Abstract
Introduction
In peripheral percutaneous (VA) extracorporeal membrane oxygenation (ECMO) procedures the femoral arteries perfusion route has inherent disadvantages regarding poor upper body perfusion due to watershed. With the advent of new long flexible cannulas an advancement of the tip up to the ascending aorta has become feasible. To investigate the impact of such long endoluminal cannulas on upper body perfusion, a Computational Fluid Dynamics (CFD) study was performed considering different support levels and three cannula positions.
Methods
An idealized literature-based- and a real patient proximal aortic geometry including an endoluminal cannula were constructed. The blood flow was considered continuous. Oxygen saturation was set to 80% for the blood coming from the heart and to 100% for the blood leaving the cannula. 50% and 90% venoarterial support levels from the total blood flow rate of 6 l/min were investigated for three different positions of the cannula in the aortic arch.
Results
For both geometries, the placement of the cannula in the ascending aorta led to a superior oxygenation of all aortic blood vessels except for the left coronary artery. Cannula placements at the aortic arch and descending aorta could support supra-aortic arteries, but not the coronary arteries. All positions were able to support all branches with saturated blood at 90% flow volume.
Conclusions
In accordance with clinical observations CFD analysis reveals, that retrograde advancement of a long endoluminal cannula can considerably improve the oxygenation of the upper body and lead to oxygen saturation distributions similar to those of a central cannulation.
Keywords
Introduction
Veno-arterial extracorporeal membrane oxygenation (VA-ECMO) is used for cardiac support. 1 In the majority of cases, the VA ECMO is placed peripherally, via a percutaneous cannulation of the femoral artery. A central placement of the VA ECMO is possible by thoracotomy.
In impaired lungs, the oxygenated blood coming from the cannula in the femoral artery has to compete with the blood coming from the physiological circulation. This so-called watershed phenomenon leads to a decrease of upper body and cerebral oxygenation. The resulting difference in oxygenation between the upper and lower part of the body is known as Harlequin Syndrome.2–4
The development of new long, flexible cannulas enables the endoluminal advance of the cannula from the femoral artery to the proximal aorta, and from the femoral vein to the pulmonary artery and left atrium. First clinical experience with left atrial approaches and transluminal aortic perfusion has been encouraging. Hatam et al. published their first positive clinical experience with such cannulas. He placed an 18 Fr long flexible cannula via the jugular vein into the main pulmonary artery for right ventricular support. 5
