Abstract
Objectives
To assess whether instantaneous wave – free ratio (iFR) value is associated with left internal mammary artery (LIMA) graft failure at 12 months follow-up post coronary artery bypass graft (CABG).
Background
Data suggests bypass to a non-significant left anterior descending artery (LAD) lesion due to visual over-estimation may lead to LIMA graft failure. Implementing iFR may result in better arterial graft patency.
Methods
In iCABG (iFR guided CABG) study patients planned to undergo an isolated CABG procedure was prospectively enrolled and iFR was performed for LAD. Coronary computed tomography angiography was performed at 2 and 12 months follow-up. The primary endpoint of this study was to determine the rate of LIMA graft occlusion or hypoperfusion at 2 and 12-months follow-up. We considered a composite secondary endpoint of Major adverse cardiovascular and cerebrovascular event (MACCE) as a secondary outcome.
Results
In total 69 patients were included with no differences regarding age, sex and risk factors. At 2 months, 50 of LIMAs with pre-CABG iFR median 0.855 (0.785 – 0.892) were patent. Hypoperfusion was found in 8 LIMAs (median iFR 0.88 (0.842 – 0.90)). While, 7 LIMAs (median iFR 0.91 (0.88 – 0.96)) were occluded (p = 0.04). At 12 months, when iFR of LAD was >0.85: just 12 (31.6% out of all patent LIMAS) grafts were patent and 24 (100.0% out of all hypoperfused/occluded) grafts were hypoperfused or occluded (p < 0.001). In terms of MACCE, no difference (p = 1.0) was found between all 3 groups divided according to iFR value.
Conclusions
Instantaneous wave – free ratio value above 0.85 in LAD is a powerful tool predicting LIMA graft failure at 1-year follow up period.
Keywords
Introduction
Coronary artery bypass graft (CABG) surgery remains the best option of revascularisation for patients with multivessel coronary artery disease. The main disadvantage of CABG is a relatively high rate of graft failure over time with failure rates up to 20% at 1 year.1,2 Mechanical defects and technical errors can occur at similar rates in arterial and venous grafts, but arterial grafts have superior patency rates as are more resistant to surgical trauma, the endothelial function and vascular wall function are better preserved compared to vein grafts, the diameter of the left internal mammary artery (LIMA) graft is closely similar to the diameter of the left anterior descending artery (LAD), which limits flow turbulences that create frictional force and shear stress – induced endothelial injury. 3 Preserved endothelial nitric oxide synthase preserves good endothelial function, allows arterial remodeling in response to flow.4,5 Nonetheless, the bypass to a non-significant LAD lesion due to visual over-estimation of the LAD stenosis may lead to competitive coronary flow (CF) between the LIMA graft and LAD, which is believed to be one of the key factors leading to early LIMA failure.6,7 Currently, intra-coronary physiology measurements are considered to be the gold standard method in determining the hemodynamic significance of coronary lesions and has been demonstrated to improve significantly outcomes when used to guide percutaneous intervention.8,9 As the iFR was proved to be noninferior to FFR in multicenter DEFINE FLAIR and iFR Swedeheart trials10-13 and was confirmed in ESC guidelines as IA class indication for intermediate stenosis assessment, the usage of iFR was found as much more comfortable for the patient and the interventional cardiologist. Coronary stenosis can be verified without the requirement to induce hyperemia with adenosine which leads to a 10% reduction in procedural time versus FFR-guided treatment (p = 0.001), while reducing patient discomfort by 90% (p < 0.01). 10 iFR is calculated by measuring the resting pressure gradient across a coronary stenosis during the diastolic wave-free period within a single cardiac cycle, when coronary resistance is low and stable. JUSTIFY-CFR study clearly demonstrated that when iFR was negative and FFR was positive, CFR >2 was noted in 97% of cases. This indicates that when iFR and FFR appear to disagree, iFR provides more accurate information about the state of coronary flow than FFR. 14 We believe, that using iFR for physiologic assessment of coronary lesions may improve the accuracy of lesion evaluation which may result in better arterial graft patency.
The objective of this study was to assess whether negative or grey-zone iFR value is associated with early LIMA graft failure at 12 months follow-up post CABG.
Methods
Patient inclusion
This was a prospective, single blinded, single center study, conducted in the Hospital of the Lithuanian University of Health Sciences Kaunas Clinics (2018–2021).
