Abstract
Background:
Studies reporting long-term outcomes of venoarterial extracorporeal membrane oxygenation–treated coronary artery bypass grafting patients are scarce. The objective of this study was to examine the survival outcomes and identify mortality risk factors for coronary artery bypass grafting patients who received venoarterial extracorporeal membrane oxygenation for postcardiotomy cardiogenic shock.
Methods:
Data from 121 consecutive venoarterial extracorporeal membrane oxygenation–treated coronary artery bypass grafting patients at the Beijing Anzhen Hospital between January 2012 and December 2016 were analyzed. Multivariable Cox regression modeling was used to identify factors independently associated with 36-month mortality.
Results:
Seventy-seven patients (64%) could be weaned from venoarterial extracorporeal membrane oxygenation, 56 patients (46%) survived to hospital discharge, and 41 patients (34%) survived to 36 months. Older age (hazard ratio, 1.06; 95% confidence interval [CI], 1.03-1.10; p < 0.001), left main coronary artery disease (hazard ratio, 1.64; 95% CI, 1.04-2.59; p < 0.001), and vasoactive inotropic score (hazard ratio, 1.09; 95% CI, 1.02-1.16; p = 0.011) were independent risk factors associated with 36-month mortality. The area under the receiver operating characteristic curve for the logistic regression model, which was constructed with three pre–extracorporeal membrane oxygenation parameters—age ⩾ 60 years, left main coronary artery disease, and vasoactive inotropic score > 60—was 0.87 (95% CI, 0.81-0.94). Age and left main coronary artery disease significantly increased the discriminatory performance of Sepsis-related Organ Failure Assessment score (0.79 vs. 0.91, p = 0.025).
Conclusions:
Older age, left main coronary artery disease, and vasoactive inotropic score were associated with 36-month mortality in coronary artery bypass grafting patients who received venoarterial extracorporeal membrane oxygenation.
Keywords
Introduction
Postcardiotomy cardiogenic shock (PCS) is a life-threatening condition characterized by severe myocardial contractile impairment and reduced organ perfusion.1–3 In the current era, mortality for coronary artery bypass grafting (CABG) patients complicated by PCS approaches 40%. 4 In patients with PCS refractory to conventional treatment, venoarterial extracorporeal membrane oxygenation (VA-ECMO) has been increasingly used as the first-line mechanical circulatory support to provide temporary circulatory and respiratory support allowing cardiac function recovery or bridging to additional therapeutic alternatives. Despite the increasing use of VA-ECMO in this patient population, this rescue therapy has not been accompanied by improved survival. 5 In addition, this technique requires considerable financial and human resources. 6 Early identification of mortality risk factors and detailed analyses of survivors’ long-term outcomes are needed. However, there are currently few studies reporting death-associated factors and long-term outcomes of VA-ECMO-treated severe post-CABG cardiogenic shock patients. Therefore, this single-center retrospective study was designed to quantify survival outcomes for CABG patients who received ECMO for PCS and to identify the risk factors associated with long-term mortality.
Methods
Patients
With the approval of the institutional ethics committee/review board of the Beijing Anzhen Hospital (2016005X), a retrospective study was performed at a university-affiliated cardiac surgical intensive care unit (ICU) over a 5-year period from January 2012 and December 2016. Informed consent for data analyses was not obtained because this observational study did not modify existing diagnostic or therapeutic strategies. All consecutive post-CABG patients who received VA-ECMO for PCS were included. ECMO was initiated intraoperatively in the operating room for circulatory instability during or immediately after weaning from the cardiopulmonary bypass (CPB) in the primary cardiac procedure. The capability to institute ECMO secondarily in the ICU, for delayed PCS or cardiac arrest, was available. The clinical criteria for PCS included the following: left atrial pressure > 15 mmHg; central venous pressure > 12 mmHg; metabolic acidosis (i.e. pH < 7.3 with serum lactate > 3.0 mmol/L); end-organ hypoperfusion (urine output < 30 mL/h); cardiac index < 2.2 L/min/m2; and systolic blood pressure < 80 mmHg despite adequate filling volumes, use of multiple adrenergic agents (epinephrine > 0.1 µg/kg/min or dobutamine > 10 µg/kg/min, norepinephrine > 0.1 µg/kg/min), or an intra-aortic balloon pump (IABP).
ECMO implantation and management
The details regarding VA-ECMO initiation and management have been described previously. 7 Briefly, all procedures were performed by trained ECMO team members.VA-ECMO support was initiated via peripheral cannulation through the femoral route with the semi-open method, and an additional 6 Fr catheter was systematically inserted distally into the femoral artery to prevent leg ischemia. ECMO blood flow was adjusted on based on clinical assessments (e.g. pre-oxygenator venous oxygen saturation, evidence of hypoperfusion, resolution of hyperlactatemia, normalization of mean arterial pressure). Intravenous unfractionated heparin was given to maintain an activated clotting time of 180–210 s, or an activated partial thromboplastin time of 1.5–2 times normal. ECMO-related complications were carefully monitored. ECMO weaning was performed in patients who fulfilled our published institutional weaning criteria7,8 and passed an ECMO weaning trial consisting in decreasing and clamping ECMO flow. In general, the patient should have a pulsatile arterial waveform for at least 24 hours; be hemodynamically stable, with baseline mean arterial pressure greater than 60 mmHg with no or low doses of catecholamines; should have left ventricular ejection fraction (LVEF) of 35%, and an aortic velocity time integral (VTI) of ⩾ 12 cm; and have recovered from major metabolic disturbances. Weaning was considered unsuccessful if ECMO re-cannulation was required within 2 days of decannulation.
Data collection and outcome variables
The following information was recorded retrospectively: age, sex, body mass index (BMI), comorbid conditions, number of diseased vessels, ST-elevation myocardial infarction (STEMI), left ventricular ejection fraction (LVEF), EuroSCORE, emergency operation, off-pump CABG (OPCABG), indications for ECMO implementation, ECMO combined with IABP, pre-ECMO Sepsis-related Organ Failure Assessment (SOFA) score, 9 pre-ECMO cardiac arrest, vasoactive inotropic score (μg/kg/min, dopamine + dobutamine + 100 × epinephrine + 100 × norepinephrine + 15 × milrinone), 10 and pre-ECMO biochemical parameters.
The primary outcome was survival at 36 months. Secondary outcomes included survival at 24 months, survival at 12 months, survival at month, in-hospital mortality, death on ECMO, ECMO duration, length of ICU stay, length of hospital stay, survival to ECMO weaning, continuous renal replacement therapy (CRRT), stroke, and leg ischemia requiring surgical intervention (fasciotomy or amputation).