Computational Fluid Dynamics (CFD) is generally used to perform calculation of fluid flow based on the Navier-Stokes equations and assumption of boundary conditions. A mesh that is used for discretisation of the computational domain can be generated from medical imaging data. 6 In the postprocessing step, results can be visualized and quantified to analyse local flow phenomena and oxygen distribution. This makes CFD an optimal tool for examining different cannula positions with various support levels.
As a basis for further clinical decision making, the aim of our work was to assess the local distribution of the flow and oxygen saturation in the ascending aorta, coronaries, and supra-aortic arteries depending on the anatomy, the endoluminal position of the cannula, and the support level.
Materials & methods
Study design
Computational Fluid Dynamics was applied to analyse the perfusion of the aorta, coronary and supra-aortic arteries for endoluminal cannulation. Two aortic geometries were used for mesh-generation. The first geometry is based on an idealized geometry, while the second one was segmented from anonymous patient MRI data. Three cannula positions were chosen: descending aorta, aortic arch, and ascending aorta (see Figure 1). The total cardiac output was set at 6.0 L/min and two different cases were studied. In the first case, the blood flow in the cannula was equal to the flow coming from the heart, each at 50% (3 L/min) of the total cardiac output. Cannula Positions for ideal and patient aorta.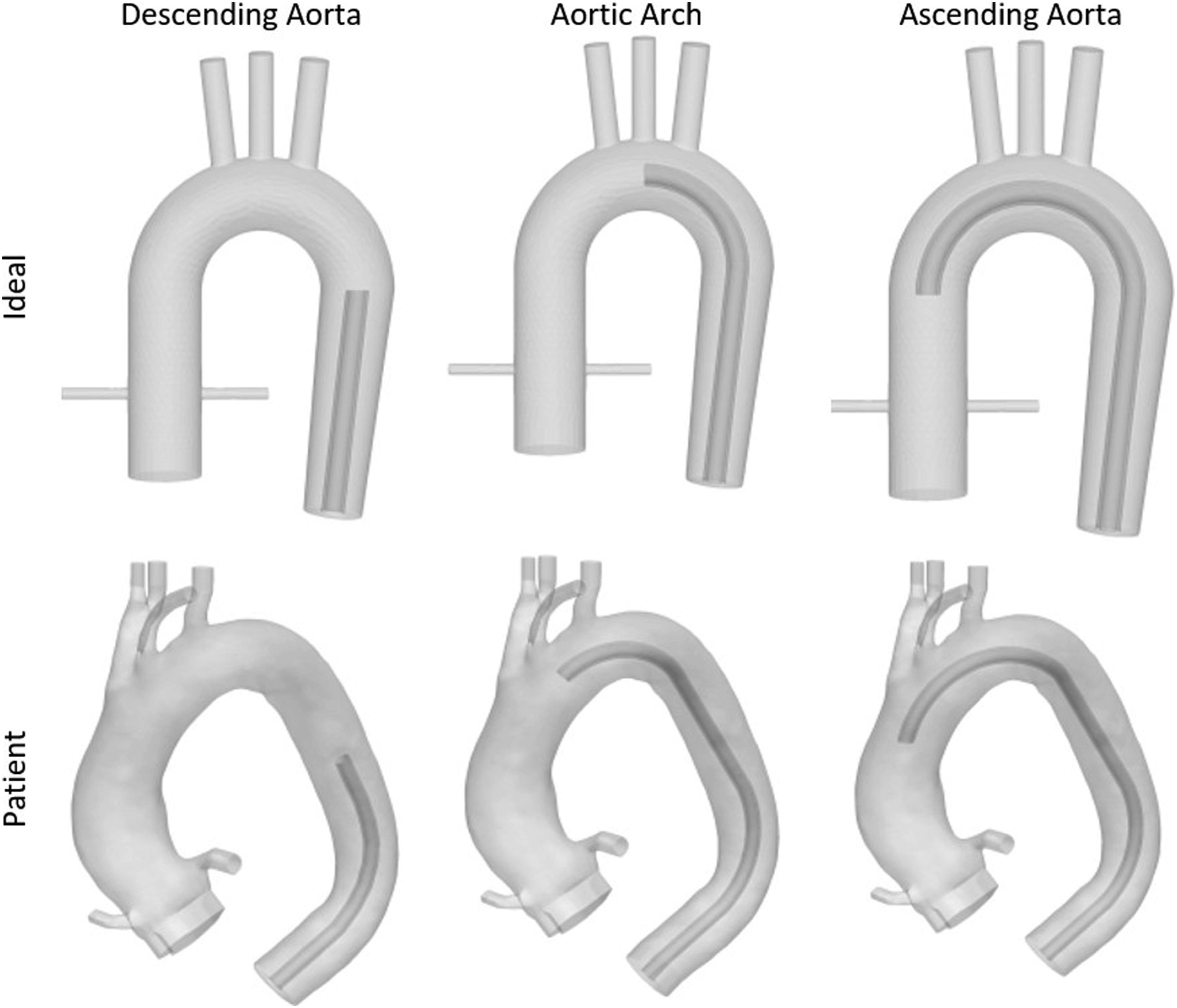
In the second case, 90% (5.4 L/min) of the total blood flow was coming from the cannula and only 10% (0.6 L/min) from the heart. The oxygen saturation of blood coming from the heart was set to 80% (which represents a clinically realistic value in patients on VA ECMO with severely impaired lungs) and to 100% for the blood leaving the cannula. The resulting oxygen saturation of the blood was averaged over mesh cells at the off-branching arteries. The flow was idealized to be non-pulsatile and due to the transient flow behaviour both laminar and turbulent computations were performed. However, the results for turbulent and laminar simulations were very similar. Therefore, only the turbulent computations will be shown in the results section.
Computational simulation
The open-source software Code_Saturne V 7.0, developed and released by Électricité de France (EDF, Paris, France), was used as a Navier-Stokes equation solver for incompressible Newtonian flow simulation. The density was set to 1050 kg/m³ and viscosity to 3.5 mPa*s. A K-omega Shear Stress Transport (SST) turbulence model and a laminar flow model were used, because of anticipated Reynolds numbers between 1500 in the aorta and 6300 in the cannula at a defined maximum volumetric flow rate. The pseudo-steady (SIMPLEC) algorithm was selected with a reference time step of 0.1s.
Mesh geometry
Two geometries were constructed: one in form of an idealized aorta and another from a patient aorta for comparison. The idealized aorta is based on a study by Paritosh Vasava. 7 Modifications include adding coronary arteries to the ascending aorta, while the ascending aorta itself was modified to have a more realistic length.
