Abstract
Background
Patients with kidney failure are at risk for lethal complications from hyperkalemia. Resuscitation, medications, and hemodialysis are used to mitigate increased potassium (K+) levels in circulating blood; however, these approaches may not always be readily available or effective, especially in a resource limited environment. We tested a sorbent cartridge (KC, K+ontrol CytoSorbents Medical Inc., Monmouth Junction, New Jersey) which contains a resin adsorber for K+. The objective of this study was to test the utility of KC in an ex vivo circulation system. We hypothesized that KC reduces K+ levels in extracorporeal circulation of donor swine whole blood infused with KCl.
Methods
A six-hour circulation study was carried out using KC, a NxStage (NxStage Medical, Inc., Lawrence, MA) membrane, blood bag containing heparinized whole blood with KCl infusion, 3/16-inch ID tubing, a peristaltic pump, and flow sensors. The NxStage permeate line was connected back to the main circuit in the Control group (n = 6), creating a recirculation loop. For KC group (n = 6), KC was added to the recirculation loop, and a continuous infusion of KCl at 10 mEq/hour was administered for two hours. Blood samples were acquired at baseline and every hour for 6 h.
Results
In the control group, K+ levels remained at ∼9 mmol/L; 9.1 ± 0.4 mmol/L at 6 h. In the KC group, significant decreases in K+ at hour 1 (4.3 ± 0.3 mmol/L) and were sustained for the experiment duration equilibrating at 4.6 ± 0.4 mmol/L after 6 h (p = 0.042). Main loop blood flow was maintained under 400 mL/min; recirculation loop flow varied between 60 and 70 mL/min in the control group and 45–55 mL/min in the KC group. Decreases in recirculation loop flow in KC group required 7% increase of pump RPM.
Conclusions
During ex-vivo extracorporeal circulation using donor swine blood, KC removed approximately 50% of K+, normalizing circulating levels.
Introduction
Hyperkalemia is a life-threatening acute electrolyte abnormality that is defined as serum potassium (K+) levels greater than 5 mEq/L and regardless of the cause can lead to cardiac dysrhythmias and increased risk of mortality.1,2 In hyperkalemic states, fast sodium channels are activated more readily, due to the cell membrane being more depolarized and the resting potential is brought closer to action potential initiation. Further increases in serum potassium levels lead to inactivation of sodium channels and prevention of action potential initiation. 1 This leads to deleterious effects in neuromuscular systems, especially in the heart, leading to lethal arrhythmias. 3 Kidney function is the most important factor in K+ homeostasis. In normal conditions, K+ is freely filtered by the glomerulus and most is reabsorbed in the proximal convoluted tubule. The bulk of K+ secretion occurs in the distal convoluted tubule, and secretion is affected by tubular fluid flow rate, aldosterone stimulation and elevated sodium and fluid loads.4,5
Hyperkalemia is one of the most common complications of acute kidney injury (AKI). 6 Renal damage caused by trauma and hypoperfusion can lead to impaired K+ secretion. 7 Hyperkalemia prevention or mitigation is particularly important during management of trauma patients as it can develop earlier or later after injury due to crush syndrome, arterial injury, use of hemorrhage control techniques (e.g. – tourniquets or intravascular catheters), or blood transfusions.8–11 In non-crush trauma patients, 29% developed hyperkalemia within 12 h after admission, with some patients having K+ levels of 9 mmol/L or higher with all of the fatalities having hyperkalemia. 10 A retrospective analysis by Stewart et al. 11 described hyperkalemic combat casualties as having higher injury severity scores, shock index, and greater likelihood of fatal AKI. The authors further emphasized that strategies to allow for early mitigation of hyperkalemia should be investigated, especially for use in scenarios of delayed evacuation from the point of injury.
Standard medical management of hyperkalemia involves the administration of drugs that affect K+ secretion and its transfer between extracellular and intracellular spaces. 3 Additionally, hemodialysis (HD) and continuous kidney replacement therapies (CKRT) are commonly applied to remove excess ions and metabolic byproducts (including K+) and mitigate the effects of AKI in the absence of normal kidney function. During HD, dialysis solution with a low K+ concentration is passed through a hollow fiber membrane in contact with blood, and K+ diffuses across the concentration gradient from serum to the dialysate. 3 These therapies have also been used in combat conditions since the Korean War 12 and, most recently, during Operation Iraqi Freedom and Operation Enduring Freedom. 12 However, specialized expertise and a significant amount of dialysate and replacement fluid (1–3 L/h, 20–40 L/day)13–15 are required, which may not be available in a remote or austere environment outside of a hospital or dialysis center. 16 These logistical challenges limit HD and CKRT to specialized Military Treatment Facilities.
