Abstract
Coal seam water injection is a coal mine safety technology used to inhibit coal dust and reduce the potential risk of gas explosions, but its effectiveness is limited by the wettability of the coal seam. Anthracite coal, due to its strong hydrophobicity, is difficult to fully combine with injected water. To improve the wettability of anthracite coal, this study systematically investigates the regulatory effects and wettability mechanisms of the non-ionic surfactant dodecyl β-
Introduction
The safe production of coal mines has always been a core concern in the mining industry, with the prevention of coal dust and gas explosions being particularly critical. As an effective coal mine safety technology, coal seam water injection is widely used to reduce coal dust generation and lower the risk of gas explosions (Li et al., 2024; Tian et al., 2024). However, its practical effectiveness is often limited by the wettability of the coal seam. In particular, for anthracite, the strong hydrophobicity of its surface makes it difficult for the injected water to adequately wet the coal seam surface, severely restricting the application performance of the coal seam water injection technology. Therefore, improving the wettability of anthracite has become an important scientific and technical challenge that needs to be addressed urgently.
Surfactants are considered effective agents for improving the wettability of coal seams, which significantly reduces the hydrophobicity of the coal surface by adjusting the chemical composition and physical properties of the coal surface, thereby enhancing the wettability (Abbood et al., 2022; Dahham et al., 2023; Du et al., 2024; Golab et al., 2024; Golabi 2012; Guanhua et al., 2019; Hao et al., 2024; Hosseini et al., 2024; Ji et al., 2023; Xiao et al., 2024; Zhou et al., 2018). For example, Wei et al. (2022) discovered through triaxial seepage and wettability experiments that surfactants can enhance the permeability and wettability of coal by altering capillary forces, thereby improving the efficiency of coal seam water injection. Zheng et al. (2021) used nuclear magnetic resonance and optical titration methods to investigate the structural parameters and wettability relationships between coal and surfactants. They established a link between the microscopic structure of surfactants and coal sample wettability, finding that ether groups, phenols or aromatic ether carbons, fatty methyl and aromatic methyl significantly affect coal wettability. Niu et al. (2021) demonstrated that surfactants can reduce the surface tension and contact angle of pure water, thereby significantly enhancing the wettability of coal dust. Chen et al. (2019) evaluated the reliability of four common methods (sessile drop measurement, sink test, capillary rise test and drop penetration test) for determining coal wettability in the presence of surfactants. They found that the sink test was the most reliable for quantitatively comparing the wettability of coal particles of different sizes. Further studies revealed that the addition of surfactants significantly improved coal wettability compared to the use of water alone. Additionally, Wang et al. (2019) studied the wettability and adsorption mechanisms of four surfactants on coal dust surfaces through surface tension and contact angle measurements. Their results indicated that both anionic and non-ionic surfactants effectively improved the wettability of coal dust.
Although the above studies provide important insights into the role of surfactants in improving coal wettability, the existing research primarily focusses on the macroscopic experimental level and the analysis of the microscopic mechanism is not yet sufficient, especially the researches revealing the mechanism of surfactants’ action on the molecular scale are still relatively scarce, lacking systematic and multi-scale analyses. This limitation has, to some extent, hindered the optimised application of surfactants in coal seam water injection technology.
Based on the above background, this study systematically investigates the regulatory effects and mechanisms of the non-ionic surfactant dodecyl β-
Unlike previous studies that predominantly employed a single experimental approach, this work integrates macroscopic physical experiments with microscopic molecular simulations to systematically investigate the wettability regulation of anthracite by the non-ionic surfactant DDM. This multiscale methodology enables a comprehensive understanding of the regulatory mechanism from macroscopic wetting behaviour to molecular-level interactions, addressing the limitations of prior studies that lacked insights into the molecular mechanisms. Furthermore, DDM was selected as a non-ionic, environmentally friendly surfactant with excellent biodegradability, low toxicity and good chemical stability. Compared with conventional cationic or anionic surfactants, DDM offers improved compatibility with coal mine safety and green mining requirements, thus exhibiting great potential for engineering applications.
Based on this, the core scientific hypothesis of this study is proposed: DDM can alter the surface chemical composition and microstructure of anthracite coal through directed adsorption, thereby significantly enhancing its hydrophilicity. To validate the above hypothesis and systematically elucidate the wettability mechanism of DDM, this study sets the following research objectives: to quantify the regulatory effects of DDM on the wettability and surface functional groups of anthracite coal through contact angle, X-ray photoelectron spectroscopy (XPS) and Fourier-transform infrared (FTIR) spectroscopy experiments. Utilise molecular dynamics simulations to reveal the adsorption configurations and energy evolution of DDM at the coal-water interface. Integrate macro-scale experimental and micro-scale simulation results to establish a multi-scale mechanism model for DDM's regulation of anthracite wettability and conduct an in-depth exploration of DDM's adsorption behaviour and micro-scale wettability mechanisms on the anthracite surface. This study aims to reveal the intrinsic mechanisms by which surfactants improve the wettability of anthracite from theoretical and technical multi-scale perspectives, providing a scientific basis for optimising coal seam water injection technology. It also offers valuable practical guidance for enhancing coal mine safety, carrying significant application value and theoretical importance.
Experiments and simulations
Experiments
Materials
Anthracite coal samples were obtained from the Zhaozhuang Coal Mine in Jincheng City, Shanxi Province, China. The coal was crushed and sieved to obtain samples between 200 and 300 mesh (particle size range of 0.074 mm to 0.050 mm). Before the experiments, the coal samples were dried in a vacuum oven at 105 °C for 2 hours until their mass stabilised, then cooled and sealed in a desiccator for use. To minimise the influence of carbonates, phosphates, clays, silica and other minerals in the coal, the samples underwent a three-step acid washing process with HCl–HF–HCl. They were then washed with excess deionised water until the filtrate reached neutral pH. After deashing, the ash content of the coal samples was reduced to below 0.3% (Chen et al., 2022; Strydom et al., 2011). To ensure environmental stability during coal sample preparation, all experimental procedures were conducted in a laboratory environment maintained at a temperature of 25 ± 1 °C and relative humidity below 50%. During sample drying and storage, a vacuum drying oven and sealed desiccator with temperature and humidity control functions were used to prevent moisture in the air from interfering with the surface properties of the coal samples.