The second geometry is an aorta model constructed from anonymized patient MRI data. Using Medical Imaging Interaction Toolkit Workbench (MITK, Medical and Biological Informatics (MBI) of the German Cancer Research Center), areas of interest were segmented and finally modified by importing them into the open-source pre-processing software Salome V 8.5 (Open Cascade S.A.S., Guyancourt, France) for further modification. Only major arteries branching off from the aorta were left attached to the geometry, whereas clean, planar inlets and outlets were made by clipping segments from the construct.
A computational mesh was generated for each aorta model and cannula position respectively consisting of approximately five hundred thousand tetrahedral cells, while extending layers consisting of triangular prism cells were added at the inlets and outlets (see dense blue regions in Figure 2). A single viscous layer of homogenous hexahedral cells was added to improve the flow computation at the vessel walls of the real patient aorta. Ideal aorta (left) and human aorta (right). Tetrahedral meshes with triangular prism extrusions.
Boundary conditions
At the inlet, Dirichlet boundary conditions were imposed as a constant velocity based on the cannula blood support level. The face from the descending aorta was defined as an outlet with homogeneous Neumann conditions imposed on all transported variables. Coronary and supra-aortic arteries were defined as negative inlets to simulate the realistic distribution of blood supply. The negative inlet velocity was calculated by considering its percentage from the total volumetric flow rate. The used percentages are based on measurements of physiological blood flow. 8 All walls were defined as rigid and unmoving with a no slip condition.
Results
Flow characterization
