Abstract
This study reports the results of a qualitative study of patient experiences of receiving treatment for wet age-related macular degeneration with ranibizumab (Lucentis)(R). Treatment involved monthly hospital visits for assessment and, where required, an intravitreal Lucentis injection. Qualitative narrative interviews were conducted with 22 patients, 18 of whom received treatment and were interviewed at two points during their treatment journey. Interviews allowed participants to reflect on their experiences of being assessed for and receiving this treatment. Overall, treated participants reported that while they had been apprehensive about treatment, the actual experience of it was far less unpleasant than they had expected. However, the data also revealed a number of issues surrounding the provision of information about treatment, as well as service delivery issues, which had considerable impact upon their experience.
Keywords
Background
Age-related macular degeneration (AMD) is the leading cause of irreversible sight loss among older people in the United Kingdom (Minassian, Reidy, Lightstone, & Desai, 2011). Of its two principal forms (wet and dry), it is the wet form that accounts for most visual disability, attributable for 90% of cases of severe sight loss (The Royal College of Ophthalmologists, 2007). Owen et al. (2012) estimate that the prevalence of wet AMD in the United Kingdom is 263,000 cases, with 37,700 new ones diagnosed each year, figures which are expected to rise as the population ages. Wet AMD causes rapid loss of central vision and usually affects both eyes, though not necessarily at the same time.
Treatment options for wet AMD have expanded in recent years. Ranibizumab (brand name Lucentis)(R) is a drug that prevents further vision loss in approximately 95% of patients and improves it in 40% (Lim, Mitchell, Seddon, Holz, & Wong, 2012). The National Institute for Health and Clinical Excellence (NICE) approved Lucentis for the treatment of wet AMD in 2008. Subsequently, there has been an exponential increase in the number of people receiving this treatment, creating financial as well as service capacity pressures in the National Health Service (NHS) (Amoaku, 2009; Keenan, Wotton, & Goldacre, 2011).
Lucentis is delivered via an intravitreal injection (an injection into the eye). Patients receiving Lucentis typically require three initial injections at monthly intervals, followed by monthly visits for assessment and further injection as necessary over approximately 24 months. After 24 months, further treatment may be given if appropriate (Harding, 2010; The Royal College of Ophthalmologists, 2009). The average number of injections received by patients has been estimated to be 14 in the first 2 years of treatment (Mitchell et al., 2009), with the optimum number and timing of injections after 24 months yet to be established. While the clinical efficacy of this treatment is well established, patients’ experiences of receiving it are less well understood. The aim of this study was to explore patients’ experiences of Lucentis treatment. Issues examined included access to treatment, the experience of undergoing assessments, expectations of the procedure and the treatment outcome, and actual experiences of, and feelings about, receiving treatment.
Method
Participant recruitment
With local ethical and NHS Research Governance approval, 22 participants with wet AMD were recruited through a hospital AMD clinic. The sample comprised 13 women and 9 men with a mean age of 77 years (range = 51–93 years).
Data collection methods
A Biographical Narrative Interpretive Method (BNIM) interview (Wengraf, 2005) was used at two points in time to explore participants’ expectations and experiences of AMD treatment. BNIM uses a single opening question, designed to elicit a narrative response, with minimal intervention from the interviewer as the participant ‘tells their story’. Once the participant has finished, the interviewer seeks further narrative responses using questions based upon the information provided in the initial response to the question. The value of narrative research methods has been demonstrated in a number of vision-related studies (Ainlay, 1989; Papadopoulos, Scanlon, & Lees, 2002; Thetford, Robinson, Knox, Mehta, & Wong, 2008, 2011). However, we found that with our participants, the resulting narratives were relatively short. We therefore modified the interview technique so that participants’ narrative responses to the initial question, as well as issues identified in interviews with other participants, were used as a starting point for further question development.
Of the 22 participants recruited, 18 were interviewed twice in order to capture expectations and feelings about the treatment and to compare this with their actual experience of the treatment. Twelve participants completed their first interview within a few days of their first injection, and 10 participants were interviewed either while they were waiting for confirmation that they would be treated or while waiting for treatment. Of these 10 participants, four did not receive treatment. Of the remaining six participants who were interviewed prior to receiving treatment, three did not know whether or not they would be treated at the time of their initial interview. Participant involvement is depicted in the flow chart in Figure 1.