Adsorption experiment
Before subsequent tests, an adsorption experiment was conducted on the acid-washed coal samples. The anthracite samples were treated with a 200-mg/L solution of the non-ionic surfactant DDM for adsorption. A mixture of 400 mg of acid-washed coal and 400 mL of DDM surfactant solution was stirred at 800 rpm for 10 hours using a magnetic stirrer set at a constant temperature of 25 °C. The adsorption experiments were conducted at room temperature, with the temperature maintained at 25 ± 1 °C. The adsorption system was stirred in a sealed container to avoid interference from airflow or humidity fluctuations. A constant-temperature water bath was used to maintain the stability of the solution temperature. All solutions and coal samples were pre-equilibrated to the target temperature prior to the experiment to ensure that the adsorption behaviour occurred under isothermal conditions. After sufficient adsorption of the surfactant onto the coal samples, the mixture was centrifuged at 4000 rpm for 20 minutes. The coal samples from the bottom of the centrifuge tube were collected, filtered three times using a vacuum pump and washed with deionised water to remove residual surfactant (Li et al., 2023). For further study, the resulting wet coal samples were dried under vacuum at 105 °C for 2 hours and stored for later use.
Contact angle measurement
Coal powder prepared from the above process was used for contact angle measurements. Three groups of pre-treated coal powder samples were prepared. For each group, 200 mg of coal powder was weighed using an electronic balance, pressed under 20 MPa for 5 minutes using a YP-15 infrared powder tablet press and shaped into circular coal discs with a diameter of 12 mm and a thickness of about 2 mm (Meng et al., 2023). The contact angles between the coal samples and deionised water or DDM monomer solution were measured using a JY-82C contact angle goniometer. To ensure the stability and reproducibility of contact angle measurements, all measurements were conducted in a constant temperature and humidity environment, with the temperature maintained at 25 ± 1 °C and relative humidity below 50%. The coal samples were allowed to equilibrate for at least 30 minutes prior to measurement to adapt to the testing environment. Each experiment was repeated three times, and the average value was calculated.
XPS measurement
X-ray photoelectron spectroscopy (XPS) measurements were performed using the Thermo Scientific K-Alpha X-ray photoelectron spectrometer with a monochromatic Al target (Kα hν = 1486.6 eV) as the X-ray source. During the test, the vacuum level of the analysis chamber was maintained at better than 5.0 × 10+7 mBar. The channel energy for full spectrum scans was set to 100 eV, with a measurement scan resolution of 1 eV. Narrow spectrum scans were performed with five signal accumulations (the number of scans varied by element), a channel energy of 50 eV, and a step size of 0.05 eV, mainly focussing on scanning the C 1s and O 1s orbitals. The binding energy was charge-corrected using contaminated carbon (C 1s = 284.8 eV) as the reference (Li et al., 2023).
FTIR measurement
FTIR measurements were performed using the Thermo Scientific Nicolet iS20 spectrometer. During the sample preparation phase, the adsorbed samples and KBr solid powder were separately placed in a vacuum drying oven to remove moisture, preventing its influence on the FTIR results. Subsequently, 2 mg of the adsorbed sample and 200 mg of pure KBr were mixed in a 1:100 mass ratio, ground evenly, placed in a mold and pressed into flakes under 10 MPa using a powder tablet press. The scanning wavenumber range for the FTIR test was set from 4000 to 400 cm−1, with 32 scans, a resolution of 4 cm−1. The moving mirror speed was set to 0.4747.
Molecular dynamics simulations
The construction of the Jincheng anthracite unit cell model was based on the experimental data of anthracite samples previously obtained by our research group (Yan et al., 2020). The unit cell dimensions of the anthracite model are 5.4 × 4.8 × 5.5 nm3 (X Y Z), and the model density is 1.43 g/cm3. The surfactant layer contains 10 molecules, and the water layer contains 2000 molecules. The models of the DDM surfactant and water molecules are shown in Figure 1. Molecular dynamics simulations were conducted using Materials Studio 8.0, with the COMPASS force field used to describe the interactions among anthracite, surfactant and water molecules. This force field has been widely used in simulation studies of coal molecular systems and surface adsorption behaviour (Bai et al., 2024; Hu et al., 2010; Yan et al., 2023; Yang et al., 2023). Prior to MD simulations, energy minimisation of all models was performed using the Smart algorithm. The NVT ensemble was then used for MD simulations with temperature control via the Nose thermostat. This temperature control method can effectively control temperature fluctuations in the system, ensure stable energy convergence and facilitate the reliable generation of equilibrium configurations in the system. The time step was set to 1 fs, and the temperature was maintained at 298 K. The total simulation time for the adsorption process was set to 1000 ps to ensure adequate equilibration of the system. For all MD simulations, the Ewald method was used to calculate long-range electrostatic interactions with a precision of 0.001 kcal/mol, and van der Waals interactions were calculated using the Atom-based algorithm with a cutoff distance of 1.25 nm. A vacuum layer of 8 nm was added on the surface of each model to eliminate neighboring layer interference. The above simulation parameters have been verified as feasible in multiple coal molecule system simulation studies (Gan et al., 2023; Meng et al., 2025) and can effectively capture the microscopic mechanisms involved in the interface adsorption process, ensuring the physical credibility and reproducibility of the simulation results. As shown in Figure 2, molecular dynamics simulations were used to study the interactions between water, surfactants and anthracite, with the study process divided into two stages. The first stage focussed on the adsorption process of the surfactant onto anthracite, aiming to analyse its adsorption configuration and interaction energies. In this stage, the surfactant layer was built on the anthracite surface using the Build Layer function, followed by a 1000-ps molecular dynamics simulation using the NVT ensemble. The second stage studied the interactions between water molecules and the surfactant-modified anthracite surface to evaluate the effect of surfactant modification on changes in the wettability of the anthracite surface. A 500-ps molecular dynamics simulation was performed in this stage. The simulation time has been verified through a series of preliminary tests and has been found to enable each system to achieve a relative balance between energy and configuration, meeting the computational requirements for adsorption analysis, energy statistics and hydrogen bond calculations. For comparison, molecular dynamics simulations were also conducted on the interactions between water and unmodified anthracite. To visually present the adsorption configurations more clearly, the unit cell boundaries were hidden in the simulation.

Structural models of DDM and water molecule: (a) DDM; (b) water.

