Abstract
71.759 surgical procedures were performed in 2019 with the aid of cardiopulmonary bypass in Germany. To adjust the patient’s body temperature on extracorporeal circulation, the application of a heater-cooler unit (HCU) is mandatory. However, in case of insufficient sanitisation of HCU, life-threatening infections can be transmitted by the device to the patients, including Legionella bacteria, Mycobacterium chimaera, Pseudomonas aeruginosa. To avoid disease transmission, as a requirement for safe medical practice established by regulatory authorities, HCUs must be regularly disinfected by hazardous chemicals posing a danger for both handling humans and the environment. Therefore, to comply with regulations, HCU manufacturers have introduced both timely and financially extensive sanitisation procedures. Our paper describes a novel, effective and easy to handle disinfection method for the above problematics without utilising hazardous chemicals. The method’s technical principle is electrolysis, resulting in drinking water quality regarding the analysed germs in the worldwide most commonly utilised heater-cooler device. The main aim of the study was to prove the efficacy and reliability of the device cleansing process. Furthermore, the economic impact of the novel method was evaluated. Therefore, we have undertaken 60 microbiological sampling series between December 2019 and November 2020 from a conventional HCU (3T LivaNova, Germany). During the total investigational period, no contamination with Pseudomonas aeruginosa or Legionellae could have been demonstrated in the HCU. The extreme slow-growing nontuberculous M. chimaera was detected only in one sample obtained from diamond electrode cleansed HCU water, and source of contamination was promptly eliminated by a simple technical modification of the device test-site. Additionally, the diamond electrode application is beneficial for eliminating potentially hazardous cleansing material from the process, which may affect otherwise both patients operated on cardiopulmonary bypass and the perfusionists.
Keywords
Introduction
There were over 100 heart valve endocarditis cases registered since 2013, caused by Mycobacterium chimaera (M. chimaera) in Europe and North-America. 1 M. chimaera was first detected in 2004, a bacterium that belongs to nontuberculous mycobacteria in the M. avium complex. 2 This germ is widely spread, occurs even in tap-water, and may result in pneumonia in immunocompromised via inhalatory pathway. Several microbiological studies focused on the potential role of HCUs in an infective process associated with cardiosurgical procedures utilising extracorporeal circulation (ECC). However, the application of HCUs is crucial in the thermoregulation of patients on ECC.
Microbiological samples derived from technical components of HCU circuits utilising filtered tap-water have demonstrated common contamination with M. chimaera. Due to technical reasons and additional aerosol production by HCU, the M. chimaera contamination can be traced back to the operative field. 3
In two patients carrying artificial heart valves, the bacterium has led to prosthetic valve endocarditis, detected at the University Hospital Zürich in 2011. Both patients have developed an infection long after the primary surgical procedure and deceased due to a septic condition. The tardive disease course is resulting from a prolonged growth rate of M. chimaera. 4 However, it is virtually impossible that the bacteria have directly entered the patients’ bloodstream; as there is no direct communication between the circulating blood and the HCU cooling water within the ECC oxygenator.
Equally, problematical are Legionella bacteria and Pseudomonas aeruginosa. Legionella bacteria are a genus of pathogenic, aquaphilic, non-sporozoite Gram-negative rods, their mobility is maintained by ciliary movement. 5
A Gram-negative and oxidase-positive rod, Pseudomonas aeruginosa, a member of Pseudomonadaceae family, is present in water and soil worldwide and causes a blue-greenish discolouration of the culture medium accompanied by a concomitant sweet-aromatic smell. The bacterium is a well-known pathogen leading to nosocomial infections, usually resulting in invasive, purulent infections with a potential expansion to generalised septicaemia.6,7
The current disinfection guidelines provided by LivaNova PLC (London, United Kingdom), applicable to the worldwide most widely utilised HCU, namely LivaNova 3T, require daily check-up of hydrogen-peroxide (H2O2) levels within the water container, a weekly water replacement within the HCU circuit, and time-consuming disinfection of water circuit by Puristeril® 340 (Fresenius Medical Care AG & Co, Bad Homburg, Germany) containing peracetate (C2H4O3), every 14 days.
Even in healthcare facilities, including the University Medical Center Regensburg (UMCR), with a spatial separation of HCUs from the operative theatres, allowing no aerogenic transmission of M. chimaera, the above-described disinfection guidelines from LivaNova must be strictly followed. Due to the application of hazardous cleansing materials in the process, a possible risk for personal health – even in this setting – cannot be excluded entirely. Furthermore, the procedure’s additional financial burden also reinforces the necessity of innovative approaches to optimise the process. An emerging promising alternative is represented by electrolytic disinfection undertaken with the aid of diamond electrodes. The investigations’ main goal was to reinforce the reliability and safety of the alternative cleansing process, with the additional economic benefits provided by the method.
