonates with pulmonary hypertension of unclear etiology and only 15% of centers for pulmonary hypertension with an identified cause. Similar to WES and WGS, these panels are often considered when neonates are not responding to ECMO as expected. Despite containing fewer genes, many of these panels cannot be performed on a rapid basis like WES and WGS and take at least 4 weeks to result. These panels also do not incorporate parental samples making variants of uncertain significance more difficult to interpret.
Cost of utilization was the primary testing barrier identified, with WGS most likely to be incorporated if cost was not a barrier. However, multiple studies have demonstrated the cost effectiveness of WES and WGS in critically ill neonates.9,16,20 Whole exome sequencing and WGS costs are also expected to decrease precipitously over the next few decades. 22 Comprehensive genetic testing has the potential to be particularly cost effective in neonates on ECMO as a better understanding of the underlying genetic etiology may improve prognostication and allow for more thoughtful discussions with families about continuing or limiting care as soon as a few days into an ECMO course. Genetic testing a priori may also limit ECMO utilization if the therapy has no potential benefit.
Historically, the turnaround time of the results of WES and WGS was a significant barrier to their use as a screening tool. This barrier remains for many centers where results may not be available for 1–2 weeks. Rapid diagnosis, especially in cases where results may be available in 1–3 days, may reduce the need for invasive diagnostic testing such as a lung biopsy while on ECMO. At most centers, testing is done sequentially with microarray followed by WES or WGS if microarray is non-diagnostic. However, this is likely to change in the future as most laboratories now offer WES and WGS with copy number analysis, making way for WES, and more likely WGS, to become first line tests. Universal screening with WES or WGS would also be more cost effective than sequential testing by reducing the number of tests sent for each neonate, decreasing time to result of testing, and theoretically reducing duration of treatment and/or duration of hospitalization. Additional barriers included obtaining testing approval, completing a comprehensive consent, specifically having the support of geneticists, and obtaining parental samples. Parental samples are important as testing sent as a trio with a sample from the neonate, as well as from both parents, is the most informative for interpretation.
Given that the majority of centers would consider incorporating universal screening with rapid WES or WGS for neonates upon ECMO cannulation if readily available, these are likely to become standard of care for neonates on ECMO. While genetic testing would ideally be sent at or before cannulation, it can be sent at any time during an ECMO course as testing is performed on white blood cells. This is contrary to common misconceptions that genetic testing cannot be sent after blood transfusions or initiation of ECMO.
This survey also found low use of autopsy for neonates on ECMO, which is consistent with a recent CHNC study that found low utilization and high variability in the use of autopsy for neonates in level IV NICUs. 23 Universal WES and WGS may provide a less invasive diagnosis for many of these neonates and provide closure and recurrence risk information for families.
The widespread availability of rapid and “ultra-rapid” WES and WGS brings up several ethical questions. In the era of rapid genetic diagnosis, there may be neonates for whom ECMO is or is not offered based on a coexisting genetic diagnosis, which may not fit the traditional criteria of reversibility. This is further complicated by the expanded use of genetic testing, which has led to increased diagnostic rates in patients with milder phenotypes who may not have received a molecular diagnosis previously and may impact who is offered ECMO. This broadening of the phenotypic spectrum for many conditions complicates the decision of who is a candidate for ECMO. There may also be diagnoses where there is not enough evidence to support or refute the utility of ECMO. There are also many interstitital lung diseases that may be diagnosed with genetic testing including alveolar capillary dysplasia and various surfactant deficiencies wherein the approach may have to be individualized. For example, a relevant diagnosis may redirect the ECMO course from expected recovery to a potential bridge to lung transplant, or may exclude patients who have a lethal diagnosis and are not transplant candidates. Thus, it is important that ECMO providers collaborate with geneticists in the interpretation of genetic testing and counseling of families.
The strengths of this study include that it is the largest, and to our knowledge, the only, description of genetic testing practices during neonatal ECMO utilization. Our survey incorporated responses from multiple children’s hospitals with a good response rate of 66%. Limitations include a survey design which may introduce bias by individual respondents. The survey only included large referral-based level IV NICUs, almost all of whom are affiliated with robust academic centers and pediatric subspecialists. Given this, our findings may not be as applicable to other non-academic based NICUs that provide neonatal ECMO, but may have limited geneticist support to offer and interpret complex test results, which may contribute to disparities in care. While this survey found that most centers would consider incorporating universal screening with WES or WGS for neonates on ECMO for respiratory failure, future work will still be needed to demonstrate benefit of widespread comprehensive genetic testing for neonatal ECMO patients.
Conclusions
WES and WGS are emerging as the first line diagnostic tests for critically ill neonates. Despite variation in the use of WES and WGS, rapid testing may offer earlier diagnosis, be useful for prognostication, and influence treatment course among neonates on ECMO. Cost is the primary barrier to utilization and most centers would consider incorporating universal screening with WES or WGS for neonates on ECMO if the testing was economical and ultra rapid testing was readily available. Additional barriers include lack of access to rapid testing with prolonged turnaround time of results.
