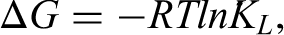Abstract
This study examines the influence of surfactants with varying hydrocarbon chain lengths—tetradecyltrimethylammonium bromide (TDAB), octadecyltrimethylammonium bromide (ODAB), and docosyltrimethylammonium bromide (DKAB)—on the structure and properties of montmorillonite (a bentonite clay) with a cation exchange capacity of 101 mmol/100 g. Structural modifications were analyzed using X-ray diffraction (XRD), infrared (IR) spectroscopy, thermogravimetric analysis (TGA), and microscopy techniques. Intercalation of these surfactants increased the interlayer spacing of montmorillonite from 1.24 nm (raw) to 1.98 nm, 2.17 nm, and 2.32 nm for TDAB-, ODAB-, and DKAB-modified samples, respectively. IR spectral analysis confirmed the incorporation of the hydrocarbon chains and a resulting decrease in clay hydrophilicity. TGA demonstrated enhanced thermal stability in the modified clays, with the highest residual mass (17.43%) observed for DKAB-modified bentonite. The decrease in the point of zero charge (pHpzc) of the clay from 9.6 (raw bentonite) to approximately 9.0 in the modified samples reflects a change in the electrochemical state of the surface. Water vapor adsorption decreased from 0.183 g/g (raw clay) to 0.086 g/g in modified samples, while benzene vapor adsorption increased. Adsorption studies with organic dyes showed a maximum sorption capacity of 10.6 mg/g for methylene blue on ODAB-modified clay and 13.6 mg/g for Congo red on TDAB-modified clay, reflecting enhanced affinity due to interlayer expansion and improved surface properties. These findings highlight the potential of surfactant-modified bentonites for improved adsorption applications.
Introduction
The incorporation of surfactants into aluminosilicate clays such as montmorillonite plays a key role in developing novel organo-clays with enhanced adsorption capacities and tailored surface functionalities. Recent reviews describe various modification strategies—including ion-exchange, surfactant grafting, and their mechanistic underpinnings—that significantly improve the interaction between montmorillonite and organic pollutants (Guo, 2020). The broad applicability of such organo-clays in sorption and environmental remediation has been further validated in studies exploring their removal of heavy metals using bentonite-based organo-clays (Perelomov et al., 2024), as well as the superior adsorption of organic micropollutants via gemini-surfactant-intercalated clays (Zango et al., 2022). Additionally, comparative analyses of surfactant types (cationic vs amphoteric vs nonionic) demonstrate how hydrophobicity and structural organization within the clay matrix directly affect adsorption performance (Lazorenko et al., 2020). To ensure compatibility between inherently incompatible organic and inorganic components, and to facilitate the penetration of large molecules into clay interlayers and onto its surface, various surfactants are adsorbed onto clays as modifiers.
Organobentonites (organically modified bentonites) are widely used in industrial formulations. They act as rheological modifiers with thickening and thixotropic properties — for instance, in cosmetics (Rana and Kim, 2024), as well as in greases and lubricants where they contribute to structural viscosity (Zhu, 2024). They are also utilized in adhesives, oil-based drilling fluids, lubricants, and polymer nanocomposites (Archibong et al., 2023; Mahmoud, 2023; Mamataliev et al., 2023). By exchanging the inorganic interlayer cations of clays with organic cations (quaternary ammonium surfactants), the clay surfaces become organophilic, improving the retention of organic contaminants from water and altering properties such as swelling, sedimentation, and rheology (Bujdák, 2023; Obradović et al., 2023). The length and structure of the surfactant's hydrocarbon chain can strongly influence the packing density within the clay interlayers and thus the extent of property modification (Seitnazarova et al., 2025).
In this study, natural montmorillonite (bentonite) was modified with three quaternary ammonium surfactants of varying alkyl chain lengths: tetradecyltrimethylammonium bromide (TDAB, C14), octadecyltrimethylammonium bromide (ODAB, C18), and docosyltrimethylammonium bromide (DKAB, C22). The influence of surfactant chain length on the structural organization within the interlayer space, the extent of interlayer expansion, thermal stability, and surface properties of the resulting organoclays was systematically investigated. Particular attention was given to adsorption characteristics, including surface charge behavior and the uptake capacity for polar (water vapor, methylene blue) and non-polar (benzene vapor, Congo red) molecules. These insights contribute to a deeper understanding of structure–function relationships in organoclay systems.
While the use of TDAB and ODAB in the modification of montmorillonite has been widely reported in the literature (Açışlı et al., 2016; Yılmaz and Yapar, 2004), DKAB-modified organobentonite has not been systematically explored. The inclusion of DKAB (C22) in this study allows us to fill this research gap and examine the effect of extended hydrocarbon tail length on structural ordering, thermal response, and sorption behavior.
Therefore, this study aims to systematically investigate the relationship between the hydrocarbon chain length of quaternary ammonium surfactants and the resulting structural and adsorption characteristics of montmorillonite-based organoclays.
The scientific novelty of this work lies in the comparative analysis of three alkyltrimethylammonium bromides with different tail lengths (C14, C18, and C22) under identical experimental conditions. This approach allows for a clear correlation between chain length and interlayer expansion, thermal behavior, surface hydrophobicity, and adsorption efficiency toward both polar and nonpolar organic molecules.
Materials and methods
Materials
The bentonite samples used in this study were enriched natural clays obtained from the Logon deposit in the Republic of Uzbekistan, with a montmorillonite content of at least 91%. The cation exchange capacity (CEC) of the raw bentonite was determined using a modified cobalt hexamine method. In this approach, 1.00 g of the sample was mixed with 20 mL of a 0.025 M [Co(NH3)6]Cl3 solution and shaken at 20 °C for 24 h to allow complete ion exchange. After equilibration, the concentration of cobalt hexamine remaining in the supernatant was measured spectrophotometrically at λ = 472 nm. The CEC was then calculated as:
The measured CEC value (101 mmol/100 g) was in good agreement with theoretical expectations and was used to determine the surfactant loading.
The surfactants used for modification were tetradecyltrimethylammonium bromide (TDAB, C14H29N(CH3)3Br), octadecyltrimethylammonium bromide (ODAB, C18H37N(CH3)3Br), and docosyltrimethylammonium bromide (DKAB, C22H45N(CH3)3Br). Methylene blue (MB) and Congo red (CR) were selected as organic dye adsorbates for testing adsorption capacity, and benzene (99% purity) was used for vapor adsorption experiments. All reagents were of analytical grade and used as received.