Adsorption process of DDM on anthracite: (a) initial configuration of Stage 1; (b) equilibrium configuration of Stage 1; (c) initial configuration of Stage 2; (d) equilibrium configuration of Stage 2.
Results and discussion
Experimental results analysis
Contact angle analysis
Contact angle is an important indicator for evaluating the wettability of coal samples, and its size can intuitively reflect the hydrophilicity or hydrophobicity of the coal surface (Yuan et al., 2021; Zhang et al., 2020). In this experiment, the contact angle between the DDM surfactant monomer solution and anthracite coal samples was measured to analyse and to assess the ability of the surfactant to enhance the wettability of anthracite coal dust.
During the experiment, all contact angle images within the time interval from when the droplet first contacted the coal sample to when the contact angle stabilised were selected. The contact angle was manually measured using ImageJ image processing software, with three repeated measurements and the average value taken. The experimental results are shown in Tables 1 and 2. The contact angle experiment showed that the angle continuously decreased as the surfactant solution spread over the coal surface. The initial contact angle of the DDM solution on anthracite was 105.3°. As the contact time increased, the droplet gradually spread across the coal surface. The contact angle gradually decreased to 38.2°, reaching equilibrium. At this point, the droplet was fully absorbed by the coal surface. In contrast, as the contact time increased, the contact angle between deionised water and the coal surface changed little and the contact angle was relatively large, indicating that deionised water had poor wettability on the coal sample. The experimental results show that the DDM surfactant significantly outperforms deionised water in regulating the wettability of the coal sample.
Contact angle parameters of DDM on anthracite over time.
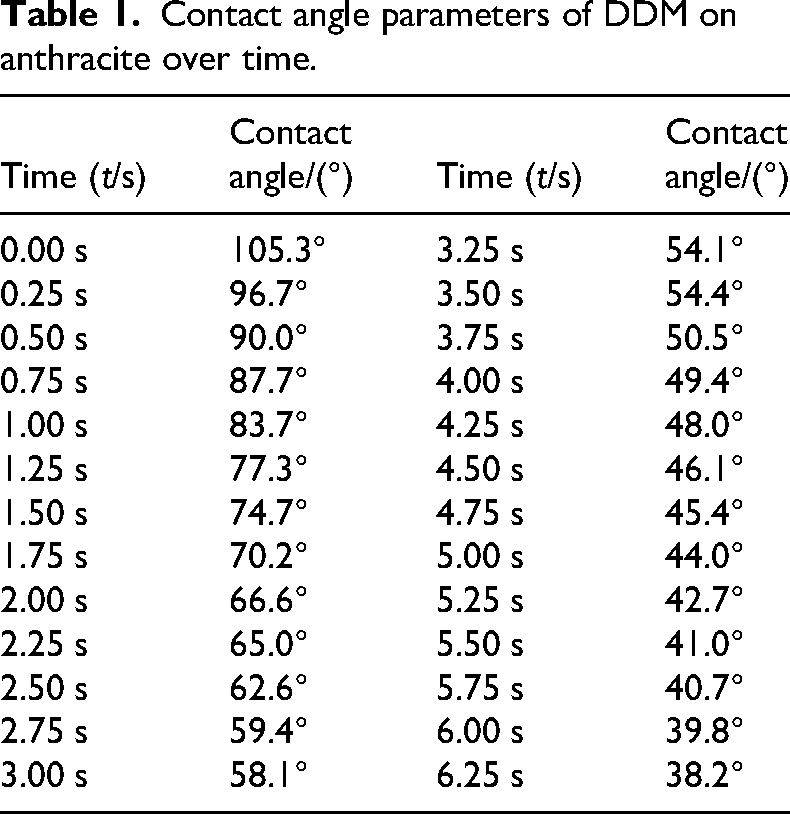
Contact angle parameters of water on anthracite over time.

To further analyse the difference in wettability between the DDM surfactant and deionised water on anthracite coal samples, dynamic contact angle experiments were conducted to measure the entire process of the DDM solution from initial contact with the coal sample to the stabilisation of the contact angle, with deionised water as a comparison. The dynamic changes in the contact angles of the DDM solution and deionised water on the surface of the anthracite coal sample are shown in Figure 3.
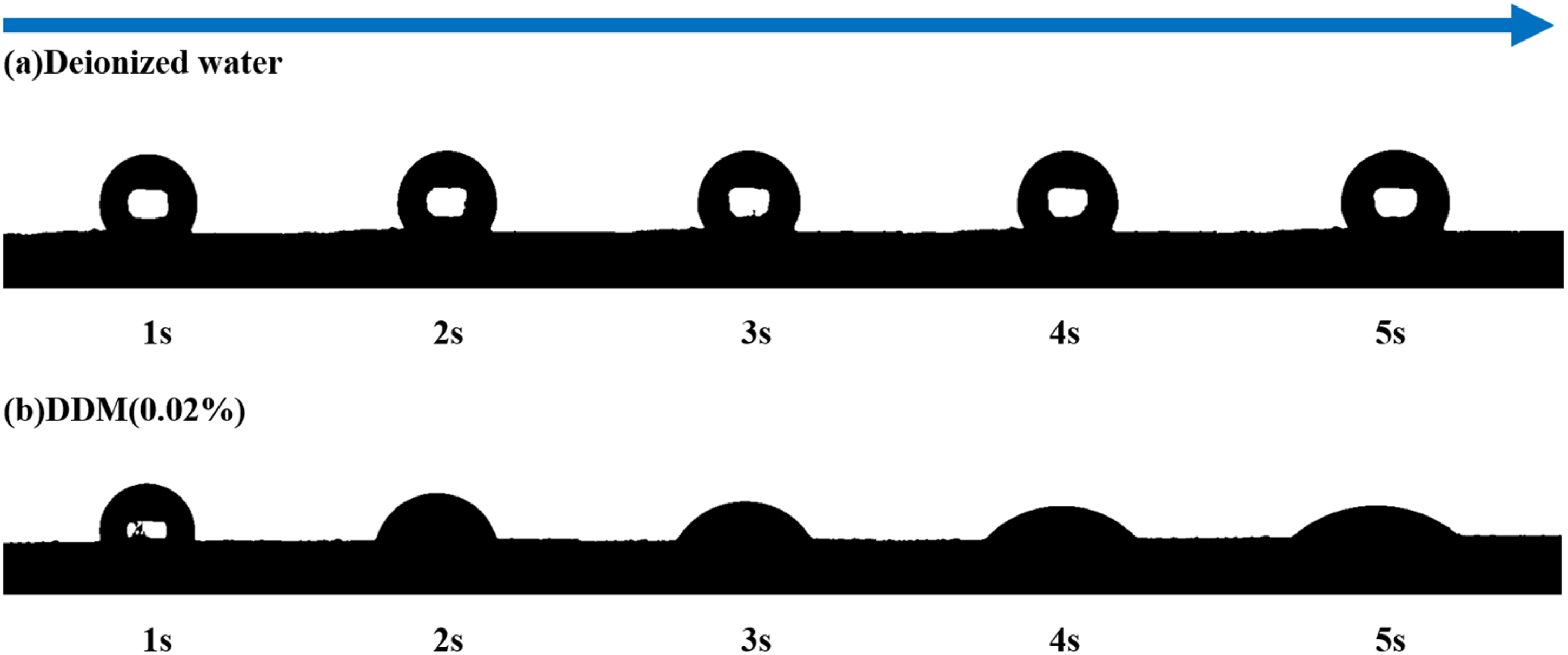
Dynamic contact angles of DDM solution and deionised water on coal surfaces: (a) water; (b) DDM.