The results show occurring watershed depending on the position of the cannula. For every position of each aorta model a high velocity retrograde flow coming from the cannula enters the aorta (red streamlines) and meets the slow antegrade blood flow (blue streamlines) coming from the heart, resulting in different watershed locations (see Figure 3). Results of velocity patterns presented by streamlines in different cannula positions and for different support levels. For better visualisation, the legend values were scaled to highlight low flow velocities inside the aorta.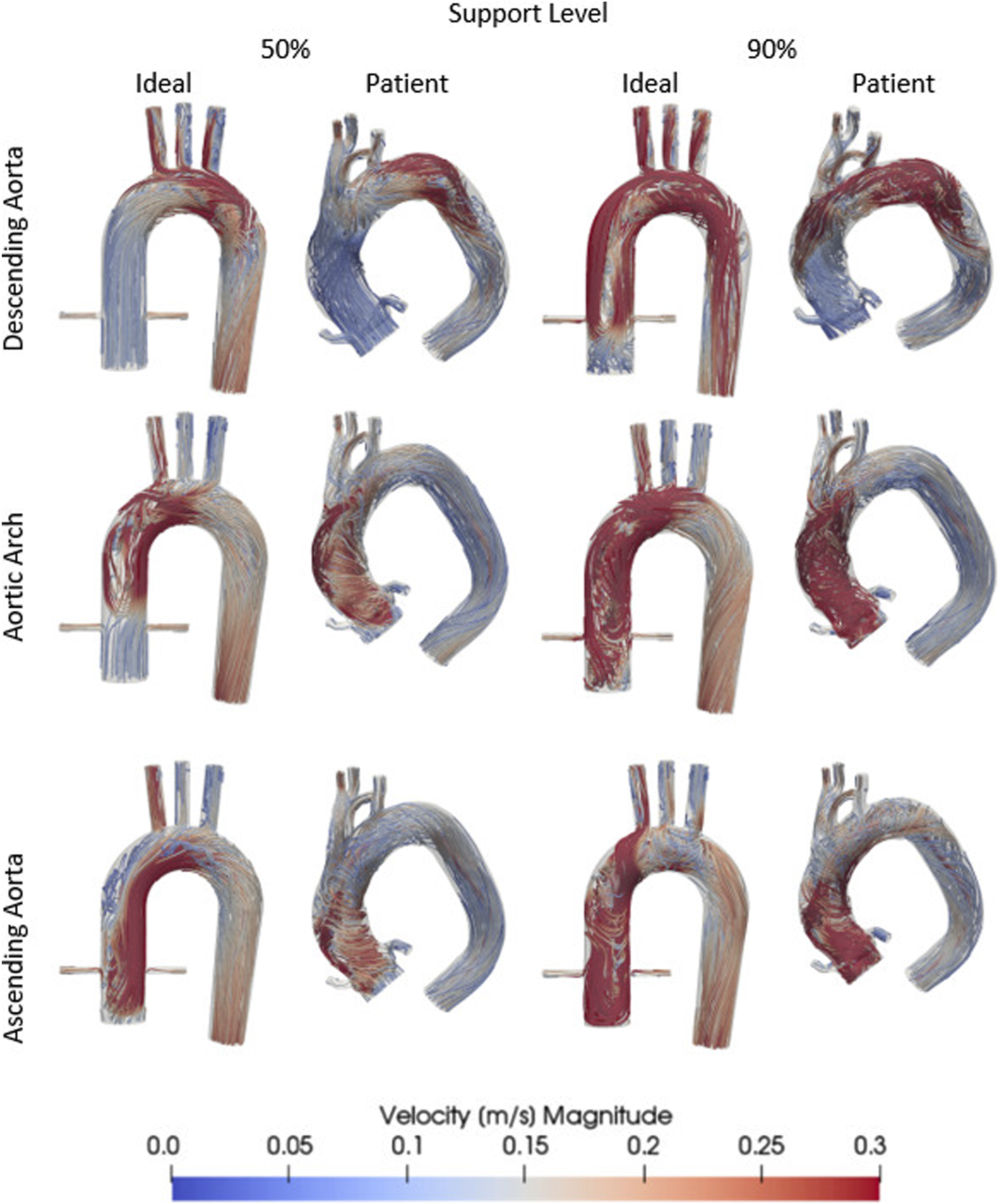
Cannula placement at the descending aorta shows watershed positions in the aortic arch between the brachiocephalic artery (BA) and left common carotid artery (LCCA) for 50% venoarterial support and at the ascending aorta for 90% cannula support. Placed at the aortic arch, watersheds appear at the ascending aorta for 50% cannula support and at the aortic root for 90% cannula support. Cannula support placed at the ascending aorta shows for both cannula support levels watershed phenomena at the aortic root. Only small differences can be seen between ideal and patient aorta results. Increasing the support level leads to a retrograde (proximal) shift of the watershed for each position.
Oxygen saturation
Based on the velocity results, the oxygen distribution at the outlets can be determined by averaging the oxygen saturation data of the mesh cells at the branching arteries.
Ideal aorta
Placing the cannula in the descending aorta leads to high oxygen saturation of the supra-aortic arteries (92.5%–96.7%), but not of the coronary arteries (see Figure 4). The cannula in the aortic arch brings about a similar oxygenation rate between 88.9% and 94.3% to the supra-aortic arteries with a higher oxygenation in the ascending aorta, but without reaching the coronary arteries. The only position that can oxygenate the right coronary artery (RCA) is at the ascending aorta. This position of the cannula shows an oxygen rich blood support of 97.3% at the RCA. At a 90% cannula support all positions show a perfusion rate between 96% and 99% at all off-branching arteries (see Table 1 in additional materials). Results of oxygen saturation in different cannula positions and for different support levels. Saturation values were projected onto the geometry’s surfaces representing exactly the saturation distributions found inside the aorta.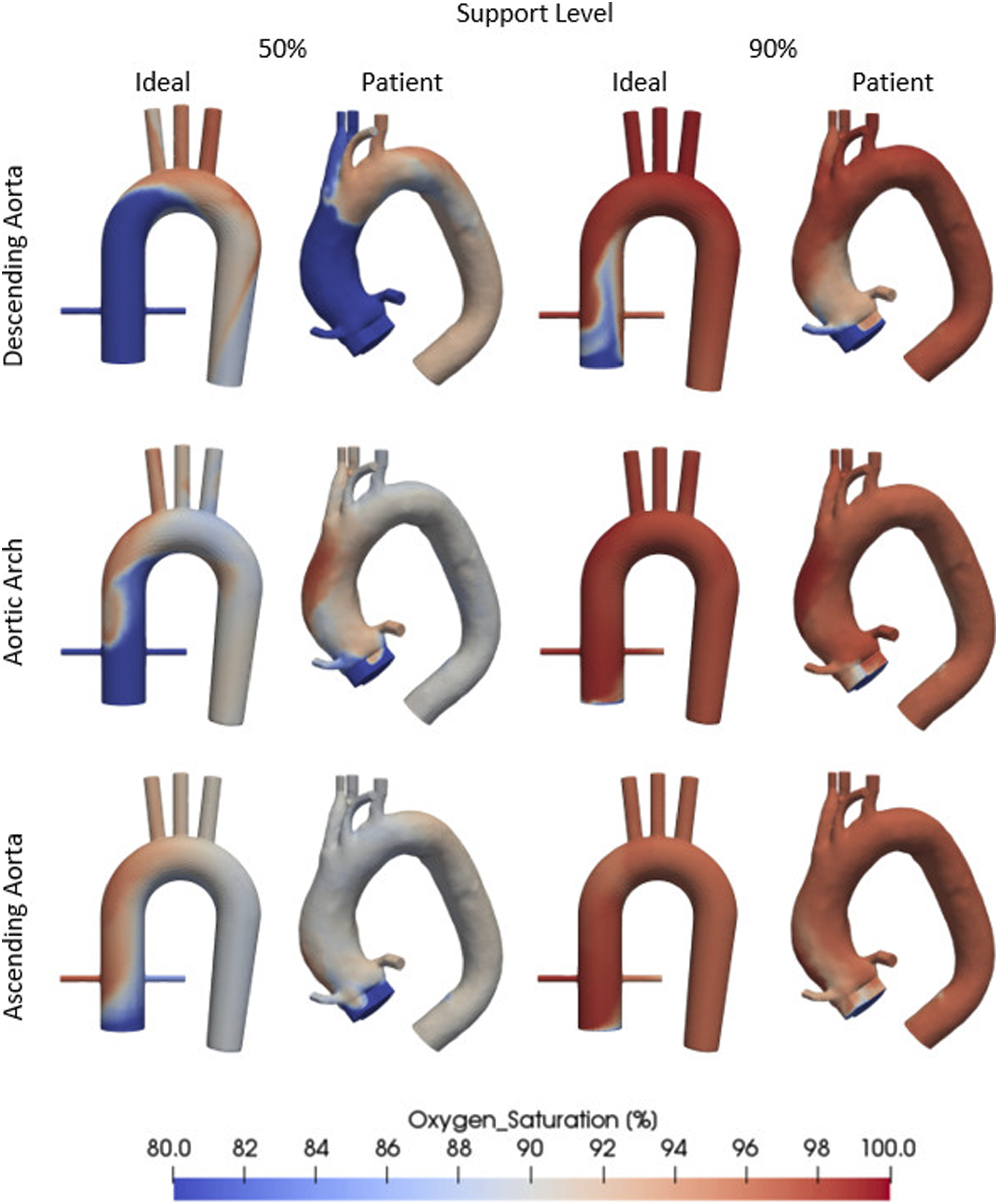
