Abstract
Objectives:
Mortality rates in patients with acute myocardial infarction and cardiogenic shock (AMI-CS) remain persistently high despite advances over the past decade in percutaneous mechanical circulatory support. This systematic review aims to analyse the existing literature to compare mortality outcomes in patients mechanically supported by intra-aortic balloon pump or percutaneous Impella 2.5/CP© for AMI-CS undergoing emergency revascularisation.
Methods:
The following MeSH terms were applied to the databases Ovid Medline, Ovid Embase, Cochrane and Web of Science: ‘Intra-aortic balloon pump’, ‘Impella’, ‘Cardiogenic shock’, ‘Myocardial Infarction’ and ‘Mortality’. This yielded 2643 studies. Using predefined inclusion and exclusion criteria, the studies were initially screened by title and abstract before full text analysis.
Results:
Fourteen studies met eligibility criteria: two randomised controlled trials (RCTs) and 12 observational studies. Data from a total of 21,006 patients were included across the studies. Notably, one study claimed reduced mortality with IABP versus control, and one study concluded that Impella© improved survival rates over the IABP. The average 30-day all-cause mortality in patients supported by IABP was 38.1%, 54.3% in Impella© groups and 39.4% in control groups.
Conclusion:
AMI-CS presents an important cohort of patients in whom conducting RCTs is difficult. As a result, the literature is limited. Analysis of the available literature suggests that there is insufficient evidence to support superior survival in those supported by IABP or Impella© when compared to control despite suggestions that the Impella© offers superior haemodynamic support. Limitations of the studies have been discussed to outline suggestions for future research.
Keywords
Introduction
Aims
Emergent percutaneous mechanical circulatory support (pMCS) devices provide an alternative to the historically used IABP. Despite advances over the past decade in the field of interventional cardiology the literature is mixed, and randomised control trial (RCT) data is limited. Furthermore, mortality rates in high-risk cohorts such as AMI-CS patients undergoing emergency revascularisation (percutaneous coronary intervention/coronary artery bypass graft) remain high at approximately 50%.1–3 Although, superior haemodynamic support associated with novel pMCS such as the Impella© has been established in porcine 4 and model studies, 5 it is unclear how this correlates with survival benefit when compared to the IABP. This systematic review aims to conduct a comprehensive analysis of the existing literature to compare mortality outcomes in patients with AMI-CS undergoing emergency revascularisation supported by percutaneous devices: IABP and Impella CP/2.5© and discuss the clinical implications of the findings.
Cardiogenic shock
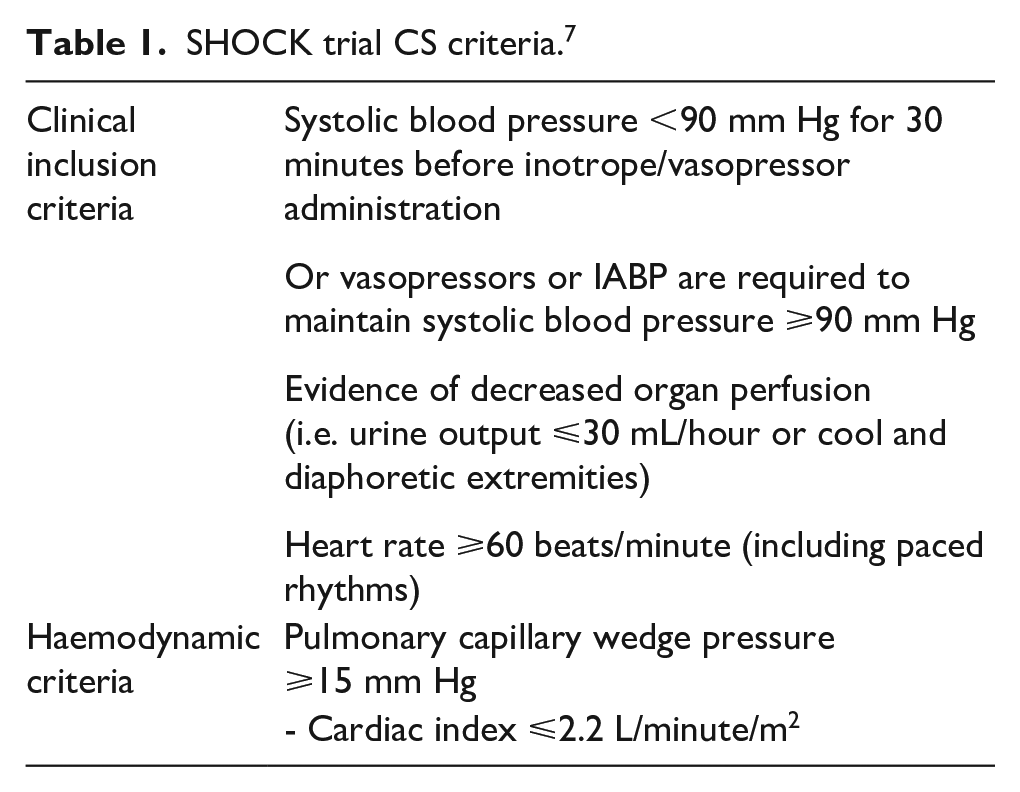
Universal agreement on diagnostic criteria for CS has not been reached. 6 The landmark SHOCK trial (1999) defined diagnostic criteria for CS based on patient clinical and haemodynamic features (Table 1). 7 This criteria has been widely recognised and frequently used in trials investigating the use of pMCS devices including the more recent ISAR-SHOCK trial. 8 Notably, serum biochemistry markers (lactate and bicarbonate) have been excluded in the SHOCK trial 7 but have been included in other subsequent trials such as the landmark IABP SHOCK II trial (serum lactate >2.0 mmol/L). 2 Serum lactate as a proxy for tissue hypoxia may be a useful diagnostic marker and indicator of the success of reperfusion therapy in AMI-CS. Other non-specific biomarkers such as creatinine and liver amino transferases indicating renal and hepatic hypoperfusion respectively should also be considered in this context. 9 For the purpose of this analysis, this definition has been broadly accepted.
SHOCK trial CS criteria. 7
Revascularisation
The SHOCK trial established reduction in mortality at 6 months associated with PCI or CABG in the setting of AMI-CS. 3 The first line recommendation in the European Society of Cardiology Guidelines for multivessel disease is revascularisation of the culprit lesion only 10 following the positive results of the CULPRIT SHOCK trial. 11
Mechanical circulatory support devices
MCS is indicated in the context of CS for bridge to decision, transplantation, recovery or durable device implantation as well as to temporarily support cardiac intervention such as PCI/CABG.12,13 Over the past decade, major advancements in this field have widened the variety of devices available. The most established and commonly used device in the UK, the IABP, 14 is rivalled by the novel percutaneous devices; Impella 2.5 and CP©, both of which were developed to supersede the limitations left by the IABP. 15 The most notable difference between the devices is that unlike the Impella© the IABP requires endogenous cardiac activity limiting its capacity to provide circulatory support to those in severe shock.16,17 Consequently, in patients with severe electro-physiological derangement or cardiac arrest rhythms, the Impella© may be preferrable.18,19 Other available devices which include Impella 5.0©, TandemHeart©, Heartmate© and Veno Arterial – Extracorporeal Membrane Oxygenator Pump (VA-ECMO) are outside of the scope of this analysis due to the difficulty of their rapid deployment in the Cath lab (Table 2).
