Abstract
Introduction:
Clot formation, infection, and biofouling are unfortunate but frequent complications associated with the use of blood-contacting medical devices. The challenge of blood-foreign surface interactions is exacerbated during medical device applications involving substantial blood contact area and extended duration of use, such as extracorporeal life support (ECLS). We investigated a novel surface modification, a liquid-impregnated surface (LIS), designed to minimize protein adsorption and thrombus development on medical plastics.
Methods:
The hemocompatibility and efficacy of LIS was investigated first in a low-shear model with LIS applied to the lumen of blood incubation vials and exposed to human whole blood. Additionally, LIS was evaluated in a 6 h ex vivo circulation model with swine blood using full-scale ECLS circuit tubing and centrifugal pumps with clinically relevant flow rate (1.5 L/min) and shear conditions for extracorporeal carbon dioxide removal.
Results:
Under low-shear, LIS preserved fibrinogen concentration in blood relative to control polymers (+40 ± 6 mg/dL vs polyvinyl chloride, p < .0001), suggesting protein adsorption was minimized. A fibrinogen adhesion assay demonstrated a dramatic reduction in protein adsorption under low shear (87% decrease vs polyvinyl chloride, p = .01). Thrombus deposition and platelet adhesion visualized by scanning electron microscopy were drastically reduced. During the 6 h ex vivo circulation, platelets in blood exposed to LIS tubing did not become significantly activated or procoagulant, as occurred with control tubing; and again, thrombus deposition was visually reduced.
Conclusions:
A LIS coating demonstrated potential to reduce thrombus formation on medical devices. Further testing is needed specialized to clinical setting and duration of use for specific medical target applications.
Introduction
Clot deposition and biofouling of blood-contacting medical devices is a major clinical concern that permeates medical practice. When blood contacts a synthetic material, the natural response is stimulation of thrombosis. Thrombus deposition on the device surfaces impairs device performance, occludes blood flow through the device, and elicits development of emboli; all of which result in significant patient complications. This issue is escalated during application of organ support technologies that involve significant blood-foreign material exposure and extended duration of use, such as extracorporeal life support (ECLS), mechanical heart valves, and ventricular assist devices. For example, according to the 2020 ECLS Organization International Summary, 11% of adult respiratory patients required a circuit replacement during the course of support. 1 This rate increased to 16 and 17% in pediatrics and neonates, respectively. In this cohort that required a circuit change, survival rate was 45–66%.
Blood fluidity is maintained in healthy blood vessels due to tunable secretion of anticoagulant agents, such as nitric oxide and prostacyclin, and a blood contacting interface composed of non-adhesive and anti-fouling surface molecules, the glycocalyx.2,3 When blood contacts a foreign surface, however, plasma protein adsorption occurs rapidly, forming a concentrated protein monolayer (up to 1000-fold higher concentration than in plasma 4 ) that serves as an adhesive boundary for platelet, leukocyte and red blood cell attachment. 5 This results in platelet activation, recruitment, and thrombus formation. Additionally, intrinsic coagulation activators such as FXII, prekallikrein and high molecular weight kininogen can adsorb to the foreign surface directly and trigger contact activation. 6
To address this challenge, non-adhesive biopolymers and surface coatings have been developed to minimize protein and cellular adsorption at the material interface. This includes micro-/nanotextured surfaces,7,8 and incorporation of lubricants via tethering or swelling9–12 to form a non-adhesive and/or repellent surface layer. One such strategy is a lubricant-impregnated surface (LIS) coating. LIS coatings have unique characteristics integrating non-adherence and self-healing that other coatings currently cannot achieve. 13 Previously, a typical LIS coating of a fluorinated substrate infused with a perfluorinated compound (PFC) liquid created a surface that demonstrated a slippery and non-adherent surface. 14 The PFC liquid has a high affinity towards the fluorinated substrate and will readily spread to cover the entire surface. 15 Therefore, the coating is capable of self-healing defects and restoring the non-adherent performance. A LIS coating formulated with a PFC has demonstrated the capability to repel blood and reduce thrombosis 15 ; however, for many blood contacting devices such as dialyzers the use of PFCs is currently restricted due to safety concerns regarding lubricant leaching. 16
From our thermodynamic analysis, the non-adherence performance of LIS coatings is not specific to fluorinated compounds. 17 A stable non-adherent LIS surface is driven by interactions among a lubricant, a substrate, and a complex media, for example, blood plasma (Figure 1(a)). Thus, different substrates and lubricants can be designed based on surface energies and surface morphology to achieve equivalent anti-thrombogenic performance in the blood stream utilizing components that do not elicit safety concerns. Thus, we developed a novel LIS coating formulated with a biocompatible lubricant that has been utilized for parenteral nutrition matched with a non-leaching textured substrate and evaluated its blood compatibility and anti-thrombogenic performance in vitro for potential ECLS applications.