Preparation of Organobentonites: The raw bentonite (LB) was first evaluated for its swelling capacity and cation exchange capacity (CEC). If the swelling volume was below 15 mL/g or the CEC was lower than 90 mmol/100 g, the sample was considered to be in the calcium form and was converted into the sodium form to enhance its intercalation ability and swelling behavior. Sodium activation was performed by treating the bentonite with 1 M NaCl solution (1:10 w/v), stirring for 24 h at room temperature. The suspension was centrifuged and washed several times with distilled water until free of chloride ions (tested using AgNO3). The resulting sodium form was dried at 80 °C, ground, and sieved.
Then, the sodium-activated bentonite was dispersed in distilled water to form a 1% (w/v) suspension. Stoichiometric amounts of each surfactant—TDAB (1.7 g), ODAB (2.0 g), or DKAB (2.3 g)—corresponding exactly to the clay's cation exchange capacity (CEC = 101 mmol/100 g), were separately dissolved in 100 mL distilled water at 70 °C. These surfactant solutions were added dropwise to the clay suspensions under continuous stirring at 70 °C. The mixtures were stirred continuously at 70 °C for 4 h to facilitate complete intercalation of the surfactants into the clay layers. After the intercalation process, organoclay products were recovered by centrifugation, washed three times with distilled water to remove residual (unadsorbed) surfactants and other impurities, and dried at 80 °C. The dried samples were ground and sieved through a 154 µm mesh to ensure uniform particle size. The resulting organomodified bentonites were designated LB-TDAB, LB-ODAB, and LB-DKAB according to the respective modifying surfactants. The raw bentonite is referred to as LB for comparison.
Determination of the Point of Zero Charge (PZC): the PZC was determined by mass titration: 30 mL of 0.001 M NaCl was placed into a 50 mL beaker, and the initial pH was measured. Subsequently, 0.05 g of the sample was added, and the suspension was stirred continuously for 10 min using a magnetic stirrer with automatic temperature regulation until equilibrium pH was reached. Additional portions of sample were then added until the equilibrium pH stabilized, independent of solid concentration. Initial pH adjustments were performed using 1 M NaOH or 1 M H2SO4, the latter chosen for its stability and minimal side-effects. Similar procedures were conducted using deionized water to verify changes in surface properties after modification. Additional specificity has been provided for all other experimental procedures and characterization methods throughout the manuscript (Zalac and Kallay, 1992).
Characterization: X-ray diffraction (XRD) patterns were recorded using a Cu Kα radiation source to determine the basal spacing (d001) of the clays before and after modification. Infrared (IR) spectra were obtained (4000–400 cm−1, KBr pellet method) to identify functional groups and confirm surfactant incorporation. Thermogravimetric analysis (TGA) was performed from room temperature to 800 °C (heating rate 10 °C/min under air) to assess thermal stability and organic content. Nitrogen adsorption–desorption isotherms at −196 °C were measured on a surface area analyzer to evaluate textural properties (specific surface area, pore volume, and pore size distribution); samples were degassed at 110 °C prior to analysis. The Brunauer–Emmett–Teller (BET) method was used for surface area calculation, while pore size distributions were derived using the Horvath–Kawazoe (HK) and micropore (MP) methods for micropores.
Determination of Surfactant Concentration in Solution: The interaction of alkylammonium surfactants with dyes in aqueous solutions has been studied primarily using methyl orange (MO, 4-(4-(dimethylamino)phenylazo) benzenesulfonic acid) (Yadav et al., 2025). In pure water, the absorption band of MO at λmax = 475 nm (Yadav et al., 2025) shifts to 370–390 nm upon complex formation with surfactants. This shift is attributed to electrostatic binding between the sulfonate group of the dye and the cationic centers of the surfactants, as well as hydrophobic and entropic effects (Sah et al., 2024; Muratov et al., 2024).
When the [MO]:[surfactant] ratio decreases from 1:10 to 1:500 in an organic medium, an additional band shift of about 20 nm is observed due to the transition of dye molecules to a parallel orientation on the micelles and π–π interactions between azobenzene moieties (Dutta and Bhat, 1993).
In our study, a spectrophotometric ion-pair method based on the surfactant–MO complex was employed for the rapid determination of residual surfactant concentration in solution after intercalation. This approach enables the estimation of the amount of unadsorbed surfactant, which is essential for calculating the loading of the montmorillonite interlayer space and the efficiency of modification. After intercalation, the samples were diluted 10–100 times depending on the initial surfactant concentration. The method is based on the correlation between the absorbance peak intensity of the surfactant–MO complex and the surfactant concentration: for standardization, an MO solution with a concentration of 0.025 mmol/L was used, while surfactant solutions were prepared in the range of 0.001–1.5 mmol/L at 70 °C under vigorous stirring. To 5 mL of each surfactant solution, 5 mL of the MO solution was added, and the mixture was transferred to a 1 cm path length quartz cuvette. Quantitative measurements were performed using scanning spectrophotometry in the range of 190–700 nm on a UV/V-5100 spectrophotometer (Shanghai Metash Instruments Co.). The λmax value for each concentration was determined from calibration spectra, and the experimental results are presented in the Results and Discussion section.
The effect of temperature was examined in the range of 25–60 °C and pH from 2 to 10. Distilled water was used as the reference medium. Based on the obtained data, a calibration curve was constructed and subsequently used to determine the surfactant concentration in solutions after separation of the synthesized organobentonites.
Adsorption Experiments: The vapor adsorption experiments were conducted using a McBain vacuum adsorption apparatus to evaluate the adsorption characteristics of the clay samples. Water and benzene vapors were used as adsorbates to assess the materials’ adsorption activity toward polar and non-polar molecules. Gravimetric and isosteric methods were employed to ensure high accuracy of the measured data. Prior to measurements, the clay samples underwent thorough activation. They were degassed under vacuum at temperatures of 200 °C and 500 °C until the residual pressure reached 10−3 mmHg. This degassing process lasted for eight hours to remove pre-adsorbed molecules and prepare the surface for subsequent adsorption.The specific surface area of the samples was calculated using both the Langmuir equation and the BET theory, allowing the assessment of adsorption capacity with respect to monolayer formation and surface structural features. Micropore volume was determined by the t-method, which is based on the thickness of the adsorbed layer and is effective for characterizing microporous structures. Adsorption isotherms of benzene vapor were measured using a high-vacuum McBain–Bakr quartz spring balance. About 1.0 g of powdered and homogenized adsorbent was placed in a sample pan suspended from a calibrated quartz spring in the adsorption column, thermostated at 20.0 ± 0.1 °C. The system was evacuated to a residual pressure of ∼1 × 10−5 mm Hg using a fore-vacuum and diffusion pump assembly, and maintained under vacuum for 6–8 h to remove pre-adsorbed species. Benzene vapor was introduced stepwise from a dosing ampoule, and the equilibrium pressure was measured with an U-tube mercury manometer. Adsorbed amounts were determined from the quartz spring elongation (sensitivity 1.78 × 10−3 kg/m). The procedure was repeated for successive equilibrium pressures until the full adsorption isotherm was obtained.
