Abstract
Low-cost activated carbon electrodes derived from Moringa oleifera seeds were developed for the efficient removal of Arsenic from contaminated water by capacitive deionization (CDI). The electrodes were synthesized using a carbon slurry composed of Moringa oleifera powder (MOP), poly(vinylidene fluoride), and carbon black. Scanning electron microscopy was used to analyze the surface morphology. In contrast, their electrochemical properties, including capacitance and electrical conductivity, were analyzed through cyclic voltammetry (CV) and electrochemical impedance spectroscopy (EIS). The capacitance values, as determined from CV and EIS measurements, were 201.3 Fg−1 and 122.62 Fg−1 for MOP (MOP-1:1 and MOP-1:2), respectively. The Arsenic removal efficiency was evaluated under optimized electrochemical conditions, achieving an efficiency of 89.38% when treating a 100 mg/L arsenite solution at an applied voltage of 1.2 V and pH 9. These findings highlight the potential of Moringa oleifera seeds-derived carbon electrodes as sustainable, cost-effective materials for Arsenic removal through CDI.
Keywords
Introduction
Arsenic contamination is a serious environmental and public health issue affecting millions worldwide. It is a naturally occurring metalloid found in groundwater, primarily originating from geological formations, industrial processes, mining activities, and agricultural runoff (Ahmed et al., 2022). Arsenic is found in two primary forms: organic and inorganic Arsenic. Inorganic Arsenic, which includes arsenite (As³⁺) and arsenate (As⁵⁺), is more toxic and commonly found in contaminated water sources (Koley, 2022). Arsenite is more mobile and difficult to remove, compared to arsenate, which is less soluble and can be more effectively removed using treatment technologies (Nicomel et al., 2016). Extended exposure to water contaminated with Arsenic has been connected to severe health consequences, such as cancer, cardiovascular diseases, and neurological impairments (Thakur et al., 2019). A recommendation limit of 10 µg/L has been established by the World Health Organization (WHO) for Arsenic in drinking water (Frisbie and Mitchell, 2022). However, many regions, particularly in South and Southeast Asia, exceed this limit, necessitating the development of efficient Arsenic removal technologies (Dudek and Kołodyńska, 2022). In Tanzania, the amount of Arsenic in water is found to be high (1–82 µg/L) in Geita (Rwamagasa village), where the mining activities take place (Irunde et al., 2022). Various conventional methods, such as adsorption (Alkurdi et al., 2021), ion exchange (Dudek and Kołodyńska, 2022), coagulation (X. Wang et al., 2021), and membrane filtration (Moreira et al., 2021), have been employed for arsenic removal. Nevertheless, these methods often suffer from high operational costs, secondary waste generation, and limited efficiency in removing arsenic at low concentrations.
To address these challenges, capacitive deionization (CDI) has emerged as an innovative and energy-efficient technology for removing various ionic species from water (Sun et al., 2023). The characteristics of the electrode materials, such as their surface area, porosity, conductivity, and electrochemical stability, have a significant impact on the performance of CDI (Folaranmi et al., 2020). Studies have explored additional efforts to improve electrode materials for CDI performance. The most electrodes used in CDI are carbon-based materials such as graphene-based material (Yan Liu et al., 2023), porous carbon (Zhao et al., 2020), activated carbon (Kyaw et al., 2021) and carbon nanotubes (Li et al., 2020), the metal organic derived carbon (Weng et al., 2021) and metal-oxides based electron such as Magnesium/Aluminum mixed oxide (Garzón-Pérez et al., 2025). Therefore, recent studies have highlighted the potential of biomass-derived carbon materials as sustainable and cost-effective electrode materials for CDI applications (L. Wang et al., 2019). Manufacturing electrodes with activated carbon obtained from biomass is a practical way to reduce expenses, minimize pollution, and facilitate the recovery of solid waste. Different scholars have shown interest in studying the performance of biomass-based carbon electrode materials for CDI application to eradicate heavy metals from water (Yong Liu et al., 2022), (Kim et al., 2021), (Yu et al., 2022), (Hadebe et al., 2022). Natural materials have attracted attention as potential adsorbents due to their availability, low cost, and environmental friendliness. Among these, Moringa oleifera seeds have emerged as a promising candidate due to their high-protein content, which facilitates coagulation and adsorption processes (Ramesh and Kumar, 2019). Additionally, Moringa seeds are widely cultivated in many regions affected by Arsenic, making them an accessible resource for decentralized water treatment. Despite extensive studies on Moringa seeds for turbidity and microbial removal, their application for Arsenic remediation via the CDI system remains underexplored. This study aims to fill this gap by evaluating the efficacy of Moringa oleifera seed-based materials for arsenite (As3+) adsorption, optimizing treatment conditions, and assessing their potential for practical application in arsenic-contaminated water.
Materials and methods
Materials
Moringa oleifera seeds, which were mature, clean, dry, and free from contaminants, were collected from trees. Chemicals, such as diethyl ether, were purchased from Prime Scientific Limited (Dar es Salaam, Tanzania). Sodium Arsenite NaAsO2 was purchased from Guiyang Zhuoyi Chemical Building Material Co., China. Polyvinylidene fluoride (PVDF) and Polytetrafluoroethylene (PTFE) were acquired from Sigma-Aldrich to enhance the stability and strength of the electrodes. Sodium hydroxide and potassium hydroxide (KOH, an activation agent) were purchased from the Merck Company (India). Activated carbon and carbon black (conductive) were purchased from Shandong Richnow Chemical Co., China.