All contact angle data are the average values ± standard deviation from three independent replicate experiments. The experiments exhibited good reproducibility, with standard deviation ranges all less than ±1.5°, and the specific values are labelled in the error bars of Figure 5. Additionally, to verify the statistical significance of the wettability differences between DDM and water, an independent t-test was performed on the contact angle data. The results indicated that the differences were statistically significant (p < 0.01), further supporting the enhanced wettability of DDM on anthracite. Based on the analysis results of Figures 4 and 5, it can be clearly observed that the initial and equilibrium contact angles of deionised water on anthracite coal samples did not exhibit significant changes over a specific time interval, with the contact angle values remaining stable between 120° and 122°. This indicates that the anthracite coal samples exhibit strong hydrophobicity, with the contact angle positively correlated to hydrophobicity; the larger the contact angle, the stronger the hydrophobicity.

Changes in dynamic contact angles of DDM and water on anthracite.

Equilibrium contact angles of DDM and water on anthracite.
When the DDM surfactant solution initially contacted the surface of the anthracite coal sample, the initial contact angle was 105.3°, which was slightly lower than the initial contact angle of 121.2° for deionised water, representing a decrease of 13.12%. This result shows that the DDM surfactant begins to exhibit a wettability effect when in contact with the anthracite coal sample, but its initial wettability effect is limited. Over time, the equilibrium contact angle of the DDM surfactant on the anthracite coal sample eventually decreased to 38.2°, demonstrating that DDM significantly reduced the contact angle of the coal sample. Within 6.25 seconds of action, DDM achieved a 63.72% efficiency in reducing the contact angle. As shown in Figure 4, the phase of rapid contact angle change was concentrated within the first 4 seconds, while the rate of change gradually diminished in subsequent periods. During the first 4 seconds, DDM achieved a contact angle reduction efficiency of 53.08%, which decreased to 22.67% in the following 2 seconds. The contact angle test results mentioned above indicate that the wettability control effect of DDM is particularly pronounced within the initial 4 seconds, demonstrating rapid spreading and the ability to form a hydrophilic interface. In contrast, the wettability of deionised water shows almost no significant change, highlighting the distinct molecular interaction differences between DDM and the coal surface. Preliminary analysis suggests that the hydrophilic groups in DDM molecules can rapidly diffuse and arrange themselves on the coal sample surface, reducing surface energy, while their hydrophobic tail chains facilitate interactions with the non-polar regions of the coal surface, enabling droplets to spread rapidly within a short time. To further elucidate the reasons behind DDM's enhanced wetting properties, subsequent analyses will be conducted using XPS and FTIR characterisation techniques.
XPS analysis
The adsorption distribution of a 200-mg/L DDM solution on the surface of anthracite coal was analysed using the XPS method, along with the surface properties of raw anthracite coal. The relative atomic content of major surface elements (excluding H) is presented in Table 3, and the corresponding XPS spectra are shown in Figure 6.

XPS spectra of raw anthracite and DDM-modified anthracite: (a) raw coal; (b) DDM–coal.
The relative content of the atomic number of the main elements (except H) on the surface of anthracite after adsorption.
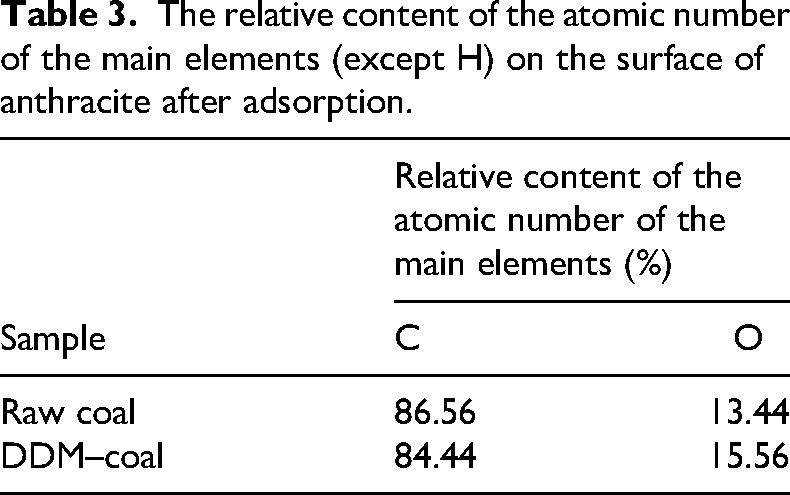
The XPS analysis results indicated that the anthracite surface contained high levels of C and O elements. Analysing the content of these elements and their corresponding functional groups effectively characterises the changes in wettability of anthracite after modification with a non-ionic surfactant. As shown in Table 3 and Figure 6, the high coalification of anthracite leads to low oxygen content, which contributes to poor wettability. Specifically, in unmodified anthracite samples, the C element accounts for 86.56%, while the O element accounts for only 13.44%.
After modification with the DDM surfactant, DDM molecules adsorbed on the anthracite surface, reducing the C content to 84.44% and increasing the O content to 15.56%. It is well known that C elements primarily exist in aromatic and aliphatic hydrocarbon structures such as –CH2, –CH3, C=C, C–C and substituted benzene rings, which are typically hydrophobic groups. In contrast, oxygen-containing functional groups, such as hydroxyl, carbonyl and carboxyl groups, are typical hydrophilic groups.