Adsorption isotherms of methylene blue (MB, C16H18NSCl, M = 320 g/mol) and Congo red (CR, C32H22N6Na2O6S2) on bentonite and organo-modified montmorillonite samples were obtained in batch mode at room temperature. Aqueous solutions of MB and CR with initial concentrations of 1, 3, 5, 10, 20, 30, and 60 mg/L were prepared, and their optical densities were measured. UV-Vis spectra were recorded in the 190–700 nm range to determine the precise λmax of each solution under the given conditions. For quantitative analysis, characteristic wavelengths of λ = 660 nm for MB and λ = 505 nm for CR were used, corresponding to the absorption maxima of these dyes within the specified concentration range, as confirmed by calibration curve construction.
To each 50 mL tube containing a dye solution, 0.05 g of clay sample was added, and the suspensions were stirred using a magnetic stirrer until equilibrium was reached (24 h at 25 °C). The mixtures were then centrifuged at 3000 rpm for 10 min. Residual dye concentrations were calculated based on calibration curves (“absorbance vs. concentration”) by measuring the optical density at the selected λ. Adsorption isotherms were plotted and processed in Langmuir coordinates using the linearized form of the Langmuir equation:
The Langmuir affinity constant (KL) was used to calculate the standard Gibbs free energy of adsorption (ΔG) according to the equation:
The specific surface area of the samples (Ssp) was calculated from the maximum monolayer capacity (A0) using the molecular cross-sectional area of the dye (am) and Avogadro's number (NA):
The total pore volume (ΣV) was determined as the product of the maximum monolayer capacity (A0) and the molar volume of the dye (Vm, cm3/mol):
Results and discussion
Surfactant uptake and calibration
The efficiency of surfactant intercalation was evaluated by determining the residual concentration of unbound surfactant in the solution after the modification process. As described in the Materials and Methods section, a spectrophotometric technique based on complex formation between methyl orange (MO) and quaternary ammonium surfactants was employed. This method enabled sensitive detection of residual surfactants through absorbance shifts in the visible region.
Figure 1 illustrates the changes in the visible absorption spectra of methyl orange (MO, 10 mg/L) upon adding increasing concentrations of each surfactant (TDAB, ODAB, DKAB) in the presence of the clay. Figure 1 shows that the main absorption peak of MO (around 460 nm) diminishes and blue-shifts progressively as more surfactant is added. The most pronounced shift is observed for the DKAB system, indicating the strongest interaction between MO and the longest-chain surfactant (DKAB). These spectral changes suggest the formation of surfactant–dye complexes, likely through a combination of electrostatic attraction (between the cationic surfactant and the anionic MO) and hydrophobic interactions, which alter the dye's electronic environment. Complexation can lead to new absorbance features or peak shape changes, reflecting a more ordered arrangement of dye molecules in the presence of surfactants (Gerasin et al., 2005).

UV-Vis absorption spectra of methyl orange (MO, 10 mg/L) in the presence of TDAB (a), ODAB (b), and DKAB (c) at varying concentrations. Spectral changes reflect the influence of surfactant type on dye–micelle interactions.
To quantify surfactant uptake, calibration curves of surfactant concentration vs. solution absorbance (with MO as an indicator dye) were established. Figure 2 presents a representative calibration for TDAB, ODAB, and DKAB in the MO system. The optical density at the MO wavelength was found to decrease linearly with surfactant concentration remaining in solution, allowing determination of how much surfactant had been removed (adsorbed by clay). The calibration plots for all three surfactants showed high linearity (R2 ≈ 0.996–0.998). This confirms that absorbance measurements can reliably track surfactant concentration. Using these curves, the amounts of surfactant intercalated into the clay were calculated. Under identical treatment conditions, approximately 77.2 mmol of surfactant per 100 g of clay (≈76.5% of the added TDAB) was intercalated into the LB-TDAB sample, 75.8 mmol/100 g (75.0% uptake) into LB-ODAB, and 74.7 mmol/100 g (73.9% uptake) into LB-DKAB. These high levels of intercalation, slightly lower for the longest-chain DKAB (likely due to steric constraints within the interlayer space), correspond to approximately 0.26 g of TDAB, 0.30 g of ODAB, and 0.34 g of DKAB per gram of clay, reflecting the increase in molecular mass of the surfactants. The progressive expansion of the basal spacing with increasing hydrocarbon chain length, as confirmed by X-ray diffraction patterns (see section “X-ray Diffraction Analysis”), provides further evidence of successful intercalation.

Calibration curves of TDAB, ODAB, and DKAB based on absorbance changes in methyl orange solution (λmax ≈ 460 nm). Symbols: TDAB (▪), ODAB (▴), DKAB (●). These curves were used to estimate surfactant uptake by montmorillonite.
The successful intercalation of each surfactant into montmorillonite is evidenced by the significant uptake percentages. The slight trend of decreasing uptake efficiency with longer chain length (from TDAB to DKAB) may be due to the increasing bulk of the surfactant molecules. Longer hydrocarbon tails can pack within the interlayer space in a more disorderly fashion or face steric hindrance, leading to marginally lower maximum loading. Nonetheless, all three surfactants achieved over 70% incorporation of the added amount, confirming effective modification of the clay.
X-ray diffraction (XRD) analysis
XRD was used to determine the basal spacing (d001) of montmorillonite before and after surfactant modification. The XRD patterns (Figure 3) show a clear shift in the d001 reflection to lower 2θ (higher basal spacing) upon organo-modification. Raw bentonite (LB) exhibits a basal spacing of 1.24 nm (d001 ≈ 12.4 Å), typical for Na+-montmorillonite in air-dried condition. After modification, LB-TDAB shows d001 = 1.98 nm, LB-ODAB has d001 = 2.17 nm, and LB-DKAB reaches d001 = 2.32 nm. These values correspond to an expansion of the interlayer by roughly 0.74, 0.93, and 1.08 nm for TDAB, ODAB, and DKAB, respectively, compared to the raw clay. The increasing expansion with longer chain surfactants indicates that the chain length governs how far apart the silicate layers can be pushed. This observation agrees with previous findings reported for TDAB- and ODAB-modified montmorillonites, where interlayer spacing increased systematically with alkyl chain length (Gerasin et al., 2005; Vidal and Volzone, 2012).In each case, the surfactant cations likely adopt a bilayer or pseudobilayer arrangement in the interlayer, with the alkyl chains more or less perpendicular to the clay surface. Longer chains (especially C22 in DKAB) promote a larger spacing, possibly forming interdigitated or tilted bilayers.