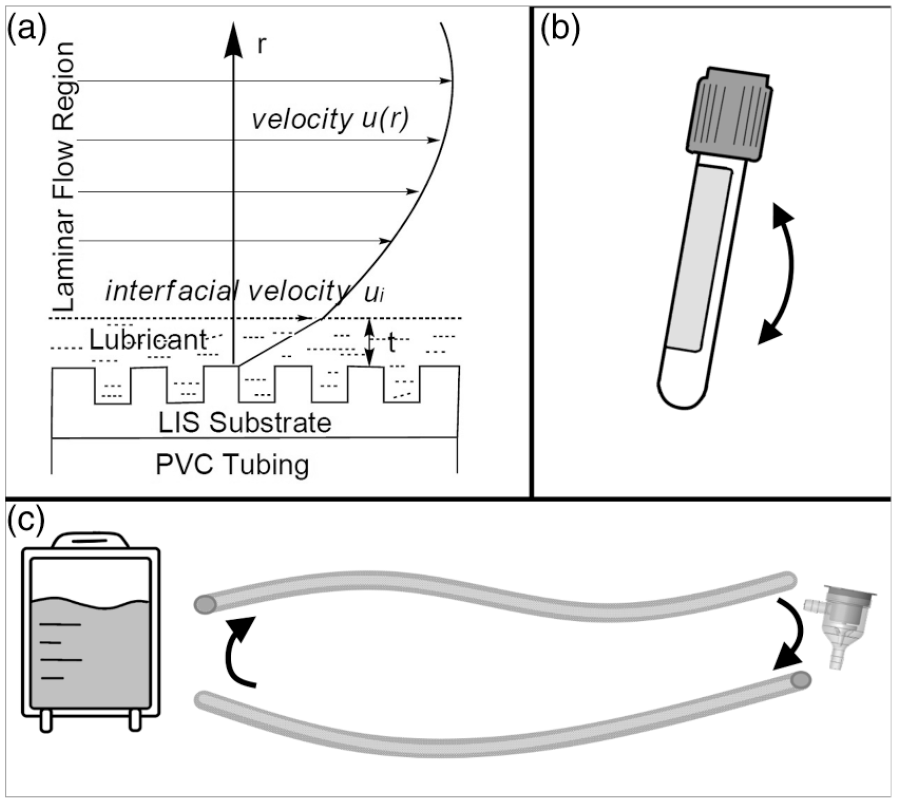
(a) Schematic of a liquid-impregnated surface (LIS) coating on an ECLS system. The LIS substrate is applied to a polymer substrate (polyvinyl chloride [PVC] tubing) to provide affinity and texture that accommodate the LIS lubricant. The interfacial velocity at the interface, ui, can be calculated based on a hydrodynamic model as a function of viscosities of the blood and the lubricant, lubricant thickness t, the diameter and length of the PVC tubing, and the blood flow rate u(r). (b) Schematic of incubation vial utilized in low-shear test models. LIS and other control coatings were applied to the interior blood-contacting lumen of the vials. (c) Schematic of circuit components in the extracorporeal circulation hemocompatibility test model including a blood reservoir, two segments of PVC circulation tubing, and centrifugal pump.
In this study, we focused on extracorporeal carbon dioxide removal (ECCO2R) as a target application. ECCO2R is performed using lower blood flow rates (<1.5 L/min) in comparison with extracorporeal membrane oxygenation (ECMO) (3–7 L/min). The low blood flow rates utilized during ECCO2R enable removal of carbon dioxide using smaller catheters than are utilized during ECMO when significant oxygenation support is required. 18 Although the use of smaller catheters during ECCO2R reduces the potential of vascular-related complications, it is unclear whether the lower blood flow rates increase the risk of thrombosis on device surfaces. Thus, we investigated application of LIS to minimize this risk of thrombosis at ECCO2R relevant blood flow rates.
Methods
To assess the hemocompatibility and efficacy of LIS modification we used three sequential models. First, a low-shear model was employed where LIS was applied to the interior lumen of blood incubation vials and exposed to human whole blood within the vials for 1 h on a vertical rocker (Figure 1(b)). Second, LIS was applied to polyvinyl chloride (PVC) medical tubing segments and exposed to isolated human fibrinogen for assessment of protein adsorption under low shear. Third, LIS was applied to full-scale ECLS circuit tubing and evaluated using clinically relevant blood flow conditions for ECCO2R in a 6 h ex vivo circulation study using swine blood (Figure 1(c)). We hypothesized that LIS reduces protein adsorption, limiting thrombus development and coagulation, in both the low-shear models and the ex vivo circulation study.
The LIS materials were prepared at LiquiGlide, Inc. (Cambridge, MA, USA). Low-shear protein adsorption and blood circulation testing were performed at the Autonomous Reanimation and Evacuation Research Program (AREVA) (San Antonio, TX, USA). Microscopy imaging was performed at Colorado State University (Fort Collins, CO, USA). Low-shear incubation vial testing was performed at the U.S. Army Institute of Surgical Research (USAISR) (protocol L-20-004) (San Antonio, TX, USA).
Vial preparation and characterization
The LIS coating is a proprietary, two-part coating consisting of a solid substrate and a liquid lubricant (see Supplemental Methods). 19 Polyethylene terephthalate (PET) blood incubation vials (ID #366408, No-additive specimen tube, 6 mL volume; Becton Dickinson, Franklin Lakes, NJ) were utilized in the low-shear in vitro hemocompatibility study. Unmodified vials (PET control) and vials with a PVC layer applied (PVC Control) served as the polymer controls. The test groups were vials with the LIS bilayer coating applied and vials with only the single substrate layer applied (Substrate).
Tubing preparation and characterization
The LIS coating was applied to PVC tubing (Tygon® ND 100-65 Medical Tubing, U.S. Plastic Corp; Lima, OH) through a multiple-dip coating process. The two-part coating consists of a liquid base layer and a liquid lubricant layer. The PVC tubings were purged with nitrogen gas to remove solvent residue. The thickness of the coating was controlled by coating concentration, dipping speed and dipping time.