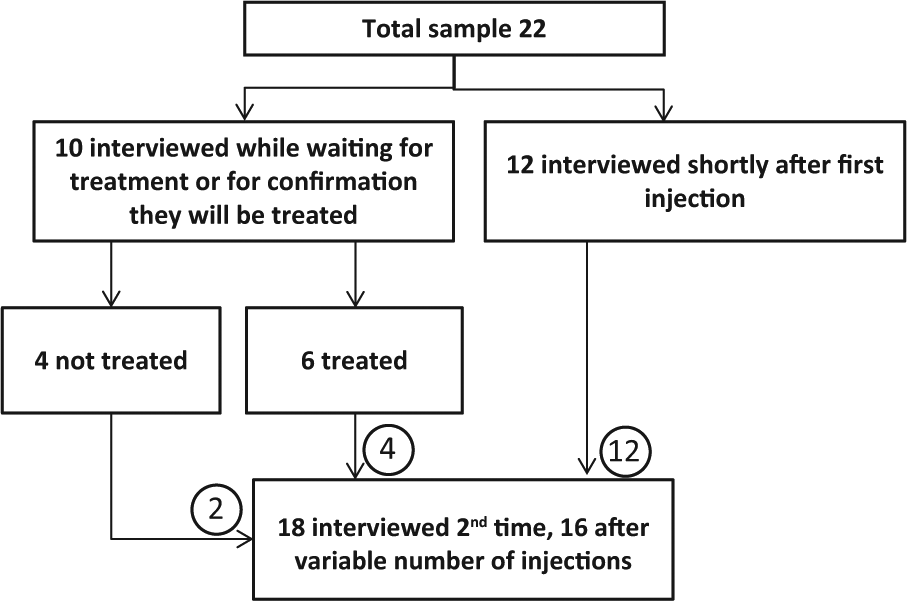
Participant involvement in the study.
The time between the first and second interviews ranged from 2 to 9.5 months, and the number of injections received varied from two to eight; only four participants received more than three injections. Interviews were conducted in participants’ homes with one exception (interview at the university). Interviews lasted between 30 min and 3 hr and were digitally recorded and transcribed. In the initial interview, the BNIM approach was used, as discussed earlier. The second interviews took a similar form to the first interviews but included more direct questioning based on the information provided in the first interviews (Wengraf, 2005).
Data analysis
Interview data were analysed thematically. In developing a coding scheme, each of three different researchers within the team independently coded three interview transcripts. The three sets of codes were then compared, identifying variation and agreement and the description of the codes assigned to text. On the basis of this comparison, a detailed coding scheme was developed and was used to code all transcripts using NVivo software. Transcripts were analysed to identify sections of text that informed understandings of the general issues for participants (Coffey & Atkinson, 1996; Silverman, 2012). Data were compared within each case (between Interviews 1 and 2) and across individuals as well as groups of participants. Each concept was assigned a descriptive code, and these codes were combined into conceptual categories that later formed broader key themes.
Results
Findings from the narrative data presented here cover four key themes: anxiety and fear of ‘the unknown’, the injection procedure, side effects, and service delivery. Pseudonyms are given for the participants and which interview the data were from (T1 or T2) is shown.
Anxiety and fear of the ‘unknown’
When discussing their feelings about treatment, participants often focussed on the injection. They reported varying levels of anxiety and apprehension about this, ranging from a small number who were very fearful, to others who were barely concerned; most reported feelings between these extremes. Pauline and Kathleen reported being very frightened at the idea of having an injection into the eye. However, the threat of losing their vision was more frightening, so they proceeded with the treatment. For Pauline, not knowing exactly what the procedure involved was most frightening – the fear of the unknown:
The procedure itself . . . again, I won’t pretend I was brave – I was absolutely petrified, that was because you just don’t know what’s going to happen. (Pauline T1) I did seriously consider not bothering because I was petrified of the idea of this injection. (Pauline T2)
Kathleen had attended the clinic for assessments for a number of months before being offered treatment. She was so frightened by the notion of an injection into the eye that she deferred treatment twice:
He (ophthalmologist) said, I think we will start treatment today, and I said, no. I was frightened. I said, not today. I said I’m babysitting. I watch my daughter’s children, but I just didn’t want it done then . . . They said, I think you should have treatment, and I said, no doctor, please, I have got to get home for these kids. God forgive me, I didn’t have them; I just didn’t want it done. (Kathleen T2)
After the second deferral, she realised that if she was to preserve her vision, she would have to be treated and returned to have the injection a month later:
When I come home and I knew the time was coming near, you know to get that first needle. And I know I have to get it done, you see people just getting in and walking out. They were alright. I [thought] when they give me the needle I would collapse in the chair and everything. No, no but it was alright. (Kathleen T2)
Similarly, for Betty, ‘the fear of having the injection was far worse than having the injection itself’ (Betty T2).