Patient aorta
For the positioning of the cannula in the descending aorta with 50% cannula support shows oxygenation of LCCA (90.2%) and left subclavian artery (LSA) (94.8%), but not of the right subclavian artery (RSA), right common carotid artery (RCCA) and coronary arteries. The second cannula position (aortic arch) can oxygenate the left coronary artery (LCA) (93.3%) as well as all supra-aortic arteries with approximately 90% oxygen saturation. At last, positioning of the cannula in ascending aorta leads to an oxygenation of 90% of the supra-aortic arteries and the coronary arteries. At 90% cannula support the aortic arch and ascending aorta position show an oxygen saturation between 93% and 97% at all off-branching arteries. The descending aorta position shows approximately 98% saturation for the RCCA, RSA, LCCA and LSA, 93.3% for the LCA and only 83.4% for the RCA (see Table 2 in additional material).
Discussion
Femoral cannulation is preferred in emergency situations because of the easy accessibility of the femoral vein/artery using the Seldinger technique. 9 However, in impaired lungs, peripheral VA ECMO can lead to watershed. 10
In order to avoid the side effects deriving from the femoral cannulation, beside veno-arterial-venous (VAV) ECMO and sternotomy for central cannulation, different solutions have been proposed. Javidfar et al. use the subclavian artery cannulation instead, as it can combine the advantages of a central ECMO with lower complications. 11 This cannulation however can lead besides bleeding due to a surgical approach, to obstruction of the vertebral artery and as a consequence to cerebral malperfusion. 12 Weymann et al. have described their technique for central ECMO cannulation, which consists of a right anterior thoracotomy as a substitution of the classic sternotomy in order to minimize the risk for bleeding and damage of the surrounding structures. 13 Nevertheless, it is a time-consuming procedure and requires surgery.
