Abstract
A Ni–Co-base superalloy was subjected to three different pack aluminising procedures (1000 °C for 3.5 h, 890 °C for 8 h and 620 °C for 10 h) to produce the Al-rich coating with simultaneously improved oxidation and wear resistance. All samples showed a multi-layer coating made up of (Ni, Fe, Co)Al phase, (Ni, Fe, Co)3Al phase and transition layer. The AT620 sample possessed the lowest oxidation rate but the highest double-edge-notched (DEN) strain during high-temperature stress rupture. The high oxidation resistance might be associated with the creation of Al2O3 layer (∼5.6 μm), contributing to an inhibition of oxygen permeation. However, a premature failure was caused by the strong deformation incompatibility between the substrate and alumina layer. The AT890 samples had higher oxidation and creep-induced-crack resistance than those of the AT1000 samples, which was the result of the effective Al inter-diffusions in the transition layer suppressing the undesirable Cr-rich σ phase.
Introduction
Due to their many benefits of excellent creep strength, high ductility, low thermal expansion efficient and good corrosion resistance, Ni–Co base superalloys, which exhibit a complex microstructure constitution of the disordered γ matrix, γ′-Ni3Al precipitates, β-NiAl phase, and various carbides, 1 have been widely used in the gas turbine aero-engines. However, one significant issue for the cyclic oxidation resistance throughout the long-term impact and erosion process 2 may result from increased environmental temperature and greater service stress. In view of this aspect, the aluminide coating 3 has been regarded as one of the most efficient and widely utilised protective layering techniques because of its low cost, simple manufacture, and superior oxidation resistance. Omer et al. 4 introduced a dense aluminide coating with a thickness of approximately 35 μm and obtain approximately 2.65 times better oxidation resistance than the non-aluminised IN625 alloys. Injection laser cladding was used by Bita et al. 5 to aluminise IN738 alloy and attain a high resistance against the hot corrosion. Generally, the coating process can be realised by different techniques, such as magnetron sputtering, 6 hot dipping, 7 laser alloying 8 and pack aluminisation, 9 which can be divided into the diffusion or overlay process.
To ensure sufficient chemical diffusion and growth kinetics with homogeneous coating, the pack aluminisation process is carried out at temperatures ranging from 750 to 1100 ◦C for a reaction time of 2 to 24 h. The characteristic of aluminide coating strongly depends on the aluminising techniques and processing conditions. Sarraf et al. 10 discussed a three-stage coating formation mechanism during reactive air aluminising in a Ni-base superalloy, and the aluminide coating of pack cementation was decided by the aluminium activity, especially the aluminising temperature and time. 11 The low-activity aluminide coating deposited at high temperatures always possessed a thick double-layered structure, 12 whilst a low depositing temperature of high activity resulted in a triple-layered coating structure in which a middle layer consisting of β-NiAl phase without precipitates due to the inward diffusion of Al. 13 Yener et al. 14 demonstrated that the macroscopic layer thickness of the IN738 alloy was significantly increased with increasing temperature owing to a higher diffusion rate. Mahmoudi et al. 15 found that an increase in aluminising temperature led to a morphological evolution from an interconnected structure to an equiaxed grain structure in the coating layer. Thus, the elemental inter-diffusion mechanism played a vital role in determining the overall microstructure and performance of Al-rich coatings. However, little attention has been paid to the coating formation in the multi-component Ni–Co-base superalloy, in which a complex interaction between various elements existed. Although Xu et al. 16 found that an aluminide coating with a thickness of approximately 2.9 μm was sufficient for protection of Fe–Ni–Co-base superalloy, they focused little on the relationship between the coating microstructure and oxidation performance.
On the other hand, although the aluminide coating can significantly improve the oxidation resistance, some adverse effects will occur to the mechanical response of the matrix alloy. Theoretically, the reaction between the diffusing Al atoms and the substrate element will cause the formation of a more strengthening γ′ phase, thus improving the creep and fatigue resistance. 17 However, Kakehi et al. 18 reported a reduction of the creep strength in the aluminised Ni-base superalloys owing to the crack initiation in the diffusion layer. Liu et al. 19 found that the fatigue life of the aluminised Ni-based superalloy depends on the phase structure and metallic bond characteristics but not the hardness of the aluminised layer. Jiang et al. 20 obtained an ultra-high nano-hardness of the interface layer in the Mg/steel bimetal assisted with hot-dop aluminising, but a brittle fracture was initiated from the interface layer. Han et al. 21 found that the deformation mismatch between the substrate and coating may easily lead to crack initiation and failures under the actual long-term creep process. However, the most accessible literature 22 was based on laboratory studies, which included diverse damage mechanisms under specific loads. Meanwhile, the performance of aluminised-coating under combined oxidation and flow stress, which corresponded to realistic service conditions, was hardly considered in the previous work. Thus, it is crucial to investigate the relationship between the crack sensitivity, coating structure and aluminising process, which provides a guide design for the Ni–Co-base superalloys with comprehensive oxidation and mechanical performance.
In this study, different procedures of pack aluminisation were adopted in a Ni–Co-base superalloy system to obtain the optimal Al-rich coating, which exhibits comprehensive excellent oxidation and crack resistance. Compared with the previous work, 1 the aim of this paper was to study the effect of surface engineering, that is, different aluminising procedure including the temperature and time, on the oxidation and crack sensitivity of aluminised superalloy, which focused on the relationship between the coating structure and the its related performance. The studied Ni–Co-base superalloy shows a higher Al content, a higher γ solvus temperature and a better resistance to stress accelerated grain boundary oxidation (SAGBO) than the traditional Ni-base superalloy due to the enhancement of β-NiAl precipitation, 23 which renders it a potential candidate for the turbine disc material with a temperature bearing capacity of 750 °C. 24 The relationship between the microstructure of the coating and the oxidation behaviour was investigated. Moreover, the crack sensitivity was evaluated based on the stress rupture life of double-edge-notched (DEN) samples to reveal the dominant damage mechanisms, which provides a guide for the design of Ni–Co-base superalloys application in higher temperatures and larger stresses.