Incubation vial low-shear hemocompatibility assessment
Blood collection and exposure
Incubation vial testing was performed with human donor blood collected in a de-identified fashion at the USAISR Research Blood Bank. Male and female volunteers (n = 12) age 18–45 with no known coagulopathies were included. Blood was collected into 3.2% sodium citrate. Incubation vials were rinsed with phosphate buffered saline (1x PBS, pH = 7.4), and then blood from each donor was distributed into the four different vials (PET Control, PVC Control, Substrate, and LIS). Vials were incubated at 37°C for 1 h on a vertical rocker.
Post-exposure blood and material analysis
Following incubation, blood from the vials was analyzed using thromboelastography (TEG) as we previously reported 9 and impedance platelet aggregometry. Briefly, for TEG (TEG 5000, Haemonetics Corporation; Braintree, MA) blood was analyzed without activating agents and the following variables were recorded: reaction time (R), initial clot formation time (K), and clot strength (MA). Platelet aggregometry (Model 700, Chrono-Log Corporation; Havertown, PA) was performed using collagen stimulation (1 µg/mL). Whole blood (500 μL) was diluted with 3 mM CaCl2 in 0.9% NaCl and incubated at 37°C for 5 min before collagen activation. A test duration of 6 min was utilized and the relative area under the curve (AUC) recorded. The remaining blood was centrifuged twice for platelet-poor plasma (3000 × g, 10°min) to measure prothrombin time (PT), activated partial thromboplastin time (aPTT) and fibrinogen concentration (STA Compact Max, Stago; Parsippany, NJ). After incubation, vials were rinsed and prepared for scanning electron microscopy (SEM) imaging (Supplemental Methods).
Low shear fibrinogen adsorption onto tubing segments
Assessment of fibrinogen adsorption onto Substrate and LIS modified PVC tubing segments (2 cm length) was assessed using previously reported methods 20 (see Supplemental Methods).
Tubing 6 h extracorporeal circulation hemocompatibility assessment
Circuit Design
The LIS-modified ECLS circulation tubing was incorporated into a benchtop blood circulation loop consisting of two tubing segments (1 m length, 3/8” ID), a blood reservoir, and a centrifugal pump (Novalung Console and DP3 pump head, Fresenius Medical Care; Bad Homburg, Germany) (Figure 1(c)). A second circuit was constructed using unmodified PVC. Blood flow rate was monitored using clamp-on ultrasonic flow sensors (Transonic Systems; Ithaca, NY). Pressure was monitored post-pump (Propaq M, ZOLL Medical; Chelmsford, MA). A Hemotherm® heater (Gentherm Medical; Cincinnati, OH) was used for temperature maintenance (38°C). Circuits were primed with 1 L normal saline at 1.5 L/min for 30 min prior to start of testing.
Blood collection and circulation
Swine donor blood was collected following approved protocols (SU0001-10-22 and SU003-09-23, University of Texas San Antonio, San Antonio, TX, USA) at the Autonomous Reanimation and Evacuation Research Program (San Antonio, TX, USA) in compliance with the Animal Welfare Act, the principles set forth in the “Guide for the Care and Use of Laboratory Animals,” and all local, state and federal guidelines for the ethical use of animals. Blood (1 L) was collected from Yorkshire swine donors (55 ± 5 kg) following an unrelated study involving polytrauma injury with 48-h follow-up. Blood was collected into low-dose unfractionated heparin (0.75 U/mL), and was divided such that PVC and LIS circuits were evaluated with paired blood from one donor simultaneously. Blood flow was maintained at 1.5 L/min. Unfractionated heparin boluses (25–50 U) were administered during circulation to maintain an ACT goal of 140–180 s.
Data collection and blood testing
At the start of circulation (BL) and hourly, blood samples were collected to measure ACT, pH, lactate, and potassium. Flow rate, pump revolutions per minute (RPMs), circuit pressure, and heparin administered were recorded. At BL, 3 and 6 h, blood samples were collected to assess coagulation and platelet activation. Briefly, blood was collected into ethylenediaminetetraacetic acid (EDTA) for complete blood cell count (CBC) and preparation of plasma by centrifugation (3000 × g, 10 min) to measure free hemoglobin. 21 Additionally, a platelet activation panel was performed by flow cytometry (CytoFLEX, Beckman Coulter; Brea, CA) as previously reported. 22 Monoclonal antibodies for CD61-FITC (clone JM2E5) and CD62P-RPE (clone Psel.KO.2.5) (Bio-Rad; Hercules, CA) were used to indicate the platelet population and P-selectin positive platelets, respectively. Bovine lactadherin-Alexafluor 647 (Haemotologic Technologies, Inc; Essex Junction, VT) was used for phosphatidylserine (PS) analysis to indicate procoagulant platelets (see Supplemental Methods). Citrated blood (3.2%) was collected for TEG using kaolin activation and heparinase cups; and for platelet aggregometry as described above using collagen (3.2 μg/mL). 22 Platelet poor plasma was prepared as described above for analysis of PT, aPTT, fibrinogen, D-dimer, von Willebrand Factor (vWF) activity, and antithrombin III (STA Compact Max, Stago; Parsippany, NJ). 23
Post-circulation tubing assessment
Following 6 h circulation, tubing and pump heads were rinsed and inspected for deposition of macroscopic clots. The size and quantity of clots was recorded, and a thrombus deposition score (Supplemental Table S1) was assigned to rank the severity of thrombus deposition from 0 (no thrombi visualized) to 5 (most severe, thrombus occluding circuitry or reducing ID of tubing lumen by >25%). Tubing was collected for visualization by SEM (Supplemental Methods).