Preparation of activated carbon derived from Moringa oleifera seeds
Moringa oleifera seeds, which were clean, free from contaminants, and dried properly, were collected from trees, followed by removing the husk of the seeds to access the inner kernels. The kernels of Moringa oleifera were pulverized with a mortar and pestle to create a powder. The Moringa oleifera powder (MOP) was added to diethyl ether, and thorough mixing was performed using a magnetic stirrer to extract fats and oils. The powder was obtained through suction filtration using a Buchner flask. It was then dried at room temperature in an open environment on a bench for 72 h, according to the procedures by (Nwagbara et al., 2022). Subsequently, the material was crushed and passed through a 90–150 µm sieve. Then, the powder was carbonized at a heating rate of 10 °C min−1 for 180 min at 600 °C in a nitrogen-filled tube furnace (CTF 12/65/550). The carbonized material was then activated (6 g of material to 6 g of KOH and 3 g of material to 6 g of KOH) at a 1:1 ratio for MOP and KOH, and at a 1:2 ratio for MOP and KOH, respectively, for 120 min at 600 °C in the same furnace with the same heating rate. After cooling to room temperature, activated carbon was removed from the tube furnace. The sample was then washed repeatedly with distilled water to equalize its conductivity to that of pure water. The sample was dried in the oven at 100 0C for 24 h. Moreover, the samples were designated as MOP-1:1 for a 1:1 ratio and MOP-1:2 for a 1:2 ratio, respectively. Figure 1 shows the schematic diagram for the preparation of Moringa Oleifera seeds biomass.

Schematic diagram preparation of Moringa oleifera seeds biomass.
Physical and electrochemical characterizations of the materials
The surface morphology structure of the materials was investigated using scanning electron microscopy (SEM, Carl Zeiss Ultra Plus). The Quantachrome Novawin, version 11.03, was used to determine adsorption/desorption isotherms at a temperature of 77.350 K to determine the specific surface area of the samples. At a temperature of 1800 °C, the prepared samples were first outgassed in 4 h under vacuum. Using the technique from non-local density functional theory (NLDFT), the pore size of the materials was calculated.
The electrochemical impedance spectroscopy (EIS) and cyclic voltammetry (CV) of the electrode materials were examined using a PGSTAT204, model number AUT50663, from Metrohm. In a three-electrode working system, the Ag/AgCl electrode served as the working electrode, reference electrode, and counter electrode, and it was measured with frequencies ranging from 0.01 Hz to 100,000 Hz. The scan voltage ranged from −0.1 to 0.1 V at a sweep rate of 10 mV/s. A total mass of 5 g of electrode was prepared from a ratio of 8:1:1 for activated carbon, conductive carbon black, and PVDF, respectively. The electrode was made from a piece of nickel form, from each of the two ratios (ratio 1:1 and ratio 1:2) and used to determine its electrochemical properties. The capacitance was calculated using equation (1) with the obtained CV results.
Where C is the specific capacitance (F g–1), Idv is the integrated area under the curve, m (g) mass of the active material deposited on the electrodes, V is the scan rate (V s–1), and Δv is the potential window.
Fabrication of electrodes derived from activated Moringa biomass
The activated Moringa electrode materials were fabricated using the procedures described by Kyaw et al., 2021. Figure 2 shows the fabrication of electrodes from Moringa Oleifera seeds. 0.8 g of activated Moringa powder with particle sizes between 90 µm and 150 µm (MP; specific area 766.995–1109.380 m2g−1) was used as the primary electrode material. 0.1 g of Carbon black was used to increase the electrode surface's electrical conductivity. 0.1 g of PTFE was simultaneously dissolved in ethanol. The obtained Moringa slurry was agitated at room temperature for 6 h to ensure homogeneity, resulting in a dough-like paste. The dough was pasted onto a graphite sheet, and using a doctor blade, it was cut into a 4 cm by 4 cm size. The prepared electrode materials were then dried at 100 °C for 10 h to remove all solvent remaining on the electrode surface.

Fabrication of electrodes from Moringa oleifera seeds.
Electrosorption of arsenic ions
A solution of 100 mg/L containing an Arsenic ion concentration was prepared from an Arsenic salt. Arsenic concentrations were measured using a PerkinElmer Analyst 400 Atomic Absorption Spectrometer equipped with a graphite furnace; the method detection limit (LOD) for Arsenic was 0.5 µg/L, and the limit of quantification was 1.5 µg/L. Calibration curves were prepared using Arsenic standard solutions (Sigma-Aldrich, 99.99% purity) at concentrations of 0, 5, 10, 20, and 50 mg/L. A CDI cell in batch mode, with a spacing of 5 mm and a circulating rate of approximately 10 mL/min, and an applied voltage of 1.2 V, was used to examine Arsenic ions in electrosorption. A volume of 30 mL of initial solution was provided in the cell by using a peristaltic pump for 90 min. The conductivity meter (GMH 340) was used to control the conductivity of the solution every 5 min to investigate the Arsenic removal efficiency. Figure 3 shows the schematic diagram of CDI experiment.