Materials and methods
The studied Ni–Co-base alloy was supplied by Shanghai Steam Turbine Factory, and the detailed chemical compositions of the material were measured by an atomic absorption spectrometer (AAS), as shown in Table 1. The 20-kg ingots were cast using vacuum induction melting (VIM) and then hot extruded into 40 mm bars with an extrusion ratio of 12:1 at about 1160 °C. The extruded material was annealed at 1120 °C for 1 h for homogenisation and then air cooled. Then, after surface cleaning, different pack cementation treatment procedures for aluminising were conducted under an argon atmosphere using a powder mixture containing 98 wt-% FeAl + 2 wt-% NH4Cl at different temperatures and times of 620, 890 and 1000 °C for 10, 8 and 3.5 h, respectively. Different aluminising temperatures were designed based on the phase diagram in order to enhance the formation of different intermetallic compounds, as shown in Figure 1(a). From the thermo-calc analysis, a lower aluminising temperature could enhance the formation of the Ni3Al (L12) phase, whilst the NiAl B2 phases were prone to nucleating at high temperatures. The coating time was selected to make sure that the outward diffusion distances of the matrix concentrations (Ni, Co, Fe and so on) were similar during different aluminising procedures. Then, a multi-step β and γ′ aging procedure was conducted for the Al-coating samples (90 mm × 15 mm × 15 mm), that is, aging at 850 °C for 4 h and air cooling, followed by aging at 725 °C for 8 h, and then furnace cooling to 620 °C for 8 h, and air cooling, in order to introduce the corresponding secondary β and γ′ phases in the matrix, respectively. Finally, the obtained samples with different aluminisation procedures were marked as ‘AT620’, ‘AT890’ and ‘AT1000’, respectively.
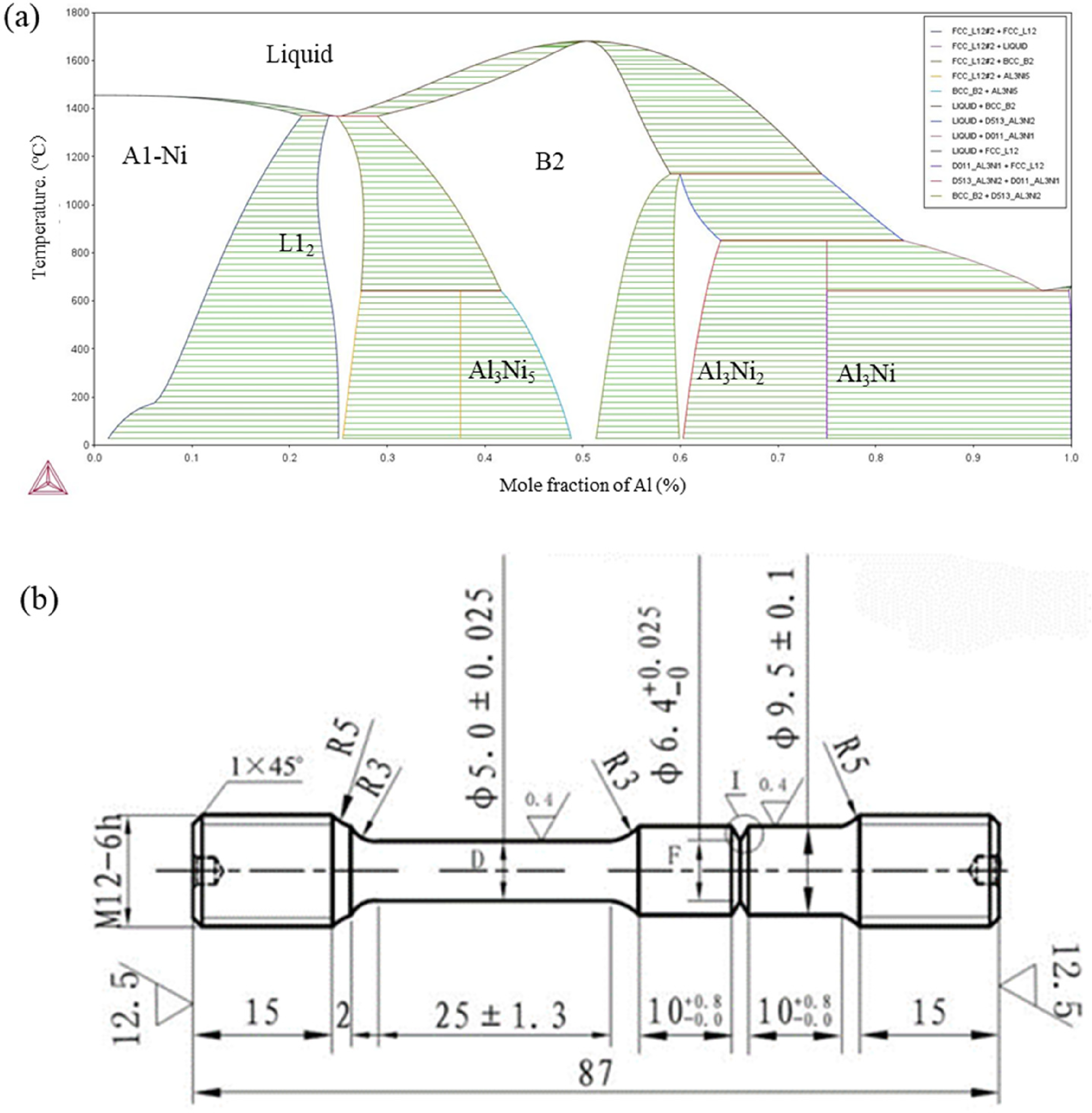
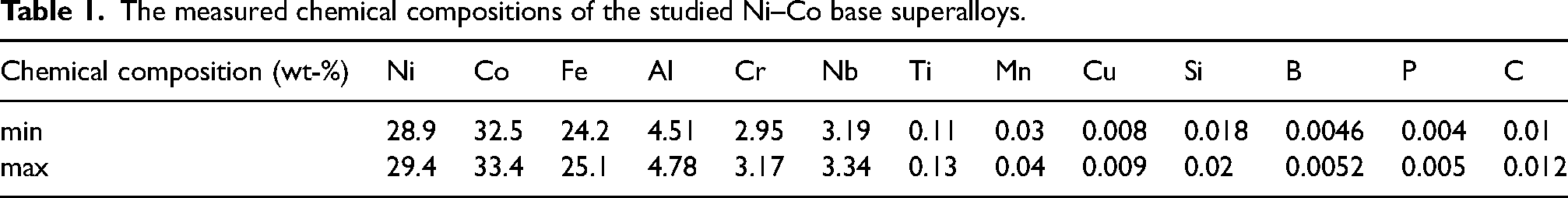
(a) The Ni–Al binary phase diagram calculated by thermo-calc software. (b) Schematic drawing of double-edge-notched specimens for high-temperature stress rupture test.
