Abstract
Magnesium hydroxide modified activated carbon (Mg-HAAC) was obtained from the raw material of activated carbon of
Introduction
In recent years, the problem of excessive heavy metals in the atmosphere, water bodies, soil, and organisms caused by wastewater from industries such as mining and metallurgy, chemical manufacturing, and mechanical electronics has increasingly attracted widespread attention (Liu et al., 2023; Rahman and Singh, 2019; Zhou et al., 2020). The excessive heavy metals in water bodies could only be treated by transferring their location and changing their physicochemical state, which might pose a significant threat to human health (Sall et al., 2020). The current treatment methods on heavy metal pollution mainly include chemical precipitation, physical adsorption, and biological methods (Mao et al., 2021; Qin et al., 2020; Vardhan et al., 2019). For example, Ciobanu et al. (2023) prepared two types of biological adsorbents including biomass and biochar from
However, the common metal ion modification methods of activated carbon usually need to be carried out at high temperatures and had drawbacks such as cumbersome processes, high preparation costs, and high pollution risks (Hafizuddin et al., 2021; Liu et al., 2023; Ma et al., 2022; Xu et al., 2020). The modification of magnesium hydroxide does not have the issues mentioned above. The modification process is simple and safe, requiring no high temperatures, high energy consumption, and the modification materials are readily available and inexpensive.
In recent years, the modification of magnesium hydroxide has attracted the attention of many scholars. For example, Duan et al. (2020) pointed out that magnesium hydroxide-modified activated carbon has the advantages of environmental protection, safety, low cost, and simple operation. Furthermore, the specific surface area and adsorption capacity of activated carbon could be significantly improved by magnesium hydroxide modification (Duan et al., 2020; Tolkou et al., 2023). Kang Wang et al. (2016) found that the cadmium (II) adsorption capacity of coal-based activated carbon modified by magnesium hydroxide method increased from 3.47 to 8.08 mg/g (Wang et al., 2016). However, there have been no reports on the preparation of activated carbon from
In this article,
Materials and methods
Materials and reagents
The
Preparation of activated carbon samples
HAAC was prepared by phosphoric acid activation method. Firstly, 300 g of the dried
The preparing process of Mg-HAAC from HAAC was described as follows. In brief, 100 g of HAAC sample was add into 1500 mL different concentrations magnesium chloride solution (1.0, 1.5, 2.0, 2.5 mol/L) to stir for 3 h at 20°C and 180 r/min. Next, the HAAC-MgCl2 was collected from the mixed solution by filtrating under the vacuum condition and dried at 90°C to constant weight. The dried sample was mixed with 1500 mL of sodium hydroxide solution of 1 mol/L and reacted for 3 h under the condition of 25°C and continuous stirring of 180 r/min. Finally, the Mg-HAAC was obtained from the reaction solution after vacuum filtering, rinsing with deionized water until the pH remains constant and no chloride ions present in the filtrate and drying at 90°C to constant weight.
Physicochemical properties and structural characterization
Determination of magnesium hydroxide loading rate
5.00 g of Mg-HAAC was mixed with 150 mL of acid solution (pH 1.0) and shaking in a conical flask for 15 h. Then filtering the mixture solution by filter paper to collect the filtrate. And the magnesium ion content in the filtrate was measured using an atomic absorption spectrophotometer (PinAAcle D900; PerkinElmer; USA). The loading rate of magnesium hydroxide was calculated using the following formula.
Determination of Copper (II) adsorption capacity under different pH
0.1 g of HAAC and Mg-HAAC was added into 100 mL solution containing160 mg/L Copper (II) with different pH (3.0, 4.0, 5.0, 6.0, and 7.0), respectively. Then filtering the mixture solution by a filter membrane to collect the filtrate after shaking at 25°C for 2 h. And the Copper (II) content in the filtrate was measured using an atomic absorption spectrophotometer. The Copper (II) adsorption capacity of HAAC and Mg-HAAC under different pH was calculated by the following formula.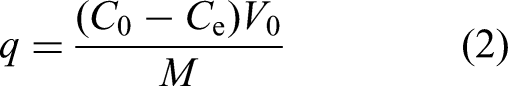
Determination of specific surface area and pore size distribution
Specific surface area and pore size distribution of HAAC and Mg-HAAC was determinate by a specific surface area and pore size analyzer (BSD-660; Beside Instrument Technology (Beijing) Co., Ltd; China) equipped with vacuum devices based on the adsorption of high-purity nitrogen by the sample at 77 K. And the results were calculated using the Brunner–Emmet–Teller (BET) and Barrett–Joyner–Halenda (BJH) methods, respectively.
X-ray diffraction (XRD) analysis
The micro-crystalline structure information of the two samples was conducted to analyze by a powder X-ray diffraction analyzer (Smartlab SE, Rigaku corporation, Japan). The testing conditions were as follows: ray of Cu Kα, filter of Ni, tube voltage of 40 kV, tube current of 30 mA, testing range of 10–80, and step angle of 0.02.
X-ray photoelectron spectroscopy (XPS) analysis
Two samples of HAAC and Mg-HAAC were placed in the chamber of the K-Alpha XPS instrument (Thermo Scientific K-Alpha, Thermo Scientific, USA) after pressing into a pellet and sticking on the sample tray. Then they were introduced into the analysis chamber when the pressure in the sample chamber was less than 2.0 × 10−7 mbar. The main testing conditions were as follows: 400μm of the spot size, 12 kV of the operating voltage, 6 mA of the filament current, and a full-spectrum scanning energy range of 150 ev with a step size of 1 ev.
Scanning electron microscopy (SEM) observation
Surface morphology of HAAC and Mg-HAAC were observed by a SEM (TESCAN MIRA; Tesken (China) Co., Ltd; Czechoslovakia) equipped with a SE2 secondary electron detector after spraying gold on a conductive adhesive for 45 s. The acceleration voltage was 3 kV, and the high and low vacuum resolutions were 1.2 and 3.5 nm, respectively.
Analysis of Copper (II) adsorption mechanism
Analysis of Copper (II) adsorption isotherm and kinetic models
Adsorption isotherm experiments were conducted at temperatures of 298.15, 308.15, and 318.15 K using a 100 mL of Copper (II) solution with different concentration gradients (10–160 mg/L, pH = 7). The Copper (II) solution was mixed with 0.1 g of activated carbon samples and subjected to shake for 2 h, then, filtered and measured. Alternatively, 0.1 g of activated carbon sample was mix with 100 mL of 160 mg/mL of Copper (II) solution and react for different times (5–120 min), and the filter was collected through filtration after the adsorption reaction was completed.
The adsorption performance of two types of activated carbon toward Copper (II) was investigated, and the Langmuir, Friendlies, pseudo-first-order kinetic model, and pseudo-second-order kinetic model were selected to fit the adsorption equilibrium data. The effects of temperature, initial Copper (II) concentration, and adsorption time on the maximum adsorption capacity of activated carbon, as well as the adsorption mechanism were analyzed as well. The calculation formulas for each model are as follows:


