Abstract
Background:
Left ventricular (LV) afterload increase with protracted aortic valve (AV) closure may represent a complication of veno-arterial extracorporeal membrane oxygenation (V-A ECMO). The aim of the present study was to assess the effects of an intra-aortic balloon pump (IABP) to overcome such a hemodynamic shortcoming in patients submitted to peripheral V-A ECMO.
Methods:
Among 184 adult patients who were treated with peripheral V-A ECMO support at Medical University Center Maastricht Hospital between 2007 and 2018, patients submitted to IABP implant for protracted AV closure after V-A ECMO implant were retrospectively identified. All clinical and hemodynamic data, including echocardiographic monitoring, were collected and analyzed.
Results:
During the study period, 10 subjects (mean age 60 years old, 80% males) underwent IABP implant after peripheral V-A ECMO positioning due to the diagnosis of protracted AV closure and inefficient LV unloading as assessed by echocardiography and an absence of pulsation in the arterial pressure wave.
Recovery of blood pressure pulsatility and enhanced LV unloading were observed in 8 patients after IABP placement, with no significant differences in the main hemodynamic parameters, inotropic therapy or in the ECMO flow (p=0.48). The weaning rate in this patient subgroup (mean ECMO duration 8 days), however, was only 10%, with another patient finally transplanted, leading to a 20% survival-to-hospital discharge.
Conclusion:
IABP placement was an effective solution in order to reverse the protracted AV closure and impaired LV unloading observed during peripheral V-A ECMO support. However, the impact on the weaning rate and survival needs further investigations.
Introduction
Veno-arterial extracorporeal membrane oxygenation (V-A ECMO) has been widely employed for refractory cardiogenic shock and cardiac arrest. V-A ECMO may be quickly assembled and implanted, has limited costs compared to other mechanical circulatory support devices and allows the attending physicians to temporarily stabilize the compromised hemodynamic conditions while providing a bridge to recovery or to other more durable treatments.1,2
V-A ECMO, therefore, is a useful circulatory support system, but has shortcomings. One of these is the effect of retrograde aortic flow from the ECMO system on left ventricular (LV) afterload and unloading. This counter-stream blood flow variably increases the afterload of the already impaired LV. 3 This can lead to reduced LV ejection and dilatation, higher pressure within the LV, left atrium and pulmonary veins, leading to stasis in the left cardiac chambers with thrombosis, clot formation and pulmonary oedema. 4 In the worst cases, the LV will be unable to overcome the ECMO-generated counter-flow and pressure, leading to a protracted closed aortic valve (AV). Based on these aspects, LV unloading during V-A ECMO, therefore, may represent either a critical aspect to be monitored or an indication for adjunctive unloading procedures. 5
Echocardiographic monitoring is a cornerstone of V-A ECMO management and, besides the information of ongoing LV function, is paramount to assessing LV unloading and defining the extent of blood stasis in the left cardiac cavities (echo contrast, named “smoke-like” effect), thereby, indicating the need of ancillary unloading maneuvers.6-9 Nowadays, there are several techniques, of varying aggression and complexity, which aim to unload the LV. 5 Those are classified either according to their surgical/percutaneous approach or considering the anatomical unloading location.
However, the optimal technique and the target patient population who will actually benefit from venting procedures remain unclear. 5
The majority of patients who are supported with V-A ECMO apparently do not require LV unloading. The prevalence of the use of the various unloading techniques is unknown. 10
Since the Extracorporeal Life Support Organization Guidelines for Adult Cardiac Failure recommend that the intra-aortic balloon pump (IABP) should be placed as an additional support to ECMO, it is used and routinely placed at the initiation of ECMO at many medical centers.11-15
From the physiological point of view, IABP should positively support myocardial ejection by reducing the increased afterload. However, many studies have not shown a consistent effect on survival and, thus, the evidence for the additive benefit of IABP therapy is limited or controversial.16-18
The aim of the present study, therefore, was to assess the impact of IABP in patients submitted to peripheral V-A-ECMO presenting protracted AV closure with or without clear signs of LV stasis and increased pressure in the left cardiac chambers due to increased LV afterload.
Methods
Patient series
In the period from January 2007 to January 2018 at the Maastricht University Medical Center, peripheral V-A ECMO support was implanted in 184 adult patients for severely impaired cardiovascular conditions.