Computational Fluid Dynamics is an important tool not only for engineering issues but also all kind of cannulation- and perfusion issues for the clinician. Many CFD investigations regarding ECMO, cannulation techniques and perfusion routes have been performed. Gu et al. conducted a primary CFD study about the hemodynamic differences between central and peripheral ECMO. 14 Nezami et al. investigated how varying levels of support provided by ECMO alter distribution of perfusion and their impacts on the location and characteristics of the watershed region. 15 Kaufmann et al. and Neidlin et al. used CFD to analyse a newly developed outflow cannula for cardiopulmonary bypass.16,17
Nowadays available and medically approved long flexible cannulas allow a percutaneous cannulation of virtually all central vessels and heart chambers for drainage and “inflow” purposes. Hence watershed (“the Harlequin Syndrome”) could be avoided by transluminal central perfusion coming from a peripheral access. Our first clinical experiences with such cannulas were promising. An improvement of the cerebral oxygenation in patients on VA ECMO suffering from poor cerebral oxygenation could be observed when long arterial cannulas were used. Hatam et al. of our group published their clinical results of a fully percutaneous RVAD, in which the arterial cannulation was performed by placing an 18 Fr long flexible cannula (REVAS R Freelife, Aachen, Germany) via the jugular vein into the main pulmonary artery. 5
In order to further prove the validity of these experiences, a CFD simulation was performed to determine flow distribution oxygenation status of upper body perfusion. Until now no such simulation has been conducted for an endoluminal approach.
Our results show similar major patterns in an ideal and real patient aorta with just small local differences. Watershed areas differ depending on the position of the cannula in the aorta. A retrograde advancement of the cannula positively affects the supply with oxygenated blood of the coronary and supra-aortic arteries by shifting the watershed position in the same direction. These results are further verified by the oxygen saturation data. The RCA, for instance, is perfused with oxygenated blood only when the cannula is positioned in the ascending aorta. If the support level from the cannula is 50%, the coronary and upper body arteries are perfused only when the cannula is positioned in the ascending aorta for the ideal aorta geometry; the results for the real patient aorta equally suggest that the best solution consists in positioning the cannula in the ascending aorta and in the aortic arch. With a little advancement and adjustment of the cannula from the aortic arch, the coronary and supra-aortic arteries would be fully perfused with oxygenated blood also for the ideal geometry. Using 90% cannula support, the blood flow from the cannula is so high that the choice between the positioning of the cannula in the aortic arch or ascending aorta does not make a difference.
Positioning a long cannula in the aorta seems theoretically and based on our CFD results an attractive method to avoid watershed phenomena. However, some limitations have to be taken into consideration.
From the CFD point of view, fixed idealized boundary conditions (non-pulsatile flow behaviour and rigid walls) as used in our CFD study could lead to differences between CFD simulations and patient measurements from Doppler Ultrasound or MRI flow scans.
From a clinical point of view, even though until now not observed clinically, positioning a long cannula in aorta could theoretically lead to possible side effects such as vessel injury from the perforation of the aorta,18–20 obstructions of supra-aortic arteries and downstream thromboembolic events due to large foreign material surfaces. If a cannula is proximal to the internal carotid artery, it can cause a stroke. 21
The results of oxygen saturation distribution from our CFD study correspond to those of an open, central cannulation or of a subclavian cannulation. In addition, this cannulation does not have the disadvantages of central cannulation such as the need for surgery or time-consuming procedures,22,23 or those of a subclavian cannulation such as cerebral malperfusion as a consequence of the obstruction of the vertebral artery. 12 Most of the abovementioned side effects of placing a long cannula in the aorta may probably be avoided if appropriate visualisation (echo fluoroscopy) is used.
In conclusion our CFD analysis reveals, that retrograde advancement of a long endoluminal cannula can considerably improve the oxygenation of the upper body and leads to oxygen saturation distributions similar to those of a central cannulation. During full flow (>90% support) there is no relevant difference in supra-aortic oxygenation with respect to the anatomic position of the cannula thus allowing a cannula position just behind the LSA. Further validations of the CFD results in a smaller animal experiment will be followed by a more detailed clinical evaluation.
Supplemental Material
Supplemental Material - Computational fluid dynamics analysis of endoluminal aortic perfusion
Supplemetary Material for Computational fluid dynamics analysis of endoluminal aortic perfusion by Daniel Malinowski, Yvan Fournier, Andreas Horbach, Michael Frick, Mirko Magliani, Sebastian Kalverkamp, Martin Hildinger, Jan Spillner, Mehdi Behbahani and Flutura Hima in Perfusion
Footnotes
Authors note
Parts of this work were presented at the ESAO 2021: 47th Congress of the European Society for Artificial Organs September 7–11, 2021, London, UK (Abstract ESAO 2021. Int J Artif Organs 2021; 44: 599).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