Schematic diagram of CDI experiment. CDI: capacitive deionization.
The electrosorption capacity (EC) of As ions from water, was obtained by using equation (2) below
where Co is the initial concentration (mg/L) and Cf is the final concentration (mg/L), V is the volume of the solution used and m (g) is the mass of the electrode.
Also the removal efficiency of As was determined using equation (3) with the initial and final Arsenic concentrations,
To investigate the performance of Arsenic ion removal from water, the electrode's charge efficiency and energy consumption were determined using equations 4 and 5, respectively.
where Z is the valency of ions, F is Faraday constant, V is the volume of the solution, Q is the total charge passed, and Co and Cf are the initial and final concentration.
where E is the energy consumption, V is the average applied voltage, Q is the total charge passed, and ΔN is the number of moles of ions removed.
Results and discussion
Surface analysis
The surface and pore morphologies of MOP-1:1 and MOP-1:2 samples, as shown in Figure 4 (a, b, c, and d), were analyzed using SEM microscopy for carbon activated at 1:1 and 1:2 MOP: KOH at different magnifications. The observed pores and rough surface across all images MOP-1:1 (a and b), MOP-1:2 (c and d) suggest a well-developed structure for ion adsorption and transport. These features are beneficial for CDI applications, as a rougher surface offers more active sites for the removal of Arsenic. However, the SEM images in Figure 4 (c and d, same ratio, different magnification) show some broken pores. KOH can cause this; higher KOH concentrations induce significant structural and morphological changes primarily due to their strong chemical activation and etching effects. KOH acts as a potent activating agent that penetrates and reacts with the carbonaceous or electrode material surface, leading to enhanced pore development. At elevated KOH concentrations, the chemical activation becomes more aggressive, enlarging existing micropores and creating new mesopores and macropores. This increases the surface area and modifies the pore size distribution, which is crucial for adsorption and electrochemical performance. Higher KOH concentrations accelerate the etching process by breaking carbon–carbon bonds or metal oxide layers, leading to increased surface roughness and the exposure of active sites. This effect improves ion accessibility and electron transfer kinetics. The interaction with KOH also introduces oxygen-containing functional groups (e.g. C-O, C = O) on carbon surfaces, enhancing hydrophilicity and adsorption affinity.
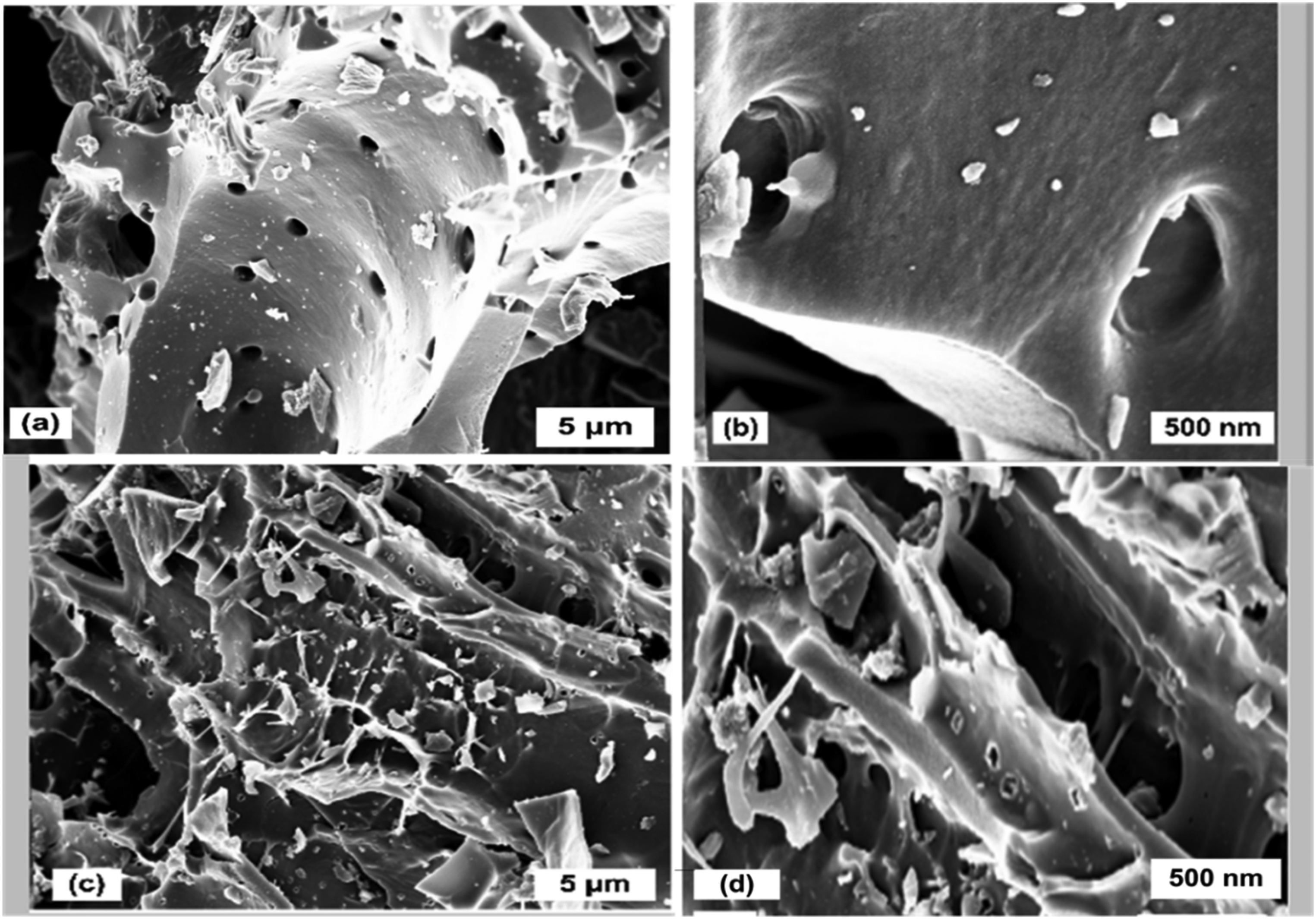
SEM images for MOP-1:1 and MOP-1:2 samples: (a, b) ratio one at 5 µm and 500 nm, respectively; (c, d) ratio two at 5 µm and 500 nm, respectively. MOP: Moringa oleifera powder; SEM: scanning electron microscopy.