Mechanical circulatory support devices.
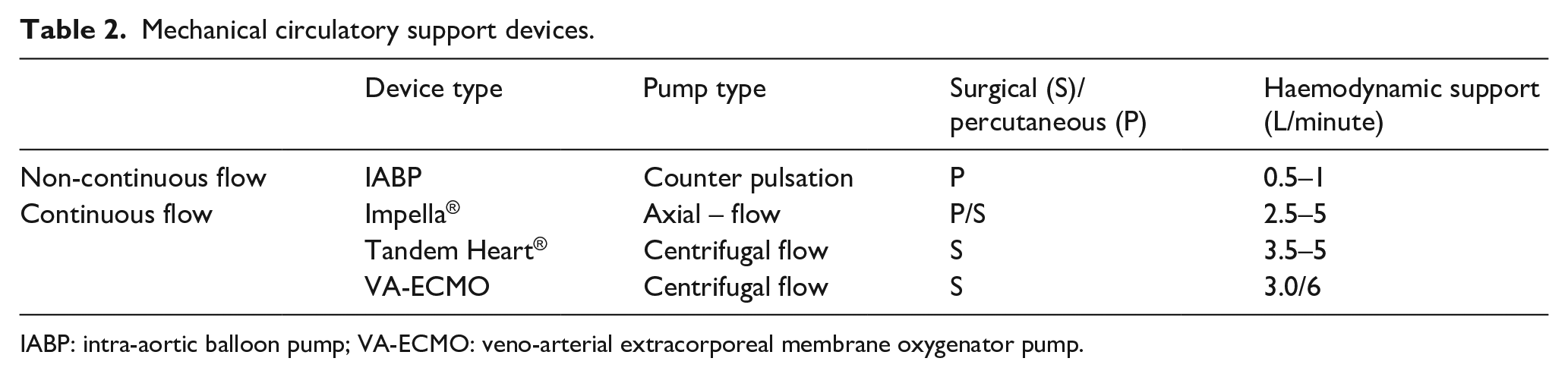
IABP: intra-aortic balloon pump; VA-ECMO: veno-arterial extracorporeal membrane oxygenator pump.
Methods
Search
In line with PRISMA guidelines a comprehensive search of the literature was conducted on 30/12/2020 using the following databases: Ovid Medline, Ovid Embase, Cochrane and Web of Science. For initial scope analysis of the literature a combination of Boolean operators and MeSH terms ‘Intra-aortic balloon pump’, ‘Impella’, ‘Cardiogenic shock’ and ‘Mortality’ were trialled. Of those trialled, only one search captured landmark randomised controlled trials answering the specific question addressed in this systematic review. The search executed for this review yielded 2643 papers and is detailed below:
- Ovid Embase: (Impella OR (IABP or ‘Intra-aortic balloon pump’)) AND (‘cardiogenic shock’) AND (‘myocardial infarction’) AND (hospital mortality/or all-cause mortality/or mortality/or cardiovascular mortality)
- Ovid Medline: (Impella or IABP) AND (‘Cardiogenic shock’) AND (‘Myocardial Infarction’) AND (Mortality)
- Web of Science: (((Impella OR (IABP or Intra-aortic balloon pump)) AND ((cardiogenic shock) (mortality) (‘myocardial infarction’))))
- Cochrane: (Impella OR (IABP or ‘Intra-aortic balloon pump’)) AND ((cardiogenic shock) AND (myocardial infarction) AND mortality)
Inclusion and exclusion criteria
To ensure the studies included in the final analysis answered the specific question posed by this review, predefined selection criteria were used and have been outlined below (Table 3):
Study selection criteria.
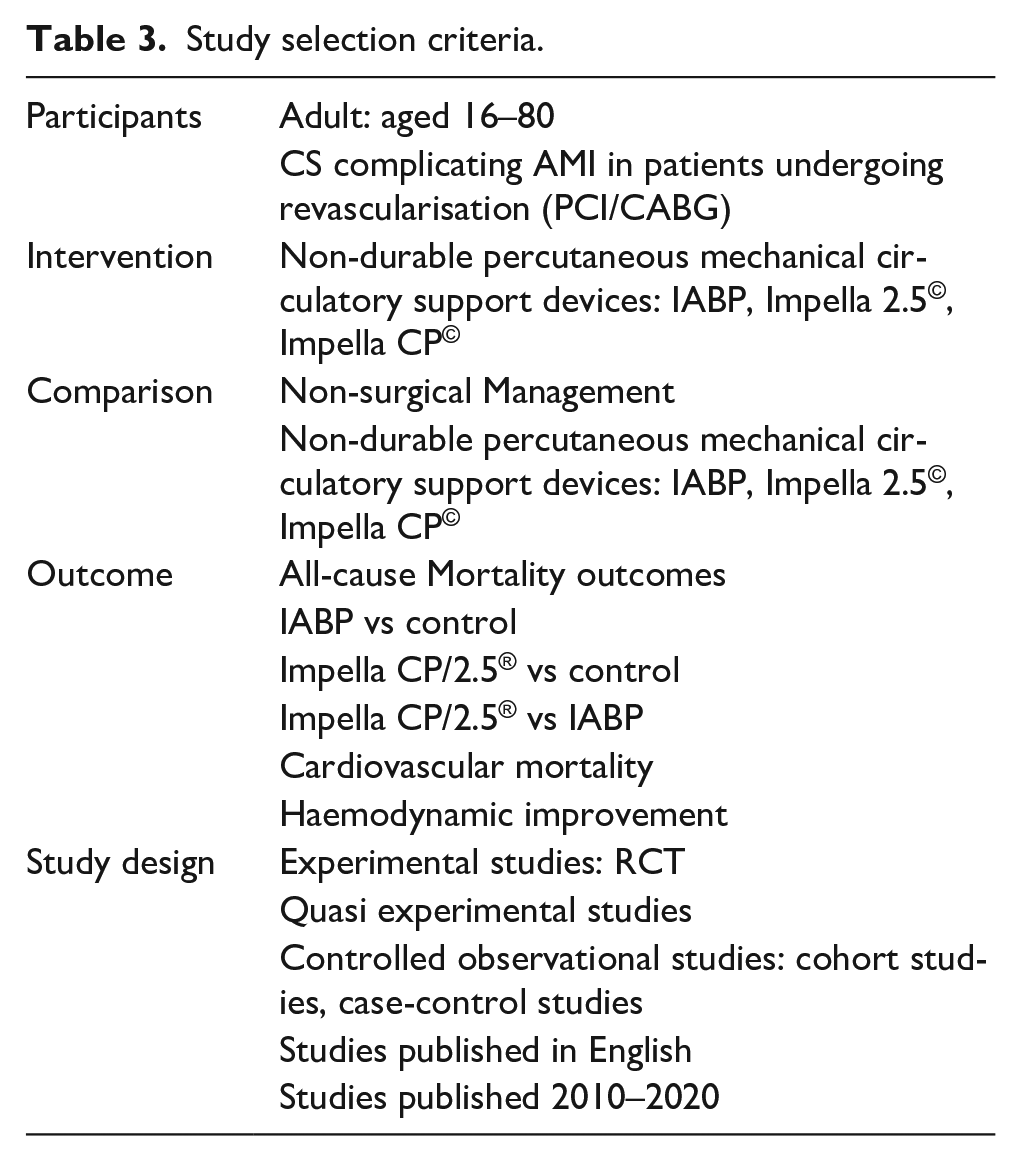
Study selection
Each study was independently reviewed initially by title and abstract by one of the authors (K.F.) with any controversy resolved by mutual agreement between authors (K.F. and P.R.). Of the studies shortlisted by initial screening, the full text was acquired and analysed, using predefined inclusion and exclusion criteria. A summary of the study selection process has been outlined via PRISMA 20 flow chart (see Figure 1). The study selection process took place from time of initial search 30-12-2020 until 04-01-2021.