Patients were enrolled after undergoing diagnostic coronary angiography and being referred by the Heart Team to undergo an isolated CABG procedure. A big number of patients (281 out of 350) were excluded from study after detailed echocardiography or not willing to participate. Just 69 patients met all inclusion/exclusion criteria as the purpose was to create very homogeneous group excluding all other circumstances which could influence MACCE or follow up period (Figure 1). An informed consent was obtained from all study participants in accordance with research ethics review board guidelines. Kaunas Regional Biomedical Research Ethics Committee approval number BE-2-89 (10.12.2018). Design of the study.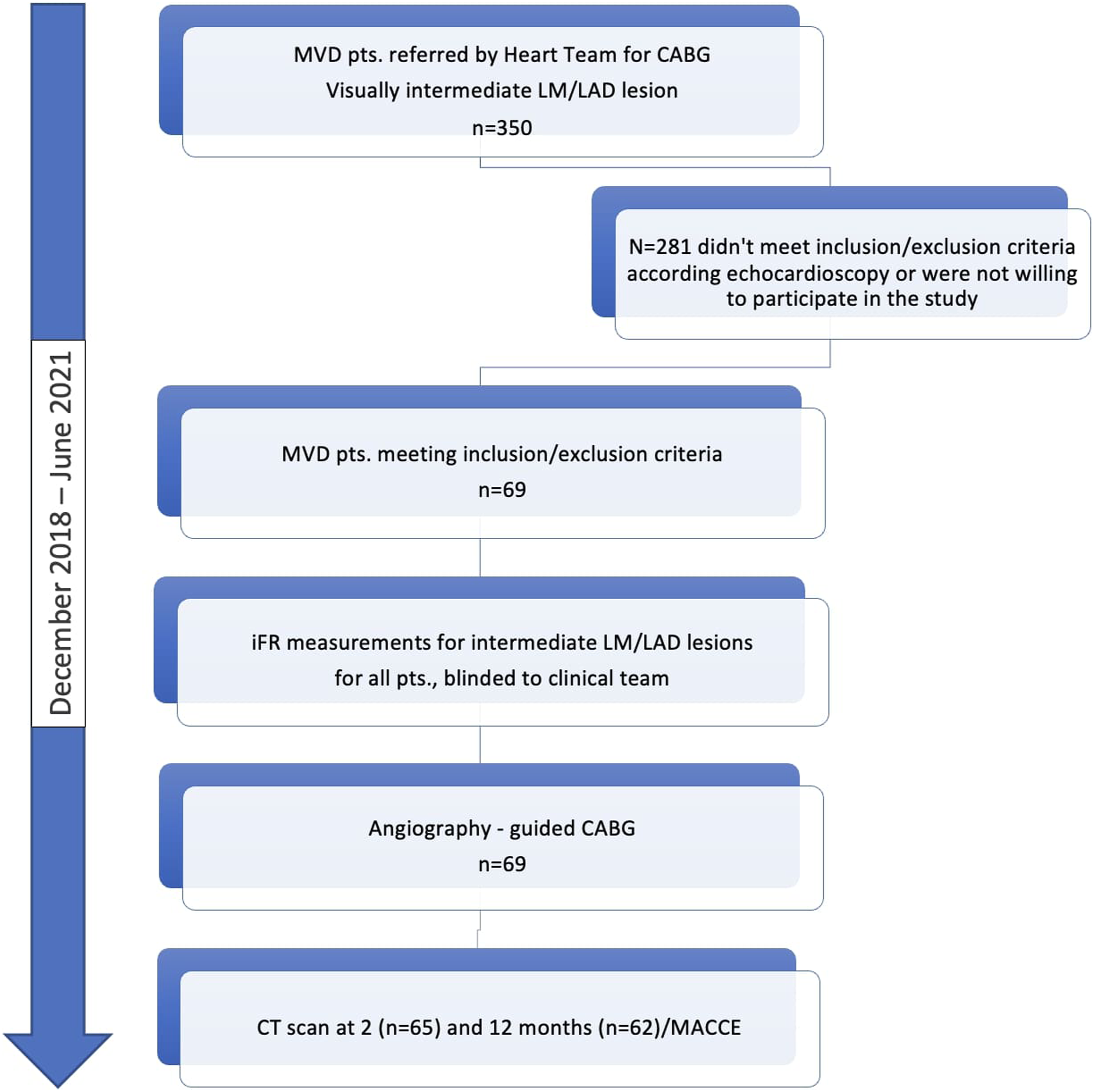
Patients included were aged over 18 years old, had stable angina and multivessel disease diagnosed by coronary angiography, defined as coronary artery disease in two or more native major epicardial vessels or their branches by coronary angiogram with visually assessed de novo coronary stenosis. Plan for CABG as optimal revascularization mode was made by the treating clinical team.
Patients were excluded from study if they were diagnosed with acute ischemia or were scheduled to undergo emergent surgery, had previous CABG, presented with moderate to severe reduced left ventricular systolic function (left ventricle ejection fraction <40%), had atrial fibrillation or were diagnosed with significant structural heart disease such as valvular heart disease. In addition, patients with a documented history of hepatic, oncologic or lung disease, allergy to contrast media and renal failure, were also excluded. Finally, candidates with history of chest radiotherapy were not enrolled in the study.
Data on patient demographics, comorbidities, in-hospital and discharge medications, coronary angiography characteristics, transthoracic echocardiography findings, clinical course after CABG (cardiopulmonary resuscitation, cardiogenic shock), short term outcomes, long-term outcomes (ischemic or hemorrhagic stroke, myocardial infarction (MI), target vessel revascularization, cardiovascular death and all-cause mortality) were obtained from hospital inpatient and outpatient records.
Quantitative coronary angiography
After enrollment, two experienced interventional cardiologists independently performed lesion quantification using quantitative coronary angiography (QCA); in case of disagreement, an unblinded third interventional cardiologist served as the adjudicator. iFR was performed for LAD with a coronary lesion of 35%–75% stenosis calculated by QCA quantification method during a planned procedure. iFR was measured at least 2 times in distal part of LAD and iFR pullback was performed not to miss a target lesion. Stenoses in LAD with iFR<0.86, were considered as hemodynamically significant, with iFR (0.86–0.89 – were called “grey zone” and with iFR>0.89 – hemodynamically nonsignificant. iFR measurements were blinded to the clinical team. Patients were assigned to coronary angiography-guided CABG per routine practice, and CABG strategy was chosen based on visual assessment of the coronary arteries (Figure 1).
CABG technique
Patients underwent CABG with the number of grafted vessels dictated by the treating clinical team as physiology measurement were blinded to surgeon. During CABG, grafts were placed onto vessels with visually intermediate or significant stenoses with an expectation with LIMA on LAD. Surgical revascularization was completed by On Pump grafting with use of Cardioplegia.
Coronary computed tomography angiography
Coronary Computed Tomography (CT) scans were performed 2 times: in early postoperative period (2–3 months) and in 12 months after CABG. The purpose of selected scan time period was important for identification when the collapse of graft happens.