Statistical analysis
The characteristics of the patients at baseline were reported as percentages for categorical variables and as medians (with interquartile ranges) for continuous variables, as appropriate. Categorical variables were compared with chi-square or Fisher’s exact tests, and continuous variables were compared with Student’s t test or the Mann–Whitney U test, as appropriate. The cumulative rates of survival after ECMO initiation were analyzed using the Kaplan‒Meier method and inter-group comparisons were performed using the log-rank test. Multivariable Cox proportional hazards regression modeling was used to identify factors independently associated with 36-month survival using backward stepwise selection of variables with univariate p values of less than 0.1. The identified predictors of survival were then included in multivariable Cox logistic regression to predict survival at 36 months. In the multivariable Cox model, age and vasoactive inotropic score were dichotomized into binary variables by assessing the discriminatory ability of clinically relevant univariate “cut-off” values to predict survival at 36 months. Model discrimination and calibration were assessed using the area under the receiver operating characteristics curve (AUROC) and the Hosmer–Lemeshow C-statistic, respectively. Model discrimination of the SOFA score versus the logistic regression model and SOFA score combined with age and left main coronary artery disease were compared using AUROC. Cox proportional hazards regression models were also used to calculate the hazard ratios (HRs) and their 95% confidence intervals (CIs) in the early (within 1 month) and later (1–36 months) follow-up periods. The analyses were adjusted for age, left main coronary artery disease, and vasoactive inotropic score. A subgroup analysis was performed to explore the characteristics and outcomes OPCABG patients and on-pump CABG patients. Values of p less than 0.05 were considered to be statistically significant. All the analyses were performed with STATA /SE 12.0 (StataCorp, College Station, Tx, USA) and SPSS 25.0 (SPSS Inc., Chicago, IL, USA).
Results
Populations
A total of 121 consecutive VA-ECMO-treated CABG patients were included (Figure 1). Baseline characteristics of the patients according to 36-month survival status are presented in the Table 1. The median age of patients was 624 months (range, 55-67 years); 95 were male (79%). The mean BMI was 25 kg/m2 (range, 22-27 kg/m2). Sixty-four patients (53%) underwent off-pump CABG. Thirty-nine patients (32%) were not successfully weaned from CPB requiring transition to ECMO. The indications for ECMO implementation were unsuccessful weaning off CPB (n = 39), delayed cardiogenic shock in ICU (n = 63), and cardiac arrest (n = 19). More than 90% of the patients received additional IABP therapy to ECMO. Median pre-ECMO LVEF was 25% (range, 20-31%), and 65 patients (54%) suffered from pre-ECMO cardiac arrest. There was significant difference between the survival group and the non-survival group in age, hypertension, left main coronary artery disease, unsuccessful weaning off CPB, delayed cardiogenic shock in ICU, SOFA score, vasoactive inotropic score, platelet count, blood lactate, and serum creatinine.
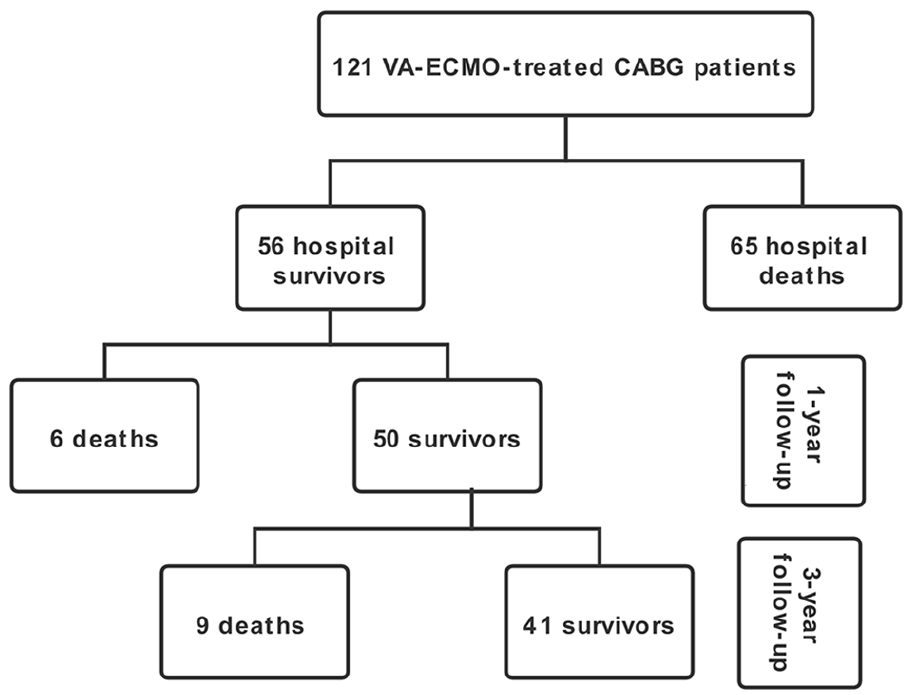
Study flow diagram.
Baseline characteristics of the patients according to 3-year survival status.