BET analysis
The BET surface area of carbon samples activated at 1:1 (KOH: MOP-1:1) was found to be 766.995 m2g−1 but increased to 1109.380 m2g−1 for the samples activated at 2:1 (KOH: MOP-1:2). The increase in surface area can be caused by stronger chemical etching due to the addition of KOH which also promoted the development of more mesoropores and micropores on the carbon materials. The pore size of the carbon samples ranges from 2 to 4 nm, indicating the microporous and mesoporous nature of the samples. Figure 5 (a) shows the relationship between the quantity of nitrogen adsorbed and relative pressure for two materials, MOP-1:1 and MOP-1:2. MOP-1:1 exhibits a higher nitrogen adsorption capacity compared to MOP-1:2 across the entire pressure range, the higher adsorption in MOP-1:1 could be attributed to improved specific surface area and availability of distribution of pore sizes which can be caused by increasing the amount of KOH during activation process. The shape of the isotherms suggests the type of porosity, where the gradual increase in adsorption at low pressure indicates the presence of micropores. Moreover, the steady rise at higher pressures suggests the presence of mesopores. Both isotherms appear to belong to Type IV, which is typical for mesoporous materials, characterized by a hysteresis loop indicating capillary condensation within the pores. (Supplementary Figures 1 and 2)

(a) Isotherms for nitrogen adsorption and desorption, (b) distribution of pore size for MOP-1:1 and MOP-1:2. MOP: Moringa oleifera powder.
Figure 5 (b) shows the pore size distribution as a function of pore width for MOP-1:1 and MOP-1:2. MOP-1:1 exhibits a higher pore distribution than MOP-1:2 in the micropore region and mesopore range (2 to 4 nm). Both materials exhibit a sharp peak below 4 nm, indicating that a significant portion of the pore structure is microporous, with some mesoporosity extending beyond 4 nm. Table 1 indicates the textural properties, such as BET surface area, pore size distribution, and pore volume of MOP-1:1 and MOP-1:2. The dominance of micropores and mesopores is advantageous for CDI applications, as micropores are beneficial for ion storage, and mesopores facilitate ion transfer (Fang et al., 2021).
Summary of porosity characteristics of MOP-1:1 and MOP-1:2.

MOP: Moringa oleifera powder.
Electrochemical characterization
The electrical potential of the ions absorbed on the electrode surface is indicated by capacitance. Capacitance was measured using CV in a voltage window of −1.0 V to 0.0 V in 6 M KOH to verify the electrical capacity of the carbon electrode. Capacitance indicates the electrical capacity of absorbed ions on the electrode surface. Figure 6 (a and b) displays the CV curves of MOP-1:1 and MOP-1:2 electrodes with varying scan rates ranging from 5 mV/s to 50 mV/s. The curves exhibit a nearly rectangular shape, particularly at lower scan rates (5 mV/s and 10 mV/s), indicating that the material exhibits excellent double-layer capacitance behavior and good ion accessibility. As the scan rate increases (25 mV/s and 50 mV/s), the curves slightly distort, indicating the presence of resistive effects or diffusion limitations. According to the theory, ionic resistance restricts the adsorption and desorption of ions at higher scan rates because the ions do not have enough time to adsorb on the electrode surface (Islam et al., 2022). The large area under the curve corresponds to a higher charge storage capacity, indicating that the material performs well in terms of capacitance. Figure 6(b) shows that the MOP-1:2 electrode material exhibits capacitive behavior but appears to have a lower specific capacitance than the MOP-1:1 electrode material. The smaller enclosed area suggests a lower charge storage capacity, which could be due to a lower specific surface area and less-developed porosity compared to the material for MOP-1:1. The greater distortion at higher scan rates highlights slower ion diffusion or poorer ion transport properties. The specific capacitance of MOP-1:1 and MOP-1:2 electrodes was calculated using equation 1, and it was found that the specific capacitance of the MOP-1:1 electrode material was 201.3 Fg−1 higher compared to that of the MOP-1:2 electrode material, which was 122.62 Fg−1, as shown in Figure 6(c). This could be attributed to a larger enclosed area under the curve for MOP-1:1, which suggests a higher charge storage capacity resulting from a higher specific surface area and well-developed porosity.