An alternative solution for hyperkalemia in the austere, resource-limited environment but also in hospitals, could be utilization of sorbent-based approaches. This type of therapy could be utilized as a “bridge” to help patients survive until they are able to receive definitive care.
To meet the above goals, a sorbent cartridge (KC, K+ontrol, CytoSorbents Medical Inc., Monmouth Junction, New Jersey) has been developed which eliminates K+, without the excessive fluid requirements used in traditional HD and CKRT systems. The KC device utilizes a hemocompatible resin adsorber polymer that is designed to bind potassium ions. It is envisioned to be used standalone with a simple peristaltic blood pump; or as an adjunct to renal replacement therapy, extracorporeal membrane oxygenation, or any combination of these therapies. In this study we tested this new generation sorbent device during ex vivo circulation using swine donor blood. We hypothesized that KC would reduce K+ levels in extracorporeal circulation of donor swine whole blood infused with KCl.
Methods
A benchtop ex vivo circulation system (Figure 1) was developed combining KC with a NxStage PUREMA Hemodialysis membrane (CAR-505 Cartridge set; NxStage Medical, Inc., Lawrence, MA) to test the efficacy of the KC cartridge on K+ removal in swine whole blood. Overview and schematic of circulation setup: (a) Shaking water bath with blood bag; (b) Masterflex Peristaltic Pump with variable RPM; (c) NxStage CKRT Membrane; (d) K+ontrol Cartridge; (e) KCl Infusion Pump; (f) Flow Meters (f1 = Main loop flow, f2 = Recirculation Flow).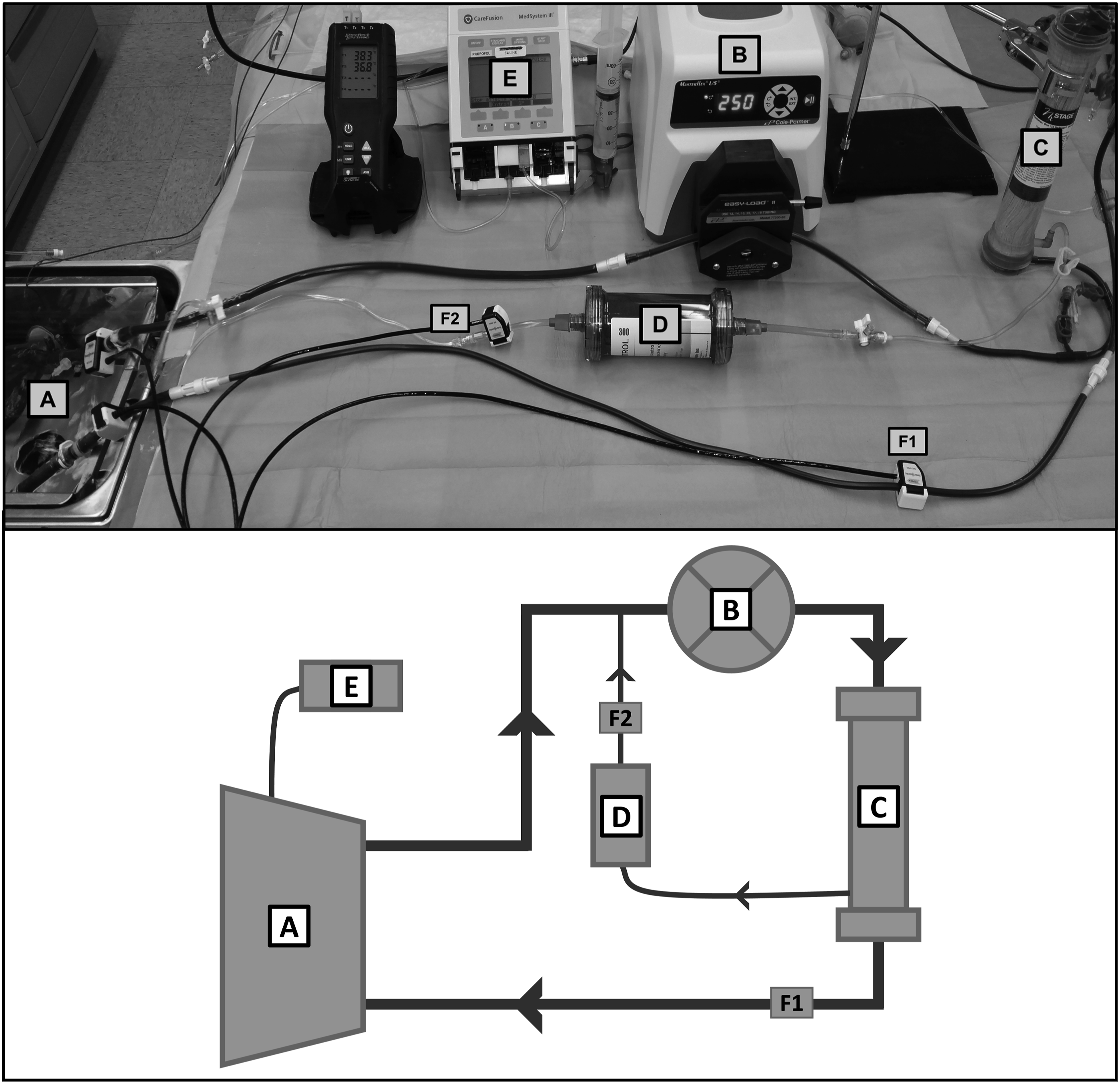
Circuit setup and study groups
A six-hour circulation study was performed using whole blood from injured swine subjected to trauma and round the clock ICU care. The test platform was constructed with a blood bag (BB), peristaltic pump (Cole-Parmer Masterflex, Vernon Hills, IL), NxStage membrane (NX), and 3/16-inch inner diameter (ID) tubing (Figure 1(a)–(c)) resulting in a circulation loop. Additionally, a recirculation loop (Figure 1(d)) was created using the 1/8-inch ID NxStage permeate line which was connected back to the main blood loop before the pump. This recirculation loop has a KC installed inline (KC group) or is a direct connection (CTRL group). Clamp-on flow sensors (ME4PXL, Transonic Systems, Ithaca NY, USA) were used to measure both main loop flow (Figure 1(f1)) and recirculation loop flow (Figure 1(f2)).
Blood collection
End of study blood collection was performed via terminal exsanguination according to an IACUC approved protocol in an AAALAC accredited facility from swine used in other studies within our Autonomous Reanimation and Evacuation Research Program (AREVA). Whole blood (∼2 L) was collected at a rate of 100 mL/min. Unfractionated heparin was added at a dose of 1000 units per liter of blood to prevent coagulation. Heparin was utilized in lieu of citrate anticoagulation to limit dilution of blood with anticoagulant. Immediately following collection, blood was transferred from the collection bags into the BB and circuit flow was initiated in a loop without the NX using a 3/16-inch ID line connected from the pump exit to the BB.
Pre-baseline circulation preparation
Heparin was added as needed to achieve an activated clotting time (ACT) greater than 999 s, measured by a Hemochron Signature Elite analyzer (Instrumentation Laboratory, Bedford, MA). The BB was placed in a shaking water bath (SW21, JULABO GmbH, Seelbach, Germany) set to 38.0°C. Pump RPM was adjusted to ensure at least 50 mL/min of flow through the permeate line, while maintaining main loop flow under 400 mL/min.
Blood gas analysis was performed using a GEM4000 analyzer (Instrumentation Laboratory, Bedford, MA). Plasma free hemoglobin (PFHb) was measured using the HemoCue Plasma/Low HB System (HemoCue AB; Angleholm, Sweden). Blood samples were obtained via a luer sampling port on the BB.