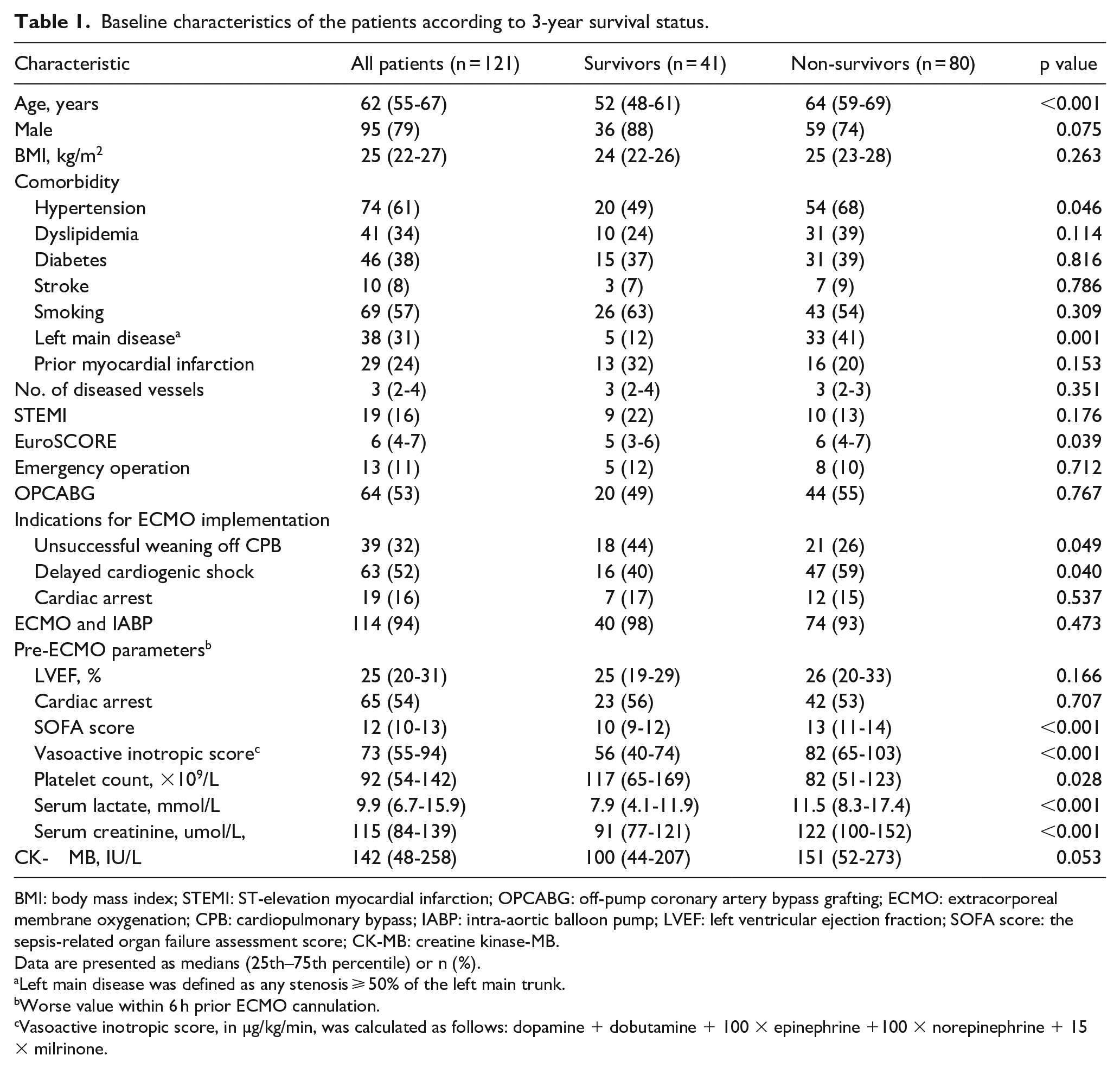
BMI: body mass index; STEMI: ST-elevation myocardial infarction; OPCABG: off-pump coronary artery bypass grafting; ECMO: extracorporeal membrane oxygenation; CPB: cardiopulmonary bypass; IABP: intra-aortic balloon pump; LVEF: left ventricular ejection fraction; SOFA score: the sepsis-related organ failure assessment score; CK-MB: creatine kinase-MB.
Data are presented as medians (25th–75th percentile) or n (%).
Left main disease was defined as any stenosis ⩾ 50% of the left main trunk.
Worse value within 6 h prior ECMO cannulation.
Vasoactive inotropic score, in µg/kg/min, was calculated as follows: dopamine + dobutamine + 100 × epinephrine +100 × norepinephrine + 15 × milrinone.
Patient outcomes
Seventy-seven patients (64%) could be weaned from VA-ECMO (Table 2). The median (IQR) time on VA-ECMO support was 4 (3-5) days. The median (IQR) length of ICU stay and hospital stay duration were 8 (5-12) and 20 (15-29) days, respectively. More patients in the non-survival group required CRRT compared with the survival group (48% vs. 15%; p < 0.001). The incidences of limb ischemia, stroke, and anoxic encephalopathy were similar between the two groups.
Outcomes according to 3-year survival status.
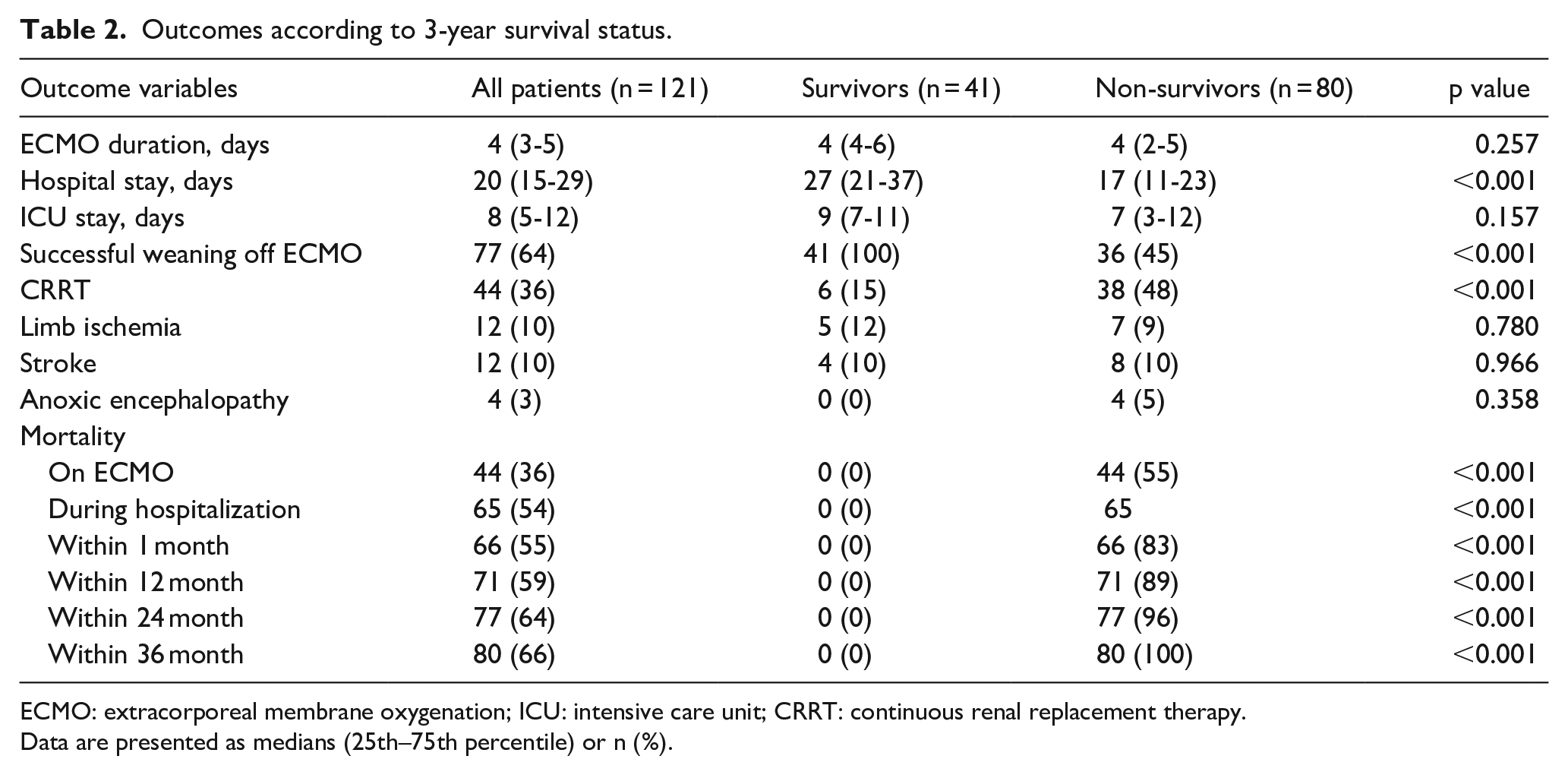
ECMO: extracorporeal membrane oxygenation; ICU: intensive care unit; CRRT: continuous renal replacement therapy.