CV results for the MOP electrodes (MOP-1:1 and MOP-1:2) with varying scan rates and the specific capacitance vs scan rates. MOP: Moringa oleifera powder; CV: cyclic voltammetry.
The EIS analysis reveals distinct electrochemical properties for the MOP-1:1 and MOP-1:2 electrode materials, as shown in Figure 7. The semicircle diameter indicates charge transfer resistance at the point where the Arsenic ionic solution and electrodes interchange. MOP-1:1 exhibits a smaller semicircle diameter, indicating a lower charge transfer resistance (Rct) of 1.87 Ω when the electrode and electrolyte come into contact, suggesting faster electron transfer kinetics. This lower charge transfer resistance (Rct), coupled with its overall lower impedance as evidenced by the extended curve along the Z’ axis, suggests superior interfacial charge transfer and ion transport properties. However, the higher charge transfer resistance (Rct) of 6.32 Ω observed in MOP-1:2 suggests low kinetics at the electrode surface, probably due to intrinsic material properties.
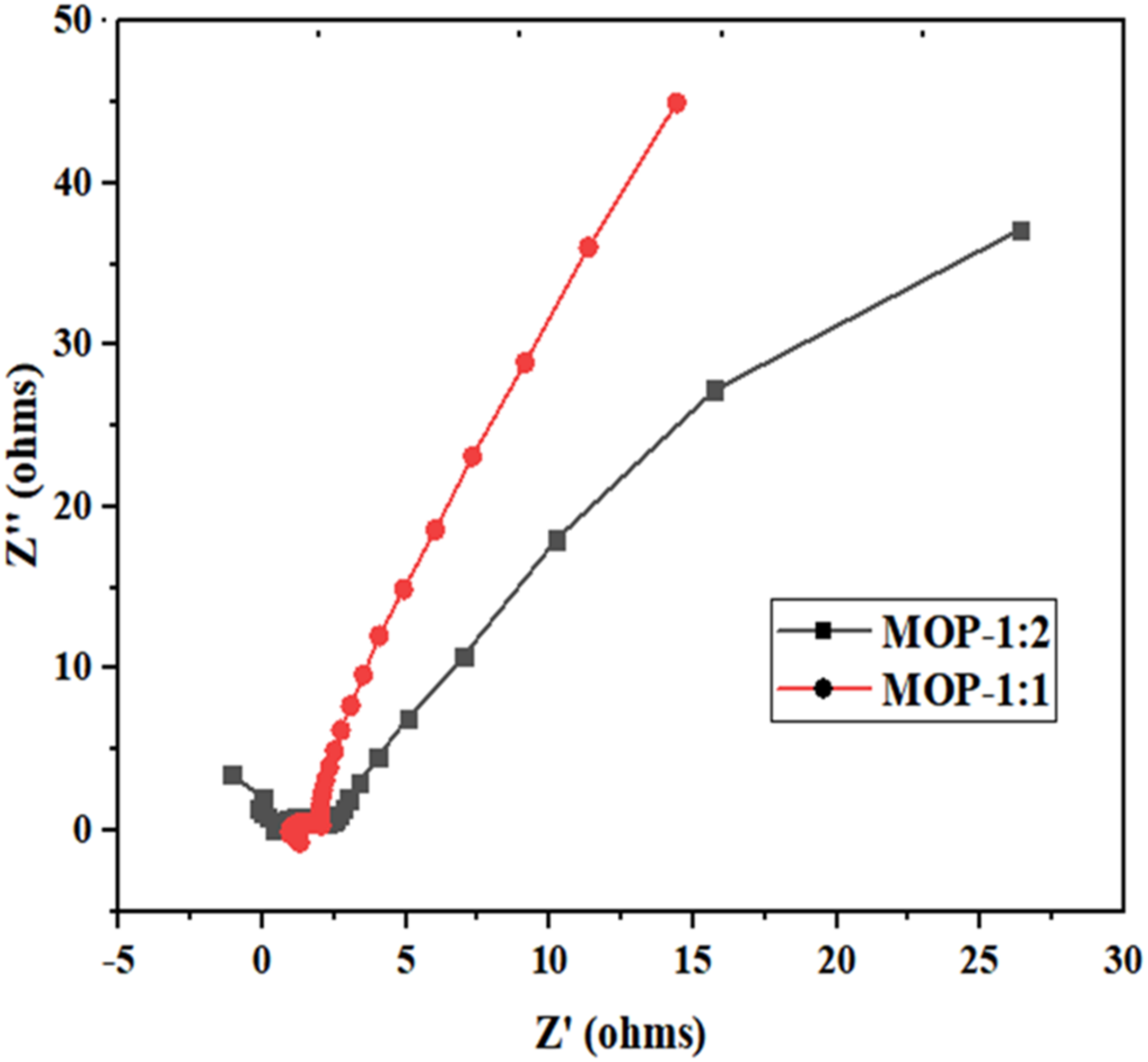
Nyquist plot of electrochemical impedance spectroscopy (EIS) for MOP-1:1 and MOP-1:2 electrodes. EIS: electrochemical impedance spectroscopy.