Material and methods
In a period from December 2019 to November 2020, 60 extensive microbiological investigation series were conducted on a conventional HCU (LivaNova 3T), to evaluate the efficacy of a Water Circuit Disinfection Device (WCDD) integrated diamond electrode in the HCU water cleansing procedure. The initial twice-a-week sampling was reduced to once a week at a later stage of the study. As there was no initial data available regarding compatibility and eventual side-effects of the novel disinfection method on both LivaNova 3T device and plastic components of the oxygenator, the study was conducted on an isolated LivaNova 3T device without any patient contact. To assess the novel cleansing procedure’s material compatibility with the heat exchanger situated within the ECC oxygenator, scanning electron microscopic (SEM) imaging were undertaken of heat exchanger surfaces of two new, unused oxygenators exposed to diamond electrode disinfected water. These two oxygenators were standard cardiosurgical items of different manufactures.
Besides evaluating efficacy of the novel disinfection method in contrast to a LivaNova guideline-driven cleansing, economic aspects of the procedures were also reviewed.
The LivaNova 3T hypothermia unit has an net weight of 110 kg and is filled with a total of 12 L of filtered water. The circulating water for LivaNova 3T fluid replacement was initially filtered through a 0.2 µm pore-diameter bacterial sieve, and then exposed to electrolysis generated by a WCDD (dimensions: 30 × 41 × 20 cm). The WCDD was attached to the HCU, tubing connections to both patient- and cardioplegia circuits were established as depicted on Figure 1.

View of WCDD functional unit on top of LivaNova 3T (left) and tubing connections of both systems during disinfection process (right).
In this process, the connections between the 3T unit and WCDD are established and remain permanent. During the surgical procedures, two lines are disconnected from the WCDD and immediately attached to the oxygenator. Thus, there is no direct connection exists between the 3T device and WCDD during the procedure; just the previously purified water passes through the 3T unit and the connected oxygenator of the heart-lung machine. Therefore, there is no interference present between the water flow and the heater-cooler function of the 3T hypothermia unit.
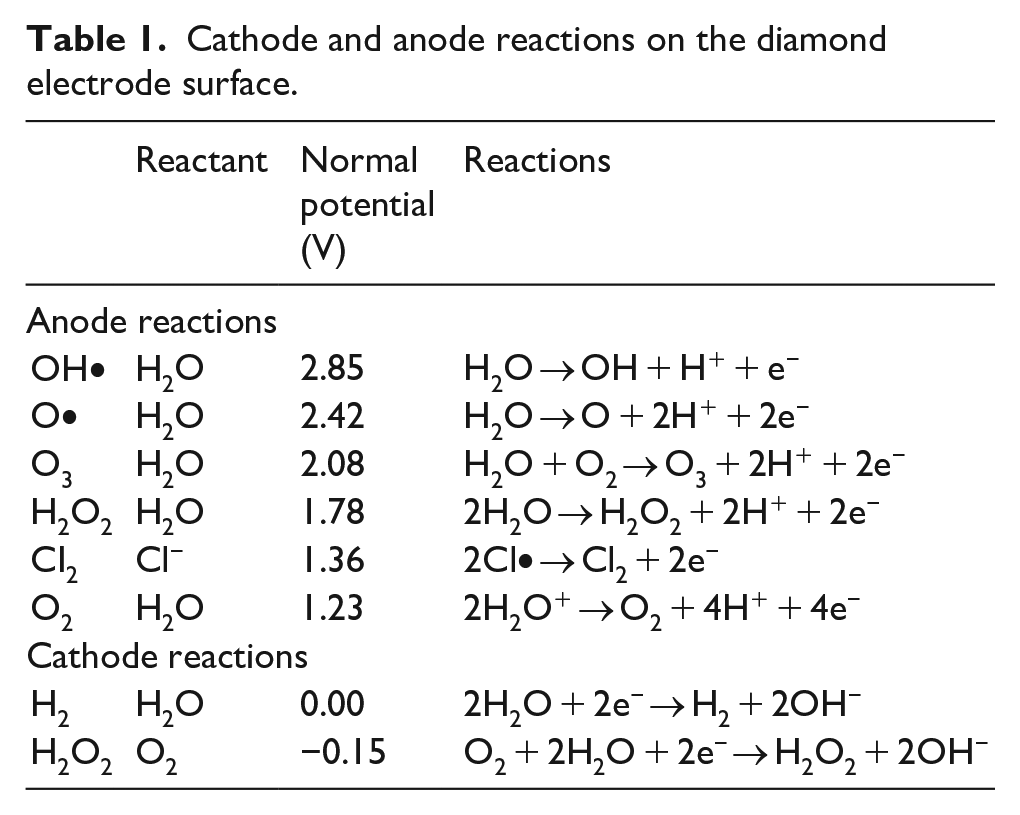
The WCDD incorporates boron endowed, electrically conductive diamonds (Pro Aqua Diamantelektroden Produktion Ltd, Niklasdorf, Austria) that produce as a result of high separation currents hydroxyl (OH•)−, oxygen free radicals (O•) and ozone (O3) in Table 1 detailed concentrations.