All patient data were analyzed in relation to etiology of cardiovascular illnesses, clinical and hemodynamic conditions at ECMO implant and thereafter, ECMO management and in-hospital outcome. In particular, echocardiographic and blood pressure curve evaluation soon after temporary support placement and subsequent examinations were reviewed in order to highlight the presence of an effective or partial AV opening or the confirmation of a permanent closure of the same valve or any other hemodynamic as well as radiographic signs of blood stasis. Efficacy of LV ejection was also assessed by velocity time integral (VTI) assessment.
Patient informed consent was waived in this study, based on the retrospective nature of the research, by the patient status at the time of data recordings (patients intubated and sedated) and by the use of routine parameter recording and diagnostic tool.
IABP and ECMO placement: method and management
For all patients, the contra-lateral femoral artery of the V-A ECMO cannulation site was cannulated for balloon placement (Seldinger method). If feasible (no major resistance at IABP passage through the small skin incision), a sheathless technique was used to reduce the incidence of leg ischemia. The tip of the balloon was placed 1 cm distal to the junction with the left subclavian artery, as assessed by echocardiographic assessment and by a mobile chest x-ray system at the bedside.
Either of the electrocardiogram (ECG) or the aortic blood pressure curve was used as a trigger; for the ECGs, the descending section of the R wave (representing closing of the AV) was used to calibrate the counter-pulsation interval, with an IABP ratio of 1:1. If the patient showed low dependence on positive inotropic drugs, the IABP ratio was gradually reduced to 1:3 accompanied by half an hour of observation; if circulation was steady, the IABP was removed.
The Rotaflow (Getinge, Hirrlingen, Germany) was the centrifugal ECMO pump used in all patients who underwent peripheral V-A ECMO.
Hemodynamic data collection
