Abstract
Exchanging circuits in patients undergoing extracorporeal life support (ECLS) for component failure can be a life-threatening endeavor if the patient has no native organ function and is fully dependent on ECLS. Traditional circuit exchanges involve replacing the entire circuit at once, leading to a sufficient loss of support that risks hemodynamic deterioration in the absence of mechanical ventilation or mechanical circulatory support. A staged approach is described using a parallel circuit configuration to reduce the risk of physiologic decompensation in patients with total dependence on ECLS. The approach involves splicing in a second primed circuit in parallel, transitioning flow incrementally from old to new circuit, and removing the old circuit. This approach provides hemodynamic and physiologic stability even in patients with absent underlying cardiopulmonary function.
Keywords
Introduction
Circuit exchanges during extracorporeal life support (ECLS) are necessary when circuit components fail but can represent a life-threatening risk if the patient is highly dependent on ECLS with little or no native organ function and physiologic reserve. Exchanges are typically performed by preparing a replacement circuit, clamping, and cutting out the old circuit near the cannulas, and splicing in the new circuit. Having two teams whereby one team manages the drainage limb while the second team simultaneously manages the return limb can minimize the time without support. Elevating the level of native organ support, for example, through mechanical ventilation or vasoactive agents can help support the patient but with the risk of additional organ injury. Despite careful coordination between teams, however, additional time is still required to clear the new circuit of prime, as high circuit flow when resuming support with a crystalloid-primed or banked blood-primed circuit to reduce this time is associated with hemodynamic instability.
The risk of this approach is greatly amplified when underlying pulmonary or cardiovascular function is totally absent. The currently expanding practice of extubating patients on ECLS1,2 further compounds the problem because there is no capability of providing support with mechanical ventilation. The time needed to complete the exchange plus the time to clear the prime can reach 2–3 minutes or longer, leading to a high risk of severe cardiopulmonary instability and cardiac arrest.
A technique is described in which the circuit exchange is staged using parallel circuits, requiring only 5–10 seconds of circuit interruption at any given time while permitting a slow crystalloid or blood prime exchange to minimize adverse physiologic effects.
Technique
The approach is to stage the circuit exchange into the following steps:
A new circuit is prepared and primed according to the institution’s normal procedures. Y-connectors are attached to the end of the new circuit (Figure 1(a)). For adult circuits, these will typically be ⅜ x ⅜ x ⅜ inch connectors and ¼ x ¼ x ¼ for pediatric circuits. A tubing clamp is placed adjacent to each Y-connector. Sequence of steps for circuit exchange. A. The return and drainage limbs are doubly clamped and the new circuit return and drainage limbs with a Y-connector is inserted into the existing circuit. B. Flow is restored through the existing circuit with the new circuit limb remaining clamped. C. The flow is then slowly transitioned from old to new circuit by adjusting clamps on both limbs (not shown). D. Once the transition is complete, the old circuit is clamped. E. The Y-connector is removed and replaced with a straight connector with a temporary clamping and cutting of the new limb. F. Flow is resumed through the new circuit and the old circuit is discarded.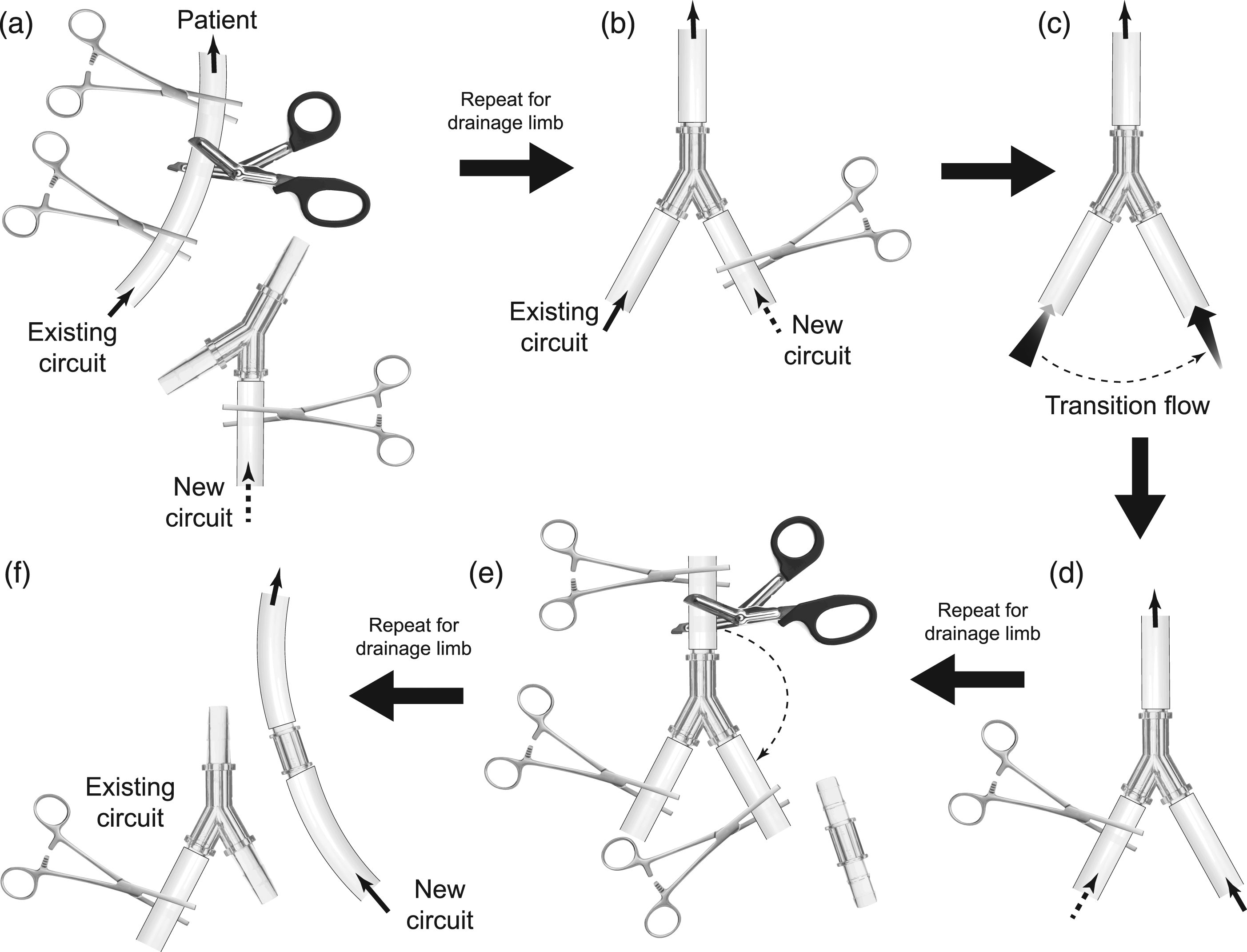