Statistical methods
All statistical tests (SAS 9.1; Cary, NC) were two-sided with alpha ⩽0.05 for significance. See Supplemental Methods.
Results
Human blood incubation vial assessment
Donor CBC summary is contained in Supplemental Results (Supplemental Table S2). The effect of exposure to the modified and control surfaces on clot formation dynamics (TEG), platelet aggregometry, and plasma coagulation tests is shown in Figure 2. Reaction time was prolonged for substrate compared to LIS. Maximum clot strength was significantly reduced in the LIS group. Platelet aggregation was elevated in the Substrate group compared to LIS and Controls. There were no differences in PT and aPTT. Excitingly, fibrinogen concentration in plasma from LIS vials was significantly higher compared to all other groups, suggesting fibrinogen adsorption onto the vial surface was reduced. This reduction in fibrinogen adsorption and potential subsequent reduction in thrombus deposition were supported by post-blood exposure SEM imaging of vial interior surfaces (Figure 3). Deposition of activated platelets, blood cells, and thrombi were limited or absent on LIS vials.
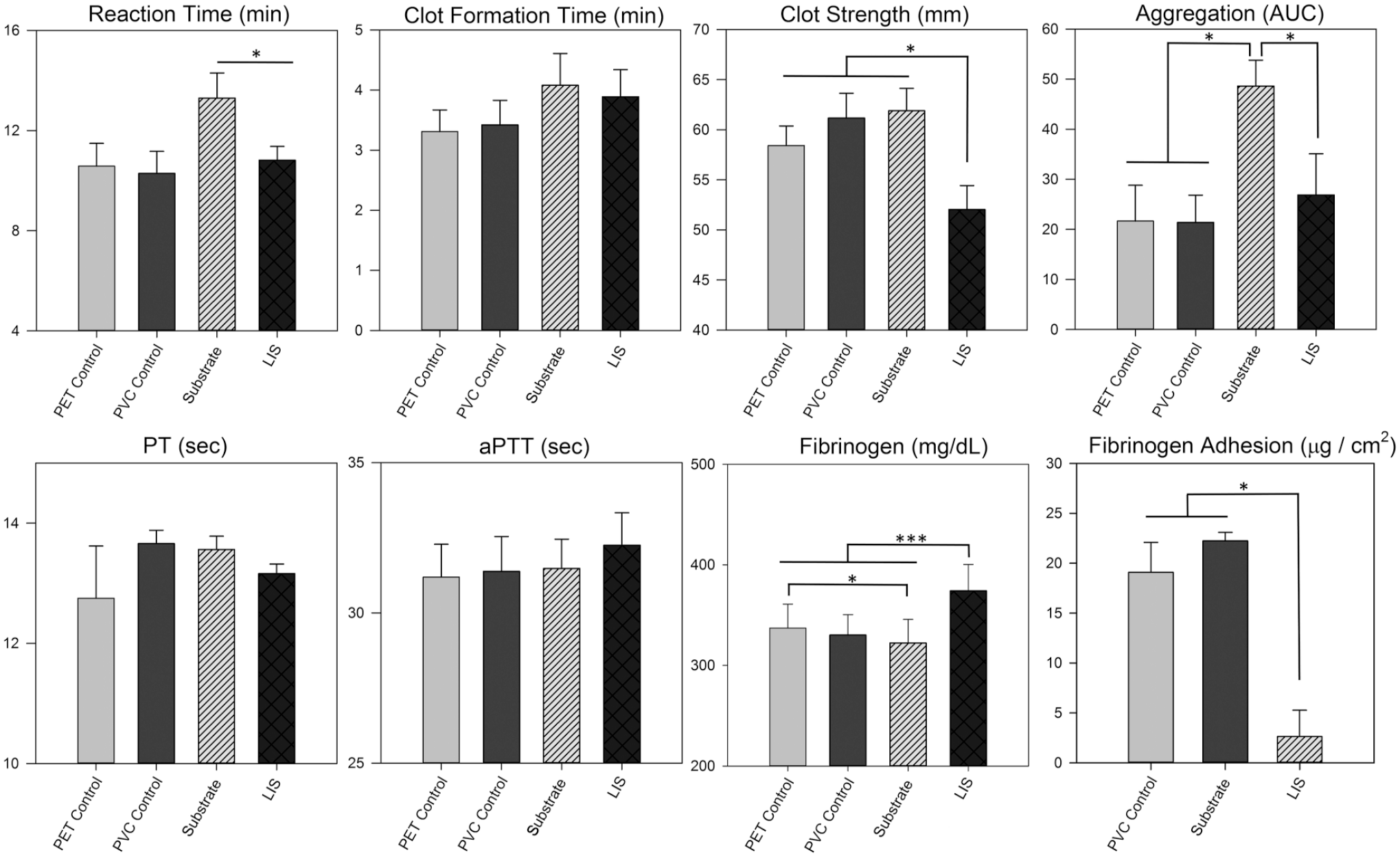
Results of thromboelastography (TEG), collagen-induced platelet aggregation, prothrombin time (PT), activated partial thromboplastin time (aPTT), and plasma fibrinogen concentration in human blood incubated in unmodified polyethylene terephthalate vials (PET Control), polyvinyl chloride coated vials (PVC Control), vials with substrate modification (Substrate), and vials with liquid impregnated surface modification (LIS), N = 12. Additionally, fibrinogen adhesion to 2 cm PVC tubing segments with surface modifications are shown. Results are expressed as mean ± standard error. Significant differences between groups are indicated: *p < .05, **p < .001, ***p < .0001.