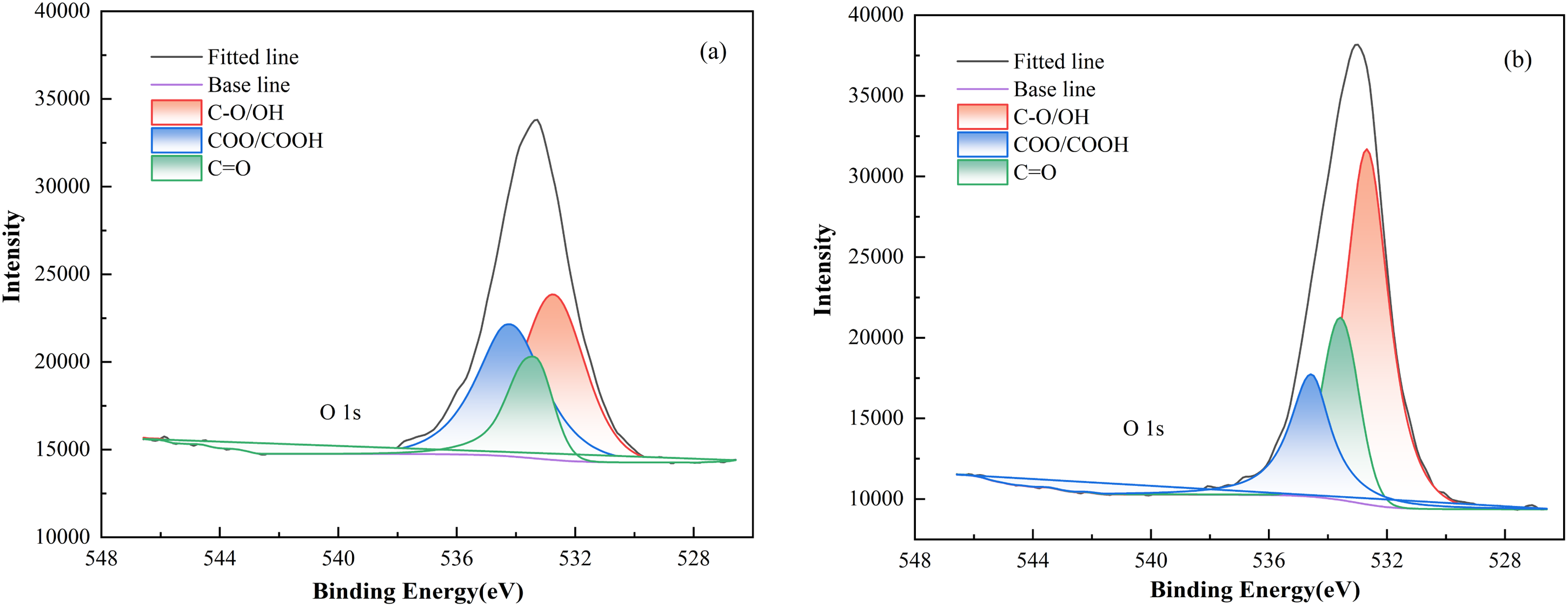
The XPS results reveal that after DDM modification, the relative content of hydrophobic groups on the anthracite surface decreases, while the relative content of hydrophilic groups increases. Thus, a comparative analysis of C and O element contents preliminarily concludes that the hydrophilicity of anthracite coal is significantly enhanced after DDM adsorption. These changes indicate that DDM adsorption not only affects the elemental composition of the coal surface but also causes hydrophobic groups to be partially masked, while hydrophilic functional groups such as hydroxyl and carbonyl groups are enriched at the interface, thereby enhancing the coal's affinity for water molecules. This structural change in functional groups is an important physicochemical basis for the enhanced wettability. Although it has been demonstrated that the DDM surfactant effectively improves the wettability of anthracite coal, further investigation into the states of C and O elements in the surface layer after surfactant adsorption is necessary. To better understand the adsorption characteristics of C and O elements in different states on the coal surface, split-peak fitting was performed on the carbon and oxygen spectra based on the binding energy differences of core electrons. The C 1s and O 1s XPS spectra of raw anthracite coal and coal samples after surfactant adsorption are shown in Figures 7 and 8. The peak positions, areas and atomic assignments for C 1s and O 1s are detailed in Tables 4 and 5.

XPS C 1s spectra of raw anthracite and DDM-modified anthracite: (a) raw coal; (b) DDM–coal.
XPS O 1s spectra of raw anthracite and DDM-modified anthracite: (a) raw coal; (b) DDM–coal.
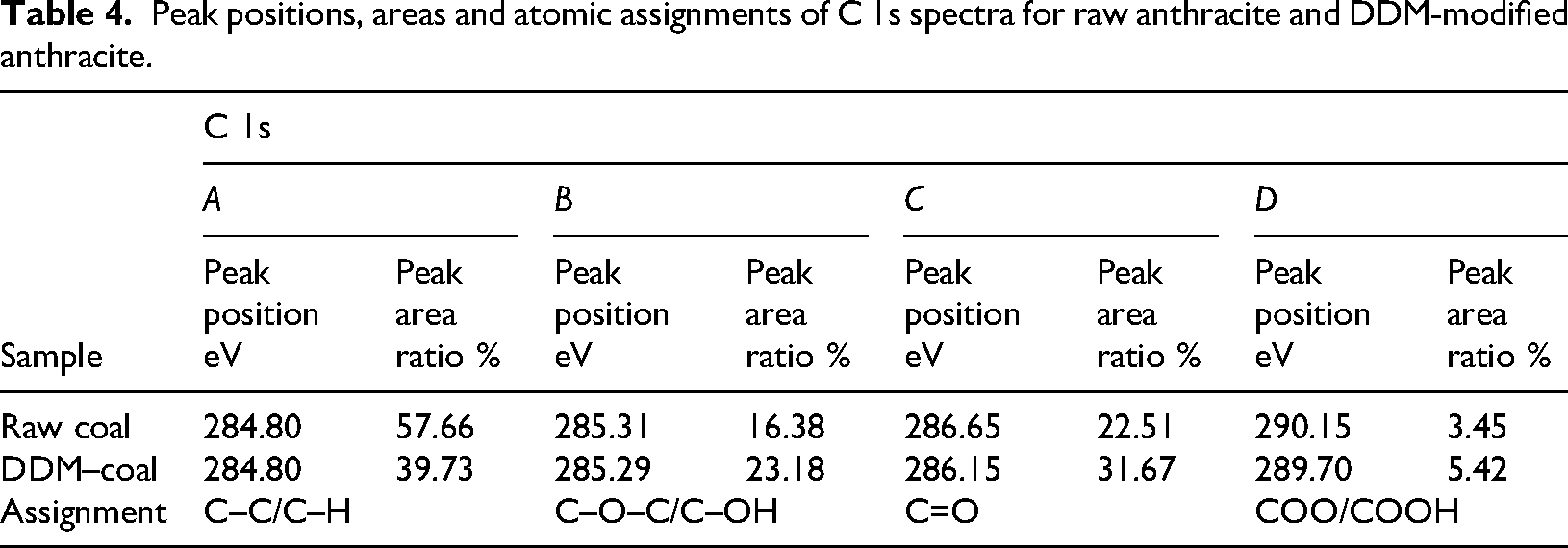
Peak positions, areas and atomic assignments of C 1s spectra for raw anthracite and DDM-modified anthracite.