The measured chemical compositions of the studied Ni–Co base superalloys.
The microstructures of the coating and substrate were characterised by combined optical micrograph (OM, ZEISS), scanning electron microscopy (SEM, TESCAN MIRA3), transmission electron microscopy (TEM, JEOL-2100F) and energy disperse spectroscopy (EDS, OXFORD). The OM and SEM samples were cut from the coated samples, mechanically polished, and then etched with electrolytic phosphoric acid to characterise the overall morphology. TEM samples were mechanically thinned to 70 μm with followed double-jet electro-polishing using a mixed solution of HClO4:C2H5OH = 1:4 at a direct voltage of 50 V at −20 °C, to characterise the chemical composition and size of nanoprecipitates.
The specimens with a dimension of 10 mm × 10 mm × 5 mm underwent ultrasonic cleaning to remove impurities before oxidation, and they were placed into alumina crucibles and maintained contact with the bottom and wall. The high-temperature oxidation test was carried out in a muffle furnace in the atmosphere at 680 °C. After oxidation for 100, 200, 500 and 1000 h, the specimens were removed and weighted by a balance with an accuracy of 10−4 g. Three specimens for each state were used to achieve an average loss value. A 580-MPa monotonic stress rupture test of the DEN sample was performed at 600 °C using an INSTRON stress rig to observe the crack sensitivity during high-temperature deformation. The dimensions of DEN specimens are shown in Figure 1. The oxidation and DEN samples were first cut to the specific dimensions from the homogenised material, then, the whole samples were aluminised by the same pack cementation treatment. The nano-indentation tests of the AT620 and AT890 samples were performed on the Agient Nano indenter G200 equipped with a Berkovich indenter to a maximum load of approximately 10 mN with a three-sided irregular pyramid tip (spring constant: 600 N m−1).
Results and discussions
Figure 2 shows the substrate structures of the Ni–Co-base superalloys under different aluminisation temperatures. All the OM images (Figure 2(a)) of the AT620, AT890 and AT1000 exhibit a constitution of γ matrix, intra- and inter-granular β phase with similar average grain size (10–15 μm), and only show little difference in the phase distributions. Moreover, the SEM maps (Figure 2(b)) show more details; that is, a homogeneous distribution of γ′ nanoprecipitates can be observed in the matrix. The TEM characterisation shows that the coherent γ′ precipitates in all samples possess a mixed spherical-like and irregular morphology with a bimodal grain size distribution of 5–10 nm and 15–80 nm, respectively. However, the nano-sized spherical γ′ precipitates dominate in the AT890 and AT620 samples. It has been demonstrated that the volume fraction and morphology of γ′-Ni3Al were closely related to the solution and aging treatment. In the AT1000 samples, the primary γ′ precipitates nucleate in the high-angle grain boundary during slow cooling process after aluminising at a high temperature above the solvus line, 25 and they show a high coarsening tendency after the subsequent recrystallisation and aging process, leading to the formation of irregular shaped γ′ precipitates. Meanwhile, some needle-like precipitates are found in the interior of the intra-granular β phase (granular-like), and they are identified as η precipitates by the selected area diffraction pattern (SAED) (Figure 2b3). However, the η precipitate in the AT620 sample shows a larger number density than those in the AT890 and AT100 samples (Figure 2c3), which is mainly attributed to a simultaneous aging effect during the low-temperature aluminisation process. Additionally, the η phase Ni3(Ti,Nb,Ta) with a D024 hexagonal structure is generally regarded as a detrimental phase for the tensile, corrosion and creep-rupture properties.26,27

The OM, SEM and TEM images of the substrate structures of the Al-coating (a1, a2, a3, a4) AT1000, (b1, b2, b3, b4) AT890 and (c1, c2, c3, c4) AT620 Ni–Co base superalloys.