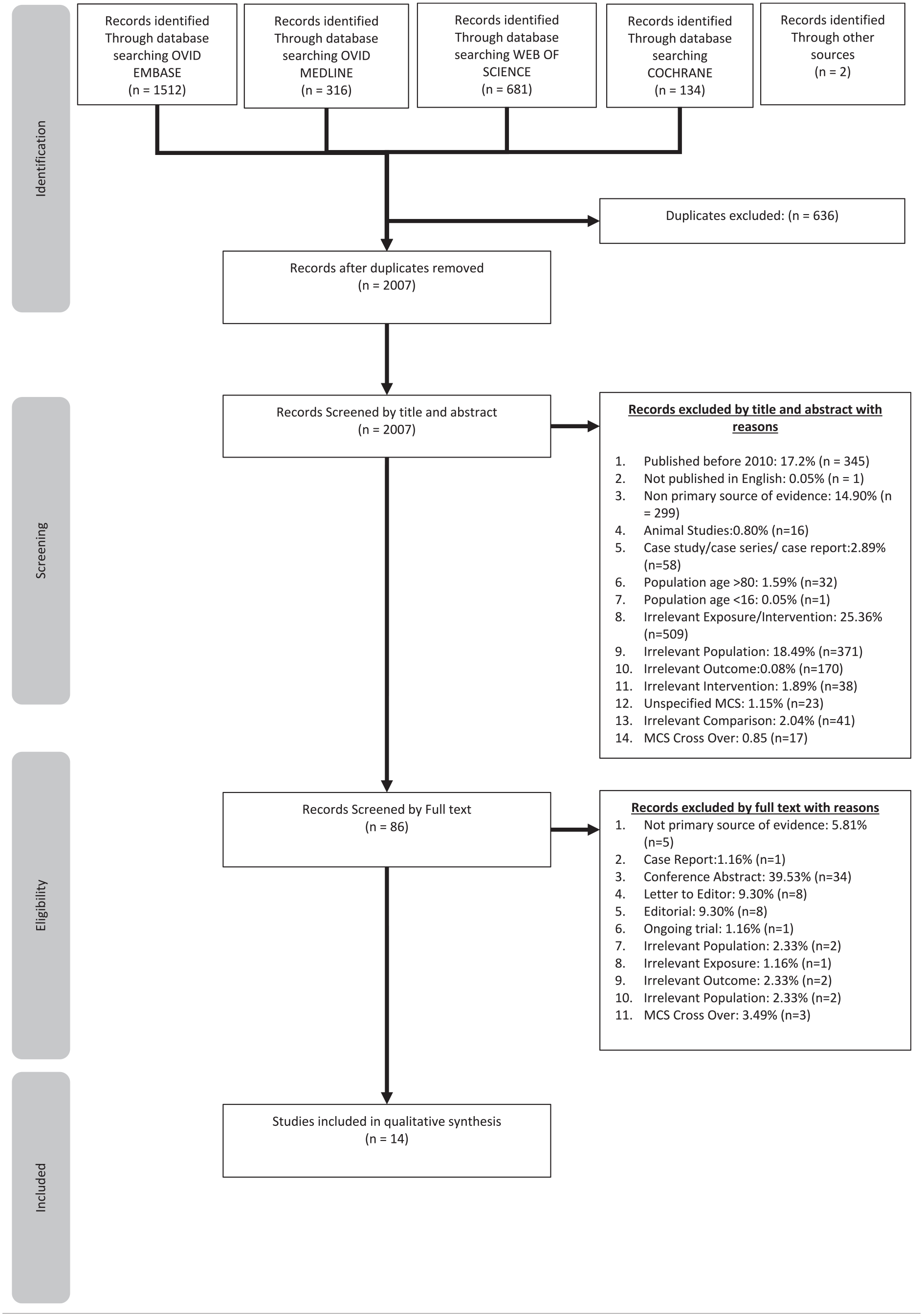
PRISMA flow chart for search results. 21
Eligibility
Fourteen studies met predefined inclusion and exclusion criteria and underwent further analysis using the following tools: NICE Algorithm 22 to assign study design and level of evidence, CASP checklists 23 to aid with critical appraisal and determine study quality for eligibility to be included in the final analysis and Cochrane ROBINS-1 24 and RoB 2.0 25 tools for the assessment and categorisation of bias risk. The results of this analysis have been presented in tabular format as seen in Table 4.
Summary table for risk of bias assessment.
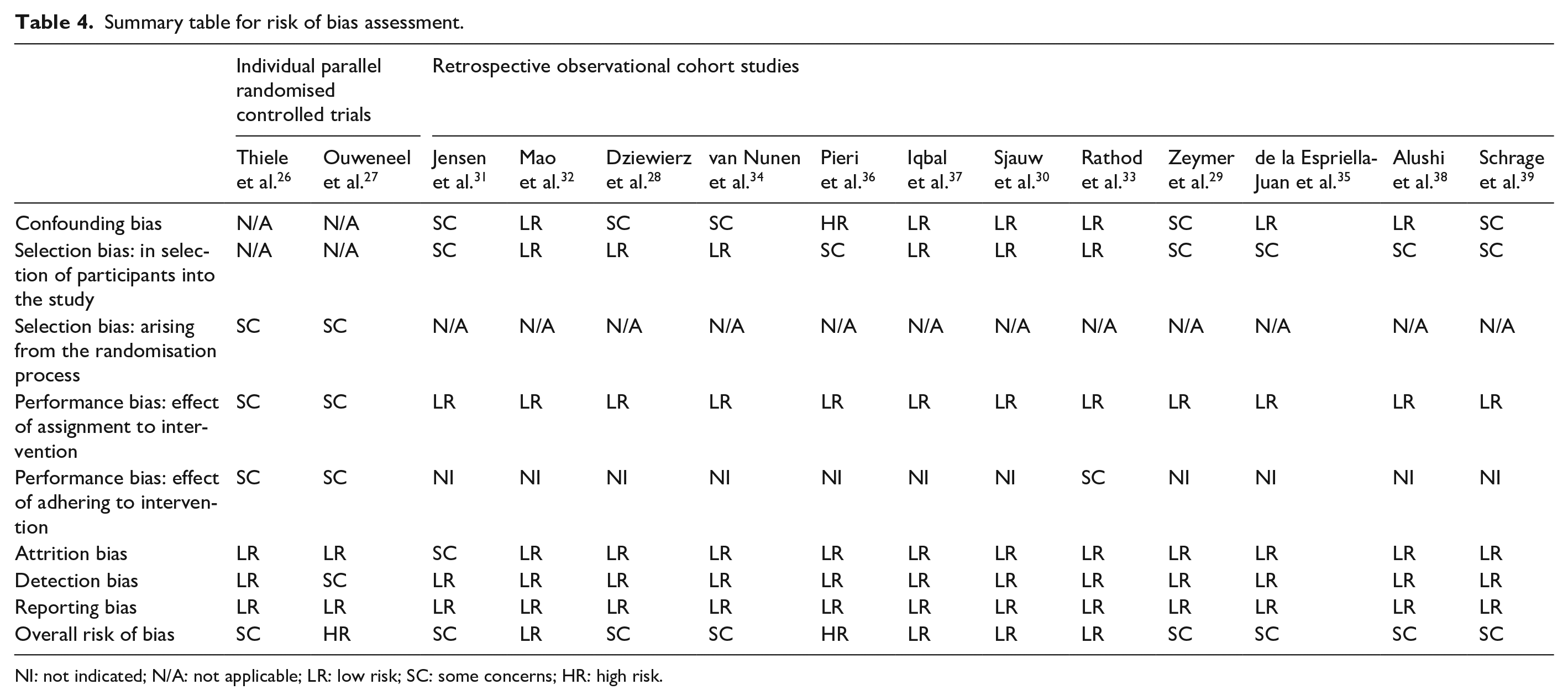
NI: not indicated; N/A: not applicable; LR: low risk; SC: some concerns; HR: high risk.