CT scans for CABG patency evaluation were performed using a 320-detector (Aquilion One ™, Toshiba Medical Systems, Japan) or a 256-detector scanner (Revolution, GE Healthcare, Milwaukee, USA) with prospective ECG tube gating. CT scans were performed using 100–120 kV tube voltage, 0.6–0.625 collimation and 280–350 ms gantry rotation time.
Image acquisition was performed in accordance with the guidelines. 15 Contrast medium (Ultravist 370, Shering, Berlin, Germany) was injected based on the patient’s body weight. Premedication with oral beta blockers and ivabradine, as well as intravenous beta blockers, was given to achieve target heart rate of <60 bpm. Images at 75% R–R intervals were used primarily for image reconstruction. If motion or other artifacts compromised diagnostic image quality, additional cardiac phases were reconstructed. Analysis of CT datasets were performed using workstation Vitrea Extend, Cardiac analysis (Canon Medical Systems, USA) or ADW 4.5, CardiQ Xpress (GE Healthcare, Milwaukee, USA). Axial images, reformatted images, multiplanar reconstructions and curved multiplanar reconstructions were independently assessed by two experienced specialists (a radiologist, 12 years of experience and a cardiologist, 6 years of experience) for bypass graft hypoperfusion, occlusion or obstructive stenosis. Interpretation and reporting of native coronary artery disease and graft patency was performed according to the latest guidelines by the Society of Cardiovascular Computed Tomography. 15 Discontinuation of contrast flow in the graft with no further flow was labelled as graft occlusion. Locally reduced contrast intensity or disruption of flow was defined as graft hypoperfusion or stenosis. In case of disagreement between readers, the images were carefully reviewed, reaching a consensus agreement.
Study endpoints
The primary endpoint of this study was to determine the rate of LIMA graft occlusion or hypoperfusion at 2 and 12-months follow-up. For evaluation, coronary computed tomography angiography (CCTA) was performed. Assessment was done by 2 experienced specialists blinded to physiology measurements.
We considered a composite secondary endpoint of MACCE, defined as any of the following events occurring within 1 year: stroke, target vessel revascularization (TVR), non-fatal MI or cardiovascular death, was chosen as a secondary outcome. Follow-up was performed during outpatient hospital consultation visits: 2 and 12 months after CABG routinely, in case of any new clinical signs – repeatedly.
Statistical analysis
Statistical analysis of the data was performed using SPSS, version 27.0 (IBM Corp., Armonk, NY, USA). Kolmogorov-Smirnov test was used to determine distribution of numerical variables. All parametric data were expressed as mean and standard deviation or median and interquartile range (IQR) 25–75%. Chi-square (χ2) was used to test categorical variables. Kruskal-Wallis and Mann-Whitney tests were used to compare non-normally distributed variables. Receiver Operating Characteristic (ROC) curve was used to predict diagnostic probability of a defined iFR value. Binary logistic regression analysis was performed to test the association between iFR and graft patency. p-value less than 0.05 was considered significant.
Results
Clinical characteristics of study patients at baseline.
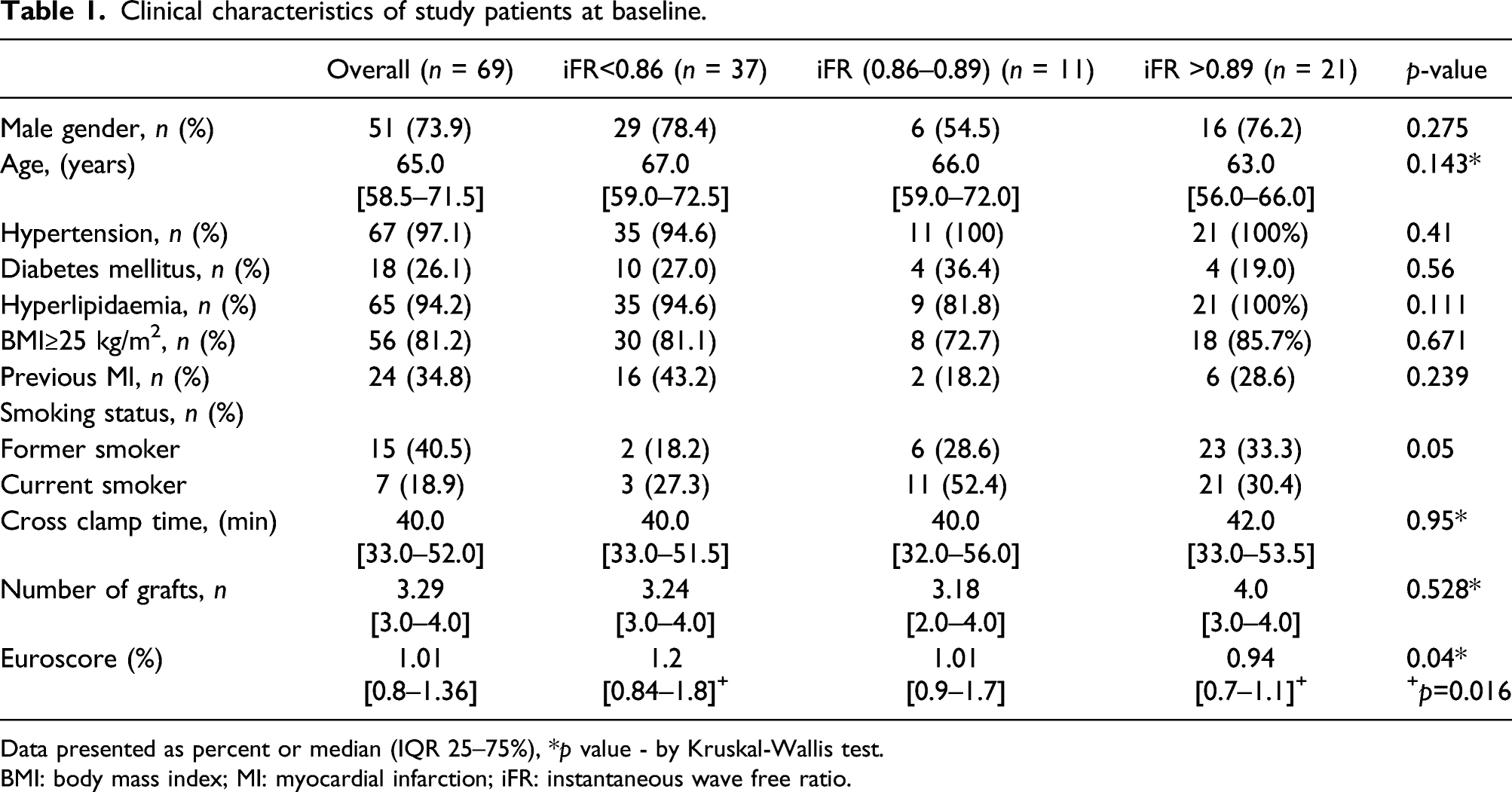
Data presented as percent or median (IQR 25–75%), *p value - by Kruskal-Wallis test.