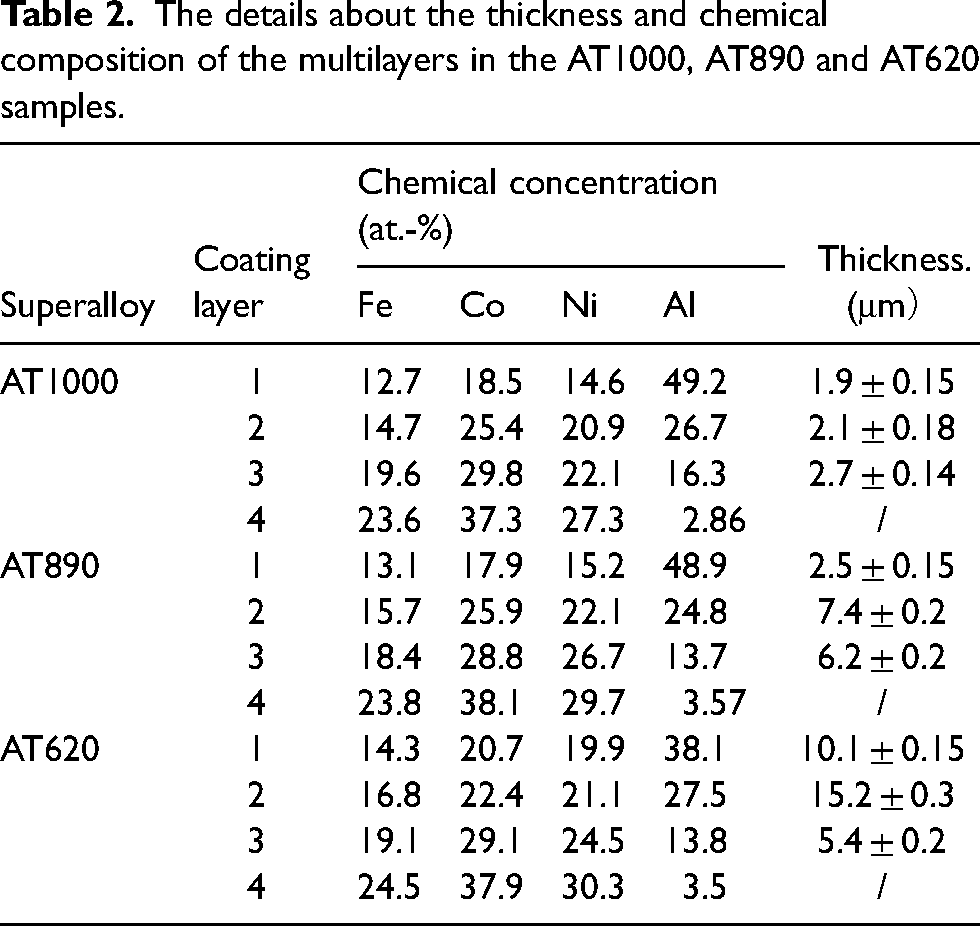
Figure 3 shows the morphologies and chemical concentration distributions of the cross-sectional Al-rich coating of the AT1000, AT890 and AT620 Ni–Co-base superalloys. The AT620 sample shows the largest thickness of Al-rich coating than those of the AT890 and AT1000 samples due to the longest aluminisation time (Figure 3(a)–(c)). Moreover, an intermediate layer between the substrate and the coating can be observed after all the aluminisation processes in the local-magnified SEM image. The elemental analysis (Figure 3(g) and (h)) also proves the existence of such multi-layer coating in the three samples, that is, the (Ni, Fe, Co)Al phase in the outer layer (layer 1), the (Ni, Fe, Co)3Al phase in the secondary layer (layer 2) and the intermediate layer (layer 3). The existence of an intermediate layer means that elemental diffusion occurs (especially for Al) between the coating layer and the substrate. The details about the thickness and chemical composition of the multilayers can be seen in Table 2.
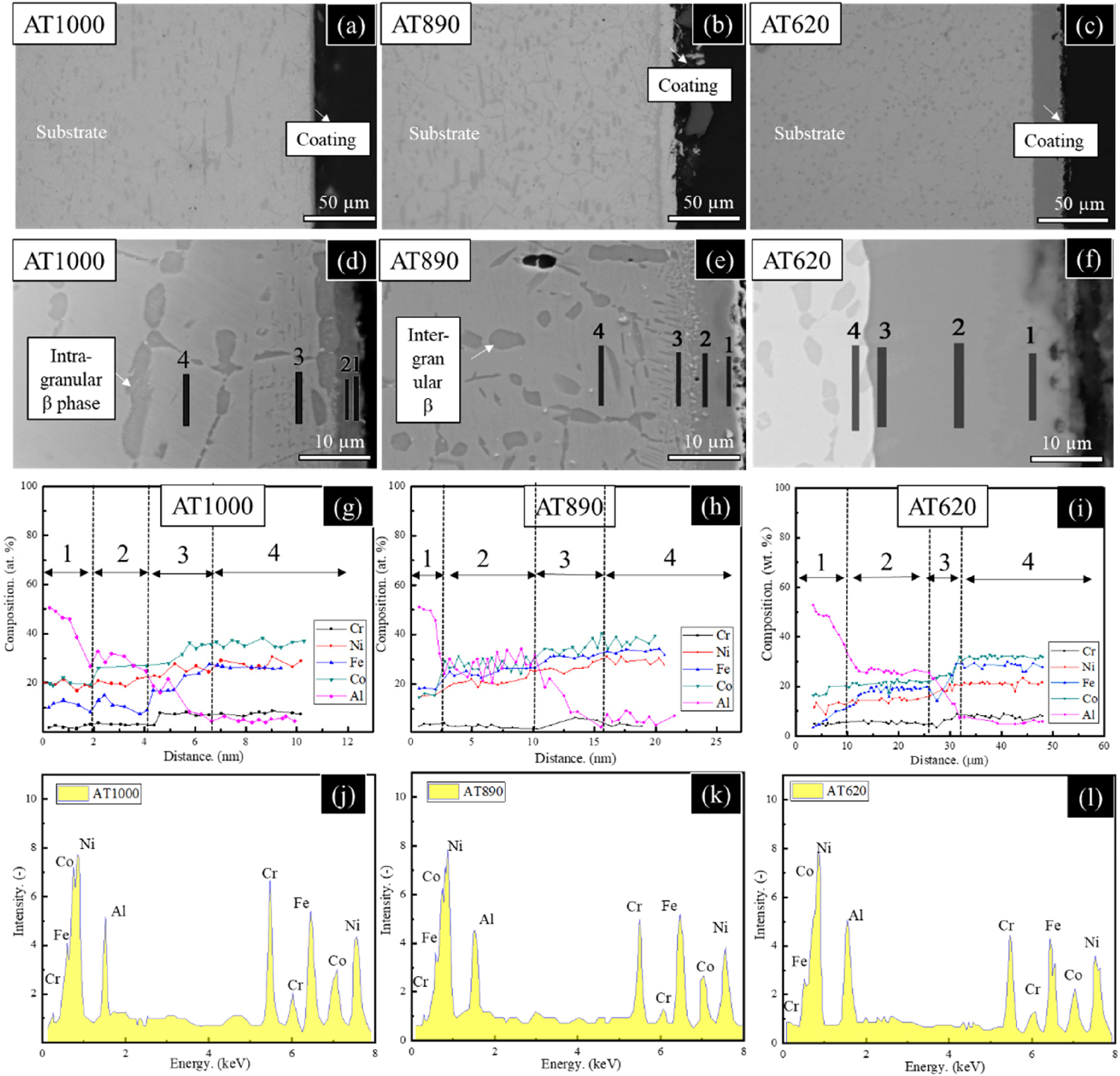
The morphologies, chemical concentration distributions and corresponding EDS spectrums of the cross-sectional Al-rich coating of the (a, d, g, j) AT1000, (b, e, h, k) AT890, and (c, f, i, l) AT620 Ni–Co base superalloys.