Study initiation
After initial blood sampling and circuit flow initiation, the NX was connected between the pump exit and BB. Afterwards, slow continuous ultrafiltration was performed in 200 mL increments followed by repeat blood sampling and analysis to increase hematocrit to 35%–40% as determined by the GEM4000 analyzer. Once the hematocrit goal was reached, the NX permeate line (the recirculation loop) was connected to the main circuit with or without a KC based on the study group. In both groups, a 5 mEq potassium chloride (KCl) bolus was added to the system to increase circulating K+ levels to 8–10 mmol/L. Additionally, in KC group, a KCl infusion pump infused 20 mEq KCl into the BB over the first 2 h (10 mEq/h) of the study. In CTRL group there is no continuous KCl infusion. Blood samples for the baseline timepoint (BL) were taken once the tubing was connected and recirculation initiated. Blood analysis (pH, pO2, pCO2, K+, Hct), pump RPM, and flow rates were recorded for BL and hourly for 6 h. In the KC group, the KCl infusion was terminated at the start of the third hour of the study. A study schematic showing the main execution steps is shown in Figure 2. Flow diagram detailing various study steps. The main difference between the two groups is the addition of a KC in the recirculation loop and continuous KCl infusion for the first 2 h of the study in the KC group.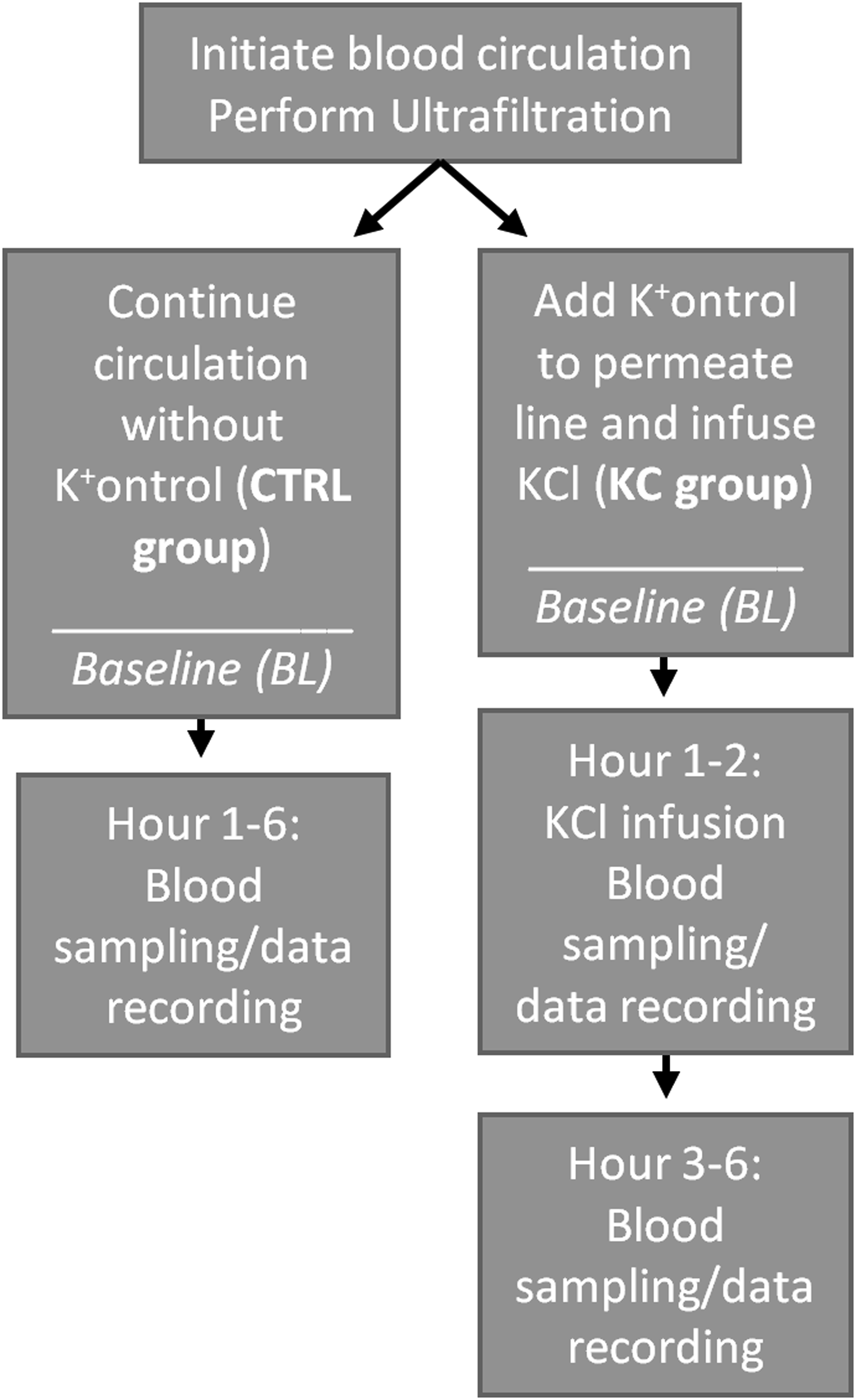
Statistical analysis
Using SAS 9.3 (Cary, NC) and an alpha of 0.05 for significance, a two-sample test for equivalence was conducted comparing BL to hourly values within each group, followed by an independent variable test to compare between groups. Data is reported as mean ± standard error of the mean.