Data are presented as medians (25th–75th percentile) or n (%).
The overall survival rates were 45%, 41%, 38%, and 34% at 1, 12, 24, and 36 months, respectively (Figure 2). The mortality on ECMO and during hospitalization for all patients was 36% and 54%, respectively (Table 2). Fifty (89%) hospital survivors survived to 12 months after ECMO initiation, 44 (79%) survived to 24 months, and 41 (73%) survived to 36 months. The Kaplan–Meier curves illustrate that most of the deaths occurred during the first 30 days (Figure 2(a)). Additional Kaplan–Meier curves were plotted for the subsequent 1- to 36-month period (Figure 2(b)) for patients who survived the first month. Patients supported with ECMO for > 3 days had significantly higher 36-month survival rate than patients supported with ECMO for ⩽ 3 days (41% vs. 20%; p = 0.014 by log-rank test; Figure 3). Among hospital survivors, patients supported with ECMO for > 3 days (9/33) had similar 36-month survival rate (4/10) with patients supported with ECMO for ⩽ 3 days (73% vs. 60%, p = 0.855). Patients with SOFA score > 10 had significantly lower 36-month survival rate than those with SOFA score ⩽ 10 (20% vs. 71%; p < 0.001 by log-rank test; Figure 4(b)).
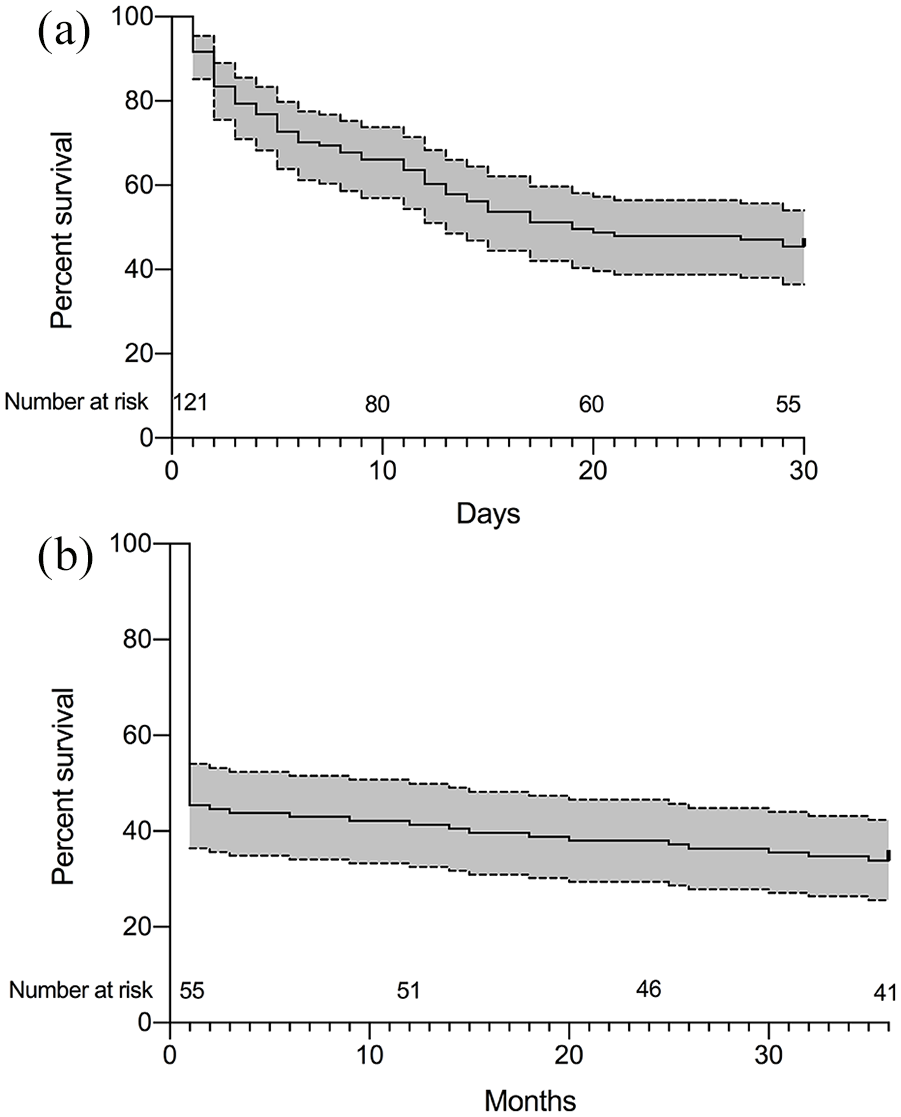
Survival curves the patients receiving extracorporeal membrane oxygenation: (a) 0 to 30 days; (b) 1 month to 36 months. Dotted lines represent 95% confidence interval for survival.