Study quality
To ascertain study quality, the above tools were used to scrutinise the shortlisted studies based on study findings, bias, statistical analytical techniques utilised and study limitations to discuss the clinical implication of the conclusions reached. The characteristics of each study have been tabulated in Table 5.
Study characteristics.
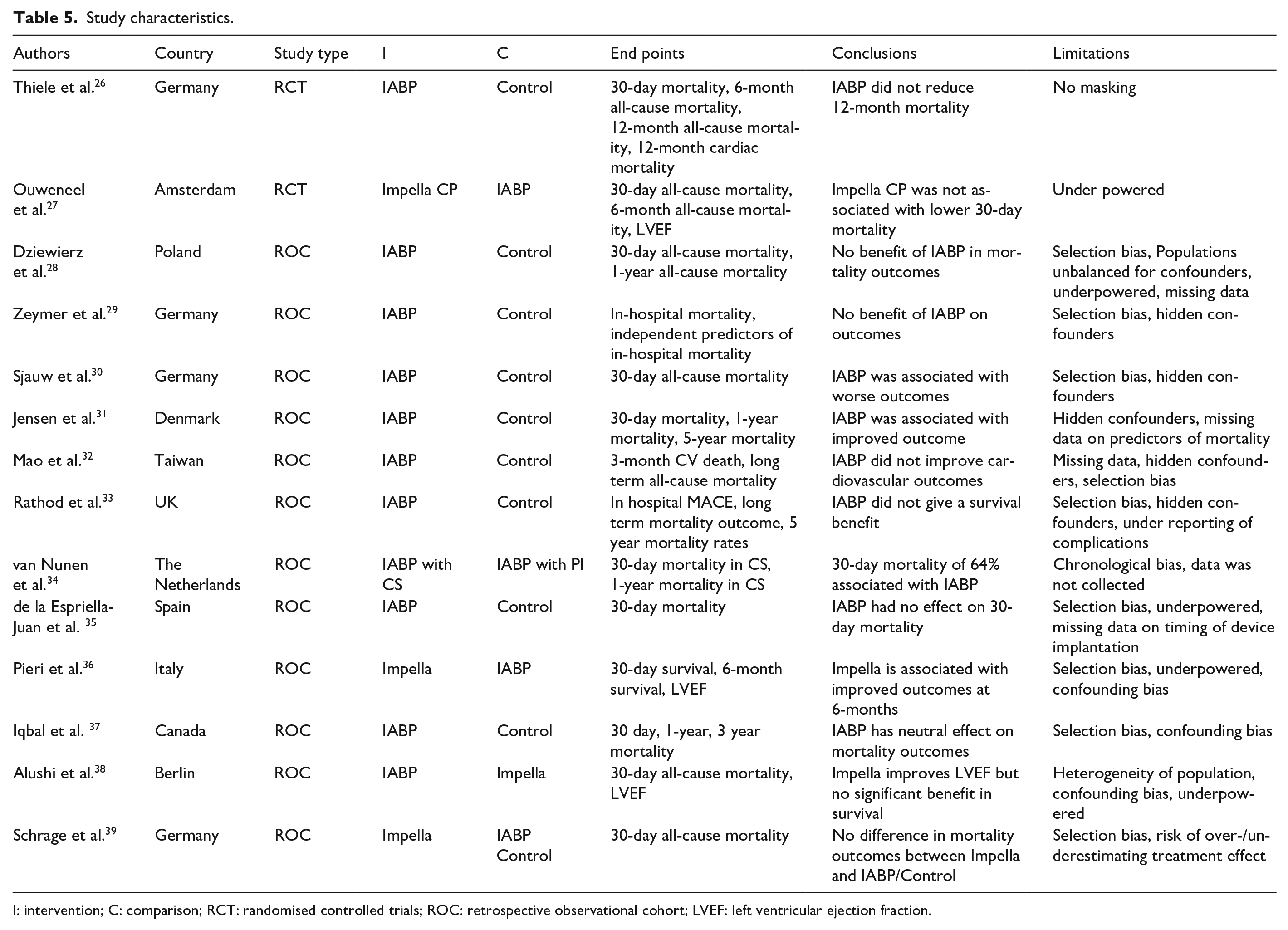
I: intervention; C: comparison; RCT: randomised controlled trials; ROC: retrospective observational cohort; LVEF: left ventricular ejection fraction.
Outcome measures
The primary endpoint was 30-day mortality; for the purpose of this study this can be defined as all-cause mortality. The secondary end points were cardiovascular mortality and haemodynamic improvement including improvement in LVEF and degree of shock.
Data extraction
Each study was analysed for data relevant to the question asked by this analysis. All available mortality data was recorded including all-cause and cardiovascular mortality. For further analysis of the study population and confounding variables that may impact the measured outcomes, several of the patient baseline characteristics were extracted such as the age, sex, presence of comorbidities, location of lesion etc. as well as the extraction of statistically significant independent predictors of mortality such as age, creatinine and cardiogenic shock.