Representative scanning electron microscopy images (1000 × magnification) of blood incubation vials after blood exposure. Unmodified polyethylene terephthalate vials (PET Control) (Panel A), polyvinyl chloride coated vials (PVC Control) (Panel B), vials with substrate modification (Substrate) (Panel C), and vials with liquid impregnated surface modification (LIS) (Panel D) are compared.
Low-shear fibrinogen adsorption on lubricant-impregnated surface-modified polyvinyl chloride tubing segments
Adsorption of fibrinogen onto LIS-modified PVC tubing segments was substantially reduced compared to unmodified PVC Control and Substrate (Figure 2, “Fibrinogen Adhesion”). This agreed with the increased fibrinogen content in plasma from the LIS blood incubation vials.
6 h extracorporeal circulation tubing assessment
To evaluate the safety and efficacy of LIS modification under clinically-relevant blood flow conditions for ECCO2R, LIS was applied to full-scale ECLS tubing and evaluated for 6 h at 1.5 L/min. Donor blood was collected from swine following polytrauma injury, imparting a challenging and clinically relevant test model as the donor blood was in a hypercoagulable state following injury stimuli. Summary of donor CBC and coagulation tests acquired at the time of blood donation are in Supplemental Table S3.
All circuits remained patent, with the exception of one experiment where both the LIS and PVC circuit occluded simultaneously at 5.5 h. Pump RPMs to maintain blood flow at 1.5 L/min were not different between groups (Supplemental Table S4). There was no group difference in ACT and quantity of heparin bolused to maintain the ACT target (at 6 h, PVC = 80 ± 40 U, LIS = 90 ± 40 U) (Supplemental Table S4). No group difference in pressure, blood pH, lactate, or potassium was observed (Supplemental Table S5).
Platelet count and degree of platelet activation and hypercoagulability, as indicated by P-selectin and PS exposure, respectively, are shown in Figure 4. Platelet count was significantly reduced in PVC circuits by 3 h; and was significantly reduced in both PVC and LIS by 6 h. Percentage of activated platelets and procoagulant platelets became elevated in the PVC group at 3 h and 6 h, respectively, while neither were elevated in the LIS group.
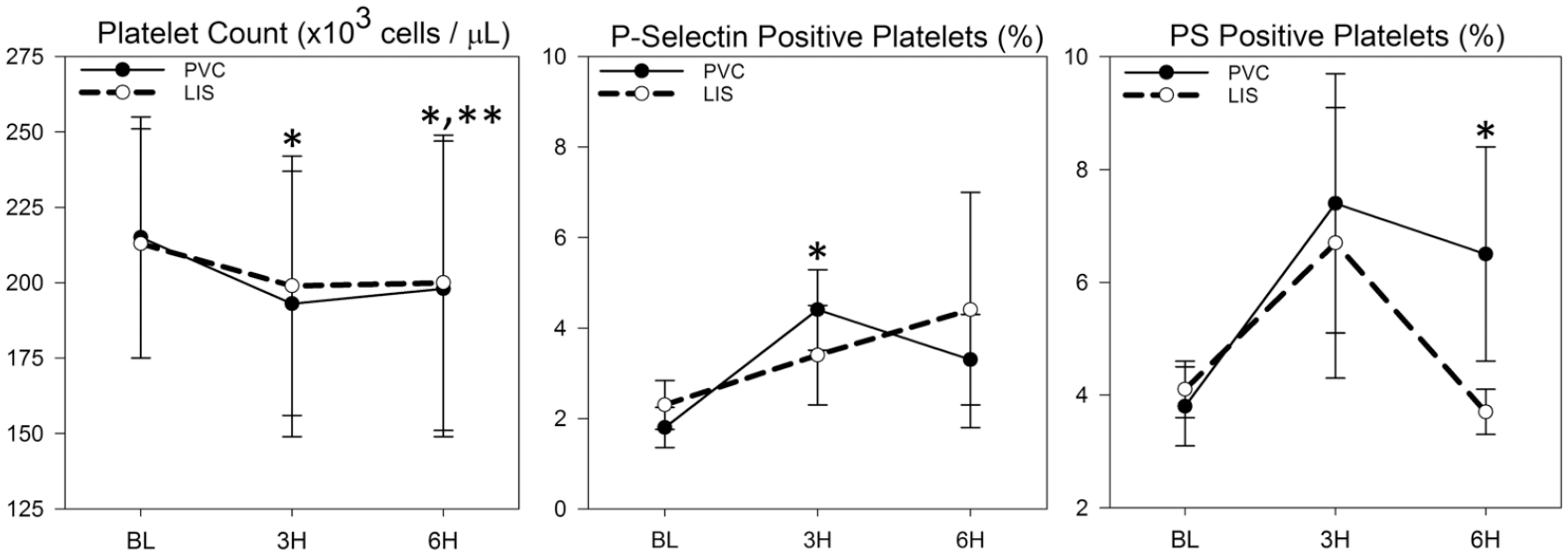
Platelet count and percentage of activated platelets (P-selectin positive) and procoagulant platelets (phosphatidylserine [PS] positive) collected over 6 h extracorporeal blood circulation in vitro using standard polyvinyl chloride (PVC) medical tubing compared to liquid impregnated surface (LIS) modified PVC tubing (n = 5/group). *Indicates significant difference within PVC group versus baseline (BL). **Indicates significant difference within LIS group versus baseline. All tests were two-sided with p < .05 for significance.