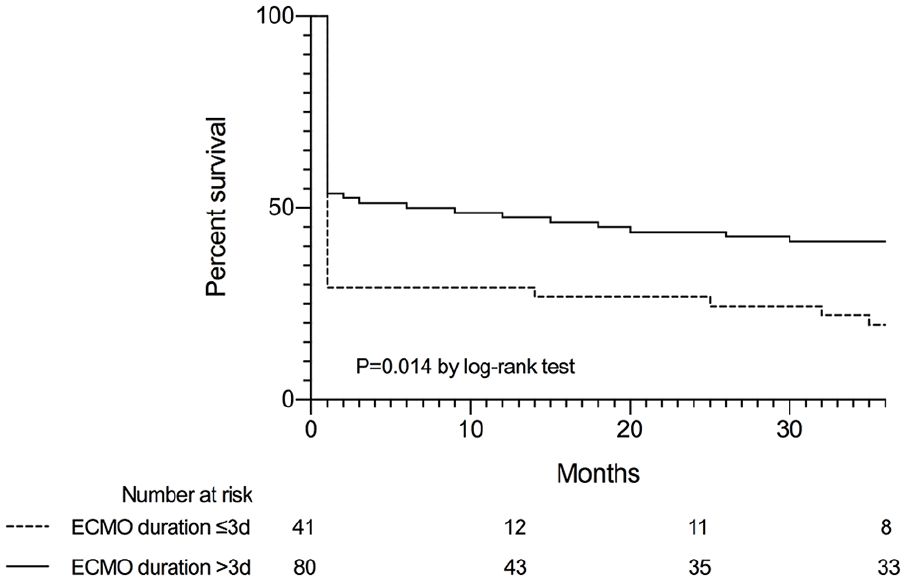
Kaplan–Meier curve showing survival for those with ECMO duration more or equal to or less than 3 days.
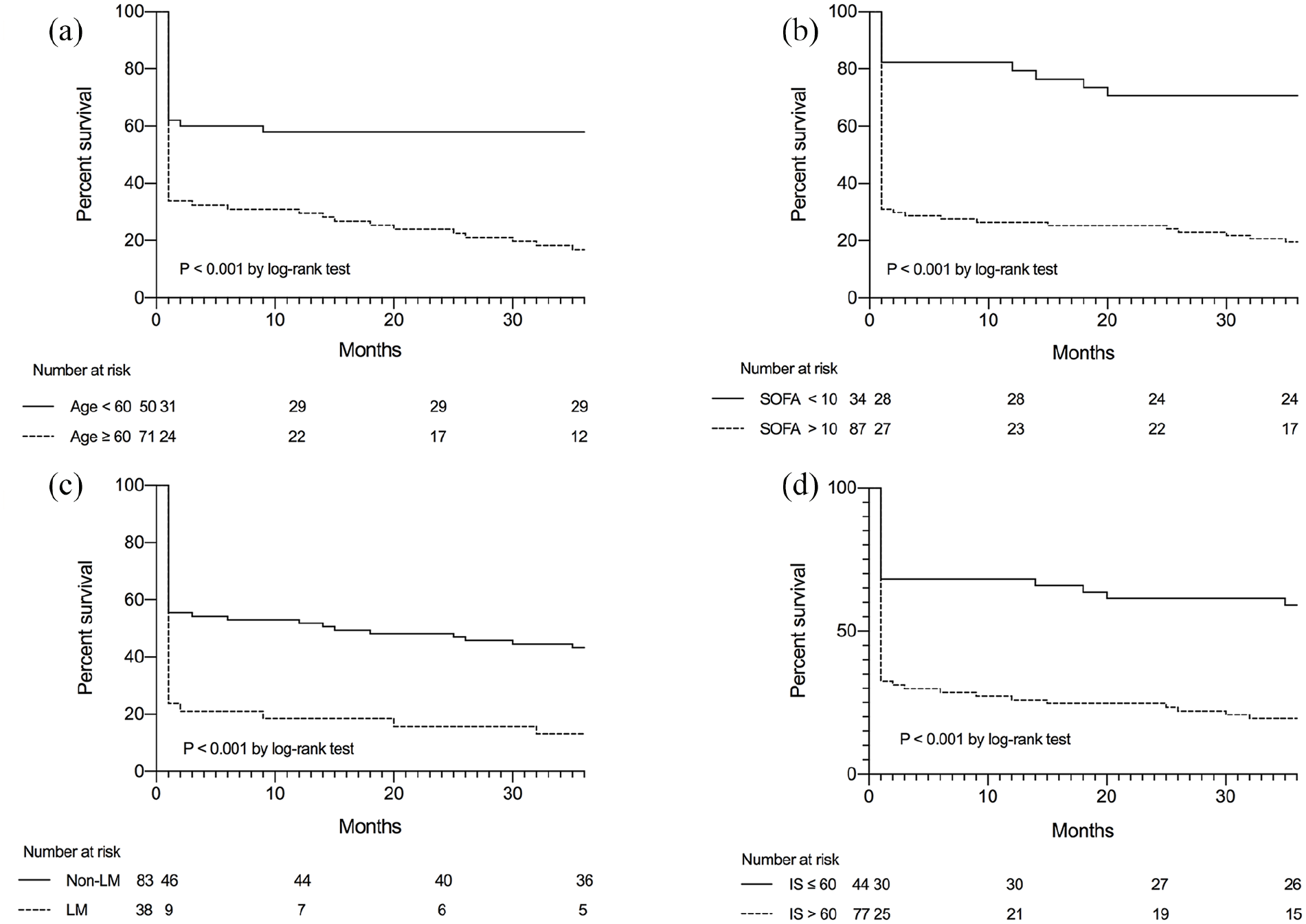
(a) Kaplan–Meier curve showing survival for those with age greater than or equal to or less than 60 (b) Kaplan–Meier curve showing survival for those with SOFA score greater than or equal to or less than 10; (c) Kaplan–Meier curve showing survival for those with or without left main coronary artery disease; (d) Kaplan–Meier curve showing survival for those with vasoactive inotropic score greater than or equal to or less than 60. LM, left main coronary artery disease; IS, vasoactive inotropic score.
Predictors of 36-month mortality
Variables associated with mortality at 36 months by univariable Cox analysis were age, left main coronary artery disease, vasoactive inotropic score, serum creatinine, and serum lactate (Table 3). Multivariable Cox analysis identified older age (HR, 1.06; 95% CI, 1.03-1.10; p < 0.001; Table 3), left main coronary artery disease (HR, 1.64; 95% CI, 1.04-2.59; p < 0.001; Table 3), and vasoactive inotropic score (HR, 1.09; 95% CI, 1.02-1.16; p = 0.011; Table 3) as independent risk factors associated with 36-month mortality. The adjusted HRs of age ⩾ 60 years, left main coronary artery disease, and vasoactive inotropic score > 60 for mortality at 1 month (Table 4) were 1.86(95% CI, 1.09-3.18), 2.09(95% CI, 1.28-3.42), and 2.77(95% CI, 1.53-5.03), respectively. The adjusted HRs of age ⩾ 60 years, left main coronary artery disease, and vasoactive inotropic score > 60 for mortality at 1-36 months (Table 4) were 8.92 (95% CI, 1.92-41.49), 3.97 (95% CI, 1.23-12.83), and 2.89 (95% CI, 0.88-9.49), respectively.