Supplemental Material
Supplemental Material - Current practices for genetic testing in neonatal extracorporeal membrane oxygenation: Findings from a National survey
Supplemental Material for Current practices for genetic testing in neonatal extracorporeal membrane oxygenation: Findings from a National survey by KT Wild, Franscesca Miquel-Verges, Natalie E Rintoul, Robert DiGeronimo, Sarah Keene, Shannon E Hamrick, Burhan Mahmood, Rakesh Rao, Nicholas R Carr, on behalf of the Children’s Hospitals Neonatal Consortium (CHNC) in Perfusion
Footnotes
Acknowledgements
Beverly Brozanski, Jacquelyn Evans, Theresa Grover, Karna Murthy, Michael Padula, Eugenia Pallotto, Anthony Piazza, Kristina Reber and Billie Short and ex-officio David Durand, Francine Dykes, Jeanette Asselin), are executive members of the Children’s Hospitals Neonatal Consortium, Inc., who developed and manage the CHND (![]() ). For more information, please contact:
). For more information, please contact:
We are indebted to the following CHNC participating institutions that serve the infants and their families; these institutions (and their site sponsors) also have invested in and continue to participate in the Children’s Hospital’s Neonatal Database (CHND):
1. Alfred I. duPont Hospital for Children, Wilmington, DE (Kevin Sullivan)
2. All Children’s Hospital Johns Hopkins Medicine, St. Petersburg, FL (Victor McKay)
3. American Family Hospital, Madison, WI (Jamie Limjoco, Lori Haack)
4. Advent Health for Children, Orlando, FL (Narenda Dereddy, Raj Wadhawan)
5. Ann & Robert H. Lurie Children’s Hospital of Chicago; Chicago, IL (Karna Murthy, Gustave Falciglia)
6. Arkansas Children’s Hospital, Little Rock, AR (Becky Rogers)
7. Boston Children’s Hospital, Boston, MA (Anne Hansen)
8. Brenner Children’s Hospital, Winston-Salem, NC (Cherrie Welch)
9. Cincinnati Children’s Hospital; Cincinnati, OH (Beth Haberman)
10. Children’s Healthcare of Atlanta at Egleston; Atlanta, GA (Anthony Piazza)
11. Children’s Healthcare of Atlanta at Scottish Rite; Atlanta, GA (Gregory Sysyn)
12. Children’s Hospital and Medical Center, Omaha, NE (Nicole Birge)
13. Children’s Hospital Colorado, Aurora, CO (Theresa Grover)
14. Children’s Hospital of Orange County; Orange, CA (Michel Mikhael, Irfan Ahmad)
15. Children’s Hospital of Philadelphia; Philadelphia, PA (Michael Padula, David Munson)
16. Children’s Hospital of Pittsburgh of UPMC; Pittsburgh, PA (Toby Yanowitz)
17. Children’s Hospital of Wisconsin, Milwaukee, WI (Michael Uhing, Ankur Datta)
18. Children’s Medical Center; Dallas, TX (Rashmin Savani, Luc Brion)
19. Children’s Mercy Hospitals and Clinics, Kansas City, MO (Julie Weiner)
20. Children’s National Medical Center, Washington, DC (Billie Short, Lamia Soghier)
21. Children’s of Alabama, Birmingham, AL (Carl Coghill, Allison Black)
22. Children’s Hospital of Los Angeles, Los Angeles, CA (Rachel Chapman)
23. Children’s Hospital of Michigan, Detroit, MI (Girija Natarajan)
24. Connecticut Children’s Medical Center, Hartford, CT (AnneMarie Golioto)
25. Cook Children’s Medical Center, Fort Worth, TX (Jonathan Nedrelow, Annie Chi, Yvette Johnson)
26. Dell Children’s Hospital, Austin, TX (Ashley Lucke, Molly Pont)
27. Hospital for Sick Children, Toronto, Ontario, Canada (Kyong-Soon Lee)
28. Le Bonheur Children’s Hospital, Memphis, TN (Mark Weems)
29. Levine Children’s Hospital, Charlotte, NC (Eugenia Pallotto)
30. Nationwide Children’s Hospital, Columbus, OH (Thomas Bartman)
31. Nemours Children’s Hospital, Orlando, FL (Aaron Weiss)
32. Oklahoma Children’s Hospital, Oklahoma City, OK (Trent Tibble)
33. Primary Children’s Hospital, Salt Lake City, UT (Con Yee Ling, Shrena Patel)
34. Rady Children’s Hospital, San Diego, CA (Mark Speziale, Brian Lane, Laurel Moyer)
35. Riley Children’s Hospital, Indianapolis, IN (Rebecca Rose)
36. Seattle Children’s Hospital, Seattle, WA (Elizabeth Jacobsen-Misbe, Robert DiGeronimo)
37. Stead Family Children’s Hospital, Iowa City, IA (Julie Lindower)
38. St Christopher’s Hospital for Children, Philadelphia, PA (Vilmaris Quinones)
39. St. Louis Children’s Hospital, St. Louis, MO (Beverly Brozanski, Rakesh Rao)
40. Texas Children’s Hospital, Houston; Houston, TX (Kristina Reber, Lakshmi Khatakam)
41. UCSF Benioff Children’s Hospital Oakland; Oakland, CA (Priscilla Joe)
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
This manuscript is original research, has not been previously published and has not been submitted for publication elsewhere while under consideration.
Author Note
Data and references can be made available upon request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