The details about the thickness and chemical composition of the multilayers in the AT1000, AT890 and AT620 samples.
The oxidation kinetics of the Ni–Co-base superalloys under different aluminisation procedures are shown in Figure 4(a), which presents the mass change per area of all samples during the oxidation test. All the curves show a similar parabolic tendency with time increasing, and the accuracy value R2 is considered reliable when it is greater than 0.85. Meanwhile, the mass change of the AT620 sample is much lower than that of the AT890 and AT1000 samples, indicating the highest oxidation resistance in the same environmental conditions. To quantify the oxidation rate, a parabolic rate constant formula is given as follows:
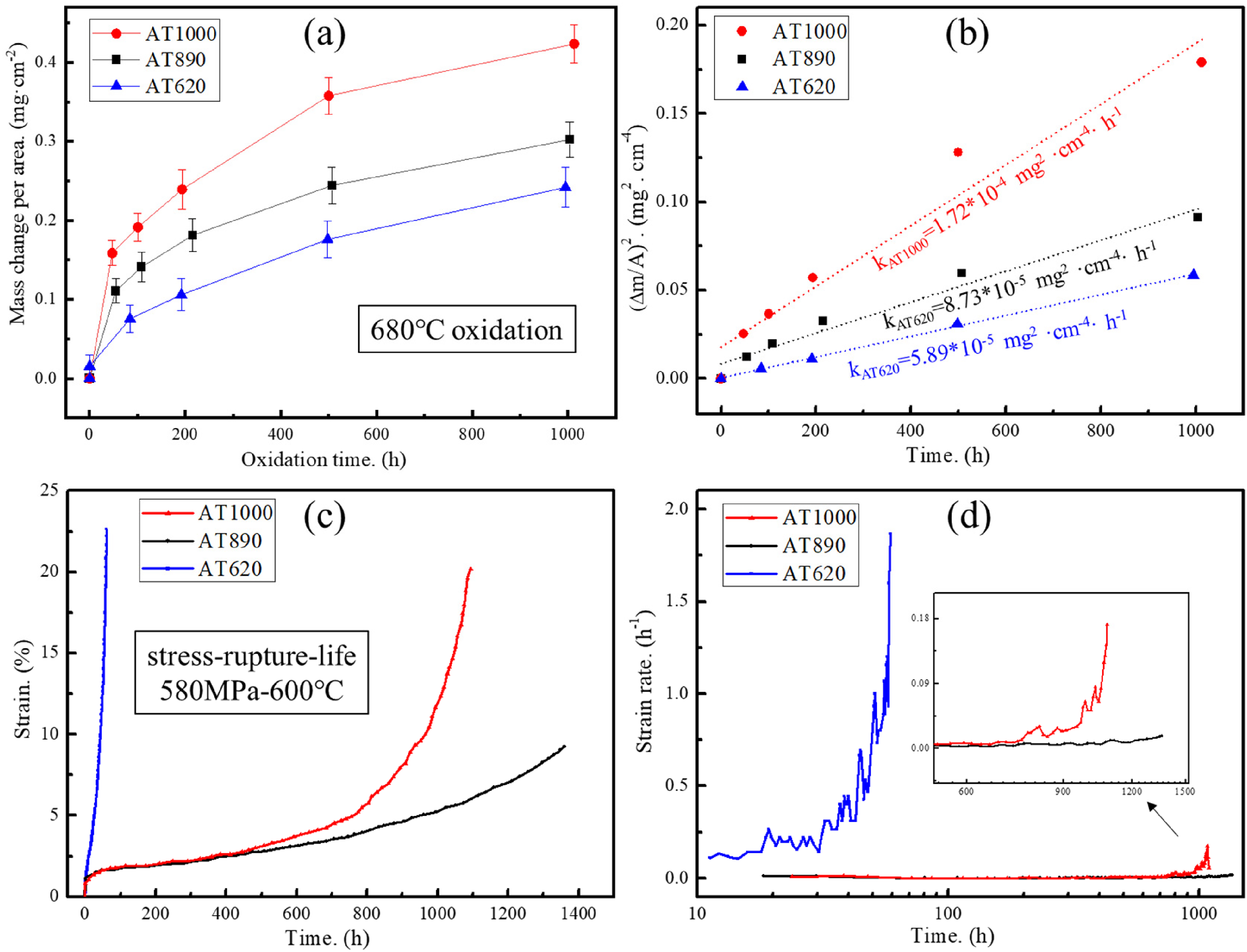
(a, b) The oxidation kinetics and (c, d) high-temperature stress rupture behaviour of DEN samples for the Al-rich coating AT1000, AT890 and AT620 Ni–Co base superalloys.