A lack of information often compounded the feelings of anxiety about the treatment:
It’s just the apprehension of not knowing, which adds to the tension and the nerves, and you know, the first time . . . not knowing what the actual procedure is going to be . . . knowing whether it is going to hurt. (Mary T2)
These participants felt that much of their anxiety was rooted in their fear of the ‘unknown’. Being told that treatment involved injections into the eye, but having little further information or opportunity to discuss the procedure in detail, appears to have fuelled their fears. However, some participants reported that they deliberately did not seek further information. In some cases, this was because they trusted the doctors to do whatever was needed:
I must admit, I don’t ask any questions. I just don’t ask any because I just put myself in their hands and say, well, they think it’s necessary to do this. (Ron T1)
For others, it seemed that managing the amount of information they had was a coping strategy. Pauline, in particular, restricted the amount of information she received about both her condition and the treatment in order to ensure that she was not overwhelmed with more information than she could process or deal with emotionally. Thus, although she reported that her initial fears and anxieties surrounded ‘the unknown’, she had purposefully limited how much information she had elicited:
I haven’t done a lot of research because I don’t want to . . . I just look . . . when I . . . I actually sat with my friend in work and I was like, you get it up on the Internet, read me a sentence at a time, and I will tell you when to stop. And she was like . . . usually expect to go three times . . . and I was like stop, stop, stop. What? More than one injection, more than one?! Right, let’s leave it. (Pauline T2)
Our data suggested that for most of our participants, it was not until they were in the treatment room that the procedure was fully explained to them, and that this explanation was provided by the nurses. Participants spoke positively of the nurses’ accessible explanations of what would happen, providing reassurance and support throughout the procedure:
I think fully explaining, I think, is when you are actually in the room where you are going to have [the injection] and they are getting you ready. ‘Cause obviously they have to cover everything else up and explain to you what’s going on. So yeah, they are very good explaining. I think the sisters are better explaining than the doctors. (Mary T2)
Participants’ actual experience of the initial injection dissipated much of their apprehension, even among those who reported high levels of anxiety. It became much easier after the initial injection as they then knew what to expect:
[the] first one, I was a bit apprehensive but other than that, once that was done there was no problem. (Jim T2) if I had known what I know now, there is no way I would have gone through all the stress of that. It’s the ordinary stuff that worries you . . . your mind starts thinking, oh do they pull your eyelid down and go through the eye, they can’t go through . . . but having the procedure next time I will actually be okay. (Pauline T2) Very apprehensive with the first one. The second one I wasn’t so bothered. (Mary T2)
While a small number of participants reported not being particularly concerned about the procedure either before or after their injection, they confirmed that any apprehension was again dispelled following their initial injection.
Injection procedure
Participants tended to break down the treatment procedure into three key components: application of eye drops, draping, and needle entry. They commonly reported that they disliked the various eye drops used in preparation for the injection, particularly where these caused a stinging sensation:
I mean, eye drops are horrible, when somebody’s poising with those to drop on you. (Ethel T1) It’s not so much of the injection, it’s the preparation . . . I’m alright with the drops but when they put the iodine in it really smarts. (Hannah T2)
A sterile drape covers the face and is used to reduce the risk of infection. While some participants reported that they had not liked having this placed over their face, it was the removal of the drape which they found most unpleasant. It was described as being like a sticky plaster, but because it was attached to the sensitive eye area and held back the eye lashes, it was more unpleasant and sometimes painful when removed. This was reported to be at least as unpleasant as the needle entry:
Of course they have to whip it [drape] off quickly. It’s like a sticky plaster and that hurts quite a bit. (Ethel T1) That [drape] hurts more than the injection . . . It sticks easy, it’s a wonder I’ve got any eyebrows left! (Beryl T2)
Most participants reported that they had been told that they would not feel the needle upon entry or, certainly, any pain. Generally, the injection was described as painless, with some participants not realising it had been completed. However, others felt pressure or a small scratch upon needle entry:
I felt it going in and I thought with the anaesthetic drops I wouldn’t. But I knew it was in but it wasn’t terribly painful. (Alan T1) I felt that little bit of a sharp thing in my eye then, and I knew then that it was done. (Hannah T1) It’s virtually painless. You do feel a little prick as it goes in but . . . it’s just a feeling, yeah. It doesn’t sting or anything. As I say, the eye drops, they sting, they are worse. (Jim T2)
Ethel reported the needle entry to be painful, though not unbearable. She considered that some pain was inevitable from having an injection into the eye and so she had no cause for complaint:
The injection of course goes right into your eye, I mean if anybody stabs you anywhere it’s going to be painful isn’t it? (Ethel T1)
Some participants reported ‘jumping’ or ‘pulling away’ at the time of needle entry, and that this was a reflexive response they felt they had no control over. Bridget ‘pulled away’ just before the needle entered her eye, resulting in her eye being scratched and the procedure being repeated. Although she felt like ‘running away’, she had the injection and despite being quite anxious about the procedure felt relieved about it afterwards:
You feel a pressure and this time I went like that, and I pulled away, which I shouldn’t have done . . . I couldn’t get up and run [away] . . . I felt like it . . . but it’s fine. (Bridget T1)
Everyone reported the procedure to be over quickly. The preparation took 10–15 min, and the injection itself and final checks just a few minutes. This was considered to be a key element that meant the procedure was bearable even if unpleasant:
It’s not that bad and at least it’s over very quickly . . . well they put some plastic thing over that side of your face, like Phantom of the Opera thing, and then sort of cut a little bit and put callipers to hold it open, but it’s all done that quickly that you are up and sort of, it’s all done before you realise it. (Burt T1)
Overall, participants described the procedure as unpleasant but manageable. More importantly, as they valued their sight so much, a large majority of this sample had not considered refusing the treatment. Indeed, most suggested that they would undergo whatever treatment was necessary to preserve their vision, regardless of whether or not it was unpleasant or painful:
If it’s going to improve my eyesight, I will go with it. I am not going to sort of say, no I am not having that injection in my eye. I will do whatever it takes like, you know, I value my eyesight, I will do everything I can to try and keep it as long as possible. (Burt T1)
Side effects
Most reports of side effects associated with Lucentis in the literature focus upon clinically serious, though rare, side effects such as stroke, serious eye infection, bleeding, retinal detachment and tear, and lens damage (Bentley, Michel, Tellis, Keeffe, & Clarke, 2012). None of our participants were affected by any of these. However, several reported a number of clinically non-significant side effects such as bloodshot or watering eyes, pain, general discomfort, and visual distortions. A small number reported pain or irritation, which impacted their experience of treatment. However, although painful and, in some cases, frightening, none of the participants indicated that these side effects would cause them to consider discontinuing treatment:
There was no problem having the injection but that evening the eye was very sore and I was getting a bit worried but after a night’s sleep it eased off and it became alright. (Jim T2) It is a little bit sore once the anaesthetic wears off but it’s only like as if you have got a bit of grit in your eye, it’s nothing drastic. (Betty T2) I didn’t feel anything, they injected my eye then I had some more drops. In the evening it was smarting a bit, you know, when the pain killer faded away it was a little bit painful. Little bit, but the next morning it was okay. (Ron T1)
A small number reported high levels of pain following the injection, which they had not expected. While unpleasant and frightening, these symptoms were short lived and usually lasted not more than a day and a half:
that night, it was horrendous, I have never had pain like it . . . it was excruciating. (Pauline T2)
Where participants reported having been advised of the possibility of these side effects, they stated that this meant they were less likely to feel panicked by them. However, when unexpected and painful, they were frightening. It was unclear from our data whether or not our sample discussed what might happen after the injection with clinical staff or whether they only received written information.
Participants reported being provided with an information sheet that detailed some of the side effects that they might experience and contact details if they had concerns. Several participants made use of these contact details out of hours and stated that they valued having this information and spoke very positively about the service they received. The information sheet advised that some of these side effects were possible, but did not cover all of the symptoms experienced.
Service delivery
Each appointment involved a number of tests with several different professionals and lasted several hours; participants reported that it was not unusual to be at the clinic all day. They spent a great deal of their interview talking about issues surrounding service delivery. Waiting times and the amount of time spent at the clinic on each assessment or treatment day featured in most accounts.