Statistical analysis
Available baseline patient characteristics have been presented in tabular format (Figure 2). Continuous variables have been displayed as means or medians, discrete variables have been summarised by count (n) and percentage (%). Statistical Package for the Social Sciences (SPSS) was used to pool hazard ratios (HR) for independent predictors of mortality and present the data via forest plot, only those with statistical significance have been included. For this analysis, a p value of <0.05 is considered significant. To compare the primary outcome measure (30-day mortality) between studies, a random effect meta-analysis was conducted using inverse variance to calculate the risk ratio and 95% Confidence Intervals for either IABP versus Control or IABP versus Impella© according to each study intervention. Heterogeneity was assessed using Higgins and Thompson I 2 and Z test for variability in overall effect, a p value <0.05 was considered significant. All findings have been presented via forest plot. Data for Secondary outcome measurements was greatly lacking, to provide further context to the findings a random effect meta-analysis was performed using inverse variance for cardiovascular mortality risk ratio and LVEF mean difference. All meta-analyses were performed with Cochrane Collaboration, Review Manager software v5.4.1 for Academic use. 40
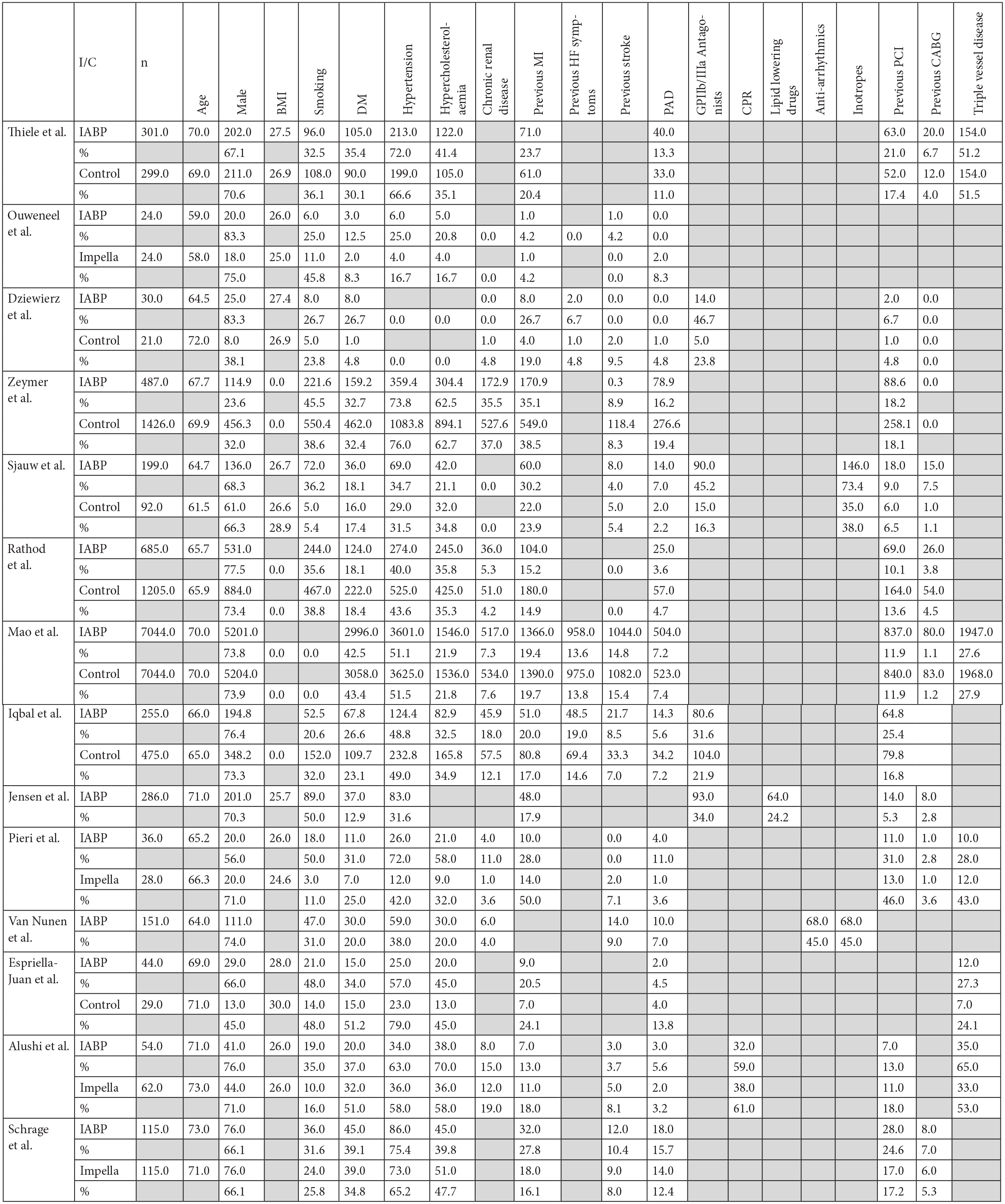
Summary of baseline patient characteristics.
Results
Search results – included studies
The final review consisted of two RCTs and 12 retrospective observational cohort studies. Data from a total of 21,006 patients were included. About 9496 were supported with IABP, 11,044 did not receive MCS and instead received medical management (control population) and 466 were supported by Impella 2.5/CP©. All patients underwent emergency revascularisation either by PCI or CABG. Observational cohort study data was collected either from registry databases or in-hospital records within the period 1997–2017. Ten of which were conducted in Europe28–31,33–36,38,39 with the remainder of data obtained from Canadian or Taiwanese records. 32,37 Both RCTs; IABP-SHOCK II and IMPRESS in Severe Shock were conducted within Europe in 2013 and 2017, respectively.
Patient baseline characteristics
From the data available the average age across the studies for the IABP population was 65.66 (IQR 64.25–69.50), versus 70.00 (IQR 66.30–71.00) for Impella© groups and 68.81 (IQR 65.93–71.00) for control groups. Males were over-represented with 15,176 (72.25%) male patients included. There was preferential use of IABP in patients with left main/left anterior descending lesions in three of the studies.
Risk of bias assessment
Cochrane ROBINS-1 and RoB 2.0 tools were used to assign bias risk for each of the following types of bias: selection, confounding, chronology, performance, attrition and detection bias. Only 2 of the 14 studies were identified as having a high overall risk of bias, 8 studies had a categorised moderate overall risk of bias, with the remaining having low overall risk (Table 4).
Primary outcome measures
All-cause mortality
The average 30-day all-cause mortality in patients treated with IABP was 38.1% versus 54.3% in Impella® groups and 39.4% in control groups. Risk ratio data associated with each intervention has been presented via forest plot, statistical analysis found results to be homogeneous between studies (Figure 3).
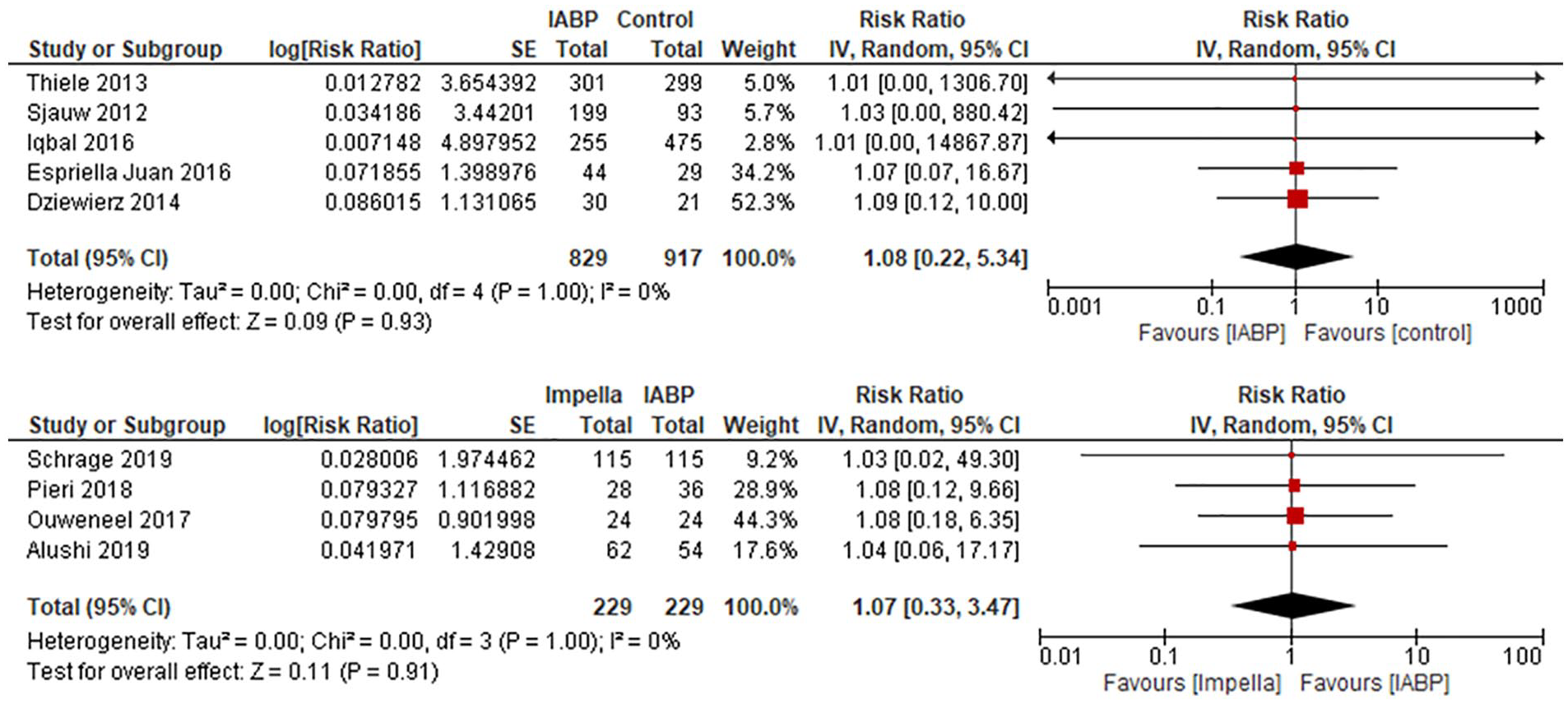
Forest plot of 30-day mortality.