BMI: body mass index; MI: myocardial infarction; iFR: instantaneous wave free ratio.
Patients IFR of LAD box Plot by graft patency in 2 months.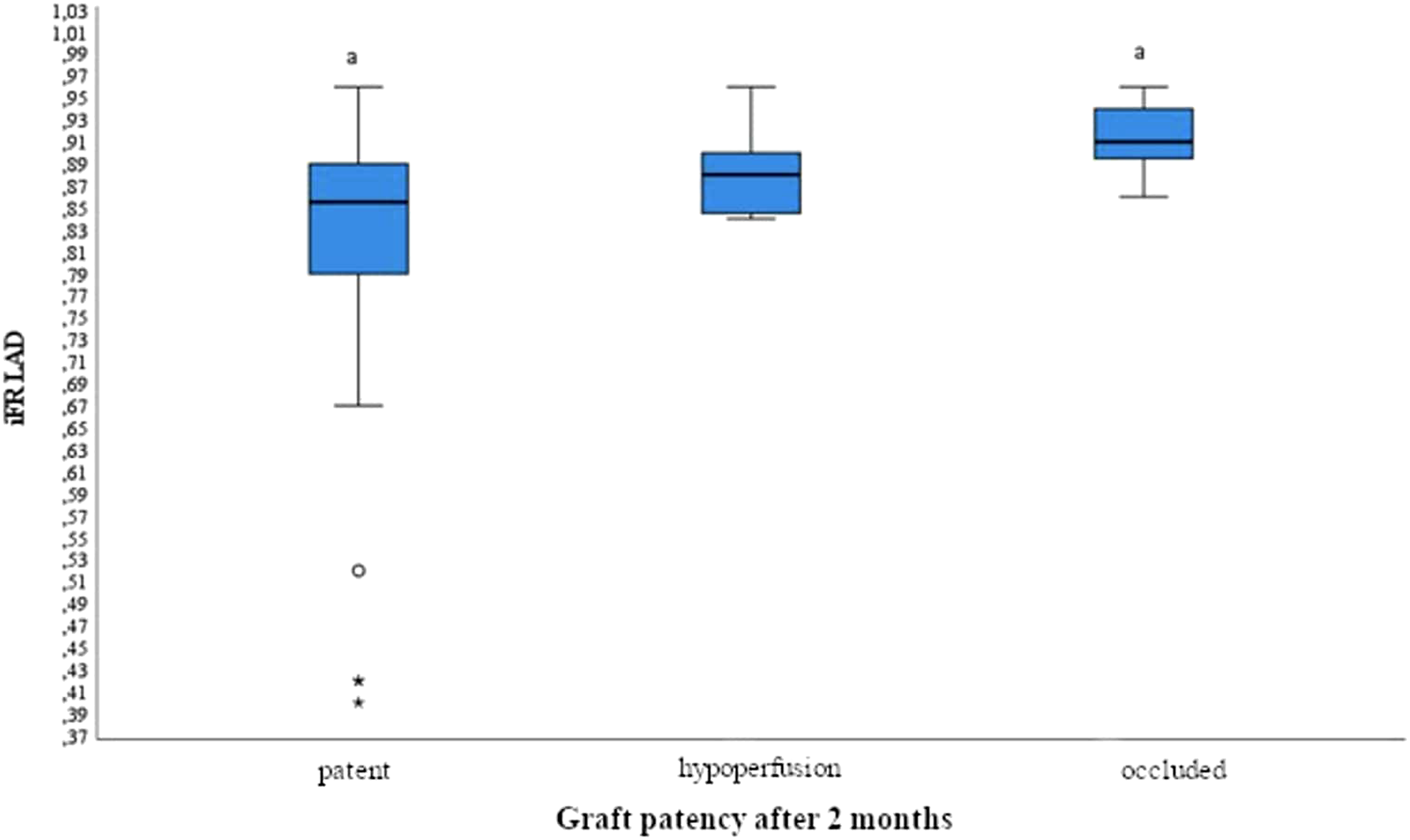
Patients IFR of LAD box Plot by graft patency in 12 months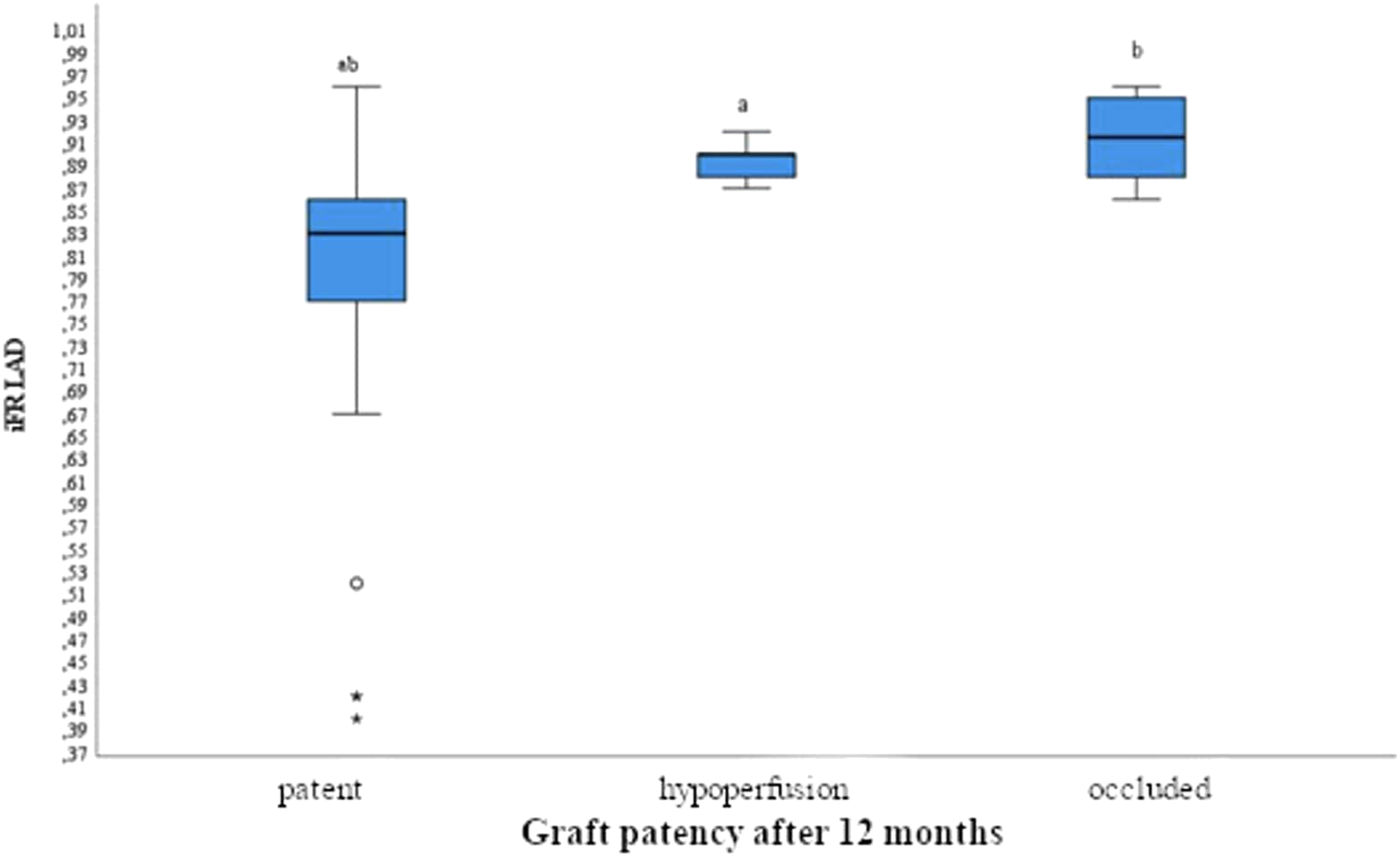
At 12 months, there was a statistically significant difference in iFR values between patent and hypoperfused groups and between patent and occluded grafts iFR values (p < 0.001 for