Results
Potassium levels and flow rates
Potassium concentration and flow rate results are shown in Figure 3. In the KC group, K+ levels decreased as soon as recirculation through the KC was initiated, and the levels normalized to about 4 mmol/L throughout the duration of the study. In the CTRL group, K+ levels were maintained ∼9 mmol/L for the duration of recirculation. Significant differences between the two groups were observed starting at hour one and continued to the end of the study. For blood pump RPM, there was no statistical difference between the two groups; however, in hours five and six within both groups, there was a significant increase in RPM compared to BL. There was a significant increase in main loop flow in KC group compared to CTRL group from hour two until the end of the study. There was no statistical difference between the groups for recirculation flow. Mean ± standard error of potassium (K+) concentration, pump revolutions per minute (RPM) and. Fluid flow rates in the main blood loop and recirculation loop. * denotes a statistical difference between BL and the hourly timepoint for the KC group, ** denotes a statistical difference between BL and the hourly timepoint for the CTRL group, † denotes a statistical difference between groups at the timepoint. P < 0.05 for significance.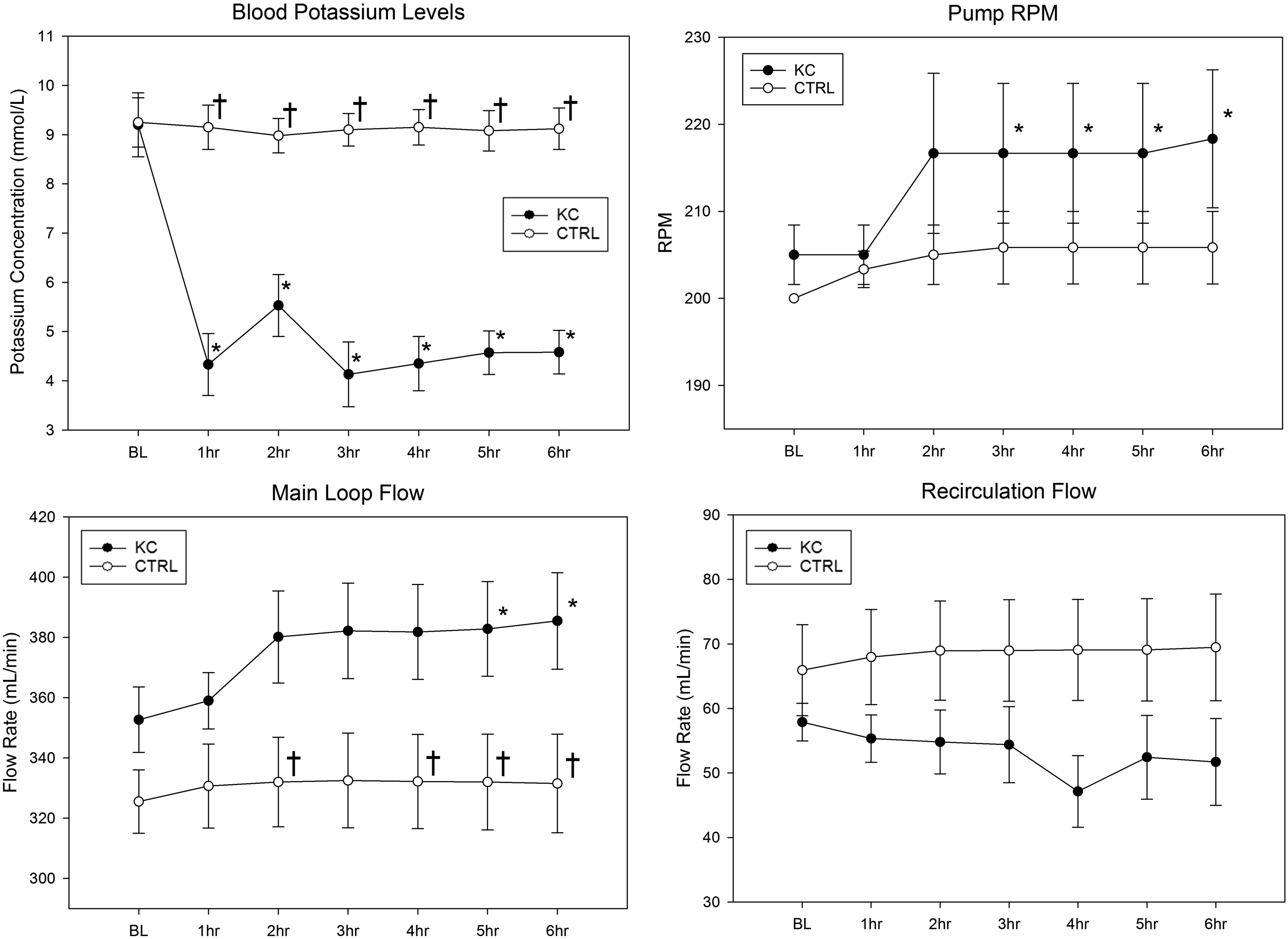
Blood analysis
Mean ± standard error describing blood activated clotting time (ACT), pH, pCO2, pO2, K+, hematocrit and plasma free hemoglobin. * denotes a statistical difference between BL and the hourly timepoint for the KC group, ** denotes a statistical difference between BL and the hourly timepoint for the CTRL group, † denotes a statistical difference between groups at the timepoint. P < 0.05 for significance.

Discussion
This study involved extracorporeal circulation of swine donor blood to which KCl was administered. We established significant and sustained K+ removal by employing a KC cartridge and demonstrated normalization of K+ levels versus controls that received KCl but no KC therapy.