Furthermore, the diffusion tail at low frequencies provides insight into the solid-state diffusion properties of both MOP-1:1 and MOP-1:2. It is more pronounced in MOP-1:2, possibly indicating poor interfaces between the carbon materials. At high frequency, the leftmost edge of the semicircle corresponds to the solution resistance (Rs), which reflects the ionic conductivity of the electrolyte. For MOP-1:1 and MOP-1:2, the solution resistances (Rs) were 0.96 Ω and 6.32 Ω, respectively. Both solution resistance (Rs) and charge transfer resistance (Rct) were represented by equivalent series resistance (ESR). The reduced charge transfer resistance (Rct) and enhanced ionic conductivity in MOP-1:1 are indicative of an improved electrochemically active surface area, coupled with improved electrolyte penetration.
Arsenic removal test
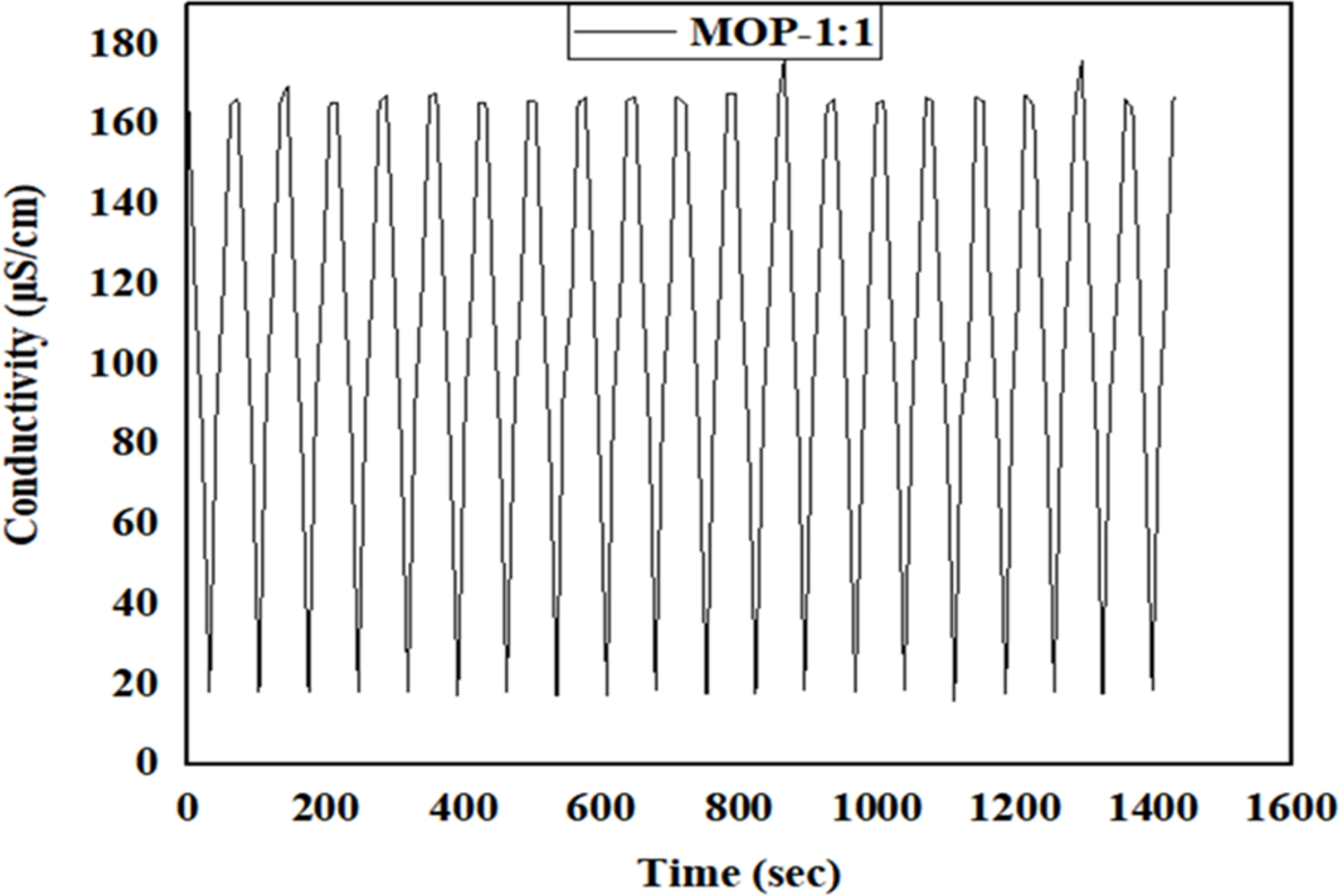
For the removal of Arsenic ions from water, the performance of the CDI based on the MOP-1:1 and MOP-1:2 electrode materials during the electrosorption process was evaluated using initial and final arsenic concentrations measured by atomic absorption spectrometry. The initial concentration of 100 mgL−1 of As3+ was prepared at a pH above 9. At pH 9, Arsenic (III) exists as H2AsO3− anions. The negatively charged dihydrogen arsenite ions (H2AsO3−) are attracted to the positively charged anode, facilitating their removal from the water. The concentrations decreased to 10.62 mg/L and 32.99 mg/L for MOP-1:1 and MOP-1:2, respectively, indicating effective deionization and removal efficiencies of about 89.38% and 67.01% for MOP-1:1 and MOP-1:2. The conductivity measurements over time, spanning 120 min, for the prepared solutions used in electrosorption at 1.2 V for MOP-1:1 and MOP-1:2 provided valuable insights into their ion removal efficiency. As shown in Figure 8, both materials exhibited a gradual decrease in conductivity from 144 µS/cm to 49.49 µS/cm for MOP-1:1 and from 162 µS/cm to 80.72 µS/cm for MOP-1:2 during electrosorption. However, notable differences are observed between the two samples; MOP-1:1 demonstrates a steeper decline in conductivity compared to MOP-1:2, suggesting that MOP-1:1 has a higher rate of ion adsorption. This suggests that MOP-1:1 exhibits enhanced electrochemical properties, likely due to superior surface characteristics, such as higher porosity or improved functional group activity.