Cathode and anode reactions on the diamond electrode surface.
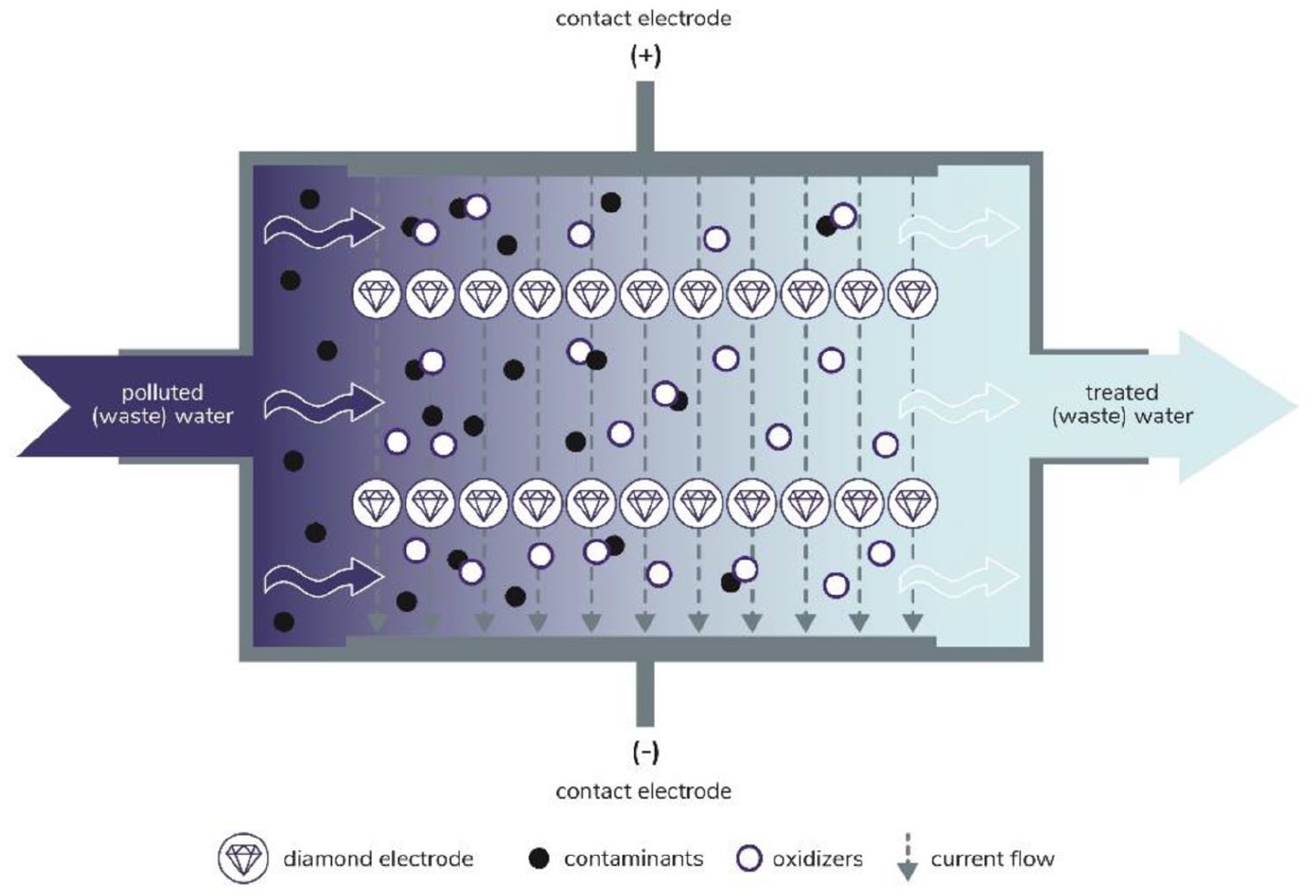
The diamond electrode represents a flow cell supplied by 30 V direct current that treats soiled circulating HCU water via electrolysis (Figure 2). During the procedure, electrolysis generates a bactericide solution in a concentration of 1 mg/L, and soiled organic water particles are going to be broken down into water and carbon-dioxide inside the flow cell. The generated oxidative constituents containing cleansed water perfuses the water circulation of 3T HCU so that a biofilm building capability of microbes both within the HCU and pipelines is effectively eliminated. The WCDD has standard cleaning and deep cleaning settings. Our study was conducted applying the deep cleaning mode exclusively.