Clinical management of hyperkalemia is dependent on the condition of the patient and the state of functioning organs available for K+ hemostasis. Regulation of potassium is largely performed by the kidneys, with some excretion (10%) performed by the gastrointestinal tract. 17 K+ levels can be maintained during acute hyperkalemia by the administration of drugs such as sodium bicarbonate or diuretics (or binders/sorbents to increase GI excretion), but most therapies require sufficient kidney function. 18 In the setting of AKI, HD or CKRT is initiated to ensure patient survival. Clearance of K+ is proportional to dialysate flow and the rate of ultrafiltration, and some therapies can see a daily replacement/dialysate fluid requirement of 30 L or more. 14 This large fluid requirement, in combination with multiple patients and a potential lack of supplies, can make these therapies difficult if not impossible to perform outside of a hospital or specialized care center.
Our goal to investigate K+ levels of 8–10 mmol/L follows closely with the high potassium levels observed in hyperkalemic trauma patients that do not survive their injuries. 10 At these levels there is a significant risk of arrhythmic complications and multiple studies have shown the link between hyperkalemia and early mortality.10,11 In the deployed setting, without access to HD or CKRT, patients that develop hyperkalemia from their injuries are at an even higher risk of death. Furthermore, at extreme levels of hyperkalemia the treatment of choice is a method of kidney replacement therapy that has a high clearance (i.e. HD). However, in the case of traumatic brain injury, a common injury in both the deployed and civilian settings, high clearances are contra-indicated due to the potential for increasing cerebral edema. 19 A method of potassium removal that could augment low clearance CKRT could be beneficial in such patients. A similar situation arises in the setting of cerebral ischemia. 20 Therefore, in addition to utility in the austere, deployed environment, the KC may have utility in the civilian setting when hyperkalemia is complicated by increased intra-cranial pressure.