Peak positions, areas and atomic assignments of O 1s spectra for raw anthracite and DDM-modified anthracite.
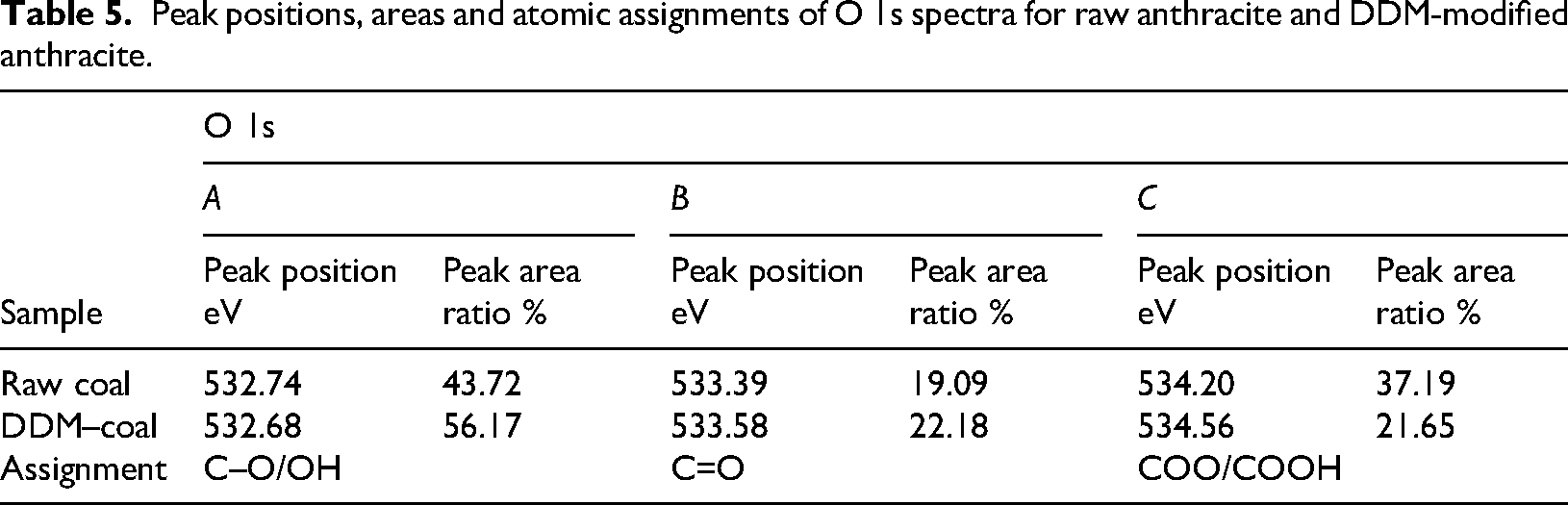
Based on the analysis of the C 1s peak area proportions in Figure 7 and Table 4, the content of aliphatic hydrocarbons (C–C, C–H) on the raw anthracite surface is the highest, accounting for 57.66% of the peak area. The content of ether bonds (C–O–C) and hydroxyl groups (C–OH) is 16.38%, carbonyl groups (C=O) account for 22.51% and carboxyl groups (COOH) make up 3.45%. After modification with DDM, significant changes are observed in the functional group proportions. The content of aliphatic hydrocarbons (C–C, C–H) decreased sharply by 17.93%, dropping to 39.73%. The content of ether bonds (C–O–C) and hydroxyl groups (C–OH) significantly increased by 6.8%, reaching 23.18%; the content of carbonyl groups (C=O) increased by 9.16%, rising to 31.67%, while the content of carboxyl groups (COOH) shows a slight increase to 5.42%. The observed phenomenon may be attributed to the adsorption of the surfactant on the anthracite surface, which forms a dense network structure. This structure encapsulates the hydrophobic groups of anthracite within the interior while exposing the hydrophilic groups outward. As a result, hydrophobic functional groups, such as aliphatic hydrocarbons, are covered by the surfactant, whereas oxygen-containing hydrophilic functional groups become the dominant components of the new interfacial layer. The surfactant-adsorbed layer, which is enriched with oxygen-containing functional groups, facilitates direct contact between water and the hydrophilic regions of the anthracite surface. Consequently, the content of oxygen-containing functional groups significantly increases, surpassing that of hydrophobic groups, leading to an overall enhancement in the hydrophilicity of the coal surface. In summary, the DDM surfactant effectively modifies the anthracite surface and significantly enhances its wettability. This conclusion aligns well with the results from the full-spectra analysis, further confirming the reliability of the experiments. Based on the analysis of O 1s binding energy, peak area and proportion shown in Figure 8 and Table 5, the relative content of oxygen-containing functional groups on the untreated anthracite surface is ranked as follows: C–O/OH > COO/COOH > C=O. Specifically, the content of C–O/OH accounts for 43.72%, COO/COOH for 37.19% and C=O for 19.09%. Upon the adsorption of the surfactant, the relative content of these groups changes significantly, resulting in the following order: C–O/OH > C=O > COO/COOH. The C–O/OH content increases by 12.45% to 56.17%, the C=O content increases by 3.09% to 22.18%, while the COO/COOH content decreases sharply by 15.54%, dropping to 21.65%.