Cox regression analyses of factors associated with 36-month death.
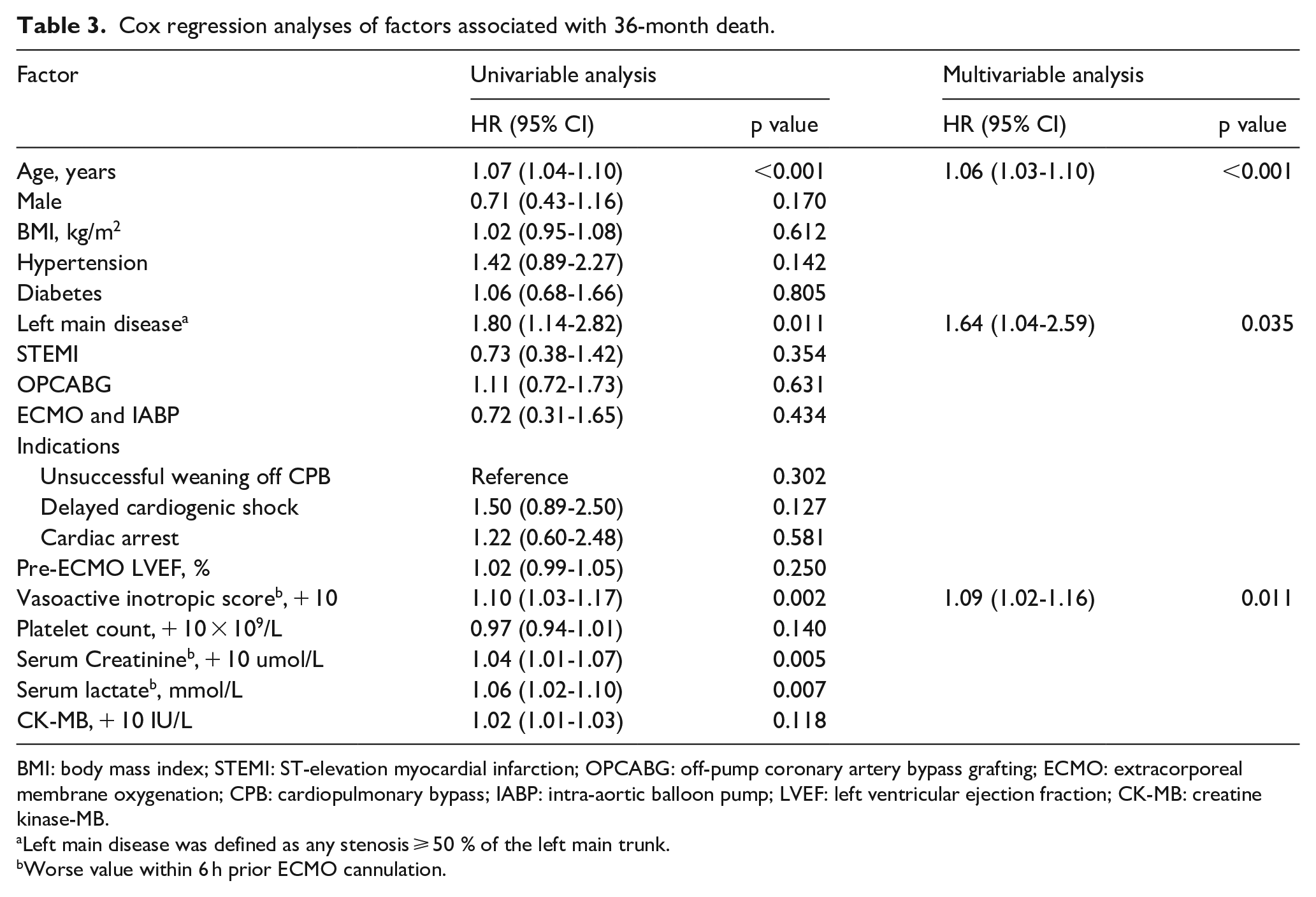
BMI: body mass index; STEMI: ST-elevation myocardial infarction; OPCABG: off-pump coronary artery bypass grafting; ECMO: extracorporeal membrane oxygenation; CPB: cardiopulmonary bypass; IABP: intra-aortic balloon pump; LVEF: left ventricular ejection fraction; CK-MB: creatine kinase-MB.
Left main disease was defined as any stenosis ⩾ 50 % of the left main trunk.
Worse value within 6 h prior ECMO cannulation.
Factors associated with mortality at 0-1 month and at 1-36 months.
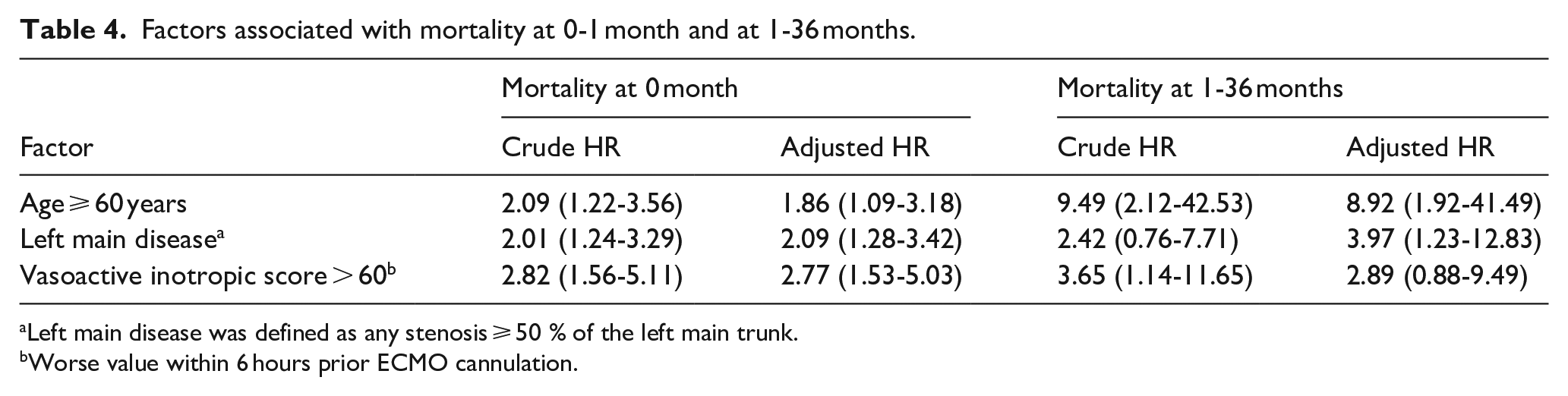
Left main disease was defined as any stenosis ⩾ 50 % of the left main trunk.