Conductivity performance for MOP-1:1 and MOP-1:2 electrodes. MOP: Moringa oleifera powder.
Over the entire testing period, MOP-1:1 achieves a lower final conductivity value, signifying better overall deionization efficiency. This performance can be attributed to improved charge storage and ion EC, making MOP-1:1 more suitable for CDI applications.
The EC of MOP-1:1 and MOP-1:2 was estimated using equation 3. It is found that the EC for MOP-1:1 is approximately 14.9 mg/g at 1.2 V, which is 25.10% higher than that of MOP-1:2. Figure 9 illustrates the electrosorption and desorption mechanism for MOP-1:1. When selecting superior CDI electrode materials, cycling stability and regeneration capacity are crucial. Arsenic solution containing 100 mg/L concentration was subjected to a direct short circuit regeneration of 20 cycles of electrosorption/ desorption for 12 h at 1.2 V, the total time per cycle (electrosorption + desorption) was 36 min and the time for each phase (electrosorption/desorption) was 18 min. When the positive and negative plates were shorted, a rise in conductivity was observed, indicating that desorption had occurred. Throughout the regeneration procedure, the MOP-1:1 electrode maintained a nearly constant regeneration rate of approximately 89%. This demonstrates how the shape of the electrode surface may impact the removal effectiveness and regeneration of the electrode materials. CV and EIS both provided more evidence for the connection between conductivity and concentration. The decrease in peak current observed in CV after Arsenic adsorption indicates a reduction in the electroactive Arsenic species in the solution. Similarly, the lower rate of charge transfer resistance (Rct) in EIS confirms that the faster charge transfer rate of Arsenic ions on the electrode surface leads to the reduction of Arsenic species in water. This suggests that MOP-1:1 electrode materials may be used to extract Arsenic ions from solution.
Electrosorption and desorption mechanism for MOP-1:1 (20 cycles). MOP: Moringa oleifera powder.
Furthermore, the energy consumption and electrode efficiency at a 100 mg/L concentration were found to be 0.09 and 0.59 KWh m−3 for MOP-1:1 and 0.98 and 0.28 KWh m−3 for MOP-1:2, respectively. This indicates that MOP-1:1 electrodes are more efficient in charging and require less energy to remove ions than MOP-1:2 electrodes. Its superior specific surface area, capacitance, and surface porosity could all be contributing factors to the strong performance of the electrode materials.
The experimental measurements, including removal efficiencies and related performance metrics, were conducted in triplicate. Table 2 reflects data variability and reproducibility for the corresponding means and standard deviation results. To rigorously evaluate performance differences between MOP-1:1 and MOP-1:2 electrodes, the independent two-sample t-tests were performed using the triplicate data for each electrode. This analysis confirmed that the observed variation in removal efficiency is statistically significant (t(≈3) = 37.32, p < 0.05). MOP-1:1 exhibited a removal efficiency of 89.38 ± 0.61% (mean ± SD, n = 3), while MOP-1:2 displayed 67.01 ± 0.84%.
Removal efficiencies for MOP-1:1 and MOP-1:2 electrodes (n = 3). Statistical significance evaluated with an independent two-sample t-test.

MOP: Moringa oleifera powder.