both). However, iFR values between hypoperfused and occluded grafts did not differ significantly. When iFR of LAD was >0.85: 12 (31.6% out of all patent LIMAS) grafts were patent and 24 (100.0% out of all hypoperfused/occluded) grafts were hypoperfused or occluded (Figure 4). Graft patency rate according to iFR value.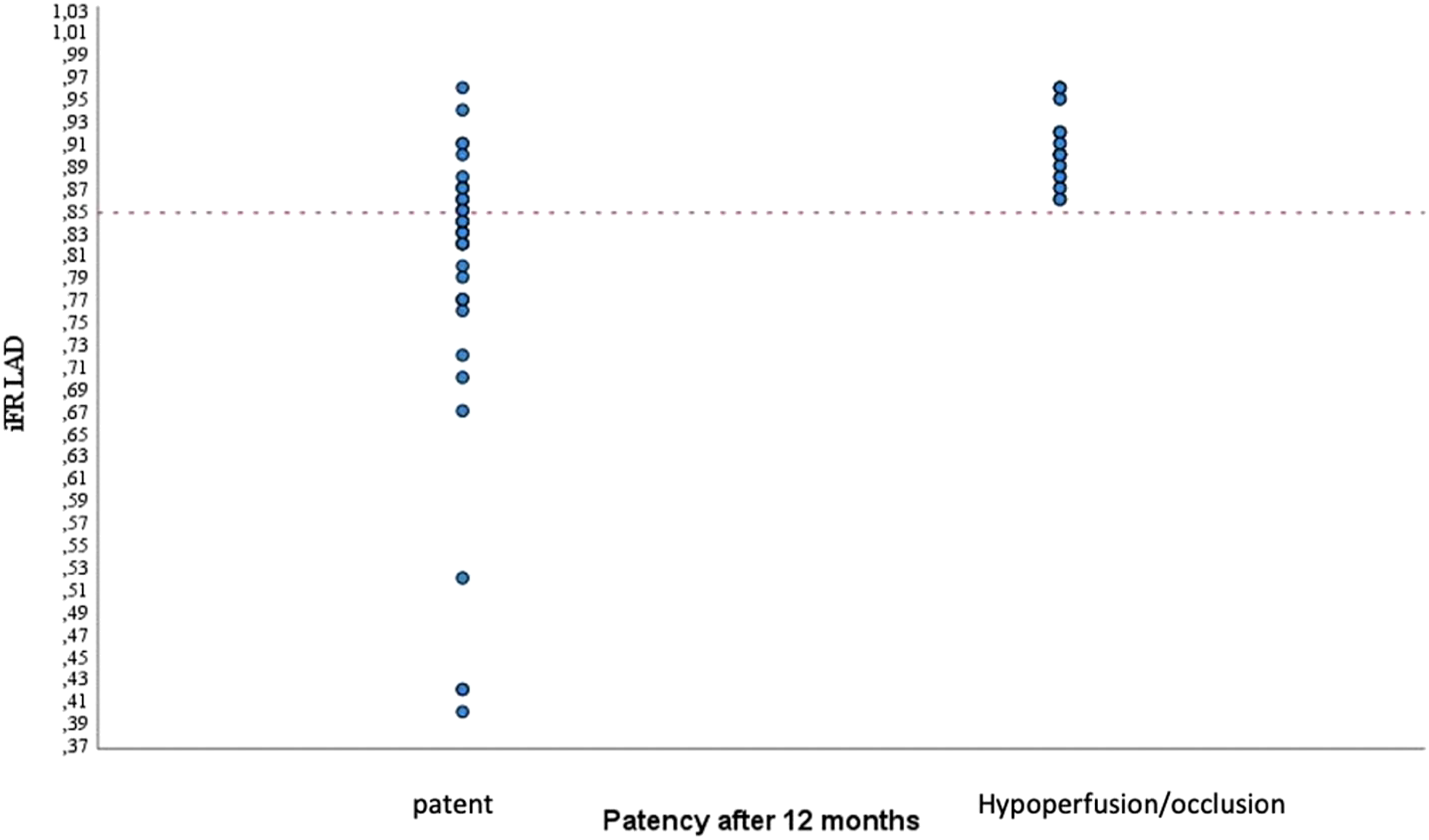
Receiver operating characteristic curve (Figure 5) was used to determine an iFR threshold value for LAD