XRD patterns of LB and surfactant-modified bentonites. Increasing d001 values reflect interlayer expansion due to surfactant intercalation: TDAB < ODAB < DKAB.
These XRD results confirm that intercalation of the quaternary ammonium surfactants took place. The presence of well-defined basal reflections for the organo-clays suggests that the surfactant layers are relatively ordered within the interlayer spaces. The trend of larger spacing for longer chains is consistent with literature on alkylammonium-modified clays (Cao et al., 2025; Lazorenko et al., 2020), where the arrangement can shift from monolayer to bilayer to paraffin-type monolayer as chain length increases; similar patterns of interlayer expansion with C14–C18 surfactants were also reported in bentonite systems by Vidal and Volzone (2012), Carmody et al. (2007), and Gerasin et al. (2005). In our case, the C14 surfactant (TDAB) likely forms a monolayer or tilt-bilayer (∼1.98 nm spacing), whereas C18 and C22 (ODAB, DKAB) achieve spacings above 2 nm, indicative of a more extended bilayer arrangement (Hedley et al., 2007; Wójcik-Bania and Matusik, 2021). This structural change is crucial, as it creates additional mesopore space and exposes more nonpolar surface area, affecting adsorption behavior discussed later.
Infrared spectroscopy (FTIR)
FTIR spectra provide evidence of surfactant incorporation and changes in the clay's surface chemistry. The spectrum of raw bentonite (LB) shows characteristic bands of the clay mineral: O–H stretching ∼3620 cm−1 (inner OH groups) and a broad band ∼3400 cm−1 (adsorbed water), H–O–H bending of water ∼1630 cm−1, Si–O stretching in the 1000−1100 cm−1 region, and Al–OH or Al–O bending vibrations at lower frequencies. Upon modification with surfactants, new bands appear and some bands change intensity. In particular, the organobentonite samples show strong C–H stretching bands around 2920 cm−1 and 2850 cm−1, corresponding to the –CH2– asymmetric and symmetric stretches of the alkyl chains. These bands were absent in raw LB and confirm the presence of long hydrocarbon chains in the modified samples. The intensity of the C–H bands is highest for LB-DKAB, reflecting its C22 chains, and somewhat lower for LB-ODAB and LB-TDAB in accordance with their shorter chains.
Additionally, the hydrophilic character of the clay surface is reduced after surfactant loading. This is evidenced by the decreased intensity of the broad O–H stretching band (around 3400 cm−1) in organo-clays, indicating less adsorbed water. The N–CH3 bending vibrations from the quaternary ammonium headgroups might also be observed as small features near ∼1470−1480 cm−1. Overall, the FTIR confirms that the surfactants are intercalated: the clay structure bands (Si–O, etc.) remain, but aliphatic C–H bands from the surfactants are now superimposed, and the relative intensity of OH bands decreases, signifying a more hydrophobic clay surface.
FTIR spectra are presented in Figure 4, which showed spectra for LB and each organo-clay. In those spectra, LB-TDAB, LB-ODAB, and LB-DKAB each exhibited the mentioned C–H stretches and diminished OH bands compared to unmodified LB.

FTIR spectra of the samples: (a) LB; (b) LB-TDAB; (c) LB-ODAB; (d) LB-DKAB.
The FTIR results support the XRD findings: the intercalation of surfactant cations replaces some of the water in the clay interlayers and bonds to the silicate surfaces via cation exchange. The reduction in hydrophilicity (fewer –OH from water) and appearance of hydrophobic –CH2– signals align with the clay's conversion to an organophilic material. Notably, no new sharp bands indicative of unwanted chemical reactions (e.g. surfactant decomposition) are seen, implying the surfactants remain intact and simply reside in the clay galleries, bound by electrostatic and van der Waals forces.
Thermogravimetric analysis (TGA)
Thermogravimetric curves (Figure 5) reveal differences in thermal stability and organic content between raw and modified clays. The raw bentonite (LB) shows a typical TGA profile with an initial mass loss below 150 °C (loss of adsorbed water), and a second gradual mass loss up to ∼600 °C (dehydroxylation of the clay lattice). By contrast, the organo-bentonites display additional mass loss steps due to the decomposition of the intercalated surfactants.