Flow cell containing integrated diamond electrodes.
Biofilms facilitate the creation of increased bacterial resistance against both antibiotics and disinfectants regarding nontuberculous mycobacteria on the surface of water conduction components in medical devices. 8 Mycobacteria that both commonly grow in water and soil have an increased resistance capability against acids and lye due to a wax component of their cell wall in comparison to other bacterium species. 9 Standard microbiological detection for nontuberculous mycobacteria consists of a positive culture of the germ, 10 that requires a unique culture medium and environment for multiplication of the microbes. In case of nontuberculous mycobacteria, a unique culture medium is represented by the Löwenstein-Jensen solution.
All microbiological samples provided for further investigations to the Institute of Microbiology and Hygienics, UMCR, was obtained from the 3T device’s water outlet, following completion of a WCDD cleansing cycle.
To assess material compatibility of diamond electrode disinfected water with commercially available oxygenator heat exchangers, LivaNova 7 TM (LivaNova PLC, London, United Kingdom) and Quadrox-i Adult (Getinge, Rastatt, Germany) oxygenators were perfused for a consecutive period of 36 h. After the latter perfusion period, the oxygenators were opened in a laboratory environment. The water-conducting elements were removed and prepared for SEM investigations. Identical unused oxygenators were additionally examined under SEM to build a control group.
The tap-water was also analysed for Legionella bacteria and Pseudomonas aeruginosa and nontuberculous mycobacteria.
As there was no initial data available regarding compatibility and eventual side-effects of the novel disinfection method on both LivaNova 3T device and plastic components of oxygenator, the study was conducted on an isolated LivaNova 3T device without any patient contact. Therefore, a Research Ethics Committee approval is waived according to local policy.
Results
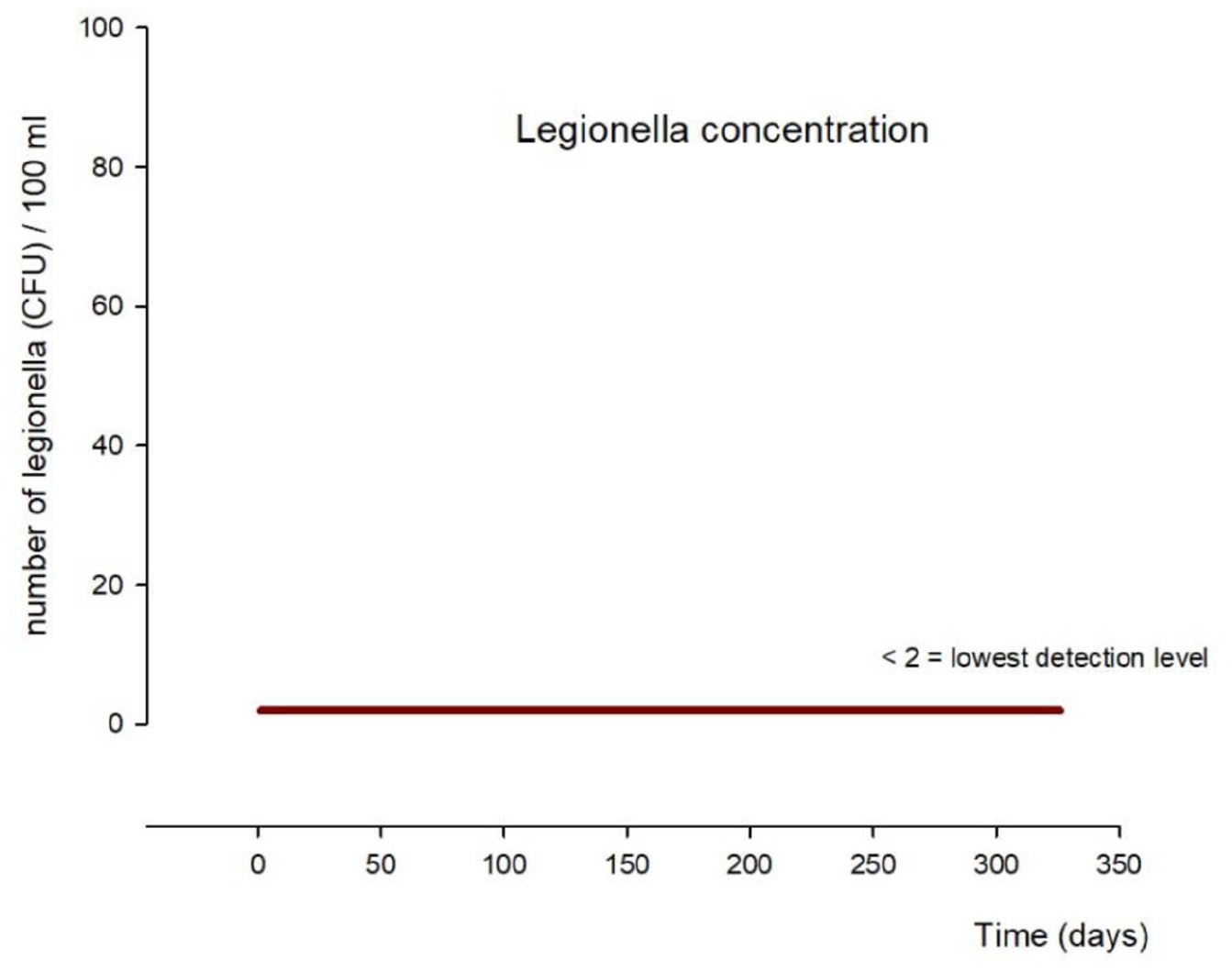
In water samples obtained from 3T HCU, the presence of Legionella bacteria was under the technical detectability of two colony forming units (CFU)/100 mL (Figure 3).