Hemodynamic parameters were collected at the following time points: a few hours before, just prior to and a few minutes after IABP placement. The following data were collected: pulsatility of the arterial systemic blood pressure, mean arterial blood pressure, systolic (SBP) and diastolic arterial blood pressure (DBP), heart rate (HR), ECMO flow and echocardiographic evaluation of the AV closure. The pre-IABP ECMO flow and the inotropic therapy reflected the average of the last six hours. By contrast, the post-IABP ECMO flow was recorded simultaneously with the echocardiographic evaluation.
Statistical analysis
The SPSS 23.0 (IBM Corp., Armonk, NY, USA) statistical program was utilized for statistical analysis. Continuous variables at different time points were examined with paired t-tests, with p<0.05 considered statistically significant.
Results
From the overall peripheral V-A ECMO population, 44 patients received combined IABP and V-A ECMO. Of these patients, 10 subjects underwent IABP implant after variable time from the V-A ECMO positioning due to the diagnosis of protracted AV closure as assessed by echocardiographic evidence of the AV dysfunction and inefficient LV unloading, as well as the absence of pulsatility at blood pressure curve monitoring.
Patient demographic data are shown in Table 1. The mean age was 60 years old and 80% were males. Patient etiologies included three postcardiotomy and two out of hospital cardiac arrests.
Demographic data.
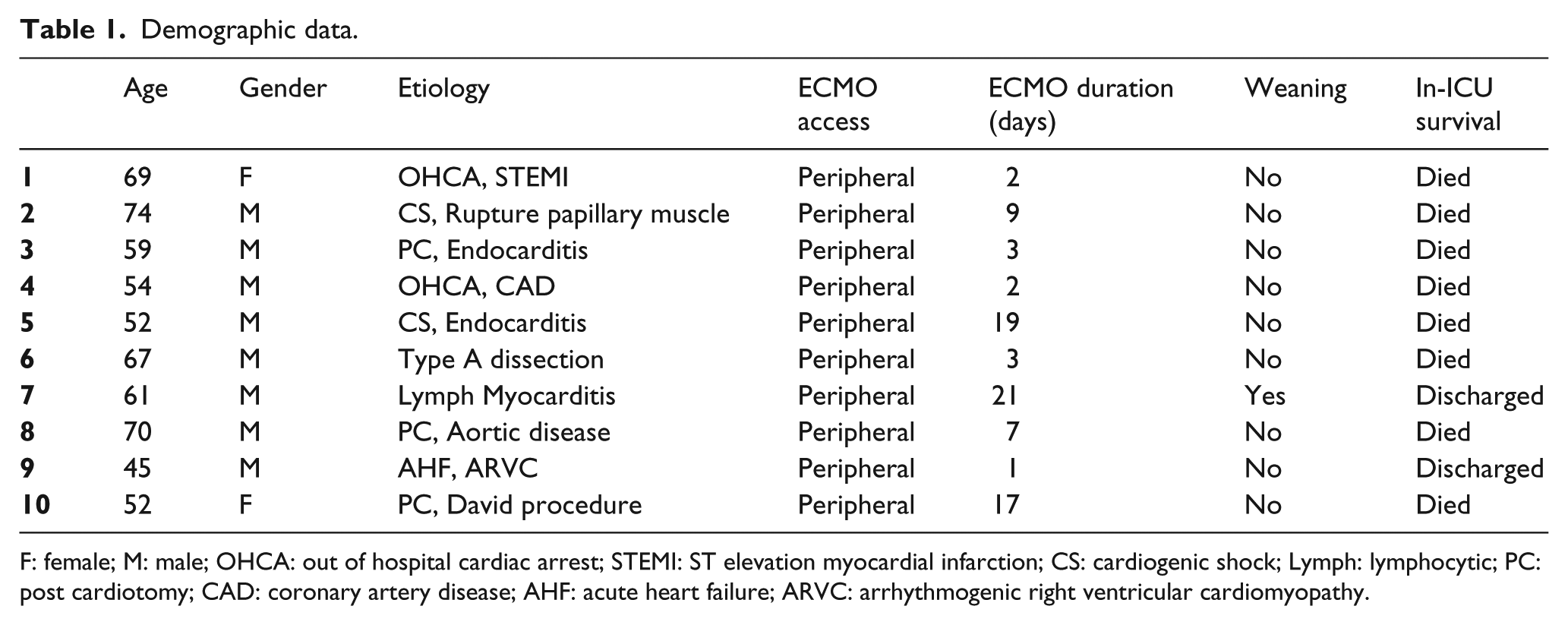
F: female; M: male; OHCA: out of hospital cardiac arrest; STEMI: ST elevation myocardial infarction; CS: cardiogenic shock; Lymph: lymphocytic; PC: post cardiotomy; CAD: coronary artery disease; AHF: acute heart failure; ARVC: arrhythmogenic right ventricular cardiomyopathy.
Table 2 illustrates the parameters collected just before and subsequent to the IABP placement.
Parameters pre and post IABP placement.
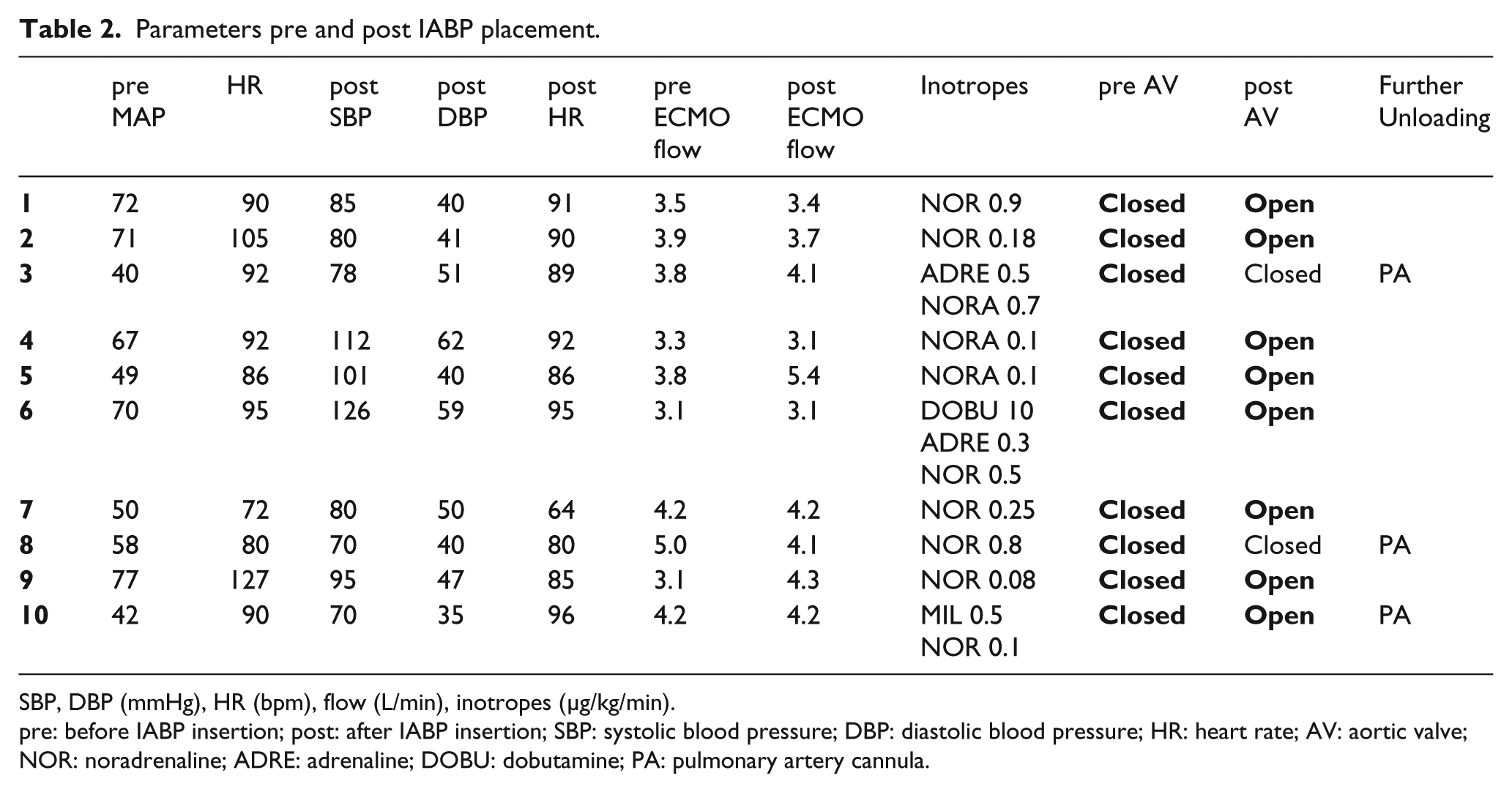
SBP, DBP (mmHg), HR (bpm), flow (L/min), inotropes (µg/kg/min).