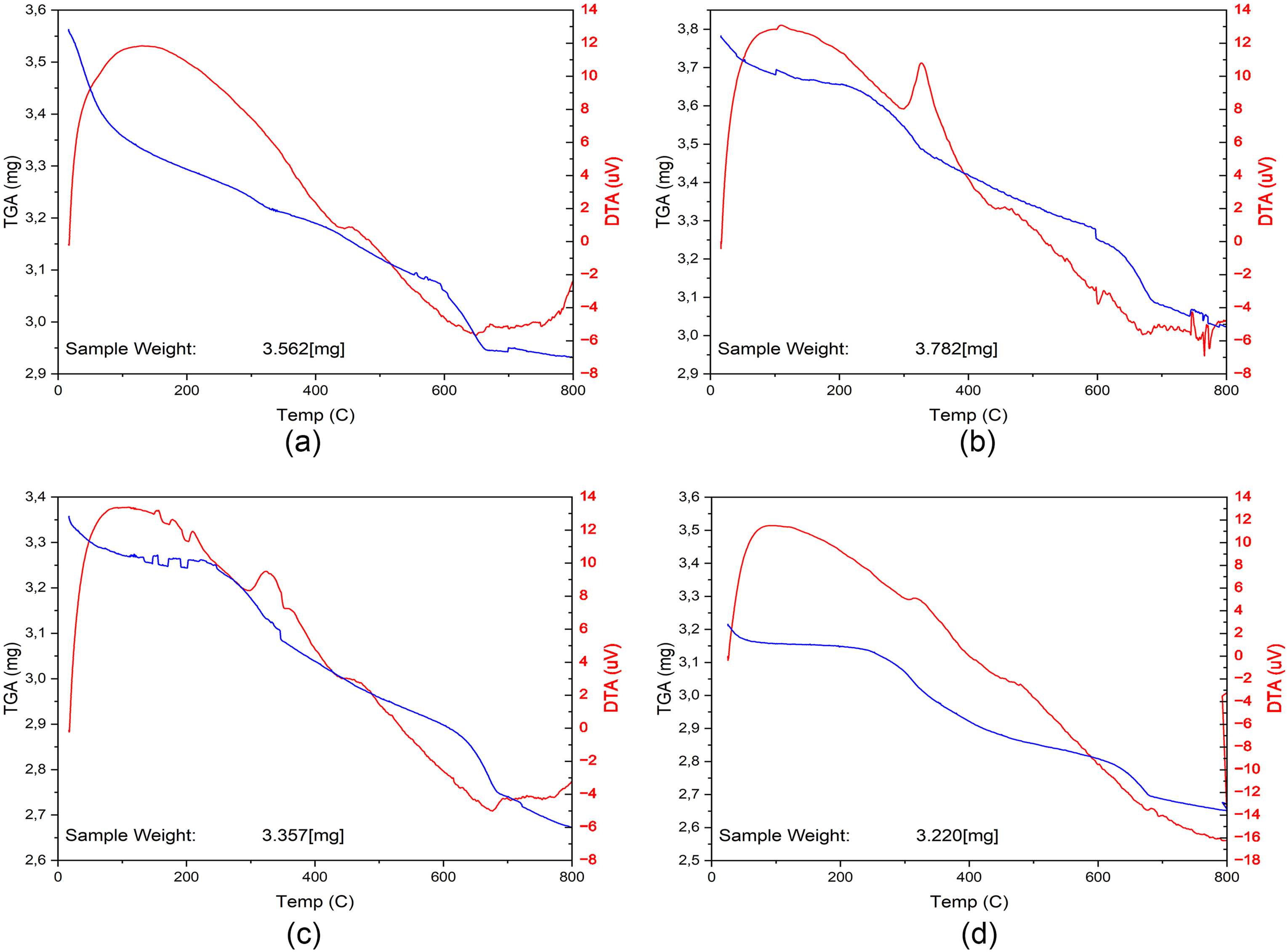
TGA and DTA thermograms of raw and surfactant-modified bentonites. (a) LB, (b) LB-TDAB, (c) LB-ODAB, (d) LB-DKAB. Organo-clays show additional decomposition steps in the 200–500 °C range, consistent with surfactant degradation.
In Figure 5, thermograms for LB and LB-TDAB, LB-ODAB, LB-DKAB are shown. All modified samples exhibit a pronounced mass loss in the range of about 200–500 °C, which is attributed to the thermal degradation of the surfactant molecules within the clay. For instance, LB-TDAB shows a main weight loss event around 250–350 °C (combustion of the C14 surfactant chains), LB-ODAB around 300–400 °C, and LB-DKAB around 350–450 °C. The shift of the degradation to higher temperature with longer chain length suggests that the larger surfactant molecules (especially DKAB) are slightly more thermally stable when confined in the clay, or perhaps decompose in multiple steps. By 600–700 °C, most organic material is burnt off, and the residual mass corresponds to the mineral fraction. Notably, the residual mass at 800 °C is highest for LB-DKAB (∼82.6% of initial mass remaining, meaning 17.4% loss) and lower for LB-ODAB (∼83.3% remaining, 16.7% loss) and LB-TDAB (∼85.0% remaining, 15.0% loss). These residuals quantitatively indicate that LB-DKAB had the greatest amount of organic content (approximately 17.4% by weight), followed by LB-ODAB (16.7%) and LB-TDAB (15.0%). This trend is consistent with the surfactant loading results and the molecular weights of the surfactants (DKAB, being the heaviest, contributes more weight percent at similar intercalation levels).
These thermal decomposition profiles are consistent with previously reported behavior of alkylammonium-modified bentonites, where mass loss in the 200–500 °C range has been attributed to surfactant degradation (He et al., 2010; Vidal and Volzone, 2012). Moreover, it has been shown that the decomposition temperature tends to increase with surfactant chain length due to enhanced van der Waals interactions and stronger confinement effects within the clay interlayer (Carmody et al., 2007; Gerasin et al., 2005).
The TGA confirms that surfactant loading increases the overall organic content of the clay and also slightly enhances its thermal stability up to a point. The modified samples maintain integrity up to ∼200 °C (no major weight loss until the surfactant burns), which implies they could be safely used in applications under moderately high temperatures that would completely dry out the clay (removing water) but not yet burn the organic modifier. The highest char yield (residual) for LB-DKAB indicates more organics were present, aligning with the notion that longer chains (with more carbon) were intercalated. After ∼500 °C, all samples converge in weight, representing primarily the inorganic oxide framework. In summary, the presence of long-chain surfactants in the interlayers notably affects the thermal decomposition behavior, with each additional –CH2– group contributing to the weight loss in the 200–500 °C range.
Surface charge (point of zero charge)
The point of zero charge (PZC) is the pH at which a material's surface has neutral net charge (equal positive and negative charges). For raw montmorillonite (which has a basic character due to alkaline cations), the PZC was found to be around 9.6. We measured the suspension pH as a function of clay addition in solutions of preset pH to determine the PZC. Figure 6 shows the dependence of equilibrium pH on the amount of raw bentonite added, for initial solution pH values of 4.0, 5.9, and 10.0. As clay is added, the pH curves converge and plateau. For the raw clay, at sufficient clay content (>0.4 g/100 mL), the pH stabilizes around 9.6 regardless of starting pH, indicating PZC ≈ 9.6.

Determination of point of zero charge (PZC) for raw bentonite (LB). The graph plots final suspension pH versus montmorillonite content (g/100 mL) added, for different initial pH values of the NaCl solution (pH 4: ▪, pH 5.9: ●, pH 10: ▴). The convergence of the curves at pH ∼9.6 reflects the PZC of the unmodified clay. At this point, adding more clay does not change the pH, indicating neutralization of surface charge effects.
For the organo-modified clays, the PZC shifted to lower values. Figure 7 compares the pH vs. clay content behavior for raw and modified samples when the initial pH is 5.9 (near neutral). The modified bentonites (LB-TDAB, LB-ODAB, LB-DKAB) all stabilize the pH at around 9.0–9.1, whereas the raw LB stabilizes at 9.6. Thus, the PZC of the modified clays is in the range ∼8.9 to 9.1. This decrease in PZC is attributed to the replacement of inorganic cations (which impart basicity) with quaternary ammonium cations. Although quaternary ammonium groups are cationic, they are buried in organic coating and do not make the clay surface as basic as Na+ does. In fact, the long hydrocarbon tails render the surface more hydrophobic and less able to attract H+ or OH– from solution. The net effect is that at any given pH below ∼9, the modified clays carry a bit more negative charge (or less positive charge) than the raw clay, consistent with their more hydrophobic, organophilic nature.