Legionella bacteria concentration along the study course.
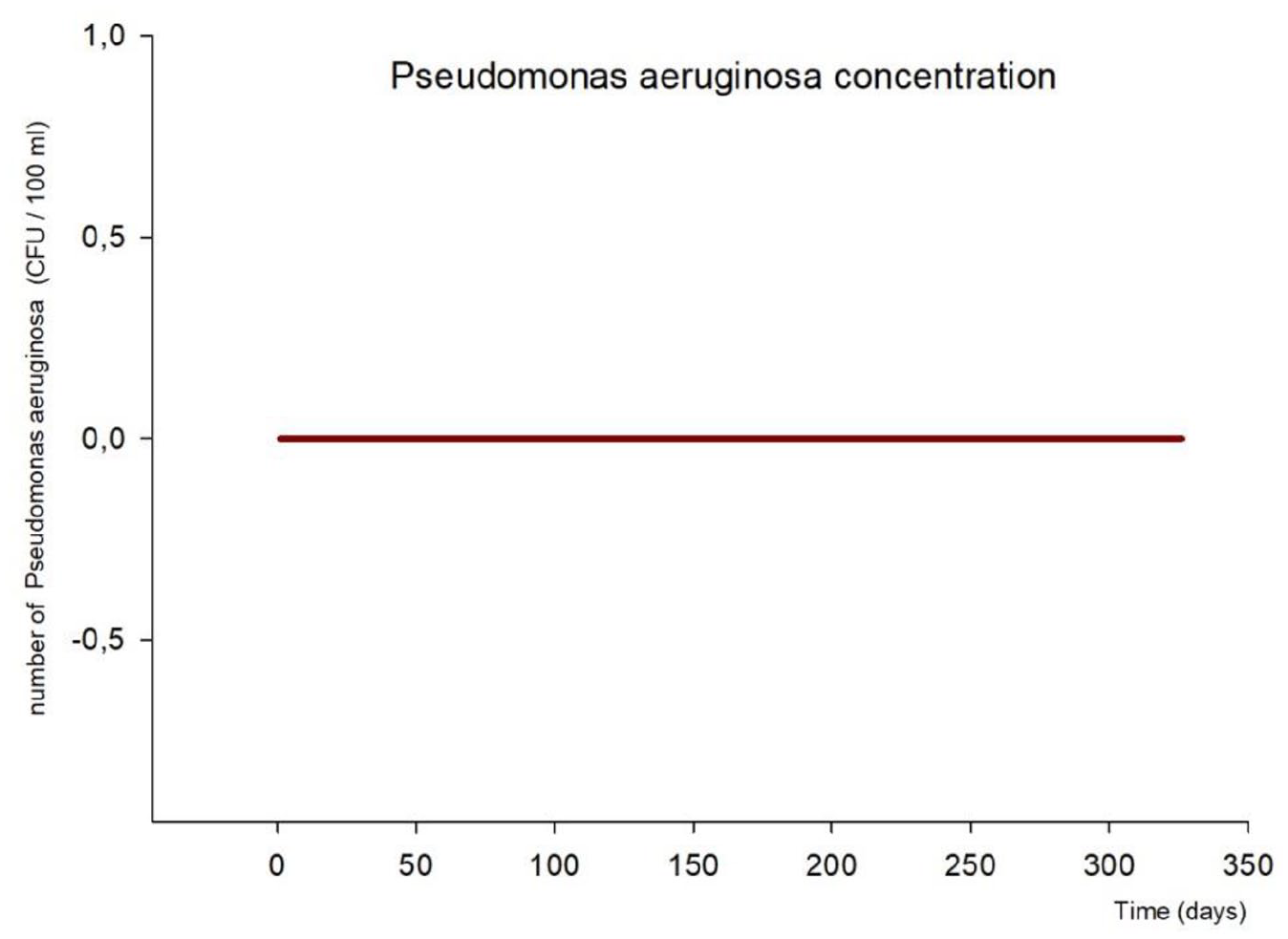
There was no Pseudomonas aeruginosa contamination of LivaNova 3T HCU water samples detected during the entire study period (Figure 4).