Secondary outcome measures
Cardiovascular mortality
The RCT conducted by Thiele et al. and the retrospective cohort study by Mao et al., were the only studies to directly measure mortality secondary to cardiovascular events. A 50% Cardiovascular mortality in both IABP and control groups were identified at 12 months in the study conducted by Thiele et al. 26 This is in contrast to the findings from the Mao et al. 32 study which found a statistically significant higher CV mortality in the IABP group (27.7%) versus No IABP (14.9%) (HR 2.07 (1.92–2.23)). Upon statistical analysis using a random effects model and inverse variance to determine overall effect of balloon pump on cardiovascular mortality, risk ratios were found to be non-significant (Figure 4).
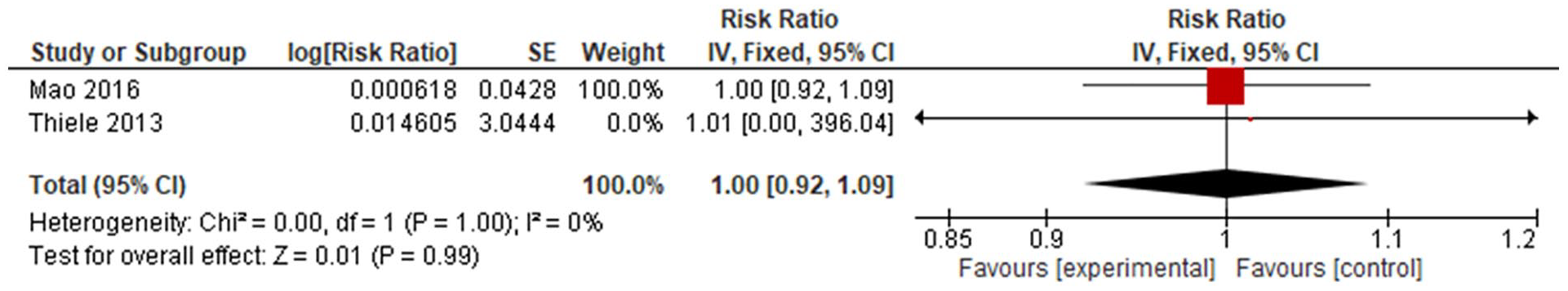
Forest plot of cardiovascular mortality.
Left ventricular ejection fraction
About 3/12 studies directly measured LVEF. One study found a minimal difference between IABP and Impella© groups with 46% versus 49% LVEF in the Impella© versus IABP groups respectively at 2.5 months. 27 The study conducted by Pieri et al. 36 found a statistically significant 20% increase in LVEF compared to 10% in the IABP (p < 0.0057), results which were mirrored in the study conducted by Alushi et al. 38 which stated a significant improvement in LVEF baseline in the Impella© groups. Statistical comparison could only be made between mean results from the studies conducted by Ouweneel et al. and Pieri et al. It should be noted this is a comparison of LVEF recorded at 2.5 months and at discharge respectively, reflecting short term outcomes. 36 Inverse variance analysis revealed favourable haemodynamic benefit associated with Impella use (Figure 5). 27
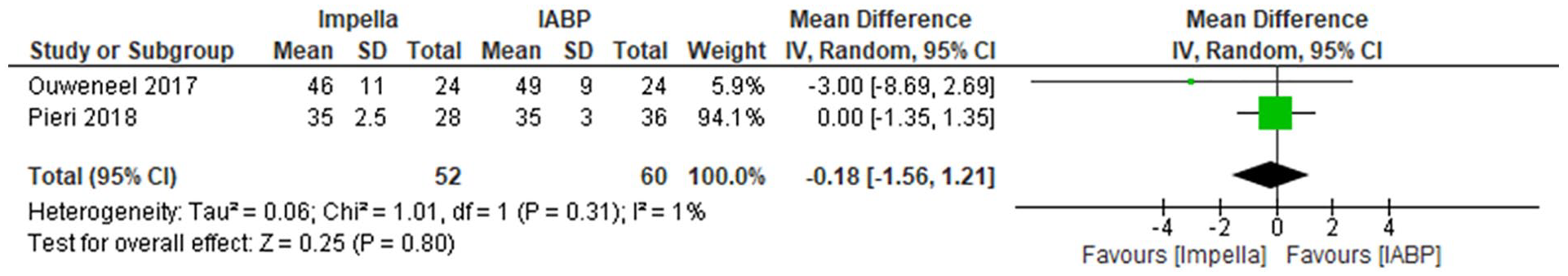
Forest plot of left ventricular ejection fraction.
Independent predictors of mortality
Identified independent predictors of mortality from each study were extrapolated and only those with statistical significance (p < 0.05) from each study have been presented via forest plot (Figure 6).