pre: before IABP insertion; post: after IABP insertion; SBP: systolic blood pressure; DBP: diastolic blood pressure; HR: heart rate; AV: aortic valve; NOR: noradrenaline; ADRE: adrenaline; DOBU: dobutamine; PA: pulmonary artery cannula.
No significant differences (p>0,05) were found, either in the hemodynamic parameters, such as the mean systemic arterial blood pressure and HR, or in the inotropic therapy or in ECMO flow details as recorded just prior to and immediately after IABP implant (p=0.48).
As shown in Table 2, IABP implant was able to reverse 80% of the cases of protracted AV closure, as confirmed by recovery of systemic arterial blood pressure pulsatility and restored AV opening at echocardiographic assessment performed after IABP placement.
Only two patients showed no benefit from IABP implant regarding AV function. Both patients immediately required a further LV unloading due to rapid progression to frank pulmonary oedema, solved by placing an additional draining cannula in the pulmonary artery. In addition, another patient, despite an initial successful restoration of the AV opening after IABP implant, required the positioning of the cannula into the pulmonary artery after 48 hours for new evidence of protracted AV closure and echocardiographic signs of intraventricular blood stasis.
Mean V-A ECMO duration was 8 days (1-21 days). As shown in Table 1, the V-A ECMO-weaning rate in this patient subgroup was only 10%, with one patient eventually transplanted at another center, leading to a final survival-to-hospital discharge of 20%.
Discussion
The case series presented in this study is unique since it is the first collection of data regarding the onset and detection of a clear hemodynamic shortcoming of V-A ECMO, namely, protracted AV closure due to the combination of poor LV function and retrograde flow generated by the ECMO system towards the AV.
Furthermore, we were able to demonstrate that IABP insertion is usually capable of overcoming this adverse event.
The resolution of such a hemodynamic ECMO-related drawback is only one of many factors which lead to a favorable or unfavorable patient outcome.