Pseudomonas concentration along the study course.
The extreme slow-growing nontuberculous M. chimaera4,11 was detected in one sample obtained from diamond electrode cleaned water of 3T HCU on day 53, in test-row number 15 (Figure 5). However, this microbiological positivity was present only in Mycobacteria Growth Indicator Tube fluid culture, not standard solid mediums. Therefore, the concentration of M. chimaera was deemed insignificant, since detailed quantification of the sample was not achievable. Furthermore, by exchanging the plastic tap of sampling outlet for a sterile poly-vinyl-chloride (PVC) tubing (Figure 6) that was then replaced weekly, no more M. chimaera positivity could have been detected.
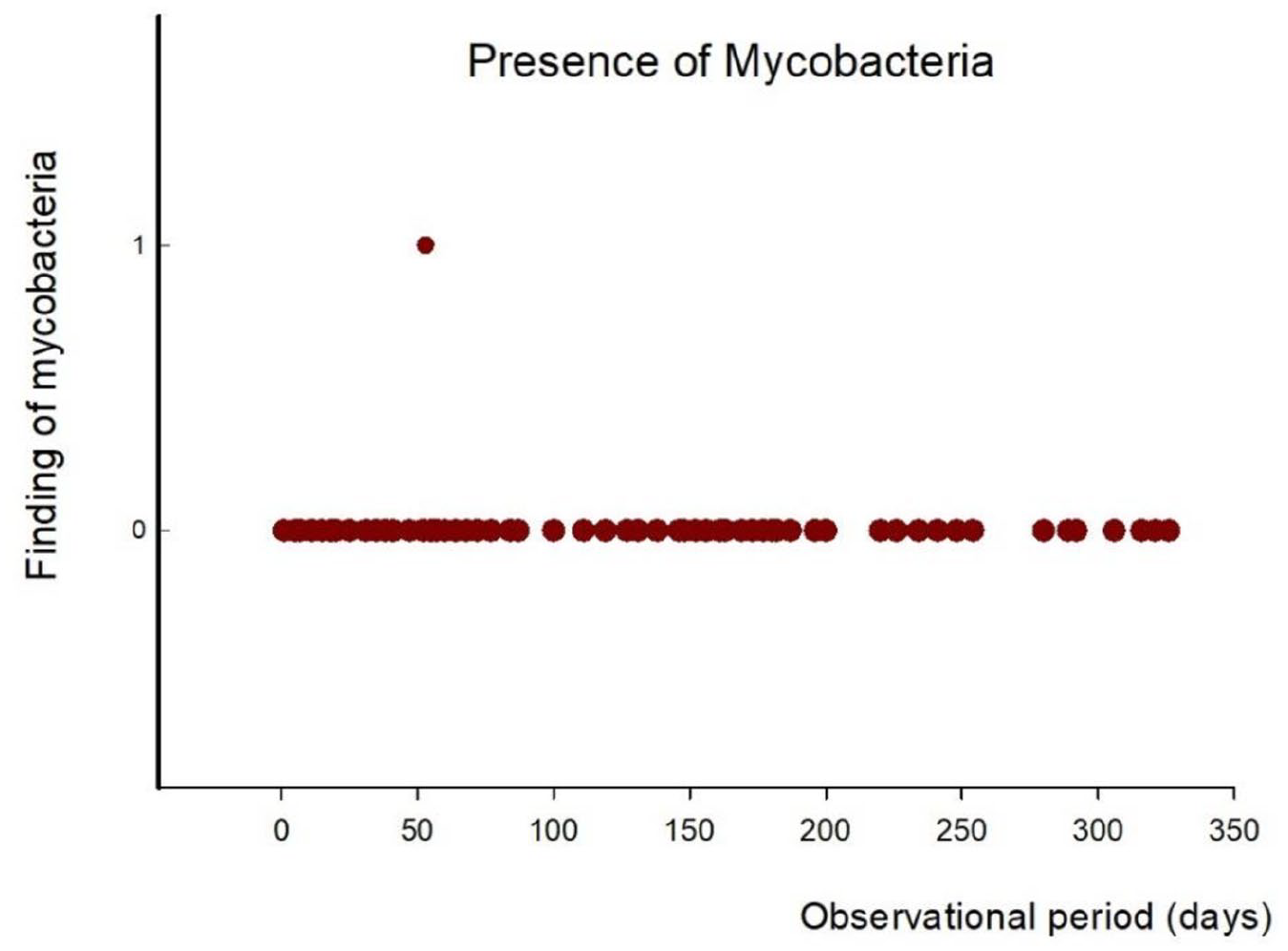
M. chimaera colonisation along the study course. Zero indicates no evidence of M. chimaera, one reveals colonisation with the germ.