CBC results and select coagulation metrics are in Table 1. In both groups, red and white blood cell counts declined, although clinically minimally. In both groups, the percentage of neutrophils decreased, and lymphocyte percentage increased at 6 h. No group difference in hemoglobin concentration, hematocrit, plasma free hemoglobin, D-dimer concentration, vWF activity, or antithrombin III activity were observed.
Complete blood cell count and coagulation metrics throughout 6 h extracorporeal blood circulation in vitro using standard polyvinyl chloride (PVC) medical tubing compared to liquid impregnated surface (LIS) modified PVC tubing (n = 5/group).
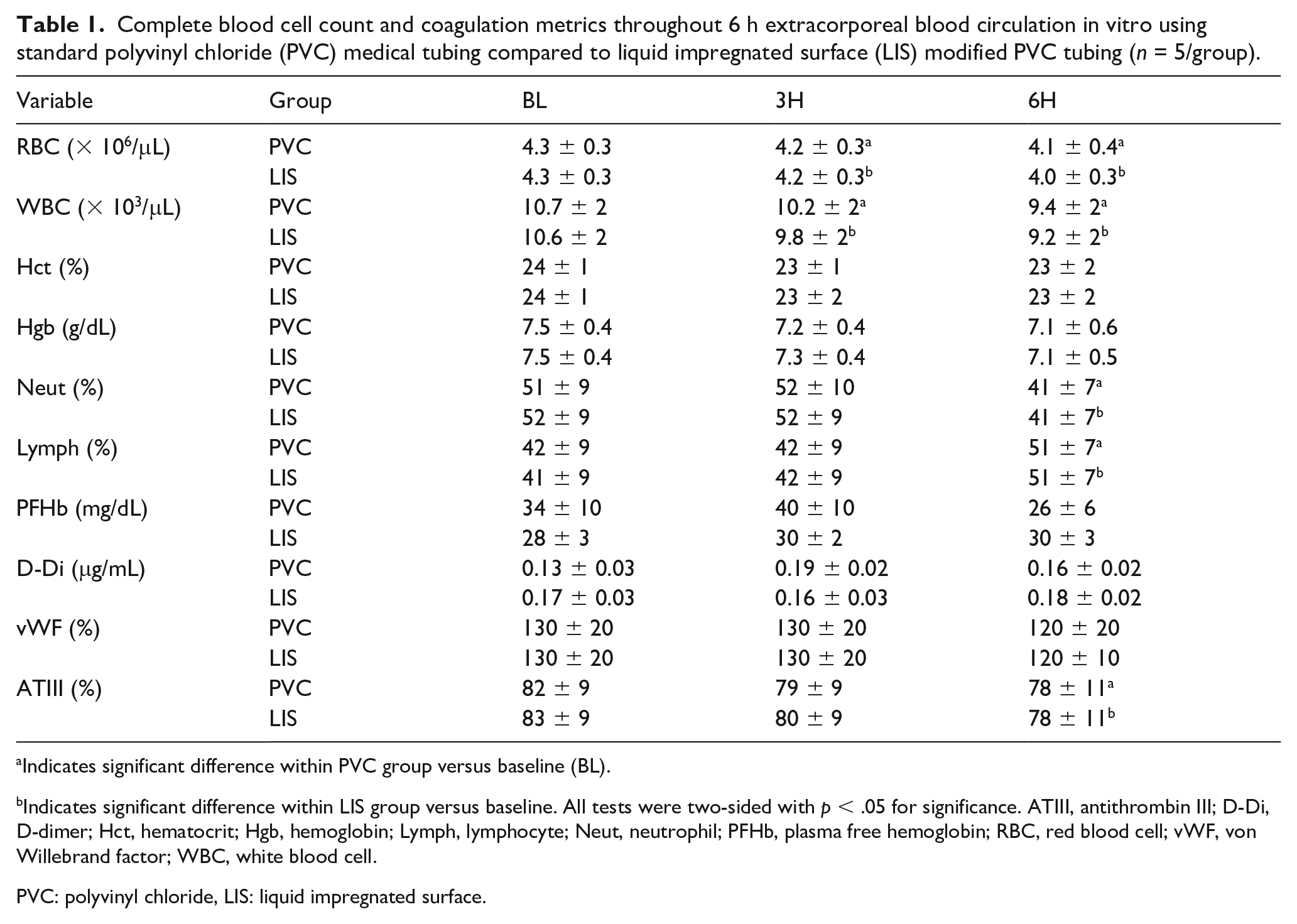
Indicates significant difference within PVC group versus baseline (BL).
Indicates significant difference within LIS group versus baseline. All tests were two-sided with p < .05 for significance. ATIII, antithrombin III; D-Di, D-dimer; Hct, hematocrit; Hgb, hemoglobin; Lymph, lymphocyte; Neut, neutrophil; PFHb, plasma free hemoglobin; RBC, red blood cell; vWF, von Willebrand factor; WBC, white blood cell.
PVC: polyvinyl chloride, LIS: liquid impregnated surface.
Coagulation and thrombus deposition scores are shown in Figure 5. No group difference in TEG, platelet aggregation, clotting times, and fibrinogen concentration were observed. Mean thrombus deposition scores for the blood pump and circuit tubing were numerically lower for LIS versus PVC. Thrombus deposition and adherence of activated platelets on post-circulation tubing samples visualized by SEM were reduced on LIS compared to PVC (see Figure S1).