For some, the amount of time waiting at the clinic to discover whether and/or when they could be treated and what the treatment would involve considerably fuelled their feelings of anxiety and agitation:
You do [get] a bit fed up sitting down waiting for your name to be called, you know, it’s a hospital so you expect them things don’t you, you’ve got to wait, but you get a bit agitated. (Hannah T2) everyone’s very wound up. At the end of the day you are waiting for them to say whether you are having an injection in your eye or not. (Pauline T2)
Despite their feelings of frustration and agitation with the service delivery, most downplayed this in the context of their gratitude for a treatment that would likely preserve their vision. Just as they suggested they would undergo almost any treatment procedure, they also indicated that they were willing to put up with what they perceived to be poor service in order to get it:
If you are going to see someone who can help you then waiting isn’t a problem. (Sarah T1) As far as I’m concerned that wipes out the day for that month. But if that keeps my eyesight good I am quite happy to do that forever. (Burt T2)
The data suggested that it was the first and second visits to the clinic which patients found most difficult (depending on whether they were attending for assessments or treatment only). First appointments involved a great deal of uncertainty, which participants reported to be anxiety-inducing, and having to wait for so long exacerbated this. These findings suggest that information and support for patients should be targeted at these early visits.
Overall, the data suggest that improvements to service delivery, particularly the amount of time spent waiting at the clinic, could potentially greatly improve patients’ experience of being assessed for and receiving treatment for wet AMD.
Discussion and recommendations
This article presents findings from an in-depth exploration of patients’ experiences of receiving treatment for wet AMD with Lucentis. This builds upon and complements the study of Beasley (2011) and Tailor, Beasley, Yang, and Narendran (2011), by exploring the issues they identified using quantitative methods in much greater depth to produce a fuller understanding of the patient experience. In particular, the qualitative approach used here has allowed us to explore
Our participants reported that their initial feelings of anxiety and apprehension about Lucentis injections were considerably reduced following an injection; many of their fears were rooted in the ‘unknown’ experience. As reported by Beasley (2011), the actual experience of the injection was far less unpleasant than participants expected it to be. None of our participants considered their experience of the procedure to be so unpleasant that it would deter them from returning for further treatment. Rather, they felt relief, and their feelings of fear and anxiety were alleviated after receiving the first injection. This finding complements previous research that assessed anxiety before and after Lucentis injections (Chua, Mitrut, Armbrecht, Aslam, & Dhillon, 2009) and found that 93% were not anxious or fearful after their first injection. Similarly, Roth, Scott, Gulati, and Prenner (2006) reported that patients experienced less discomfort than anticipated when receiving intravitreal injections for other conditions, an observation also reported for cataract surgery patients (Foggitt, 2001; Nijkamp et al., 2004).
However, although for participants in our study the actual experience of treatment was less unpleasant than feared, there were aspects of the process that caused pain and discomfort, particularly, the application of eye drops, the use of surgical drapes, and needle entry itself. Again, these findings mirror those of Tailor et al. (2011). Our participants found the removal of the drape particularly uncomfortable. Participants in Tailor et al.’s (2011) study disliked the application, rather than removal of the drape. This difference may be a reflection of the different types of drapes used. As in Beasley (2011) study, some participants in our study also experienced a degree of pain at needle entry. This suggests patients should be asked about the sensations they experience during the procedure and, where appropriate, offered alternative or additional anaesthesia.
Beasley (2011) began to explore issues surrounding patient information and anxiety levels for people receiving Lucentis, linking, as the participants in our sample have done, anxiety levels with information and communication. The provision of information about wet AMD, assessments, prognoses, and what is involved in treatment with Lucentis was highlighted as a key issue by our participants. It is clear from our data that patients’ need for information about their condition, prognosis, and treatment is highly varied, and so provision should be individualised, a finding also reported with cataract patients (Nijkamp et al., 2002) and in more general health-care settings (Beisecker & Beisecker, 1990; DeVoe, Wallace, & Fryer, 2009).
The literature also suggests that the information and communication needs of older patients may be different to those of younger patients (Greene, Adelman, Friedmann, & Charon, 1994; Robinson, White, & Houchins, 2006) which is of particular relevance in the case of treatment for wet AMD as the vast majority of patients are older (the average age of our sample was 77 years). The literature suggests that effective doctor–patient communication in ophthalmic settings is a key component of patient satisfaction (Mozaffarieh, Krepler, Heinzl, Scacu, & Wedrich, 2004; Murphy-Cullen & Larsen, 1984; Nijkamp, Nuijts, van den Borne, & Hendrikse, 2000; Nijkamp et al., 2002; Wooley, Kane, Hughes, & Wright, 1978). Inadequate provision of information has been highlighted as an issue in general AMD clinics where patients are diagnosed but not normally treated because dry AMD remains untreatable (Mitchell & Bradley, 2011).