Problem hot-spot sampling tap (left) and optimised sampling access (right).
SEM imaging comparison of oxygenator surfaces which were perfused by disinfected water for 36 h, and non-perfused oxygenator surfaces (control) revealed no effect of treated water on polyurethane surfaces in terms of perforations or micro ruptures (Figure 7). Nevertheless, salt deposits resulting from exposure to disinfection process are visible at a magnification power of 3000. The cleansing process by diamond electrodes requires a mixture of 99.8% water and 0.2% inorganic salts circulated in the LivaNova 3T HCU. These soluble salts have resulted in a fine coating on heat exchanger surfaces throughout a long experimental perfusion exposure. After 11 months of constant application, there were no degradation marks present on any surfaces, including the cooling heat exchanger and the heating coil of the 3T unit, that came into contact with the disinfected water produced by the WCDD (Figure 8).

Water-bearing surface of an unused Quadrox-Oxygenator (left) and after 36-h long contact with WCDD disinfected water in scanning electron microscope (right).

The inspection of internal 3T circuit components revealed no changes on the surfaces that came into contact with the disinfected water produced by the WCDD.
In an economic point of view, the following must be considered comparing conventional, by LivaNova recommended mandatory disinfection guidelines, requiring H2O2 concentration analysis, water replacement and disinfection by peracetic acid (C2H4O3), and the novel diamond electrode approach. Utilising the conventional method, the same cleansing process must be undertaken every 30 days in a calculated period of 6.7 working hours (400 min), see detailed in Table 2. In contrast, the WCDD cleansing course requires only 4.0 working hours (240 min) per HCU device, applying the diamond electrode. Furthermore, utilising the WCDD method, other, cleansing process non-related clinical activities can be performed even during the automatic purification activity.
Comparison of time demand regarding the LivaNova cleansing procedure and the WCDD.

Discussion
Tap water contamination in HCU for open-heart surgery can pose a considerable risk to patients; data in current medical literature has identified several microorganisms such as Legionella bacteria, Pseudomonas aeruginosa or Mycobacterium chimaera involved in the process. The main problem is the appropriate disinfection of biofilms in the mandatory heater-cooler units.
Biofilms facilitate adhesion and multiplication of bacteria such as Legionellae, which can hazardously soil the water content of HCU even in a small concentration. 12 Therefore, the concentration of Legionella bacteria cannot exceed 100 CFU/100 mL in drinking water as per German water purity regulations. 13 In all study samples, the Legionella concentration was situated below the 2 CFU/100 mL minimum measurement threshold. Hence, the WCDD disinfected water shows-up drinking water quality regarding Legionellae burden.
Pseudomonadaceae, especially Pseudomonas aeruginosa, is the most common pathogen causing nosocomial airway, urinary tract and wound infections. 14 This germ can lead to sepsis, and as a chronic infection may result in lung fibrosis in patients suffering from mucoviscidosis. The latter chronic condition ends-up in a fatal respiratory failure, the most common cause of mortality in mucoviscidosis. 15 Additionally, Pseudomonas is known to have the ability inducing a biofilm generation. 12 In view of the microbe’s capabilities as mentioned above, the German water purity regulations establish an acceptable limit of 0/250 mL of any species of Pseudomonadaceae genus in drinking water.13,16 Outcomes of WCDD disinfected 3T HCU water completely fulfils the tap water criteria.
Another biofilm-producing germ, M. chimaera has long incubation periods, even up to 5 years. Therefore, according to Swiss Medical Journal recommendations, unclear infection signs following heart surgery could be considered a nosocomial infection resulting from slow-growing, nontuberculous mycobacterial burden. 11
Our study conducted at UMCR revealed mycobacterial presence only in one culture obtained from WCDD treated water for HCU, which resulted from a sampling plastic tap built in the system that was later replaced by a sterile PVC tube connector. As a feature of plastic tap construction, the distal portion of the locking mechanism could have never been reached by the disinfectant water solution, so colonisation by mycobacteria was feasible. Following replacement by a sterile PVC sampling tube, mycobacteria were no longer detected.