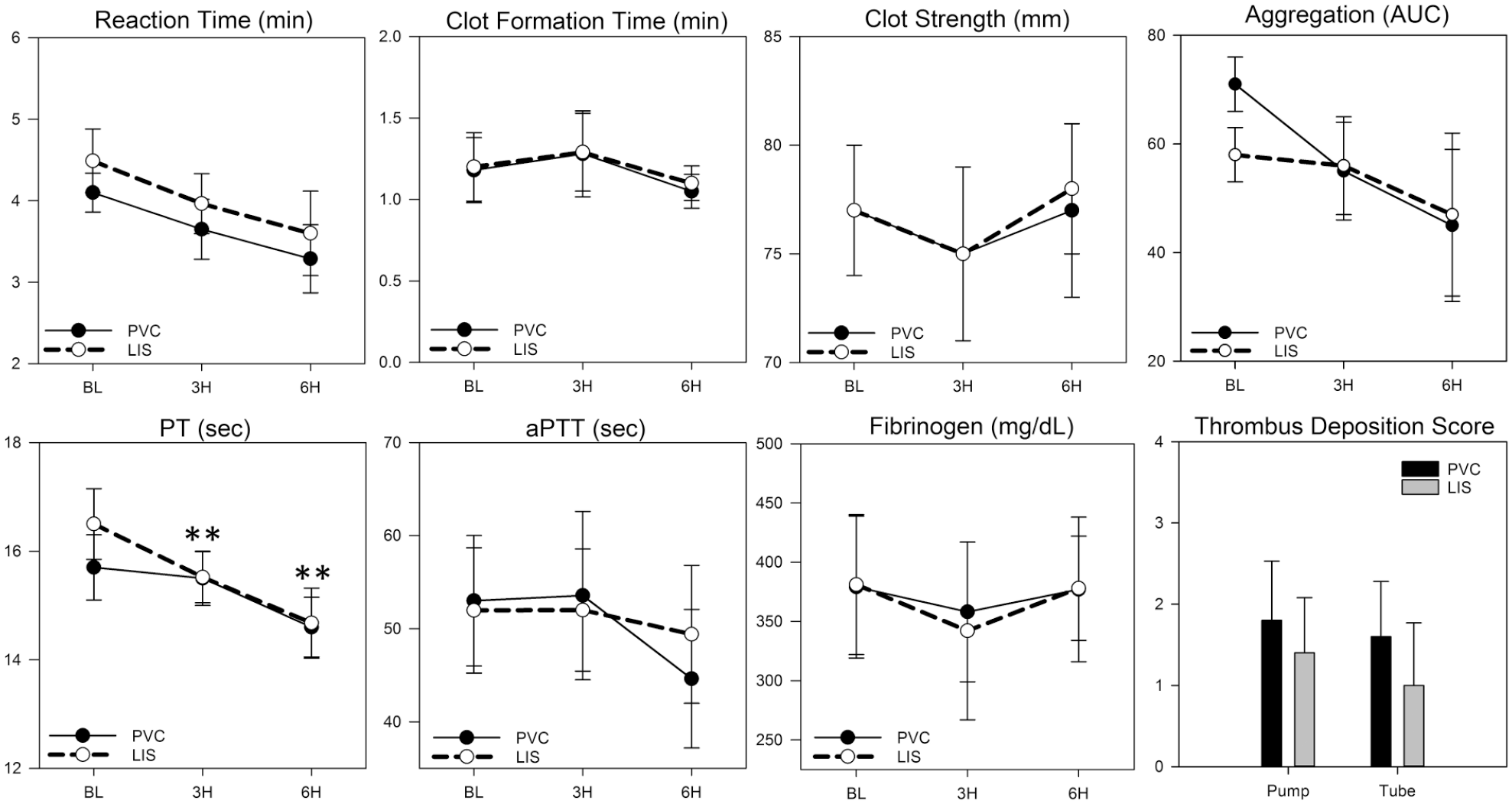
Coagulation data collected over 6 h extracorporeal blood circulation in vitro using standard polyvinyl chloride (PVC) medical tubing compared to liquid impregnated surface (LIS) modified PVC tubing (n = 5/group). *Indicates significant difference within PVC group versus baseline (BL). **Indicates significant difference within LIS group versus baseline. All tests were two-sided with p < .05 for significance. PT, prothrombin time; aPTT, activated partial thromboplastin time; AUC, area under curve.
Discussion
This in vitro study demonstrated the potential of LIS to reduce fibrinogen adsorption, limit thrombus development and potentially reduce platelet activation when applied to blood-contacting medical polymers. In the low-shear study, concentration of fibrinogen was preserved in the blood from the LIS vials, and minimal thrombus deposition was observed following blood exposure. Additionally, plasma coagulation and platelet aggregation upon stimulation were not altered, suggesting safety of the material. The reduction in clot strength measured by TEG when blood was not stimulated by an activating agent may suggest reduced platelet activation and prothrombotic tendency. The low-shear protein adsorption study showed a dramatic reduction in fibrinogen adhesion on LIS modified PVC tubing compared to control. Finally, in the 6 h ex vivo extracorporeal circulation model, thrombus deposition on full-scale LIS tubing was minimal or absent, and there was a trend towards reduction in platelet activation compared to PVC control.
These results agree with previous studies which use LIS-coatings for extracorporeal circuits and indwelling medical devices, which reduce protein adhesion, platelet adhesion, and surface thrombosis. 10 However, these studies rely on coatings consisting of perfluorocarbons, which are still restricted for blood contact applications and subject to environmental concerns. 23 Here, by using our thermodynamic algorithms, we demonstrate that anti-fouling performance can be achieved using an intravenous safe coating formulated with natural ingredients, as shown by the significant reduction in fibrinogen absorption and blood thrombosis. Further, as the LIS-coating requires a thin, micron thick liquid layer, the total volume of liquid on the coating is significantly less than safety limits for an intravenous therapeutic.