Worse value within 6 hours prior ECMO cannulation.
In the multivariable logistic regression analysis, age ⩾ 60 years (odds ratio, 10.86; 95% CI, 3.60-32.80; p < 0.001; Table 5), left main coronary artery disease (odds ratio, 11.18; 95% CI, 2.85-43.85; p = 0.001; Table 4), and vasoactive inotropic score > 60 (odds ratio, 15.01; 95% CI, 4.56-49.42; p < 0.001; Table 5) were identified as independent predictors of 36-month survival. The Kaplan–Meier curves (Figure 4(a)-(d)) confirming differences in survival for patients stratified by the binary levels. The logistic regression model calibration was good (Hosmer–Lemeshow χ2 = 7.952; p = 0.242, Table 5), and the AUROC for the logistic regression model was 0.87(95% CI, 0.81-0.94). The SOFA score (0.79; 95% CI, 0.71-0.88; p = 0.156; Figure 4) and SOFA score combined with age and left main coronary artery disease (0.91; 95% CI, 0.86-0.96; p = 0.408; Figure 5) exhibited good performances similar to the logistic regression model. However, the AUROC for SOFA score combined with age and left main coronary artery disease was significantly better than that of SOFA score alone (p = 0.025; Figure 5).
Results of multivariable logistic regression.
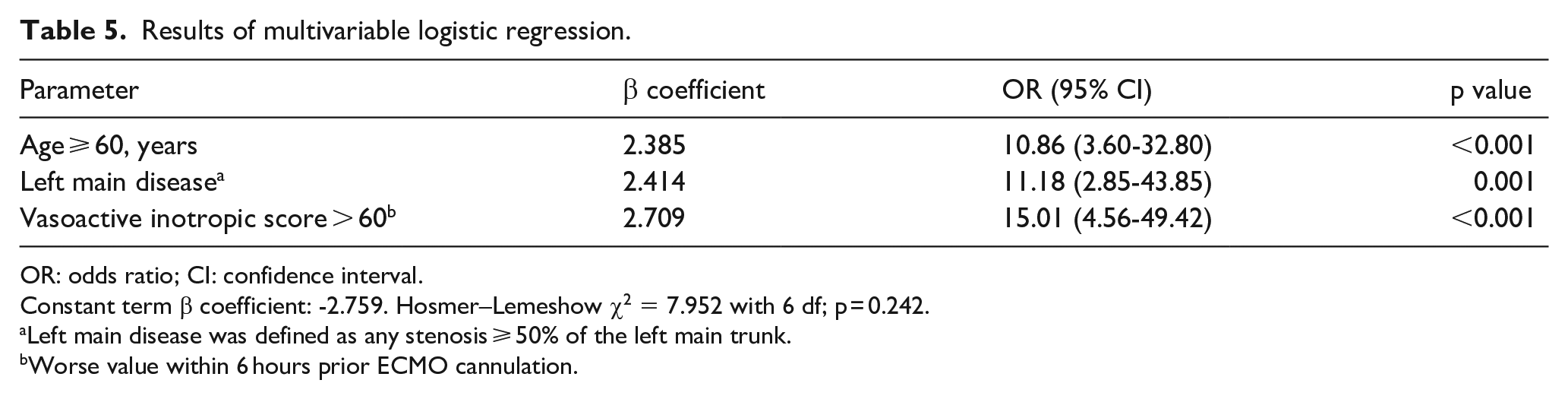
OR: odds ratio; CI: confidence interval.
Constant term β coefficient: -2.759. Hosmer–Lemeshow χ2 = 7.952 with 6 df; p = 0.242.
Left main disease was defined as any stenosis ⩾ 50% of the left main trunk.
Worse value within 6 hours prior ECMO cannulation.
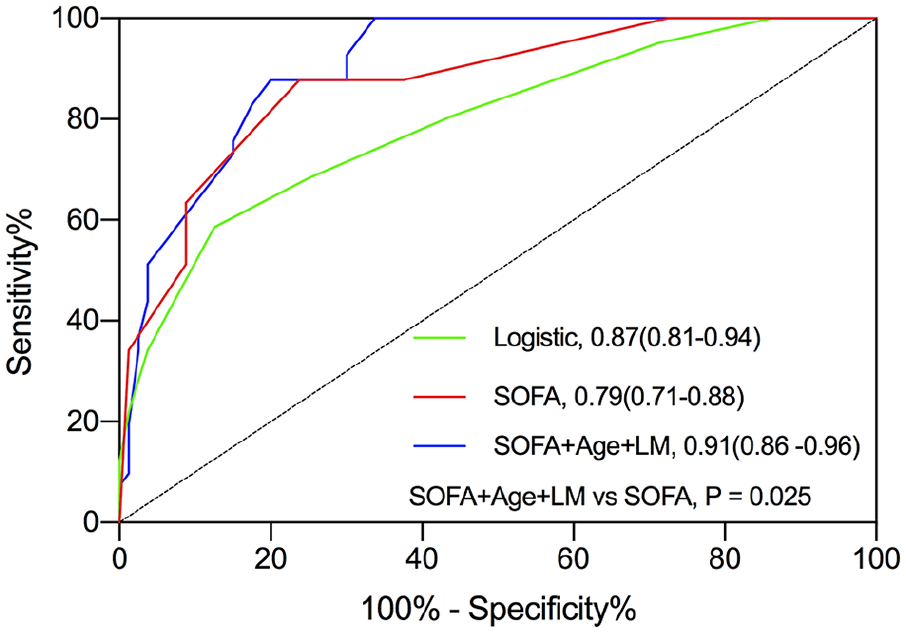
The areas under the receiver operating characteristic curves (AUROC) for predicting unsuccessful weaning. SOFA score, the sepsis-related organ failure assessment score; LM, left main coronary artery disease. The AUROC for SOFA score combined with age and left main coronary artery disease was significantly better than that of SOFA score alone.