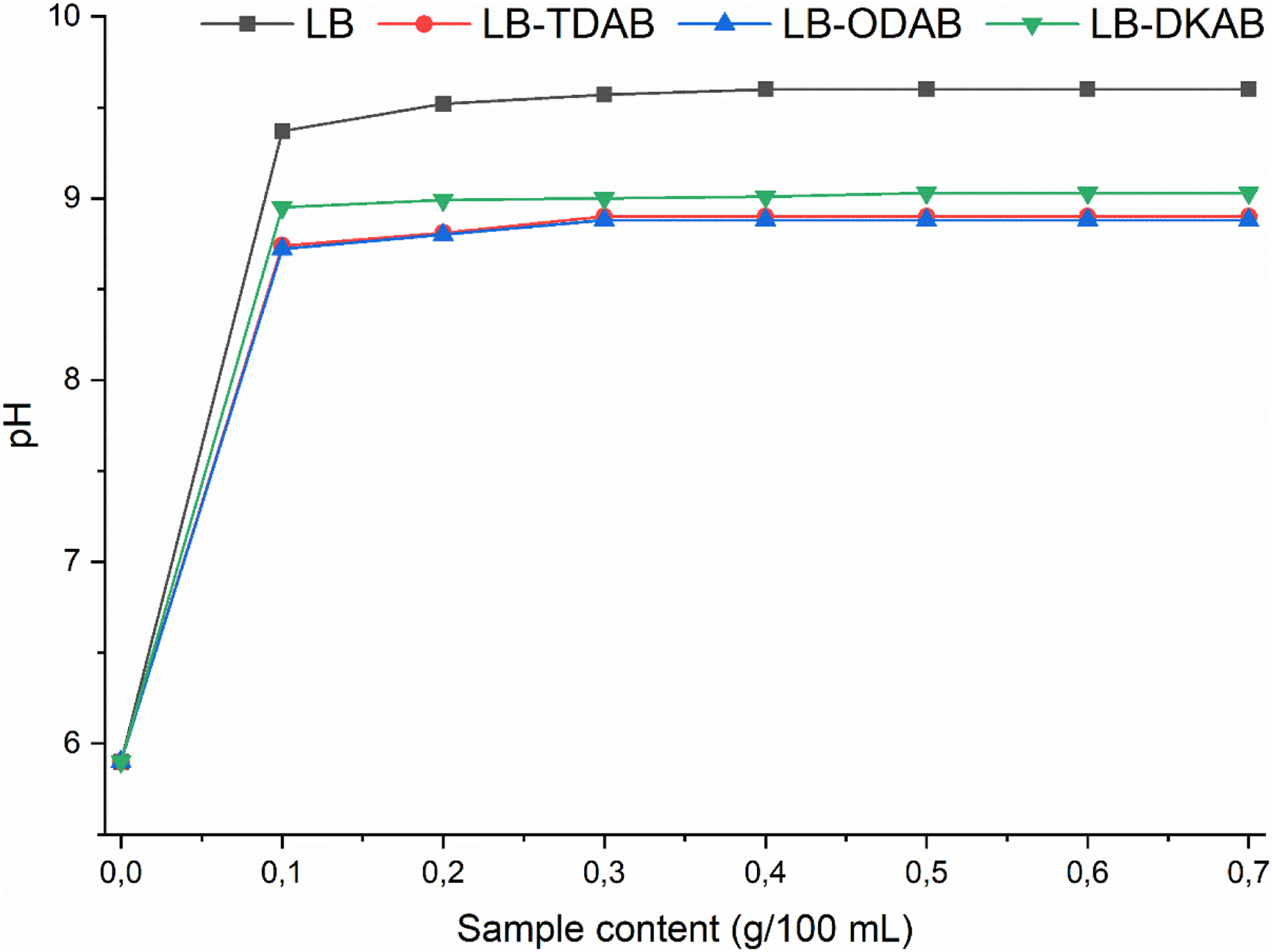
Ph stabilization curves for raw and surfactant-modified clays (initial solution pH = 5.9 in 0.01 M NaCl). Equilibrium pH is plotted against added sample mass (g/100 mL) for LB (▪), LB-TDAB (●), LB-ODAB (▴), and LB-DKAB (▾). The raw clay reaches pH ∼9.6 at sufficient dosage, whereas all modified clays level off around pH 9.0–9.1, indicating a lower PZC after surfactant modification.
The decrease in PZC from 9.6 to ∼9.0 may seem insignificant, but it is important to confirm the change in surface charge characteristics.
A lower PZC means the clay surface becomes neutral at a lower pH, so above that pH it is more negatively charged than before. This is advantageous for adsorption of cationic species or for remaining dispersed in basic environments. In practical terms, the organo-clays will have slightly different colloidal behavior and interaction with ions compared to the raw clay, an aspect important in processes like water treatment or catalysis where surface charge plays a role.
Porosity and surface area
Nitrogen adsorption–desorption isotherms (Figure 8) were measured to evaluate the textural characteristics (surface area and porosity) of the samples. The raw bentonite (LB) exhibited an isotherm of type IV(a) with a noticeable uptake at low relative pressures (P/P0 < 0.1) due to interlayer microporosity, followed by a continuous increase in adsorption at higher relative pressures, indicating the presence of mesopores or slit-shaped interparticle voids. The BET specific surface area of LB was found to be about 31.4 m2/g (consistent with literature values for montmorillonite). The total pore volume (at P/P0 ∼0.99) for LB was around 0.085 cm3/g, with an average pore diameter of ∼41 Å (calculated by BET method). These numbers indicate a porous structure comprising interlayer gaps and inter-aggregate voids.

Nitrogen adsorption–desorption isotherms of raw and modified bentonite samples at 77 K. Reduced N2 uptake after modification indicates decreased accessible surface area.
Upon surfactant modification, a notable decrease in surface area was observed. LB-TDAB showed SBET ≈ 15.6 m2/g, roughly half that of raw LB. LB-ODAB and LB-DKAB had even lower surface areas, ∼14.7 and 11.6 m2/g, respectively. This trend correlates with the increasing surfactant chain length: longer chains lead to greater blockage of surface sites and filling of pores. The surfactant molecules residing in the clay's interlayer and on external surfaces effectively reduce the accessible surface area to N2. Moreover, longer chains may cause particles to pack more tightly, further reducing external surface area. The pore volume likewise decreased (e.g. from 0.00614 cm3/g for LB to 0.00414 cm3/g for LB-ODAB, etc.), supporting the idea of pore filling/blockage by surfactants (Table 1).