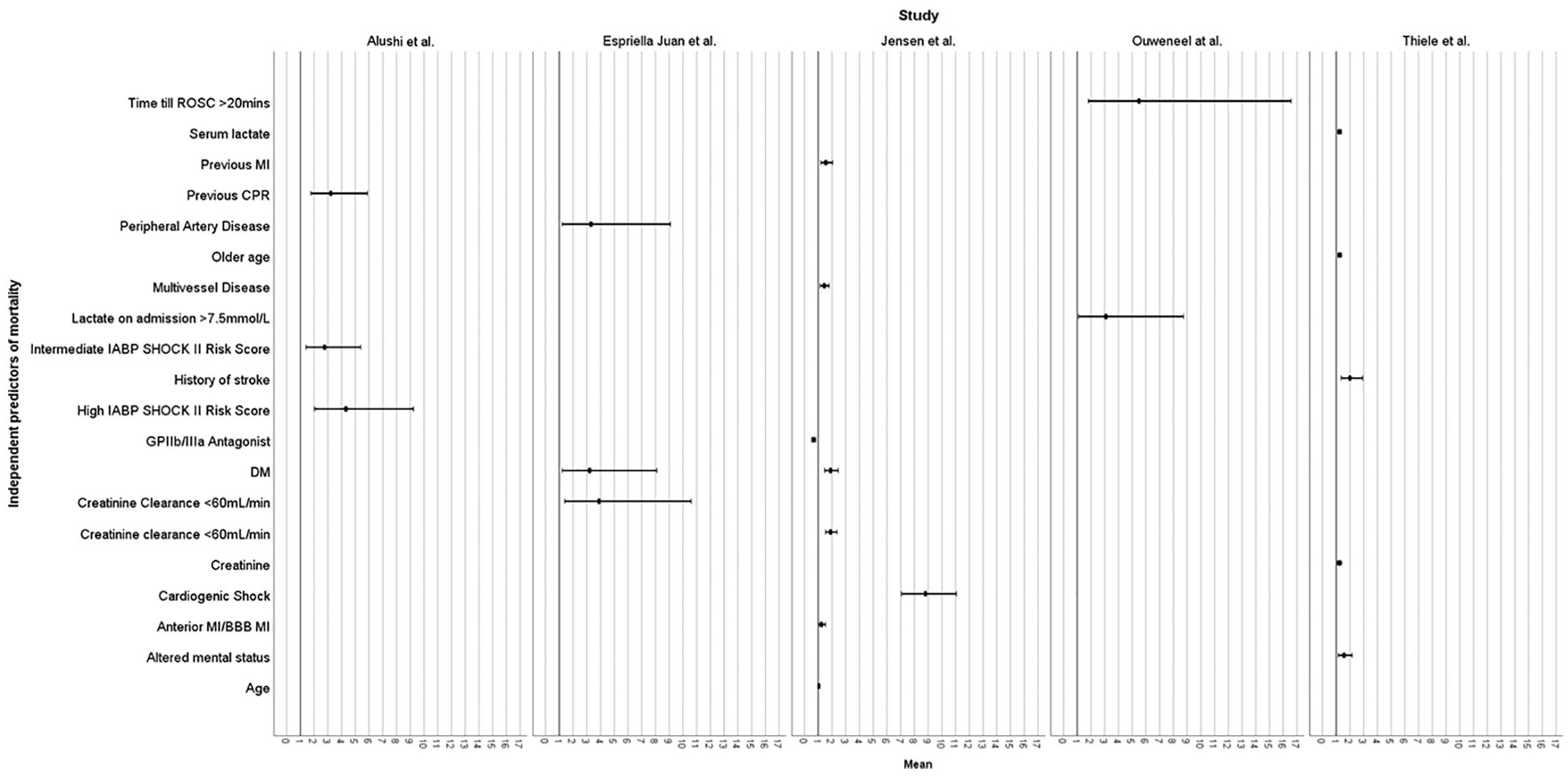
Box plot summary of independent predictors of mortality.
Discussion
This critical appraisal of the literature aimed to compare the primary outcome of short-term mortality associated with IABP versus Impella 2.5/CP© support as well as other secondary end points of cardiovascular mortality and haemodynamic benefit in AMI-CS patients undergoing emergency revascularisation. From the studies collated in this review it is apparent the evidence is not definitive.
The literature is limited and RCTs investigating this high-risk cohort is greatly lacking. Consequently, weaker studies (retrospective observational cohort studies) form the bulk of the literature investigating this demographic.
The findings of this analysis have revealed mixed results. Twelve observational cohort studies and two RCTs were included in this analysis. Of the nine observational cohort studies investigating IABP versus control, eight found that IABP was not associated with increased mortality28,29,31–33,35,37,39 (a finding also reached by IABP SHOCK II trial 26 ), one claimed statistically significant improvement in outcomes associated with IABP use 31 and one found worse mortality outcomes in the IABP population. 30 The observational study without a comparison intervention group also quoted high mortality rates (64%) in their IABP-CS population. 34 One RCT, 27 and two observational studies38,39 failed to establish significant survival benefit associated with percutaneous Impella© use when compared to IABP. One study found superior survival outcomes in patients supported by Impella© when compared to IABP. 36 One study compared Impella© to a control group, however this concluded comparable results in 30-day mortality between groups. 39
These inconclusive findings are consistent with the wider literature. The findings from the IABP SHOCK II trial included in this analysis are inconsistent with the historical BCIS-I trial as this found improved patient outcomes associated with IABP when compared to control groups. 41 It should however be noted that like many studies investigating this cohort, the BCIS-I trial was underpowered to draw statistical significance from the findings. Presently, only two complete RCTs have been conducted directly comparing outcomes in AM-CS undergoing revascularisation supported by Impella© versus IABP. The ISAR-SHOCK (2008) 8 and Impress in Severe Shock trials (2017) 27 which found comparable 30-day mortality rates between Impella© and IABP groups (46% in Impella© and IABP groups in ISAR-SHOCK and 50% in IABP versus 46% in Impella CP© group in IMPRESS in Severe Shock). Again, both studies were under powered – a recurrent limitation of other studies of this nature (PROTECT II 42 and IMPRESS in STEMI 43 ) resulting in premature termination. Thiele et al. conducted a meta-analysis of randomised controlled trials comparing IABP to pMCS which encompassed other devices out with the scope of this analysis such as TandemHeart©. Only four RCTs were included in this analysis further highlighting the difficulty of conducting RCTs investigating pMCS devices. Meta-analysis found neutral effects associated with the IABP across each study, with no survival advantage associated with IABP when compared to other pMCS including percutaneous Impella (2.5/CP)©. 44
Despite the randomised design it is difficult to draw meaningful conclusions from the results. There lacks a consensus on patient characteristics/criteria indicating the use of either the IABP or Impella©. Consequently, there will be a tendency towards selection bias due to the subjectivity of patient characteristics considered eligible for device implantation often left at the discretion of the physician. Furthermore, in such a high-risk group of patients, total adherence to assigned intervention or control arms is nearly impossible, therefore introducing a risk for performance bias due to cross over between devices or intervention effect confounded by delays in deployment of the device. This coincides with the ethical concerns surrounding conducting trials in a largely unstable population. The nature of the intervention prevents blinding of both the operator and the patient, again increasing susceptibility to selection bias. As a result, this presents many advantages in conducting retrospective observational cohort studies.
Twelve retrospective observational cohort studies were included in this analysis. Whilst this avoids ethical limitations posed by RCTs, this analysis identified that the nature of study design introduces bias from multiple domains. Only four studies used predefined inclusion and exclusion criteria, making it difficult to ascertain if appropriate statistical analysis has been undertaken to mitigate confounding bias. Total enumerative sampling was used in four of the studies30,33–35 to recruit a cohort more representative of the total population and avoid the bias associated with convenience sampling. 45 As a result, greater statistical power can be achieved, a limitation less easily attenuated in the context of RCTs. Using retrospective data carries its own risks of chronological bias as well difficulties in data acquisition leaving some confounding variables un-accounted for.
ESC Guidelines no longer recommend IABP for use (Class III recommendation), and other devices such as the Impella© have been assigned a Class IIb recommendation for ‘may be considered’ in the context of myocardial revascularisation in CS. 10 The findings from this analysis support this current guidance. Most of the studies found, that whilst IABP use is not associated with worse mortality outcomes when compared with control, the results lack statistical significance to attribute a survival benefit. In addition, one study claimed worse outcomes in the IABP group. 30 There is limited data exploring the survival benefit of Impella© use and IABP perhaps due to its limited use and greater familiarity amongst physicians in using the IABP given the Impella© was only approved for EU use in 2004. 46 However, this analysis captured four studies which compared Impella© to IABP,27,36,38,39 one of which also compared Impella© to control groups. 39 A significant improvement associated with the Impella© over the IABP was not observed, although potential reasons for ESC preference in its use can be seen when analysing the secondary end points.