In addition to reducing protein adsorption and thrombus deposition, LIS coatings have the potential to reduce shear stress during flow applications. 24 This may explain the observed increase in platelet activation and platelet PS exposure that occurred during the ex vivo circulation model in the control group that did not occur in the LIS group. Non-adhesive superhydrophobic surfaces including LIS have a lower surface-free energy than unmodified PVC. The result is a reduction in drag force at the blood-polymer interface, reducing the wall shear stress. 25 Reduction of shear stress is critical during extracorporeal circulation, as the mechanical forces and supraphysiological shear conditions within ECLS circuits and other medical devices can cause activation of platelets and clotting factors, as well as damage to blood cells including hemolysis.26,27 Interestingly, an in vitro blood circulation study that used similar blood flow rates (0.3–1.5 L/min) and tubing dimensions as performed here demonstrated that when a near-superhydrophobic coating was applied to the interior surfaces of PVC, the coating significantly reduced the rate of hemolysis at all flow rates evaluated compared to unmodified control. 28 The promising dual-action potential of LIS to reduce both contact-mediated and mechanical force-mediated coagulation and blood cell damage during ECLS requires further investigation in a circulation setting that includes all circuit components (catheter, membrane, and tubing, pump) evaluated for extended duration using similar tests of coagulation, platelet activation, aggregation, and hemolysis as performed here.
A limitation of this study is that the results generated at the blood flow rate tested may not be directly applicable to other medical devices that use significantly higher or lower blood flow rates. Another limitation of this pilot study is the short duration of blood exposure of up to 6 h only. This was due to the limited viability of donor blood in an in vitro setting; and was also to enable comparison with standard hemocompatibility methods in the literature which are performed for the same duration. 29 Prior to making claims to clinical care, it will be essential to evaluate LIS for the duration of intended clinical use. For extracorporeal organ support, for example, the average duration of support for adult respiratory indications in 2020 was 466 h. 1 This discrepancy in duration of hemocompatibility tests and duration of clinical target medical device applications is an unfortunate custom in biocompatibility research. Following promising results in early screening models as observed here, progression to in vivo testing for extended duration, and in the absence of the confounding variable of anticoagulant use, is essential. For example, we previously demonstrated successful performance of a different non-adhesive coating during a similar low-shear hemocompatibility screening as performed here, 9 as well as in a 6-h in vivo extracorporeal circulation model in swine using standard ECLS circuits to which the coating was applied. 30 The coating initially demonstrated substantial benefit compared to standard circuits; however, when we extended the test duration to 72 h and withheld systemic anticoagulation, the coating no longer demonstrated benefit. 23 While, we observed some hemocompatibility benefit with application of LIS compared to unmodified PVC in the ex vivo circulation model in this study where heparin anticoagulation was used in both circuits, we posit that this benefit may be amplified when the test duration is extended and the confounding variable of heparin anticoagulation can be eliminated during future in vivo testing.
In conclusion, LIS coating demonstrated potential as a candidate to improve the blood compatibility of blood-contacting medical devices such as ECCO2R. A material solution to prevent thrombosis and reduce or eliminate requirements for systemic anticoagulation during use of extracorporeal organ support therapies would dramatically improve safety and utilization. Further testing for extended duration to assess the safety and efficacy of this approach for multi-day organ support applications is necessary. Additionally, investigation of LIS application to other circuit components including catheters, blood pumps and membranes is underway.
Supplemental Material
sj-docx-1-prf-10.1177_02676591221095469 – Supplemental material for In vitro hemocompatibility screening of a slippery liquid impregnated surface coating for extracorporeal organ support applications
Supplemental material, sj-docx-1-prf-10.1177_02676591221095469 for In vitro hemocompatibility screening of a slippery liquid impregnated surface coating for extracorporeal organ support applications by Teryn R Roberts, Raymond P Seekell, Yanyi Zang, George Harea, Zheng Zhang and Andriy I Batchinsky in Perfusion
Supplemental Material
sj-tiff-1-prf-10.1177_02676591221095469 – Supplemental material for In vitro hemocompatibility screening of a slippery liquid impregnated surface coating for extracorporeal organ support applications
Supplemental material, sj-tiff-1-prf-10.1177_02676591221095469 for In vitro hemocompatibility screening of a slippery liquid impregnated surface coating for extracorporeal organ support applications by Teryn R Roberts, Raymond P Seekell, Yanyi Zang, George Harea, Zheng Zhang and Andriy I Batchinsky in Perfusion
Footnotes
Acknowledgements
Authors acknowledge the intellectual and mentorship support of Dr. Leopoldo C. Cancio, MD, COL (ret.). The authors also acknowledge the support of the USAISR Research Blood Bank as well as the technical support of AREVA research technician Ms Ji Lee, and USAISR personnel Ms Marianne Thrailkill, BS, and Mr Kevin Gladin, BS.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Assistant Secretary of Defense for Health Affairs endorsed by the Department of Defense, through the Peer Reviewed Medical Research Program – Technology/Therapeutic Development Award, under award W81XWH-18-2-0048 (PI: Andriy I. Batchinsky, Co-PI: Teryn R. Roberts). This work was also supported by the Combat Casualty Care Research Program, Project X_041_2020 (PI: Andriy I. Batchinsky, Co-PI: Teryn R. Roberts).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