Interestingly, the average pore diameter (by BET) slightly increased for the modified clays (e.g. LB-ODAB ∼48.8 Å, LB-DKAB ∼47.1 Å vs. LB ∼41.0 Å). This can be rationalized by the creation of some larger voids due to layer expansion, even though many smaller pores are filled. Essentially, while the total number of pores (and total volume) drops, the distance between layers increases, so any remaining pores in the interlayer region are wider. In LB-DKAB, the presence of the longest chains likely forces interlayer spacing large enough to qualify as mesopores (20–50 Å), thereby raising the “average” pore size even as overall porosity is reduced.
The reduction in surface area and pore volume upon surfactant treatment has implications for adsorption performance: fewer adsorption sites are accessible for gases like N2 (and possibly for adsorbate molecules in solution). However, the newly organic-rich surface may have a higher affinity for certain organic contaminants, potentially compensating for the loss of surface area in those specific cases. The slight increase in average pore width for ODAB and DKAB modified clays suggests that some of the clay layers are held further apart (as confirmed by XRD), providing spaces that could accommodate larger adsorbate molecules than the unmodified clay could.
Adsorption of organic molecules
Despite the surface area reduction, the organo-montmorillonites showed improved adsorption capacity for certain organic molecules, owing to the enhanced organic compatibility and expanded interlayers. Two probe dyes of different size and chemistry—methylene blue (MB, cationic) and Congo red (CR, anionic)—were used to evaluate adsorption in water. Similar improvements in dye adsorption due to surfactant intercalation have been reported for other organo-montmorillonite systems (Lee et al., 1989; Zhu et al., 2003), where increased interlayer spacing and surface hydrophobicity enhanced affinity for organic molecules.
For methylene blue, which is a cationic dye, the raw bentonite has a decent adsorption capacity due to cation exchange and surface charge attractions. The modified clays, however, present a more hydrophobic and positively charged surface (from quaternary ammonium groups), which might seem counterintuitive for uptaking a cationic dye. Indeed, the equilibrium adsorption tests showed that LB-ODAB had the highest MB uptake (∼10.6 mg/g), slightly outperforming LB-TDAB and LB-DKAB (which were around 8–9 mg/g). Raw LB reached about 8.85 mg/g at similar conditions (Figure 9, Table 2). Thus, ODAB-modified clay was the most effective for MB. This can be explained by a balance of factors: ODAB's interlayer spacing is significantly larger than raw clay (facilitating MB entry), and its surface, while bearing cationic sites, may still adsorb MB through π–π interactions with surfactant alkyl chains or via residual negative sites not occupied by surfactant. Such mechanisms have also been proposed in studies of cationic dye uptake on organoclays (Zhu et al., 2015). Meanwhile, TDAB may have less available space for MB (smaller expansion), and DKAB, although expanded, might have fewer accessible charged sites (because its high surfactant packing could neutralize most negative sites). The net result is an optimal chain length (C18) for MB adsorption in this system.
For Congo red, an anionic dye, the organo-clays showed substantially higher adsorption than the raw clay. Raw LB, being negatively charged at typical pH (∼9), repels the anionic CR to an extent (unless CR intercalates as a planar molecule). The organo-clays, however, have positively charged surfactant headgroups that can strongly attract the sulfonate groups of CR. Among the modified samples, LB-TDAB exhibited the highest CR uptake (∼13.6 mg/g), followed by LB-DKAB and LB-ODAB (both a bit lower). This trend suggests that while all surfactant-modified clays bind anionic dyes well, the one with shorter chains (TDAB) performed best, likely because it had the most accessible quaternary ammonium sites per unit area (since TDAB's shorter chains occupy less space, allowing more surfactant molecules per layer or more exposure of charges). This observation aligns with findings in earlier studies, where shorter alkyl chains provided higher site availability and enhanced anionic dye adsorption (Açışlı et al., 2016). LB-DKAB, despite having more total surfactant by mass, might have its quaternary amines somewhat buried under a thick layer of C22 tails, making some of them less reachable to the dye. LB-ODAB falls in between. These results align with the Langmuir isotherm modeling: LB-TDAB had the highest monolayer capacity (A0) for CR and a high affinity constant, whereas LB-ODAB and LB-DKAB had slightly lower A0.
The adsorption isotherms for CR (Figure 10) were well fit by the Langmuir model with correlation coefficients R2 > 0.98 for the modified clays (Table 3), indicating monolayer adsorption on homogeneous sites. The calculated Gibbs free energy changes (ΔG) for adsorption were on the order of −21 to −23 kJ/mol for the organo-clays, more negative than that of raw LB (–10.3 kJ/mol), signifying that CR adsorption is significantly more favorable on the surfactant-modified surfaces. The stronger uptake of CR by organo-clays underscores the importance of tailoring clay surfaces for target pollutants: introducing cationic surfactants creates binding sites for anionic contaminants.

Adsorption isotherms of methylene blue on organobentonites (pH 6.8–7.2).