Subgroup analysis
Baseline characteristics of the patients between the OPCABG group and the on-pump CABG group are in Supplemental Table S1. Most of baseline characteristics were similar between the two groups. There was significant difference between the OPCABG group and the on-pump CABG group in unsuccessful weaning off CPB, delayed cardiogenic shock in ICU, cardiac arrest, vasoactive inotropic score, and platelet count. In the OPCABG group, 47 patients needed ECMO for delayed PCS in ICU, and 17 patients needed ECMO for cardiac arrest; in the on-pump CABG group, 39 patients needed ECMO for unsuccessful weaning off CPB, 16 patients needed ECMO for delayed PCS in ICU, and 2 patients needed ECMO for cardiac arrest. No significance was observed in the 36-month survival rate between the two groups (31% vs. 37%, p = 0.518). The crude HR of OPCABG for 36-month mortality was 1.11 (95% CI, 0.72-1.73) (p = 0.631).
Discussion
In this single-center, retrospective study involving CABG patients with very severe PCS, we found that 46% of such patients survived to hospital discharge, and 34% of the included patients survived to 36 months. In addition, older age, left main coronary artery disease, and vasoactive inotropic score were identified as independent risk factors associated with 36-month mortality.
Although the use of VA-ECMO for PCS has increased substantially over the past decade, 11 detailed information on long-term survival is lacking. In a recent meta-analysis involving 2,877 PCS patients undergoing ECMO from 20 observational studies, the pooled survival rates at hospital discharge and 36 months were 34.0% (95% CI, 30.0%-38.0%) and 18.0% (95% CI, 11.0%-27.0%), respectively. Biancari et al. 12 included 781 adult patients who required VA-ECMO for cardiopulmonary failure after cardiac surgery and found that the survival rates at hospital discharge, 12 months, and 60 months were 35.6%, 32.8%, and 28.5%, respectively. In another study, Biancari et al. 13 analyzed the outcomes of 148 patients who received VA-ECMO therapy due to cardiac or respiratory failure after isolated CABG, and found that 12-, 24-, and 36-month survival rates were 31.0%, 27.9%, and 26.1%, respectively. These published reports of postcardiotomy ECMO (PC-ECMO) are mainly single-center experiences, and the long-term results were disappointing, frequently not identifying the predictors for long-term mortality, or combining valve surgery and CABG patients in the same series. In the present study, survival rates were 46%, 41%, 38%, and 34% at hospital discharge, 12 months, 24 months and 36 months, respectively.
Despite the low overall survival of PC-ECMO patients, the long-term survival of patients who survive to discharge appears favorable, with the vast majority of patients still alive at 12-month follow-up. 14 Saxena et al. 15 showed that survival was 69% at 36 months, and 51% at 60 months among patients discharged from hospital, confirming good post-discharge prognosis of PC-ECMO patients, even in the elderly (age > 70 years). Similarly, in the present study, survival was 89% at 12 months, 79% at 24 months, and 73% at 36 months among those hospital survivors. The Kaplan–Meier curves showed that most of the deaths occurred during the first month or hospitalization, potentially owing to complex perioperative course of PC-ECMO patients. We also found that patients who required ECMO for > 3 days had significantly lower 36-month mortality than those who required ECMO for ⩽ 3 days, which was similar with the results of the extracorporeal Life Support Organization (ELSO) registry. 16 Most of the patients who required ECMO for ⩽3 days could not be weaned from ECMO, which might account for our findings. The reasons for discontinuation in the early period included hemorrhage, organ failure, and family request. 17 Since ECMO cost was not covered by medical insurance in China, there are four patients possibly with limitation of the therapeutic efforts. Our study did not suggest that weaning should occur on a particular day in order to maximize survival. The duration of VA-ECMO depends on the underlying disease process.
Many prediction models of ECMO survival after PC-ECMO have been published over the past decade.18–20 Pre-ECMO factors, post-ECMO events, and the patient’s physiological response to circulatory support, all play a critical role. Age, female sex, obesity, diabetes, ischemic heart disease, renal insufficiency, cardiac arrest, hemodynamics, and laboratory markers of hypoperfusion were found in previous studies to correlate with survival. 11 However, most of these studies are limited by absence of risk factors associated with long-term survival. In this study, we identified older age, left main coronary artery disease, and vasoactive inotropic score as independent risk factors associated with 36-month mortality, and these variables were predictors for 1-month mortality, which was similar to other studies. The logistic regression model showed the similar results and exhibited good performance. The Kaplan–Meier curves confirmed the differences in survival for patients stratified by age (60 years), vasoactive inotropic score (60 points), and left main coronary artery disease. Survival was also significantly higher in patients with SOFA score ⩽ 10. The AUROC for SOFA score was (0.79; 95% CI, 0.71-0.88), exhibiting good performances similar to the logistic regression model. Older age and left main coronary artery disease might suggest the complex perioperative course. Vasoactive inotropic score and SOFA score were always used to roughly estimate the severity of the pre-ECMO status. We created a new model incorporating SOFA score, age, and left main coronary artery disease, and found that age and left main coronary artery disease significantly increased the discriminatory performance of SOFA score. Since vasoactive inotropic score was collinear to SOFA score, the performance of SOFA score combined with vasoactive inotropic score was not evaluated.
There are several limitations in this study. First, it was a single-center, retrospective study which may limit the generalizability of our results. Second, because left ventricular assist devices were not registered in China, no patients were transferred to another heart device underwent ventricular assist device. The long-term survival rate might have therefore been underestimated. Third, indications and thresholds for institution of ECMO might have changed during the study period and have influenced the observed outcomes. Fourth, since only patients requiring VA-ECMO after isolated CABG were included in our study, our logistic regression model may less accurately predict long-term survival in patients who receive VA-ECMO for other indications. Fifth, some characteristics between OPCABG patients and on-pump CABG patients were different, and we analyzed these two cohorts together, which might have affected the results. However, off-pump CABG was not associated with outcomes in the present study. Finally, patients’ quality of life and ECMO costs were not available in the present study; further studies are warranted to evaluate the cost-effectiveness of this lifesaving technology for PCS.
Conclusion
In our cohort of patients undergoing isolated CABG complicated by PCS requiring VA-ECMO, hospital and 36-month survival rate were 46% and 34%, respectively. Older age, left main coronary artery disease, and vasoactive inotropic score were identified as independent risk factors associated with 36-month mortality.
Supplemental Material
Supplemental_digital_content – Supplemental material for Survival following venoarterial extracorporeal membrane oxygenation in postcardiotomy cardiogenic shock adults
Supplemental material, Supplemental_digital_content for Survival following venoarterial extracorporeal membrane oxygenation in postcardiotomy cardiogenic shock adults by Fei Chen, Liangshan Wang, Juanjuan Shao, Hong Wang, Xiaotong Hou and Ming Jia in Perfusion
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by the National Key Research and Development Program of China (2016YFC1301001, to X Hou).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