Moreover, the OM and SEM images of the oxidised cross-sections have been given in Figure 5(a)–(d). Under an equivalent oxidation condition, it can be seen that the AT1000 sample possesses a much larger thickness of oxidation layer than that of the AT620 sample. The magnified SEM images show that the permeation of the oxidation layer has passed through and overlapped the Al-coating layer of the AT1000 samples, whilst it is hindered by the outer coating layer of the AT620 sample. To investigate the elemental variation beneath the oxidation layer, the chemical concentration distributions of alloy elements are shown in Figure 5(e) and (f). A stepwise decrease in the oxygen concentration is observed along the cross-section of the AT620 samples, indicating an effective inhibition of oxygen diffusion across the multi-layer coating. The formation of the outer alumina layer may be attributed to the elemental inter-reaction in the outer thick (Ni, Fe, Co)Al phase, and it also contributes to enhanced oxidation resistance. However, the AT1000 sample shows a smooth decrease in the oxygen content, which demonstrates a low ability of preventing oxygen permeation. Briefly, the oxygen penetrates all the sample surface during the early oxidation stage, however, the formation of the outer alumina layer in the AT620 samples can inhibit its further diffusion to the substate. In contrast, the in-depth diffusion of oxygen occurs in the AT1000 samples due to a thinner Al-rich coating, especially the outer (Ni, Fe, Co)Al phase, which plays an important role in the oxidation resistance.
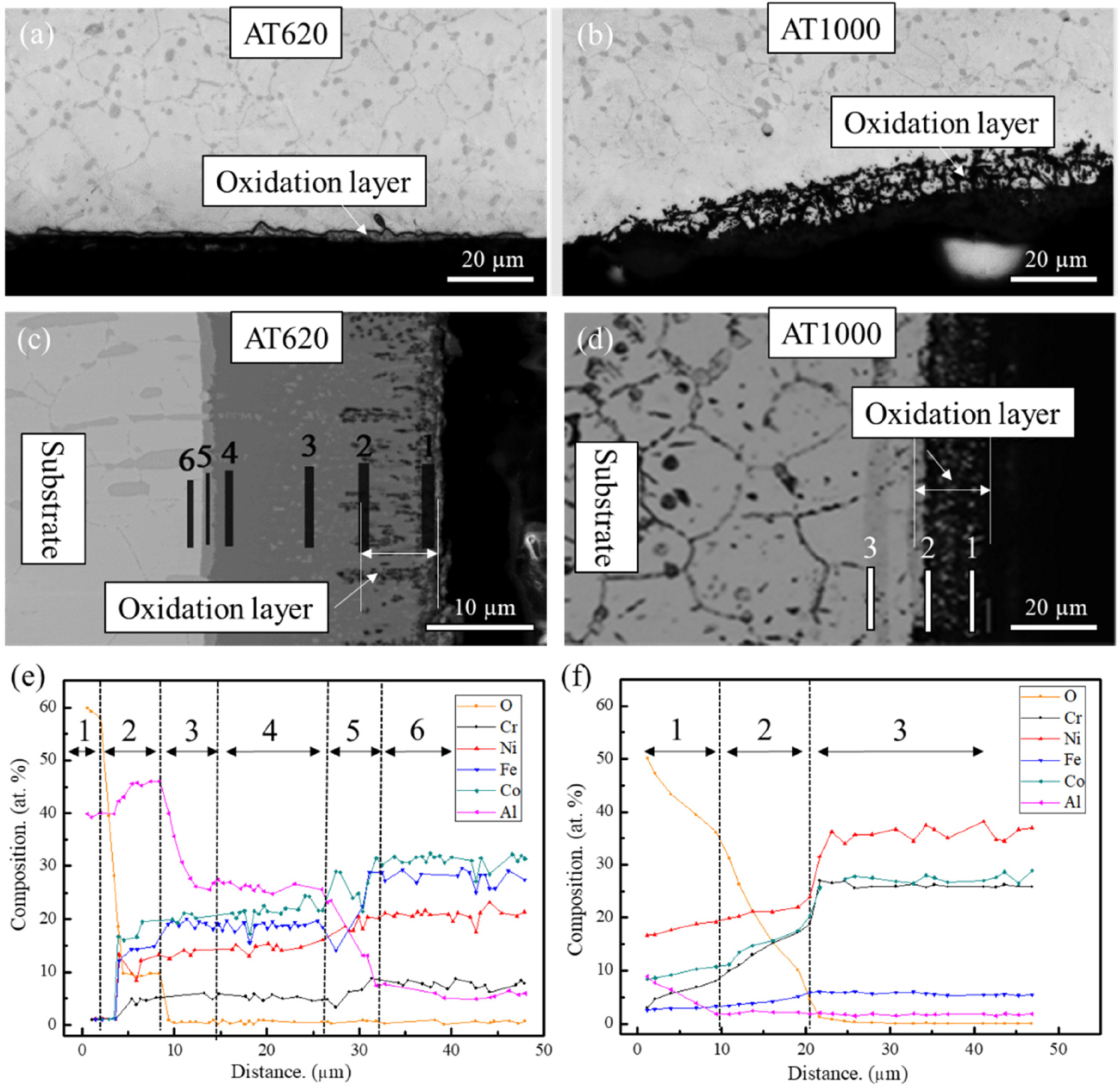
The morphologies and chemical concentration distributions of the cross-sectional Al-rich coating of the (a, c, e) AT620 and (b, d, f) AT1000 Ni–Co base superalloy after oxidation at 680 °C for 500 h.