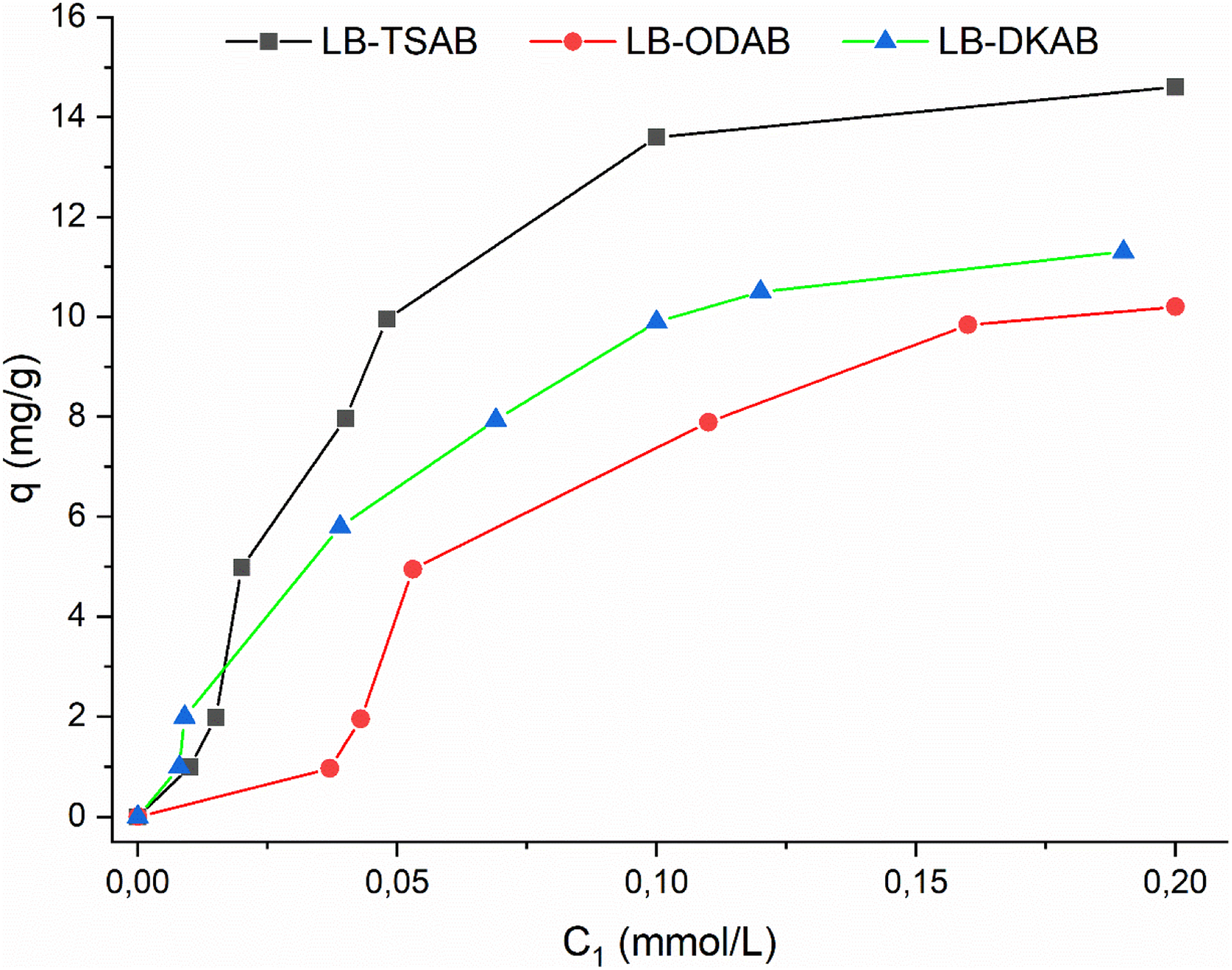
Adsorption isotherms of Congo red on organobentonites (pH 6.8–7.2).
Textural characteristics of the samples.

Adsorption parameters according to the Langmuir equation for Methylene Blue.

Adsorption parameters according to the Langmuir equation.

It is noteworthy that even though LB-DKAB has the largest interlayer spacing, it did not yield the highest adsorption in every case (it was highest for TGA residuals and second-best for CR uptake). This suggests that beyond a certain chain length, additional increases in interlayer spacing do not translate to proportionally greater adsorption, possibly due to steric hindrance or reduced accessible surface. In contrast, LB-TDAB, which had the smallest spacing among modified clays, achieved the greatest CR adsorption, likely due to an optimal combination of accessible charged sites and still sufficient gallery size to accommodate dye molecules in a planar orientation.
Overall, the adsorption studies demonstrate that organo-montmorillonite systems can be fine-tuned by selecting appropriate surfactant chain lengths. Shorter chains (C14) leave more active sites exposed and are highly effective for larger anionic dyes, whereas intermediate chains (C18) maximize uptake for smaller cationic dyes in this case. Longer chains (C22) greatly increase hydrophobicity and thermal stability but can overcrowd the surface. The enhanced benzene vapor adsorption observed qualitatively (not shown in a figure) also confirms that all modified clays are much more organophilic than raw clay: after surfactant treatment, the clays readily absorbed benzene (a nonpolar solvent) in their pores, whereas raw clay adsorbed very little. This property is beneficial for applications like organic pollutant removal from air or oil spill cleanup, where organoclays often outperform unmodified clays (Smith et al., 1990; Zhu et al., 2003).
Conclusion
As a result of this study, it is evident that intercalation of cationic surfactants with different hydrocarbon chain lengths into montmorillonite significantly alters the clay's structure and adsorption properties. In particular, the basal spacing of montmorillonite increased from 1.24 nm (unmodified) up to 2.32 nm for the longest surfactant (DKAB), confirming that longer hydrocarbon radicals produce greater clay layer expansion. This expansion, however, is accompanied by a decrease in specific surface area and total pore volume, especially pronounced for longer-chain modifiers. The surfactant-modified clays exhibit reduced hydrophilicity and a slight lowering of the point of zero charge (from pH 9.6 to ∼9.0), reflecting the more hydrophobic, organophilic surface introduced by the surfactants.
Despite a reduction in overall surface area, the organo-clays show improved adsorption performance for organic molecules. The modified bentonites had markedly higher affinity for anionic dye (Congo red), with LB-TDAB achieving the highest adsorption capacity (∼13.6 mg/g), owing to the presence of quaternary ammonium sites that bind anionic species. Cationic dye (methylene blue) adsorption was also favorable on the modified clays, particularly for the ODAB-modified sample, which balanced interlayer accessibility and available adsorption sites. Thermogravimetric analysis indicated that surfactant modification increases the organic content and enhances thermal stability—DKAB-modified clay had the greatest residual mass at high temperature, aligning with its higher organics loading.
In summary, the hydrocarbon chain length of the surfactant plays a crucial role in determining the structural and adsorption characteristics of organo-montmorillonite systems. Shorter chains (C14) are effective at introducing functional adsorption sites while maintaining more of the clay's surface area, whereas longer chains (C22) maximize layer expansion and hydrophobicity at the expense of some porosity. The intermediate chain (C18) offers a balance of these effects. For practical applications in separation science and technology, this means one can select a surfactant of appropriate chain length to target specific contaminants: for example, a TDAB-modified clay for removing large anionic pollutants, or an ODAB-modified clay for smaller organic cations or for use in organic solvent environments. The modified clays developed in this work show potential as effective adsorbents for environmental remediation, with tunable properties based on the choice of surfactant. The clear trends observed provide valuable guidance for designing clay-based adsorbents and catalysts in which organophilicity and structural openness must be optimized.
Footnotes
ORCID iDs
Funding
The study was carried out within the framework of the budget project of the Colloid Chemistry and Industrial Ecology Laboratory, Institute of General and Inorganic Chemistry, Academy of Sciences of Uzbekistan – “Synthesis and analysis of hierarchically structured adsorbents: interrelation between structure, adsorption capacity, kinetics and adsorption thermodynamics”.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.