With a single team, the return (or drainage) limb is aseptically prepared near the cannula, clamped, divided, and the Y-connector inserted in a “wet-to-wet” manner to remove air according to institutional conventions (Figure 1(b)). Flow is immediately restored through the existing circuit with the clamp maintained on the new circuit, and the patient allowed to recover from this brief interruption. The same sequence is applied to the drainage (or return) limb. If two teams are present, then both limbs can be inserted simultaneously. Each sequence takes only 5–10 seconds, and physiologic impact is minimal.
While support is maintained on the existing circuit, the clamps are gradually opened on the new circuit as flow is initiated at low flow, for example, 250 mL/min in an adult, or correspondingly lower value for pediatric patients, and continued until the patient’s blood has replaced the prime, typically 4–5 minutes. This allows gradual introduction of the prime without negative physiologic impact. Flow in the new circuit is then ramped up to full support as flow in the existing circuit is simultaneously ramped down (Figure 1(c)). The old circuit is clamped adjacent to its connection to each Y-connector (Figure 1(d)). The technique is completed by clamping and cutting out the two Y-connectors along with the connected limb of the old circuit (Figure 1(e)) and replacing with straight connectors (Figure 1(f)), sequentially, or simultaneously if two teams are present. This step can also be completed with 5–10 s each, allowing for recovery after each exchange.
Comments
This new technique was first applied in a 38-year-old post-partum non-intubated spontaneously breathing female patient with acute respiratory distress syndrome due to COVID-19 pneumonia. The Institutional Review Board (IRB) deemed this project as exempt from IRB review (IRB project #0002029). She was on venovenous support via single lumen femorojugular cannulation for 20 days and developed progressive loss of oxygenator function due to clotting. She had complete opacification of her lungs with no residual native lung function. During the circuit exchange, her arterial saturation dropped less than 10% during each stage of the procedure but promptly recovered, and she remained hemodynamically stable through the procedure.
The advantages of this staged parallel circuit approach are several: • It can be applied sequentially by a single team with an operator and assistant, or simultaneously with two teams. • It greatly reduces circuit interruption time and can be performed in patients with absent native organ function and cardiopulmonary reserve. • It eliminates the need for rapid prime infusion into the patient, maintaining hemodynamic stability. • It can be performed in any type of cannulation configuration including venovenous (VV), venoarterial (VA), venovenoarterial (VVA), and venopulmonary (VP).
A disadvantage of the technique is that two connector insertions are required for each circuit limb as opposed to one with the traditional technique. This increases the risk of air entrainment into the circuit, but the reduction in risk from cardiopulmonary instability may offset this risk. Another concern is the risk of spallation from cutting into the circuit tubing. Spallation from cutting tubing has not been addressed in the literature, and spallation is most commonly associated with protracted mechanical stress of tubing compression in roller pumps. Nonetheless, this risk cannot be excluded. The safety of this technique cannot be fully established until the procedure has been more widely integrated into practice, or systematically studied.
Parallel circuits have been employed in the past to elevate support in patients with refractory hypoxemia and high cardiac output state,3,4 but these involve dual cannulation strategies. An intraoperative circuit change from an ECLS circuit to a cardiopulmonary bypass circuit using a similar staged approach has been reported. 5 There have been no prior reports of using parallel circuits for high-risk ECLS circuit exchange. This technique is a safer option for circuit exchange in patients who are fully ECLS dependent.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