The performance of Moringa-based electrode materials (MOP-1:1 and MOP-1:2) shown in Table 3 for Arsenic removal was rigorously evaluated and benchmarked against existing CDI technologies and electrode materials. Table 4 provides a summary of selected literature data. Researchers have explored various biomass sources to fabricate electrodes for Arsenic removal via CDI, emphasizing their sustainability and structural advantages for water purification (Liu et al., 2022). This work aligns with the growing area, utilizing Moringa oleifera seeds as a sustainable precursor for electrode fabrication. Activated carbon powder derived from biomass has demonstrated high Arsenic removal capabilities in CDI systems (Lee et al., 2016). For instance, specific biomass-derived carbons have shown promising electrosorption capacities for Arsenic, although specific removal efficiencies vary widely depending on the biomass type, activation methods, and experimental conditions. In this study, the MOP-1:1 Electrode achieved 89.38% Arsenic removal, and the MOP-1:2 electrode demonstrated 67.01% removal. This performance is highly competitive when compared to other biomass-derived or synthetic carbon materials in CDI applications for Arsenic. For example, some studies on biomass-derived activated carbons have reported Arsenic removal efficiencies ranging from 60% to 90% under various conditions. An active MnO2/biochar composite/AC electrode achieved 94% efficiency in reducing Arsenic (Cuong et al., 2022). The rGO/NCDs obtained an EC of 15.07 mg/g at 1.0 V (Xu et al., 2022). An iron-manganese composite-based electron achieved a removal capacity of 411.22 mg g-1 (Ni et al., 2022). Low-cost carbon composite electrodes achieved 98.8% Arsenic removal from groundwater (Lee et al., 2016). Studies on activated carbon biomass for removing As3+ from water are scarce; however, the stated electrode materials suffer from high costs. The high removal efficiency of MOP-1:1 underscores its potential as an effective electrode for Arsenic remediation, particularly considering the simplicity and sustainability of its synthesis route from Moringa oleifera seeds.
Comparison between MOP-1:1 and MOP-1:1 performance.

MOP: Moringa oleifera powder; CV: cyclic voltammetry; EIS: electrochemical impedance spectroscopy.
The electrode materials used in CDI for the removal of arsenic from water.
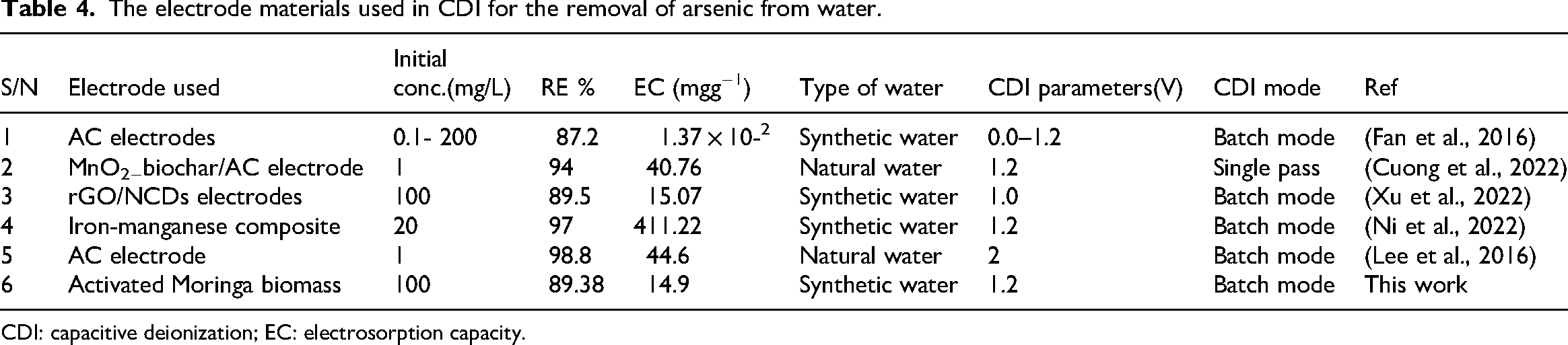
CDI: capacitive deionization; EC: electrosorption capacity.
Conclusion
This research effectively demonstrated that porous carbon materials have been successfully prepared from Moringa oleifera seeds through carbonization and subsequent chemical activation using KOH. The materials were utilized as CDI electrodes for the removal of As3+ ions from solution. The activated carbons obtained exhibited substantial mesoporosity and presented high BET surface areas of 766.995 m²/g for MOP-1:1 and 1109.380 m²/g for MOP-1:2. Notably, MOP-1:1 displayed favorable electrochemical characteristics, including fast charge transfer and high electrical conductivity, which contributed to an enhanced electrosorption capability for arsenite during CDI operation. MOP-1:1 achieved a removal efficiency of 89.38%, while MOP-1:2 reached 67.01%. These findings indicate the potential of MOP-1:1 electrodes for effective As (III) removal and underscore their suitability for CDI processes in removing heavy metals from water.
Supplemental Material
sj-docx-1-adt-10.1177_02636174251387199 - Supplemental material for Arsenic removal from water using capacitive deionization electrode materials derived from Moringa oleifera seeds
Supplemental material, sj-docx-1-adt-10.1177_02636174251387199 for Arsenic removal from water using capacitive deionization electrode materials derived from Moringa oleifera seeds by Magdalena Mwangesi, Revocatus L. Machunda and Yusufu Abeid Chande Jande in Adsorption Science & Technology
Footnotes
Acknowledgment
Gratitude is extended to Kondoa Town Council for the opportunity to pursue MSc degree.
Authorship
LRLM contributed to writing—review & editing and supervision. YACJ contributed to writing—review & editing and supervision. MM contributed to writing—original draft, methodology, investigation, formal analysis, conceptualization.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.