Service delivery issues impacted greatly upon experiences of being assessed for and receiving treatment for wet AMD. In particular, the amount of time spent at the clinic at each appointment was a particular problem and added to feelings of anxiety and agitation. Tailor et al. (2011) found that patients considered waiting to have their injection to be one of the most unpleasant aspects of the whole process, while waiting times in general AMD clinics have also been identified as a source of patient dissatisfaction (Mitchell & Bradley, 2009). The timing and number of Lucentis injections are under considerable scrutiny in clinical trials. Amoaku (2009) highlighted the capacity pressures that monthly follow-up visits place upon clinics. Given that monthly visits place a substantial burden upon patients as well as clinical services, the patient’s perspective should be given due importance in these considerations. It should also be recognised that this patient group, comprising predominantly older people, is less likely to complain and more likely to report high levels of satisfaction than others (Ware et al., 2003).
Recommendations
Delivery of this service remains in its infancy; in developing future services, the issues identified here should be considered and improvements made where possible. Overall, participants’ experiences of receiving treatment were positive. However, two key issues were highlighted as areas where improvements might be made to improve patient experiences. The first issue, also identified elsewhere (Beasley, 2011; Chua et al., 2009; J. Mitchell & Bradley, 2011; Morrell, 2001; Mozaffarieh et al., 2004; Nijkamp et al., 2004; Trento et al., 2006), relates to communication and the provision of information to patients. This includes what will happen at the clinic when they attend for appointments, but particularly about exactly what the procedure and treatment regime involve, as well as expected outcomes. Improved and innovative ways of providing information are likely to reduce anxiety. Morrell (2001) proposed that patients who are more anxious about ophthalmic surgery are less able to remain still and keep their eye in the required position, something for which there was some evidence in our study. This reinforces the need to address patient anxiety levels.
The data suggest that the written information sheets could be improved, and that this information should also be delivered verbally, providing opportunities for patients to ask questions. This approach would allow for the information to be tailored to the individual. Other approaches such as explanatory videos (as have been used for cataract surgery; Pager, 2005; Yuen, Cheng, & Chan, 2005) and peer support (other patients who have had the treatment themselves) might play a role. However, our findings suggest that these should be used to supplement, rather than replace the provision of information and reassurance. The timing of the provision of such information is, however, likely to remain problematic, particularly at ‘one-stop’ clinics where patients’ suitability for treatment is often assessed on the same day as they are offered treatment.
The second issue surrounds service delivery; the amount of time spent at the clinic at each appointment, much of which was reported to be spent waiting, featured in most accounts. Not only is this waiting frustrating, but for some it also fuelled feelings of anxiety surrounding the procedure. For people who have other health and mobility problems, or who require a family member to accompany them to their appointment, this presents additional challenges. In addition to undergoing a number of assessments with different staff, the waiting was reported to be tiring and for some, overwhelming. This further complicates the best timing for provision of information as it is unlikely that patients will be able to understand and fully consider a lot of information.
UK eye clinics have had to respond to a huge increase in demand for treatment very quickly. As such, services and physical environments involved in the delivery of this treatment are still evolving; with an ageing population and further development of treatments, this pressure will only increase in the future (Amoaku, 2009). This is set within the context of a huge growth in a wide range of day surgery (Gilmartin & Wright, 2007; Mitchell, 2003), and so the findings of this study are likely to have wide applicability. Meeting these demands, particularly during difficult economic times, will be a continuing challenge. However, given the impact of service delivery issues upon treatment experiences identified here, it is necessary to explore ways in which this could be improved. For example, if the amount of time spent at the clinic cannot be reduced, then a waiting lounge with more comfortable facilities might be appropriate.
Study limitations
This study recruited participants from one eye clinic in England. Some of the issues surrounding service delivery in particular may be specific to this clinic, or to this model of service delivery. Similarly, many of the issues reported may have application in other ophthalmic settings, such as general eye clinics. Without conducting further research across other clinics, we cannot be sure of the degree to which these issues are generalisable. However, it is likely that most of these issues have applicability elsewhere.
Footnotes
Acknowledgements
We would like to express our gratitude to the eye clinic staff in assisting with recruitment of participants and in reviewing clinical data. We would especially like to thank the participants who took part in the study who gave their time to share their experiences for the benefit of others.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This project was funded by the Macular Society.