During peripheral V-A ECMO, LV afterload significantly increases. Such a condition may be poorly tolerated by an already dysfunctional LV, leading to AV dysfunction and left cardiac chamber distension. 19 Evidence from patients undergoing LV assistance device (LVAD) implant indicate the biomechanics of the AV is often altered in this condition with the appearance of AV insufficiency.20-22 LVAD-related continuous flow may also induce protracted AV closure which, if present for months, may ultimately lead to leaflet fusion and, therefore, to irreversible AV closure. 23 This pathophysiological condition is similar to what is observed in V-A ECMO patients regarding protracted AV closure. The ECMO pumps blood directly into the aorta, increasing the aortic pressure. This leads to an increased pressure difference between the aortic root and the LV, also known as the transvalvular pressure (TVP) of the AV.22,23 The valve opens when the TVP is near zero and closes under pressure load. The continuous increased aortic pressure from the V-A ECMO raises the TVP; consequently, if the LV function is poor, the AV opening will be impaired. As a result, the latter is an early sign of insufficient LV unloading. Obviously, the usual short duration of V-A ECMO is not sufficient to induce structural valve changes as observed in LVAD patients, but may still lead to dangerous pre- or post-valvular blood stasis in extreme cases of protracted AV closure.
Echocardiography can detect and help manage such complications and hemodynamic alterations that may arise during ECMO support.24-26
We propose that the echocardiographic evaluation of LV loading should have two components. First, the anatomic evaluation should briefly check the AV, the LV and the LA distension and the vena cava inferior dilation. Second, discrete parameters should be collected. The velocity time integral might be measured at the LV out-flow tract. An estimation of the pulmonary artery pressure should be recorded by defining the maximal jet velocity of the tricuspid regurgitation. 6
Nevertheless, taking all into consideration, we speculate that AV function may allow classification of the degree of the LV overload (Table 3).
Definition of left ventricle overload.
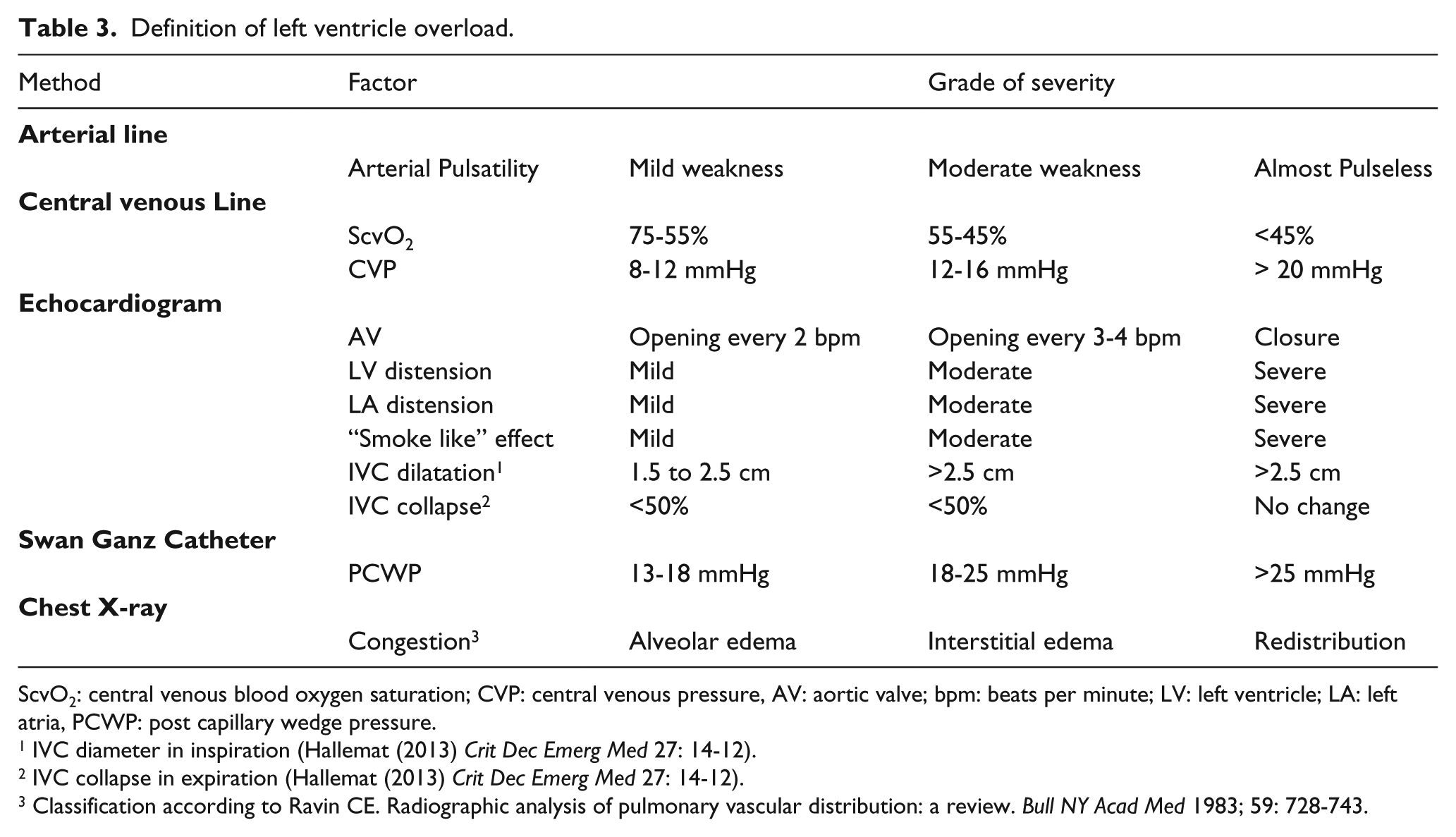
ScvO2: central venous blood oxygen saturation; CVP: central venous pressure, AV: aortic valve; bpm: beats per minute; LV: left ventricle; LA: left atria, PCWP: post capillary wedge pressure.