This phenomenon may be due to the surfactant forming a coating layer on the anthracite surface, with its hydrophilic groups oriented outward. The surfactant contains abundant free hydroxyl groups, which manifest as a significant increase in C–O/OH content during the adsorption process. The reduction in COO/COOH content may result from strong interactions between the surfactant and the COO/COOH groups on the untreated anthracite surface, leading to tighter binding and partial coverage of these groups, thereby reducing their surface content. Additionally, such interactions likely contribute to the relative increase in C=O content.
FTIR analysis
FTIR spectroscopy was used to analyse the surface characteristics of raw anthracite coal and the adsorption distribution of 200 mg/L DDM surfactant solution on the anthracite coal surface. The corresponding FTIR spectra are shown in Figure 9.

FTIR spectra of raw anthracite and DDM-modified anthracite: (a) raw coal; (b) DDM–coal.
As shown in Figure 9(a), two minor absorption peaks are observed at wavenumbers 3756 cm−1 and 3662 cm−1 on the surface of raw anthracite coal. These peaks are primarily attributed to –NH2 stretching vibrations and hydroxyl structures. Within this wavenumber range, no significant peaks were observed after the adsorption of DDM surfactant on anthracite coal, indicating that nitrogen is only present on the surface of raw coal. The reduction in –NH2 content suggests that the surfactant is uniformly distributed on the coal surface, covering a substantial number of active –NH2 sites, which indirectly confirms the surfactant's effective adsorption. In the wavenumber range of 3500 to 3200 cm−1, both samples exhibit a prominent absorption peak; however, the peak area is significantly larger for the anthracite coal after DDM surfactant adsorption compared to the raw coal surface. The absorption peaks in this range correspond to the vibrations of OH and COOH groups found in alcohols, phenols, carboxylic acids and peroxides. The increased peak area may be attributed to the exposure of hydroxyl groups from the surfactant after adsorption, leading to a significant increase in –OH content. Additionally, absorption peaks appear at wavenumbers 1628 cm−1 and 1659 cm−1, which are primarily attributed to the stretching vibrations of aromatic rings (C=C). The peak areas at these two positions are similar, indicating that the adsorption of the surfactant on anthracite coal has minimal influence on this type of vibration. The absorption peaks in the range of 1380 to 1460 cm−1 correspond to the stretching vibrations of alkyl groups (–CH2, –CH3 and C–H) in aliphatic hydrocarbons. As shown in Figure 9, the raw anthracite coal exhibits two prominent peaks in this region, whereas the sample surface after surfactant adsorption only shows weak and irregular peaks, indicating a significant reduction in alkyl content. The wavenumber range of 500 to 900 cm−1 corresponds to the vibrational peaks of aromatic rings. The raw coal surface exhibits several distinct absorption peaks, whereas the sample after surfactant adsorption shows only very weak or negligible peaks. This indicates that the number of aromatic rings on the anthracite coal surface is significantly reduced after surfactant adsorption, further demonstrating the surfactant's ability to effectively adsorb onto the coal surface and cover its aromatic ring structures. FTIR analysis indicates that DDM surfactant exhibits a significant adsorption effect on the anthracite coal surface, effectively altering the distribution of surface functional groups and thereby enhancing its wettability.
Molecular dynamics simulation
Hydrogen bond analysis
The number of hydrogen bonds formed between water molecules and the modified coal surface is a critical parameter for explaining differences in hydrophilicity. According to geometric criteria, hydrogen bonds are defined as having an intermolecular donor-acceptor distance of less than 0.25 nm and a donor-hydrogen-acceptor angle greater than 135° (Li et al., 2019; Zhao et al. 2024). Table 6 summarises the number of hydrogen bonds formed between water molecules and the coal surface as well as the modified coal surface in both systems.
The number of hydrogen bonds in different systems.

As shown in Table 6, the number of hydrogen bonds formed between water molecules and the anthracite coal surface is only 17 without the addition of surfactant, while the number of hydrogen bonds increases significantly to 83 after the addition of DDM surfactant. The significant increase in the number of hydrogen bonds indicates that DDM modification has significantly enhanced the ability of the coal surface to form hydrogen bonds with water molecules. Since hydrogen bonds are an important form through which water molecules form stable adsorption layers on solid surfaces, an increase in their number implies enhanced surface polarity and improved hydrophilicity. This result is consistent with the observed significant reduction in contact angle in the experiment, providing preliminary evidence for the molecular-scale mechanism by which DDM enhances wettability. Additionally, this suggests that the hydrophilic end of DDM may be exposed at the interface in a specific conformation, enhancing its interaction strength with water molecules. To confirm whether this interaction is primarily dominated by electrostatic attraction, we further calculated the interaction energy to determine the dominance of electrostatic and van der Waals forces.
Interaction energy calculation
The interaction energy can be used to evaluate the adsorption strength between surfactants and anthracite coal. The more negative the interaction energy, the stronger the adsorption strength and the more stable the structure after adsorption (Chen et al. 2021; Li et al. 2021; Liu et al. 2020).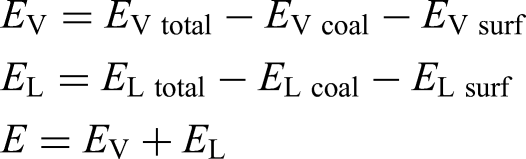
Table 7 summarises the calculated results of total interaction energy, electrostatic interaction energy and van der Waals interaction energy for the two adsorption systems. The table shows that the total interaction energy for both systems is negative, indicating that water molecules can spontaneously adsorb onto the coal sample surface.
Interaction energy between water and raw or DDM-modified anthracite surface.

By comparing the total interaction energies of the two systems, it shows that the interaction energy between water and raw coal is −561.31 kcal·mol−1 while that between water and modified coal is −678.10 kcal·mol−1. The more negative interaction energy in the modified system confirms that DDM improves the coal's surface hydrophilicity. This result is in agreement with the previous conclusions regarding the increase in the number of intermolecular hydrogen bonds.