Within the first hour, we observed a significant 50% decrease in K+ concentrations when comparing CTRL group (∼9 mmol/L) to the KC group (∼4 mmol/L), with the levels maintaining until the end of the study. We conclude that KC was able to remove K+ from the circulating blood, even while KCl was being infused. We noticed a slight increase in K+ levels at hour two of the study. We believe that this is a combination of two factors: this blood sample is taken while the continuous KCl infusion is still in progress, and the KC absorbed a large amount of K ions quickly during the first hour. This increase is no longer apparent from hour three until the end of the study, so the excess ions are eventually absorbed and the K+ levels equilibrate to normal levels between 4 and 5 mmol/L. To compare with current clinical results: patients who received oral K+ binding agents achieved normokalemia between 24 and 48 h after treatment. 21 In cases of severe hyperkalemia requiring hemodialysis, treatment can take from one to several hours to stabilize K+ levels and reduce mortality. 22
While the use of swine blood from donors that were subjected to traumatic injuries, blood loss, anesthesia and up to 72 h of ICU therapy is highly relevant to clinical care; the donor blood was potentially hemodiluted and there was variability between donors. We attempted to standardize the blood hematocrit levels by performing ultrafiltration prior to BL. To further comment on blood viability and quality throughout the study: within each group we observed a significant decrease of pH and pO2 values starting at the second hour, compared to BL. We believe this is due to the ex vivo nature of the experiment, as there is no lung or oxygenator to replenish oxygen and a slow decrease is expected. At the third hour and fourth hour for KC and CTRL groups, respectively, we also observed a significant increase in PFHb values compared to BL. For both groups the increase continued to the end of the six-hour study. The PFHb levels for both groups were highly variable between study repetitions, underscoring the variability of donor blood. No between-group differences were shown.
Flow throughout the main loop was numerically higher in the KC group, ∼400 mL/min, compared to ∼350 mL/min in the CTRL group. This is because an increase in pump RPM was required to ensure at least 50 mL/min of flow through the KC in the recirculation loop. Still, the flow rates were not statistically different, and we do not believe that the flow rate increase in the main loop influenced KC performance and K+ adsorption, as shown by the significant differences in K+ levels between the two groups. One way to ensure that the flow rates are the same in future experiments is to use a sham device with similar pressure drop to KC. Despite this increase in RPM, there were no differences in PFHb between the two groups implying that this did not result in increased hemolysis. We did not incorporate an additional pump for independent control recirculation loop flow rates. To note, the NxStage console and therapy system has the capability to utilize multiple pumps for blood flow, replacement fluid and permeate fluid; and each can be controlled independently. The circulation system described in this study has benefits in simplicity for potential use cases in portable, austere, or other non-hospital settings where simplicity and miniaturization are key features. An additional limitation of this study is the lack of pressure monitoring; however this will be incorporated in future studies.
While the KC device has been examined in two prior swine models (one involving a potassium infusion 23 and one involving hemorrhage, ischemia/reperfusion, and crush 24 ), the present study adds to the literature in four ways. First, it confirms these prior studies with regards to the efficacy of the KC for reducing hyperkalemia. Second, it demonstrates that utilizing the device does not increase the risk of hemolysis. Third, by using an ex vivo system this study allows for a better characterization of the device’s ability to remove potassium separate from the organism’s adaptive response (as in the infusion study 23 ) and variable K+ generation (as in the crush model 24 ). Last, ex vivo studies allow for easier optimization of the set up. This could entail developing methods to run the KC concurrently with traditional CKRT or to utilize CKRT systems (rather than a non-FDA approved blood pump) to implement the therapy.
The K+ontrol cartridge may be a promising new adjunct for metabolic management of injured patients with ischemia reperfusion injury and multiorgan failure. Future studies will involve in vivo testing of the cartridge in conjunction with extracorporeal life support in large animals with combat relevant trauma and multiorgan failure, carried out to 72 h following injury.
Footnotes
Acknowledgments
The opinions and assertions expressed herein are those of the authors and do not necessarily reflect the official policy or position of the Uniformed Services University or the Department of Defense.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.D.G., M.G., T.K., T.G., P.P.C. are employed by CytoSorbents Corporation and receive salary and stock compensation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Congressionally Directed Medical Research Program under via award W81XWH-17-2-0013, administered via subaward to The Geneva Foundation, P.I. Andriy Batchinsky, MD.