IVC diameter in inspiration (Hallemat (2013) Crit Dec Emerg Med 27: 14-12).
IVC collapse in expiration (Hallemat (2013) Crit Dec Emerg Med 27: 14-12).
Classification according to Ravin CE. Radiographic analysis of pulmonary vascular distribution: a review. Bull NY Acad Med 1983; 59: 728-743.
In the presence of severely impaired function or protracted closure of the AV, it is mandatory to act in order to prevent or limit complications. 5 In our experience, the first step is IABP insertion in the case of AV opening impairment and absence of arterial blood pressure pulsatility. The continued presence of other LV overloading signs, such as LV distension, smoke-like effect or severe pulmonary congestion, should drive the decision-making towards more aggressive LV unloading techniques.
Although the sample size is limited, our findings indicate that the IABP has the ability to promote AV opening. Pulsatility was resumed in almost all patients, indicating that the IABP may represent a valuable solution.27,28 Den Uil and colleagues showed a positive effect of IABP in a patient submitted to V-A ECMO and surgically implanted venting. 29 Although some other studies experienced a positive IABP effect on the final outcomes,30,31 these were not consistently achieved in our study and one patient required an additional LV unloading technique. Little is known about the meaning of AV dysfunction during V-A ECMO and how this might be considered as an early stage of the “overload continuum”. The latter could begin with a mere hemodynamic impairment, followed by LV structural and anatomic alterations, ending up with irreversible complications, such as thrombosis and pulmonary edema.
It is still unclear which LV unloading technique is indicated at what stage. 5 Further studies are urgently needed in order to define the actual rate of severe impaired LV unloading, the benefit of various venting manoeuvres, the impact of LV unloading on timing and efficacy of LV functional recovery and the influence of these on patient outcome.
Limitations of the study
This study represents a retrospective analysis of a 10-year, single-centre experience with a peripheral V-A ECMO configuration. The conduct of patient monitoring and hemodynamic recording, including AV assessment and detection of any sign of impaired LV unloading, received a gradual increase of attention and report during the study period. It is, therefore, likely that the actual extent and rate of protracted AV closure or partial opening, as well as incidence of blood stasis phenomena, have been underestimated.
Some patients had already received a LV venting (post cardiotomy) and this may have reduced the blood stasis formation, although not impacting the AV behaviour.
Patient causes of death were due to neurologic complication, multi-organ failure or sepsis. As is well known, ECMO patient course and outcome is influenced and impacted by numerous events and mechanisms, making any relationship between AV behaviour and ultimate patient outcome extremely difficult.
Conclusion
In conclusion, we were able to demonstrate that, in a limited number of patients, the ECMO-related flow, with or without the association of an extremely poor LV function, may completely impede forward trans-aortic flow, thereby, inducing a protracted closure of the valve, leading to blood stasis upstream or just downstream of the AV itself. Besides several clinical and radiographic clues, the diagnosis of such an adverse event is made by lack of blood pressure curve pulsatility, followed by transthoracic or transesophageal echocardiography, in association with other clinical or diagnostic clues. The IABP implant will restore AV valve opening in the majority of cases. Resolution of AV dysfunction, however, may not influence patient prognosis and needs further investigations.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