The cross-section coating structures of the DEN samples after stress rupture tests for 900 h are shown in Figure 6(a)–(c). An obvious micro-crack can be found in the AT620 samples, and it nucleates in the outer coating layer and propagates into the transition layer. However, the cracks are hardly observed in the AT890 and AT1000 samples, corresponding to a higher resistance to rupture cracks. Moreover, Figure 6(d) and (e) reveals the TEM substructures of the AT890 and AT1000 samples after creeping for 900 h. In the AT1000 samples, the rod-like σ phase can be observed in the substrate close to the coating, and the double beam diffraction proves that some dislocation networks occur in the interfaces between the σ phase and surrounding γ′ precipitates. Meanwhile in the AT890 samples, the MC phase nucleates after an equivalent testing time, also accompanied by large amounts of dislocation tangles. Thus, the difference in crack sensitivity between these two samples can be attributed to the impacts of different precipitations on the stress rupture behaviour.

(a, b, c) The cross-sectional coating structures and (d, e) the TEM substructures of the substrates close to the coating of the DEN samples after stress rupture tests for 900 h.
As illustrated, the aluminisation procedure plays an important role in the oxidation performance of the outer coating structure and also significantly influences the high-temperature stress rupture behaviour and damage mechanisms. Firstly, a higher aluminisation temperature is not necessary for a thicker Al-rich coating, which also strongly depends on the diffusion time. Although the formation of alumina is generally considered as a protective phase to prevent in-depth high temperature oxidation, Fahamsyah et al.
28
found that the occurrence of phase transitions of alumina at appropriate temperature and time strongly affected the oxidation rate of an aluminised Ni-based single-crystal superalloy. Jalowicka
29
attributed the formation and growth of alumina during oxidation to the Al depletion in the NiAl coating of an aluminised Ni-based superalloy. In this work, the AT620 sample possesses the lowest oxidation rate (
Moreover, the DEN AT620 sample shows the highest rupture strain at elevated temperatures. Although the multi-layer aluminide of the AT620 sample possessed a significantly higher thickness than that of the AT890 and AT1000 samples (Figure 3), the micro-cracks were prone to nucleate in the dense aluminide layers of the latter. This phenomenon can be explained as follows: firstly, the abundant elemental interaction during high-temperature rupture would result in the formation of an outer alumina layer, which exhibits a strong deformation incompatibility 21 with the superalloy matrix due to a significant difference in hardness. Figure 7 shows the change of nano-hardness with the distance far from the film surface of the AT620 and AT890 samples after stress rupture tests for 900 h. The results indicated that a larger hardness difference existed between the different aluminide layer in the AT620 sample, and the outer alumina layer possessed a lower hardness value than the intermetallic layer. Therefore, a strong strain concentration occurred in the soft alumina layer during the stress rupture. The micro-cracks were prone to nucleate in the weak interfaces, propagated into the internal substrate, and led to a premature failure. Moreover, a larger amount of detrimental η phase in the AT620 sample (Figure 1) might also result in a brittle-intergranular fracture. 26
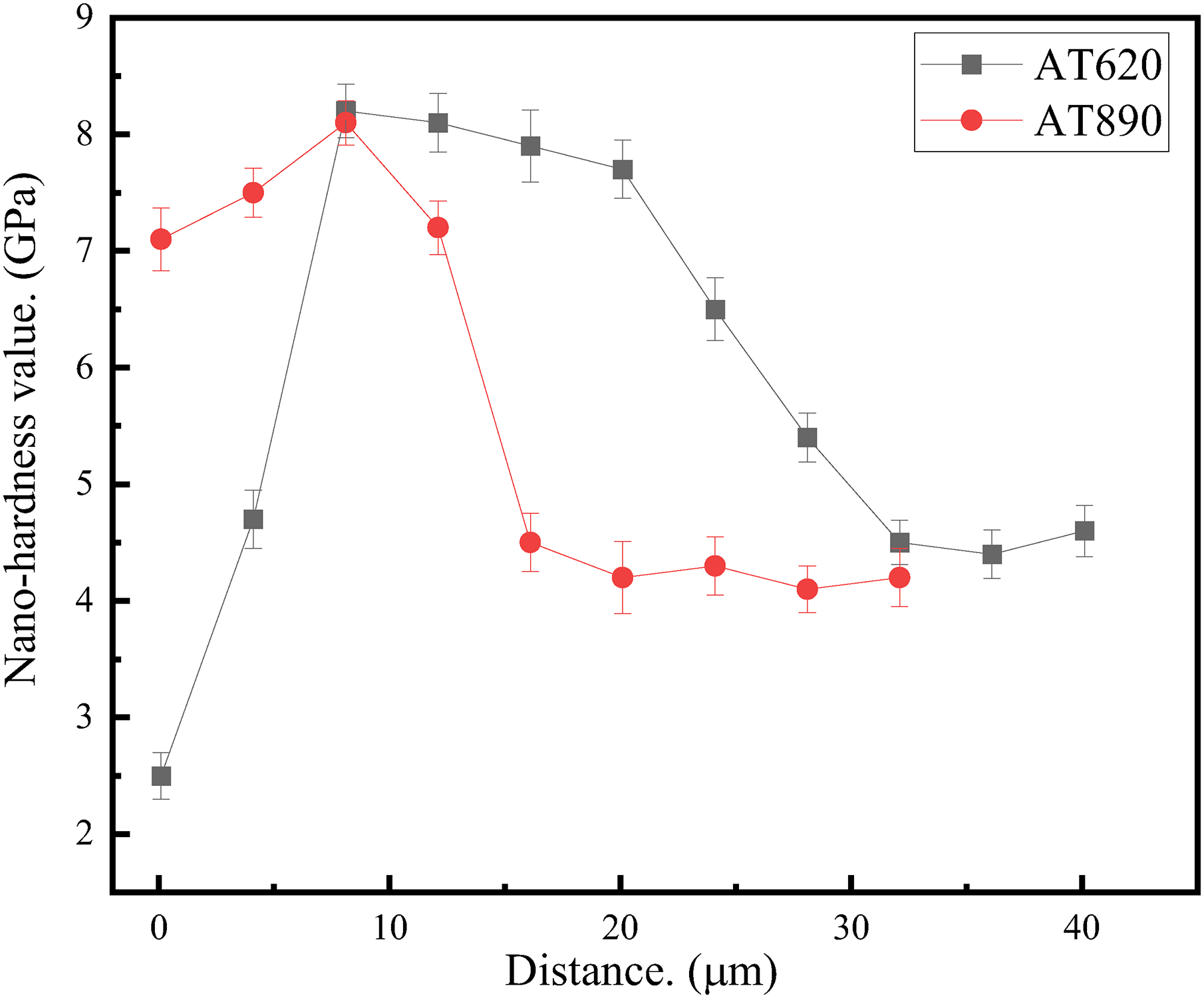
The change of nano-hardness with the distance far from the film surface of the AT620 and AT890 samples after stress rupture tests for 900 h.