At an extremely long, under normal operative circumstances not occurring 36 h perfusion period with WCDD cleansed water, there were no traces of any physical changes detected in the heat exchangers of investigated Getinge and LivaNova oxygenators. This fact well supports the compatibility of the cleansing method with extracorporeal circulation elements.
As the water cleansing technology applying diamond electrodes of WCDD is an entirely novel technical approach, all patient-connected HCU systems were disinfected as per manufacturer standards for safety considerations.
Due to their total weight of 122 kg, the LivaNova 3T HCU units cannot be routinely transported for disinfection from the operating theatre area, for example, to a remote disinfection department. Therefore, cleansing in the operating area by perfusionists trained in the cleaning procedure is required. This manufacturer’s water cleansing process consists of daily H2O2 concentration measurements, weekly system-water replacement and a time-consuming water circuit disinfection procedure every 2 weeks. Just the disinfection procedure itself for a single 3T HCU requires 1.5 h for an experienced perfusionist, and no further tasks can be undertaken during this period (Table 2). Supposed water replacements in a length of 30 min and daily H2O2 level check-up durations of approximately 5 min are added-on; in that case, a monthly cleansing requirement for a single 3T unit necessitates a 6.7 h working time investment. There are four HCUs in UMCR cardiosurgical services enrolled, the projected working time demand for water cleansing purposes on all devices measures nearly 27 h for a trained perfusionist per month.
In contrast, daily application of WCDD on LivaNova 3T HCU requires only 7 min for a short cleansing and 25 min for a deep cleansing procedure commencing on the first working day following a weekend. For WCDD cleansing purposes, the 3T HCU unit does not leave the operative theatre, so that during a cleansing procedure other tasks, that is, preparations for ECC runs can be performed. In addition to significant time saving, direct costs for Puristeril® 340 cleansing solution and H2O2 can also be eliminated. However, regarding the WCDD process, 3 year lifetime of diamond electrodes, and water level top-up costs, including inorganic salt constituents, must be considered as a result of sampling losses. Safe disinfection of 3T HCU without our unique device can only be performed according to manufacture guidelines, because of health and safety requirements. Puristeril® 340 contains peracetate; a clear, foul-smelling solution that poses a definite risk of health for HCU cleansing professionals. According to Directive 1272/2008/EC, 17 peracetate is categorised as a corrosive, irritative, inflammable and environmentally hazardous substance. Therefore, safe handling of dangerous materials requires suitable personal protective equipment, including special clothing, face shield or goggles. On the contrary, with application of WCDD, personal and environmental health and safety risks are effectively eliminated.
According to manufacturer recommendations, disinfection method of 3T devices in a space without active ventilation, as present in most operative theatres, shall be replaced only by a safe procedure from occupational health and safety point of view.
A key advantage of the WCDD for clinical routine is that the disinfection procedure can be interrupted at any point. Thus, the 3T device is immediately available for use again in the event of a cardiosurgical emergency case. The emergency conversion is not possible with LivaNova’s 3T cleaning procedure and a replacement device must be set-up. It must be recognised that our study was conducted on a conventional 3T heater-cooler that underwent complete disinfection by LivaNova prior to the investigations. Furthermore, it is not entirely clear yet and should be evaluated by further studies, in which extent are by diamond electrodes produced free radicals capable of eliminating massive, pre-existing biofilms within heater-cooler systems. Additionally, further investigations shall clarify if equivalent outcomes are achievable just by a standard cleansing process, as with the deep cleaning mode. Our study analysed the efficacy of the deep cleansing mode exclusively.
In summary, the novel circulating water disinfection method with the aid of WCDD integrated diamond electrodes for cleansing purposes of worldwide most utilised HCU in cardiac surgery, LivaNova 3T is a safe, effective and economical procedure. The electrolysis produces drinking water quality from a microbiological viewpoint for HCU, proven by the investigated bacterial burden obtained from water samples. This fact definitely reduces the risk of procedural infection in cardiosurgical patients and eliminates health and safety issues for perfusionists involved in HCU cleansing procedures.
According to a report issued by Federal Authority for Pharmaceutical and Medical Products, Germany (Bfarm), HCUs from other manufacturers show-up also a potential for bacterial contamination of contained circulating water in their systems. 18 Since a cleansing procedure of these devices poses a similar burden for utilising institutions as LivaNova 3T HCU, an adaptation of the purification process by diamond electrodes is under current elaboration.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