Further analysis of the proportion of electrostatic and van der Waals interaction energies reveals that van der Waals interaction energy accounts for 68% of the total interaction energy when water directly adsorbs onto the anthracite coal surface, dominating the process. After the introduction of DDM, during the adsorption process between water molecules and the modified coal surface, the proportion of van der Waals interaction energy significantly decreases to 40%, while electrostatic interaction energy increases to 60%, becoming the primary contributor. Additionally, in unmodified coal, the system primarily relies on van der Waals forces to maintain water–coal surface adsorption. However, after the introduction of DDM, the enhanced surface polarity significantly strengthens the electrostatic interactions between water molecules and the surface, making electrostatic interactions the dominant contributing factor. This shift in interaction mode indicates that DDM enhances surface polarity, thereby significantly strengthening the water–coal surface bonding force. This not only explains the fundamental physical driving force behind the increase in hydrogen bond numbers but also validates the rationality of the decrease in contact angle and the enhancement of surface wettability from an energy perspective.
Mass density
As shown in Figure 10, when water molecules adsorb onto the surface of raw coal surface, the water layer starts at 5.14 nm and the density of the water layer reaches 0.91 g/cm3 at 8.2 nm from the coal surface. After the introduction of DDM, the starting point of the water layer shifts back along the Z-axis to 6.12 nm, and the water layer density increases to 0.93 g/cm3 at 8.2 nm from the coal surface. This indicates that the introduction of DDM causes the water layer to shift along the Z-axis while significantly increasing the aggregation of water molecules near the coal surface. This change can be attributed to the presence of microporous structures on the coal surface and its surface chemical properties. On the unmodified coal surface, the presence of numerous hydrophobic groups results in fewer hydrogen bonds between coal and water, leading to a lower aggregation of water molecules near the coal surface. In contrast, after adding DDM, the hydrophilic head group of DDM forms numerous hydrogen bonds with water molecules through oxygen atoms, significantly increasing the aggregation of water molecules near the modified coal surface.

Mass density distribution of raw anthracite and DDM-modified anthracite: (a) raw coal; (b) DDM–coal.
Additionally, further analysis of the distribution characteristics of the hydrophilic head groups and hydrophobic tails of DDM molecules reveals that the hydrophobic tails are distributed within the range of 6.22 to 8.03 nm, showing a uniform distribution in this region. The hydrophilic head groups are distributed within the range of 6.62 to 8.03 nm, primarily concentrated around 7.1 nm. This distribution characteristic indicates that the hydrophobic tails are closer to the coal surface, partially filling the pores on the coal surface, resulting in the water layer shifting backward along the Z-axis. Meanwhile, the hydrophilic head groups are closer to the water phase, forming more hydrogen bonds with water molecules and enhancing the interaction between the coal surface and water.
The mass density analysis indicates that DDM molecules form a spatially layered and ordered interface structure on the coal surface: the hydrophobic tails are located near the coal surface to fill pores, while the hydrophilic heads face the liquid phase and form hydrogen bonds with water molecules. This ‘head-tail oriented arrangement’ self-assembly behaviour facilitates the enrichment of water molecules on the modified surface, forming a higher-density water layer, which manifests as improved surface wettability of the coal at the macroscopic scale. Notably, this distribution trend aligns with the exposure of hydrophilic functional groups observed in XPS and FTIR, validating the complementary nature of experimental and simulation results.
Radial distribution function
To gain a deeper understanding of the interactions between water molecules and the anthracite surface, as well as between water molecules and the DDM-modified coal surface, the radial distribution function (RDF) was used to study the spatial arrangement of oxygen atoms in water molecules near both the raw and modified coal surfaces. The results are shown in Figure 11.

Radial distribution functions of water molecules in the two adsorption systems.
From Figure 11, it can be observed that the RDF curves of oxygen atoms in water molecules on both the anthracite surface and the DDM-modified coal surface exhibit similar shapes, with a distinct peak appearing at 0.27 nm. This indicates that the adsorption interactions between both coal surfaces and water molecules share a specific spatial characteristic.
The intensity of the RDF peak reflects the strength of the adsorption interaction between water molecules and adjacent atoms on the coal surface. A higher peak indicates a stronger interaction between water molecules and coal surface atoms. Upon further comparison of the g(r) peak values, it was found that after the addition of DDM, the g(r) peak value for water molecules was 22.9, significantly higher than the value of 18.7 when water directly interacts with raw coal. The g(r) peak value for water molecules on the modified coal surface is significantly higher than that on the raw coal, indicating that the introduction of DDM enhances the adsorption of water molecules on the coal surface. At the same time, this also suggests that the aggregation of water molecules near the modified coal surface is higher, and the binding ability between the surface and water molecules is stronger, further confirming that the affinity of the coal surface for water molecules is enhanced due to the DDM modification.
The results of the RDF clearly reveal that the wettability of the coal surface is significantly improved after the addition of DDM. This conclusion is consistent with the previous results obtained through hydrogen bond analysis, interaction energy calculations and mass density analysis, providing comprehensive theoretical support for the mechanism by which DDM modification enhances the wettability of anthracite.
Conclusion
This study employed a combination of physical experiments and molecular simulations to investigate the effect of the DDM surfactant on the wettability of anthracite from a macroscopic perspective. The adsorption characteristics and wettability mechanisms of DDM on the coal surface were also analysed from a microscopic perspective. The conclusions are as follows:
Contact angle experiments show that the addition of DDM significantly reduces the contact angle of anthracite, demonstrating the superior regulatory effect of DDM on the coal's wettability. XPS and FTIR spectral analysis results reveal a decrease in the C element content and the absorption peak area of hydrophobic groups, while the O element content and the absorption peak area of hydrophilic groups increase. After DDM adsorption on the anthracite surface, the wettability improvement is mainly attributed to the reduction in the relative content of hydrophobic functional groups and the increase in the relative content of hydrophilic functional groups on the coal surface. Molecular dynamics simulations revealed that DDM effectively promotes interactions between water molecules and the anthracite surface. After modification with DDM, the anthracite surface exhibited increased polarity, a higher number of hydrogen bonds formed between water molecules and the modified coal, and electrostatic energy became the dominant factor in the adsorption process. Furthermore, the adsorption of DDM on anthracite forms a stable interfacial structure, with the hydrophobic tail of DDM attaching to the coal surface and the hydrophilic head orienting toward the water phase. This results in a higher aggregation of water molecules near the modified coal surface, an increased binding capacity of the coal surface for water molecules and an overall enhancement in anthracite wettability.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Shanxi Province (grant number 201901D111071).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