On the other hand, the AT890 samples exhibit simultaneously higher oxidation and crack resistance than the AT1000 samples. The cracks were hardly found in the aluminide coating of both samples. Thus, the crack resistance of both samples was closely related to the roles of various precipitates. Both the AT1000 and AT890 samples consisted of the γ′ phase, β phase and η phase. Amongst these, (i) the γ′ precipitates acted as strengthening phase to inhibit the early crack development during creep, 30 (ii) the β-NiAl phases in GBs were demonstrated to enhance the resistance to crack propagation by preventing oxygen diffusion 31 and (iii) the plate-like η phase precipitates26,27 were likely to be detrimental for the tensile, corrosion and creep-rupture properties. On the basis, a significant coarsening of the irregular γ′ phase would result in its decreased number density and strengthening effect, thus leading to easy crack formation in the AT1000 samples after long-term stress rupture. However, the contribution of the β and η phases to the crack sensitivity could be ignored due to a little difference between the AT1000 and AT890 samples.
Moreover, the Cr-rich σ phase nucleated in the AT1000 samples whilst the MC carbides were mainly observed in the AT890 samples during stress rupture. The deleterious Cr-rich σ phase 32 not only consumes the neighbouring γ′ precipitates in the AT1000 samples, but also becomes the nucleating site of dislocation networks and strain concentration. However, an effective diffusion of Al results in a thicker transition layer and a higher average Al content in the neighbouring substrate of the AT890 samples (Table 2), thus suppressing the formation of the σ phase, whilst the precipitation of MC carbide would not cause premature failure during stress rupture. On one hand, the MC carbide can provide additional precipitation strengthening and pin the grain boundary to increase the creep performance, 1 On the other hand, a high interface coherency of the spherical MC carbide contributes little to the stress concentration due to dislocation pining-ups during deformation, which improves the stress rupture properties during long-term service. 33 The difference in crack sensitivity between the AT890 and AT1000 samples can be attributed to the impacts of different precipitations on the stress rupture behaviour.
Conclusions
This work studied the role of the aluminisation procedure on the microstructure evolution, oxidation resistance and crack sensitivity of a Ni–Co-base superalloy system. The high-temperature oxidation behaviour was quantitatively evaluated and analysed based on the thickness and chemical composition of the Al-rich coating. Moreover, the damage mechanisms during the stress rupture test were revealed in terms of substructure evolution. The main conclusions can be drawn as follows:
The AT620 sample shows the largest thickness of Al-rich coating than those of the AT890 and AT1000 samples due to the longest aluminisation time. All the substrates exhibit similar constitutions of the γ matrix, intra- and inter-granular β phases. A multi-layer coating, consisting of (Ni, Fe, Co)Al phase, (Ni, Fe, Co)3Al phase and transition layer, exists in all samples. The parabolic curves indicate that the oxidation rate of the AT620 sample is much lower than that of the AT890 and AT1000 samples. The obtained k values for the AT1000, AT890 and AT600 samples are 1.72 × 10−5, 8.73 × 10−4 and 5.89 × 10−4 mg2 cm−4, respectively. The highest oxidation resistance of the AT620 sample may originate from the combined advantages of a large coating thickness (∼28.6 μm) and the additional excellent oxidation-resistant Al2O3 layer, which may be associated with the Al inter-diffusion in the outer (Ni, Fe, Co)Al phase. The AT620 DEN sample exhibits the highest high-temperature rupture strain, and the micro-cracks nucleate only after testing for 90 h due to the strong strain incompatibility between substrate and coating. However, the high-temperature strain rate of the AT890 sample almost remains stable even until 1200 h, demonstrating a strong resistance to creep-induced crack, which can be attributed to the suppression of the σ phase due to an effective inter-diffusion of Al in the transition layer.
Footnotes
Acknowledgements
The authors are grateful to the financial support of the National Nature Science Foundation of China (No. 52001213). Also, this work is sponsored by the Shanghai Sailing Program (20YF1447200) and “Chen Guang” project(20CG65) supported by Shanghai Municipal Education Commission and Shanghai Education Development Foundation, Natural Science Foundation of Shanghai (20ZR1455300), and the introducing talents project of Shanghai Institute of Technology (YJ2021-4).
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