stenosis, prognosing graft hypoperfusion or occlusion, according to 12 months CCTA graft patency. In patients with LAD iFR <0.85, the odds of a patent LIMA graft at 12 months were three times higher (OR 3.0 95% CI (1.89–4.762). Receiver operating characteristic (ROC) test for LAD iFR threshold predicting dysfunction of LIMA graft according to 12 months CCTA graft patency: iFR value >0.85 in LAD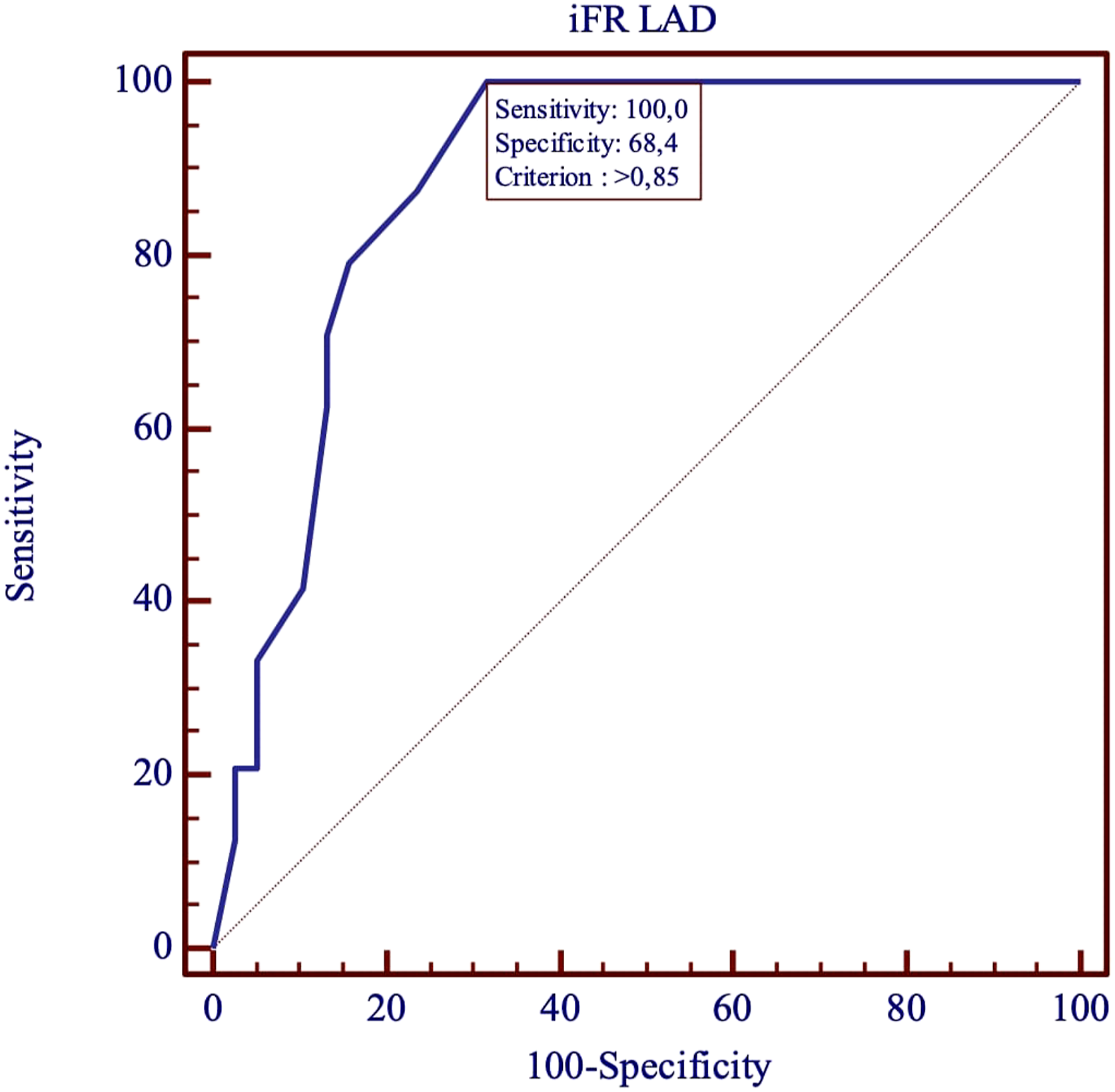
MACCE during 12 months follow up period.