Two of the three studies included in this analysis found statistically significant improvement in LVEF. One quoted an improvement of 20% in the Impella© group compared to 10% in the IABP group, 36 and another found that despite poorer LVEF at baseline in the Impella© group, the degree of improvement was significantly greater than in the IABP group. 38 This has been demonstrated in a model controlled mock circulation to simulate CS with the Impella© being significantly more effective in unloading the left ventricle. 5 Similar results have been achieved in porcine studies with significant improvement in haemodynamic support found in the Impella 2.5© group. 4 Notably, the first RCT conducted in this population also found superior haemodynamic benefits in the Impella© group, as reflected by a greater improvement in cardiac index (CI) (Change in CI Impella© group = 0.49 (0.46 L/minute/m2 in Impella© versus 0.11 ± 0.31 L/minute/m2 (p = 0.02) in the IABP group). 8 However, this was not recreated in the IMPRESS in Severe Shock Trial which showed no overall difference between IABP and Impella© groups. 27 Despite this, it may be inferred that the Impella offers superior haemodynamic support when compared to the IABP which has important prognostic implications. LVEF may be interpreted as a proxy for myocardial recovery. LV unloading devices such as the Impella© allow for the acute recovery of salvageable ischaemic myocardium and some LV function 47 through increasing microvascular perfusion of the infarcted myocardium. 48 Restoration of LVEF following AMI has downstream effects in increasing end organ perfusion and established reductions in readmission and 1-year mortality. 49
As previously stated, current guidelines lack explicit criteria for pMCS device implantation. NICE Guidelines 46 currently recommend Impella 2.5/CP© for use in supporting high risk PCI which they have defined as multi vessel disease, left main stem lesion, poor LVEF and refractory ischaemia based on findings by Jones et al. 50 These guidelines are consistent with the findings from this analysis. When taking into consideration the collated baseline patient characteristics, it was determined that IABP use was more commonly seen in patients with left main stem or left anterior descending lesions across three studies.28,31,35 Furthermore, in one of the studies, Impella© use was associated with those categorised as undergoing high-risk PCI. 36 The left main and its branch the left anterior descending artery supply a significant myocardial territory and consequently carry significant mortality risk if affected 51 and yet the mortality between groups has no statistically significant difference. This may imply an indirect benefit in survival associated with these devices when compared to control.28,36
Overall, study populations between intervention groups were balanced. Persistent statistically significant differences in baseline patient characteristics observed were age, male gender,31,33 hypertension,30,35,36,39 smoking history,30,36,38 hypercholesterolaemia30,34,36 and previous CABG.30,34 No extrapolatable associations between characteristics and device implantation were found. Noteworthy correlations can be seen in the studies conducted by Schrage et al. and Alushi et al.; patients supported by IABP had more cardiovascular risk factors such as current smoking, 38 hypertension, hypercholesterolaemia and previous MI 39 when compared to the Impella© group. The significance of which is unclear as these factors have not been proven to be associated with increased risk of mortality in AMI-CS supported by left ventricular assist devices. 52
When considering the baseline patient characteristics across the studies there are consistencies in characteristics found to be independent predictors of mortality. This data has been presented as forest plots with the statistically significant (p < 0.05) characteristics included (Figure 3). Therefore, the following characteristics that should be included in baseline patient characteristics measurements to allow for the identification of confounders for use in multivariate analysis and for establishing independent risk factors of mortality are age, creatinine clearance <60 mL/minute, presence of bundle branch block, cardiogenic shock and diabetes mellitus. When compared to a recent review 53 and meta-analysis 54 investigating predictors of mortality in AMI-CS patients supported by IABP undergoing emergency revascularisation, an established predictor of mortality; timing of device implantation has failed to be recorded across all studies. Despite this, the consensus amongst the studies that measured timing of implantation included in this analysis is that delayed insertion of the IABP 26,30 or post PCI implantation of IABP/Impella© was not associated with increased mortality risk 27 and therefore should also be measured in future studies.
Limitations
This review was conducted through a systematic search of the available literature across four different databases. The searches have been clearly outlined for reproducibility and the inclusion and exclusion criteria have been defined. Despite these measures, admittedly some significant studies may have been inadvertently excluded from our analysis due to failure to be captured by our initial search. To mitigate this risk, an initial scope analysis of the literature, recent systematic reviews and meta-analysis was performed to establish landmark trials for inclusion in the overall analysis. This led to several trials of search combinations until confidence was reached that the final search captured the most relevant studies available in the literature.
The primary focus for this analysis was to compare short term mortality in AMI-CS populations mechanically supported by the IABP versus percutaneous Impella© devices to avoid problems of attrition bias in observational studies where data may be missing. Therefore, we cannot comment on the longer-term outcomes in these populations including mortality after 30-days, reinfarction rates or re-admission rates and degree of morbidity.
Very few randomised controlled trials have been performed in this cohort therefore, most of the studies included are retrospective observational cohort studies. This greatly limits the conclusions that can be reliably drawn from analysis of the studies as they are prone to significant chronological, selection and confounding bias. Statistical analytical techniques to account for these biases have been performed in some of the studies but this will never fully recreate randomisation. Furthermore, it is difficult to determine the degree with which confounding has been accounted for as certain patient characteristics could not be obtained from database or registry data stores.28,32,33,37 Due to the retrospective design of these studies it is difficult to ascertain the quality of data despite some data quality assurance measures being in place, 32 especially for the single centre studies. Thus, creating an issue of over reporting and mistakenly correcting for confounders or under reporting caused by hidden confounding variables.32,33
Only two RCTs have been included in this analysis. The nature of the intervention prevents masking of the patient and operator. The population investigated in this analysis (AMI-CS) represents an extremely unstable cohort, as a result cross over between groups is inevitable as deteriorating patients will be upgraded to a pMCS device from medical management or to another device introducing performance bias. The absence of RCTs in the literature highlights the difficulty in performing these studies in this group of patients.
Conclusion
Whilst there does not appear to be a short-term benefit in survival outcomes seen between IABP and Impella© intervention groups when compared to each other and to control groups (non-surgical management), it appears that Impella© offers superior haemodynamic support.
This systematic review highlights the need for further RCTs in the context of pMCS in AMI-CS and PCI. Furthermore, consistent independent predictors of mortality have been identified for inclusion in future studies. It should be noted that the strength of the conclusions reached in this analysis are limited and despite validating ESC guidelines on device recommendation, further research is needed to determine the absolute prognostic benefit of Impella© in a population with such a high mortality.
Supplemental Material
sj-doc-1-prf-10.1177_02676591211037026 – Supplemental material for Intra-aortic balloon pump versus percutaneous Impella© in emergency revascularisation for myocardial infarction and cardiogenic shock: systematic review
Supplemental material, sj-doc-1-prf-10.1177_02676591211037026 for Intra-aortic balloon pump versus percutaneous Impella© in emergency revascularisation for myocardial infarction and cardiogenic shock: systematic review by Kristina Frain and Paul Rees in Perfusion
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