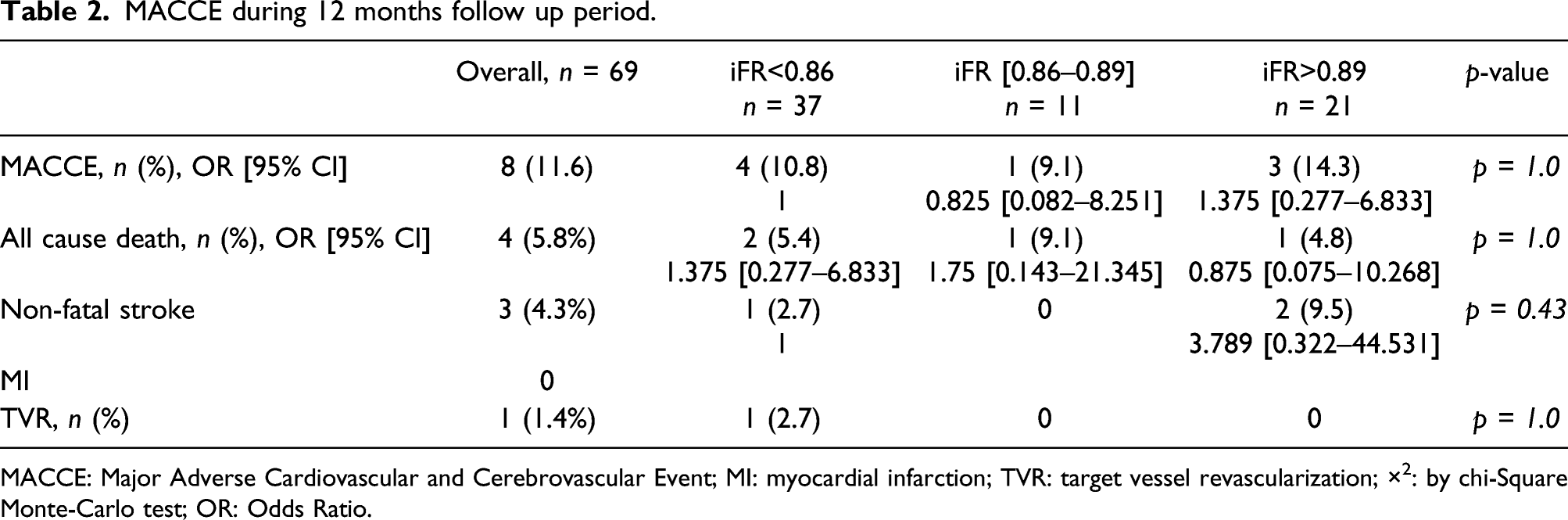
MACCE: Major Adverse Cardiovascular and Cerebrovascular Event; MI: myocardial infarction; TVR: target vessel revascularization; ×2: by chi-Square Monte-Carlo test; OR: Odds Ratio.
Discussion
iFR is a tool to assess hemodynamic significance of intermediate coronary artery lesions and has demonstrated to improve outcomes when used to guide percutaneous coronary interventions.16,17,18 The SWEDEHEART and DEFINE-FLAIR randomized controlled trials showed that iFR-guided revascularization method was non-inferior to FFR-guided revascularization in terms of major clinical outcomes within 1 year after procedure.19,20 FFR-guided surgical revascularization is associated with a lower number of grafts, shorter aortic clamp time, shorter hospitalization time, but no difference in MACE compared to angiography guided CABG. 21 However, the role of iFR guidance on CABG strategy is still being evaluated. iCABG study is one of the first studies to evaluate prospectively the impact of preoperative iFR measurements of LAD on LIMA patency. Twelevemonths follow up CCT scans revealed that hypoperfusion and occlusion of arterial grafts were more frequent if preoperative iFR of LAD indicated haemodynamically insignificant lesion including “grey zone”. Competitive coronary flow is considered to be the main cause of LIMA graft failure. 22 This phenomenon appears/manifests when stenosis of targeted coronary artery is mild and the resistance at the arterial graft is high, blood predominantly flows through the native coronary artery. In addition to that, decreased flow through the arterial graft can cause deterioration of flow-mediated vasodilatation and lead to arterial graft failure. In PREVENT IV trial, Harskamp and colleagues revealed that target vessel (LAD) stenosis <75% predicts LIMA graft failure. 23 Furthermore, studies based on preoperative FFR evaluation showed significantly higher rate of patent arterial grafts at 1 year, 24 6 years follow up if target coronary artery was with functionally significant lesion. Although hemodynamically insignificant target coronary artery stenosis can predispose the competitive coronary flow, The GRAFFITI (GRAFt patency after FFR-guided vs angiography-guided CABG) trial failed to show any difference in both arterial and venous graft patency in FFR-guided and angiography-guided groups. 25 However, in GRAFFITI trial all the LAD (arterial grafts) were with physiologically confirmed significant stenoses, no “grey zone (0.75–0.80)” of FFR was singled out, and acute coronary syndromes were included into the study, which could impact the graft attachment quality. In iCABG trial, LAD-LIMA grafts were evaluated more detailed according iFR values including “grey zone”, no acute coronary syndrome patients were involved, all the results of iFR were blinded, so even negative iFR value of LAD was grafted if angiographicaly approved. Although, iFR threshold value optimizing revascularization strategy is still questionable. The iFR “grey zone” varied in different studies: from 0.86–0.93 in the ADVISE study, 0.83–0.86 in the CLARIFY study to 0.89 in SWEDEHEART and DEFINE-FLAIR.16,19,20,26 Modi N.B. and colleagues aimed to prove that optimal iFR threshold value 0.89 is less accurate compared to 0.86. 27 Our study revealed that iFR<0.85 as a very strong prognostic value for LIMA graft patency within 12 months after CABG (Figures 2 and 3).
Data from IRIS-FFR registry showed that revascularization of coronary artery with gray zone FFR (0.75–0.8) did not show better clinical outcome, or a reduced risk of MACE. 28 In iCABG study, revascularization of “grey zone” also did not improve clinical outcomes. Furthermore, at 12 months follow-up, LIMA graft dysfunction occurred significantly more often in patients with iFR values 0.86–0.89 than iFR<0.86. Although the loss of graft patency did not cause angina pectoris because the native blood flow wasn’t compromised: the stenosis of native artery was hemodynamically insignificant, the flow was native and sufficient.
In our study iFR guidance on LIMA graft did not show benefit in patients’ clinical outcomes at 12 months follow-up. Despite of LAD iFR value, graft patency, symptoms of stable angina and incidence of MACCE did not differ significantly; no myocardial infarction occurred and just single elective target vessel PCI was performed. Botman et al. 25 revealed that there was no difference in angina class or repeat interventions despite of bypass graft failure at 1 year follow up. The GRAFFITI trial demonstrated no difference in incidence of death, myocardial infarction, stroke or target vessel revascularization in FFR-guided and CA-guided groups despite graft patency, but it is associated with a simplified surgical procedure as fewer grafts were performed in FFR-guided group. 27 Although only 65% of the patients (111/172pts.) completed the investigation by CCT scan, it could definitely impact the rate of graft patency. Contrary to our findings, S. Fournier et al. showed FFR-guided CABG was associated with significantly lower rate of death, myocardial infarction and MACCE.26,27 However, his findings were retrospective and lasted for 6 years and revealed that the patency of angiography-guided grafts seems to progressively decline over the years, unlike in the FFR-guided grafts where no occlusion occurs in the 3 first years.
Preoperative iFR guidance may simplify CAGB procedure or even change the revascularization tactics (PCI vs CABG) though there is lack of evidence of superior clinical outcomes in patients with no or patent versus occluded arterial graft because of concurrence flow.
Limitations
The present study has a number of limitations.
First, it was carried out in a single medical center. Single-center studies lacks external validity required to support widespread changes in practice which is considered a major limitation. On the other hand, multicenter studies provide a better basis for the subsequent generalization of its findings. This arises from the possibility of recruiting the subjects from a wider population and of various ethnical groups. Moreover, multicentral studies gives the potential for a wider range of clinical judgements concerning treatment strategies, since treatment strategies and surgical technologies varies from center to center.
Second, we measured IMA graft failure at 2–12 months after CABG, which didn’t reveal the precise timing of IMA graft failure before the CCTA was done.
Third, we had some missing data due to loss of follow-up, which might also have affected our results. Finally, the small number of study participants, may have been underpowered to detect a modest but clinically important relationship between IMA graft failure and subsequent clinical outcomes.
Conclusions
Performing iFR preoperatively may aid in guiding surgical revascularization through arterial grafts. An iFR cut-off value of 0.85 in LAD is a powerful tool predicting LIMA graft failure at 1-year follow up period and can aid cardiac surgeons to decide whether to bypass LAD or not. During 12 months follow-up period, LIMA failure didn’t cause any deterioration in patients’ clinical outcome, which might be attributed to the availability of native coronary blood flow.
Perspectives
• What is known? FFR-guided CABG is associated with a lower number of grafts, shorter aortic clamp time, shorter hospitalization time, but no difference in MACE compared to angiography guided CABG. However, the role of iFR guidance on CABG strategy is still being evaluated. • What is new? iCABG study is one of the first studies to evaluate prospectively the impact of preoperative iFR measurements of LAD on LIMA patency. Twelevemonths follow up CCT scans revealed that hypoperfusion and occlusion of arterial grafts were more frequent if preoperative iFR of LAD indicated haemodynamically insignificant lesion including “grey zone”. • What is next? Multicenter studies are needed to reveal long term results comparing angiography guided CABG to iFR guided.
Impact on daily practice
• FFR-guided CABG is associated with a lower number of grafts, shorter aortic clamp time, shorter hospitalization time, but no difference in MACE compared to angiography guided CABG. However, the role of iFR guidance on CABG strategy is still being evaluated. • iCABG study is one of the first studies to evaluate prospectively the impact of preoperative iFR measurements of LAD on LIMA patency. 12 months follow up CCT scans revealed that hypoperfusion and occlusion of arterial grafts were more frequent if preoperative iFR of LAD indicated haemodynamically insignificant lesion including “grey zone”. • Multicenter studies are needed to reveal long term results comparing angiography guided CABG to iFR guided.
Supplemental Material
Supplemental Material - Instantaneous wave free ratio value impact on left internal mammary artery graft patency: iCABG study
Supplemental Material for Instantaneous wave free ratio value impact on left internal mammary artery graft patency: iCABG study by Rasa Ordiene, Ramunas Unikas, Ali Aldujeli, Rimantas Benetis, Povilas Jakuska, Indre Ceponiene, Antanas Jankauskas, Jurgita Plisiene, Tadas Lenkutis, Gabriele Rudokaite, Rima Braukyliene, Mindaugas Stonis, Justin Davies and Prakash P Punjabi in Perfusion
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