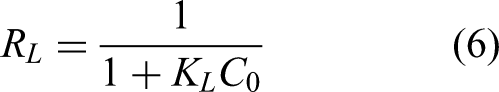


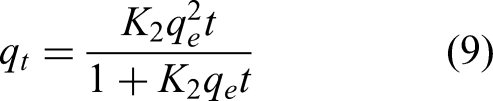
Thermodynamic study of adsorption
The Gibbs free energy, standard enthalpy, and standard entropy changes of Copper (II) adsorption on activated carbon were determined using the Van't Hoff equation. The thermodynamic properties of chromium ion adsorption on two types of activated carbon were investigated to examine the influence of temperature on adsorption and determine if the adsorption process is spontaneous. The Van't Hoff equation can be expressed as follows.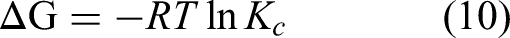

Results and discussions
The effect of magnesium hydroxide loading on the modification of HAAC
This study investigated the effects of different concentrations (1.0–2.5 mol/L) of MgCl2 solution on the Mg (OH)2 loading rate and Copper (II) adsorption capacity of Mg-HAAC modified from HAAC, and the result was shown in Table 1. With the increase of magnesium chloride concentration, the loading capacity of magnesium hydroxide on Mg-HAAC gradually increases to a maximum value of 3.92%. The adsorption capacity of Copper (II) reaches the maximum value of 23.88 mg/g when the magnesium chloride concentration is 2.0 mol/L. Magnesium hydroxide also exhibits a certain adsorption capability toward Copper (II). After being loaded onto activated carbon, magnesium hydroxide subdivides the macropores and mesopores of the activated carbon into more micropores, thereby increasing the specific surface area of Mg-HAAC. However, magnesium hydroxide also consumes some of the adsorption sites of the activated carbon. As shown in Table 1, when the loading amount of magnesium hydroxide is 3.77%, the total adsorption capacity of activated carbon and magnesium hydroxide for Copper (II) reaches the maximum value of 23.88 mg/g. When the loading amount of magnesium hydroxide is less than 3.77%, although magnesium hydroxide occupies fewer adsorption sites of activated carbon, it cannot generate more micropores within the macropores and mesopores of activated carbon to increase the specific surface area of activated carbon. Therefore, the total adsorption capacity of activated carbon and magnesium hydroxide for Copper(II) is less than 23.88 mg/g and decreases with the decrease of magnesium hydroxide loading amount. When the loading amount of magnesium hydroxide exceeds 3.77%, the occupation of adsorption sites of activated carbon by magnesium hydroxide increases, excessive magnesium hydroxide particles may also block some micropores of activated carbon, reducing the specific surface area of activated carbon and leading to a decrease in the adsorption capacity for Copper (II). At this point, the total adsorption capacity of activated carbon and magnesium hydroxide for Copper (II) decreases to 23.47 mg/g. Similar results have been reported in previous studies (Öter and Selçuk Zorer, 2020; Wang et al., 2016).Therefore, 2.0 mol/L magnesium chloride solution was chosen to modify HAAC in order to obtain Mg-HAAC with maximum Copper (II) adsorption capacity.
Magnesium hydroxide loading of modified activated carbon prepared under different conditions.

Analysis of XPS and XRD
The XPS spectra of HAAC and Mg-HAAC were shown in Figure 1(a). The carbon (C1 s) and oxygen (O1 s) photoelectron peaks were observed at binding energies of 284.05 and 531.64 eV, respectively, which all indicated the presence of a large amount of carbon and oxygen atom in both activated carbons samples. While a small peak at the binding energy of 1304.08 eV in Mg-HAAC illustrated a small amount of magnesium (Mg) was successfully incorporated into the surface of Mg-HAAC (Öter and Selçuk Zorer, 2020). The binding energy of O in Mg-HAAC increased due to the loading of magnesium hydroxide. In addition, the binding energies of C remain unchanged between HAAC and Mg-HAAC, suggesting that there was no coordination interaction between the carbon framework and Mg. This indicated that the interaction between activated carbon and magnesium hydroxide was mainly based on van der Waals forces after the precipitation of magnesium hydroxide formed by the reaction of magnesium chloride with sodium hydroxide.
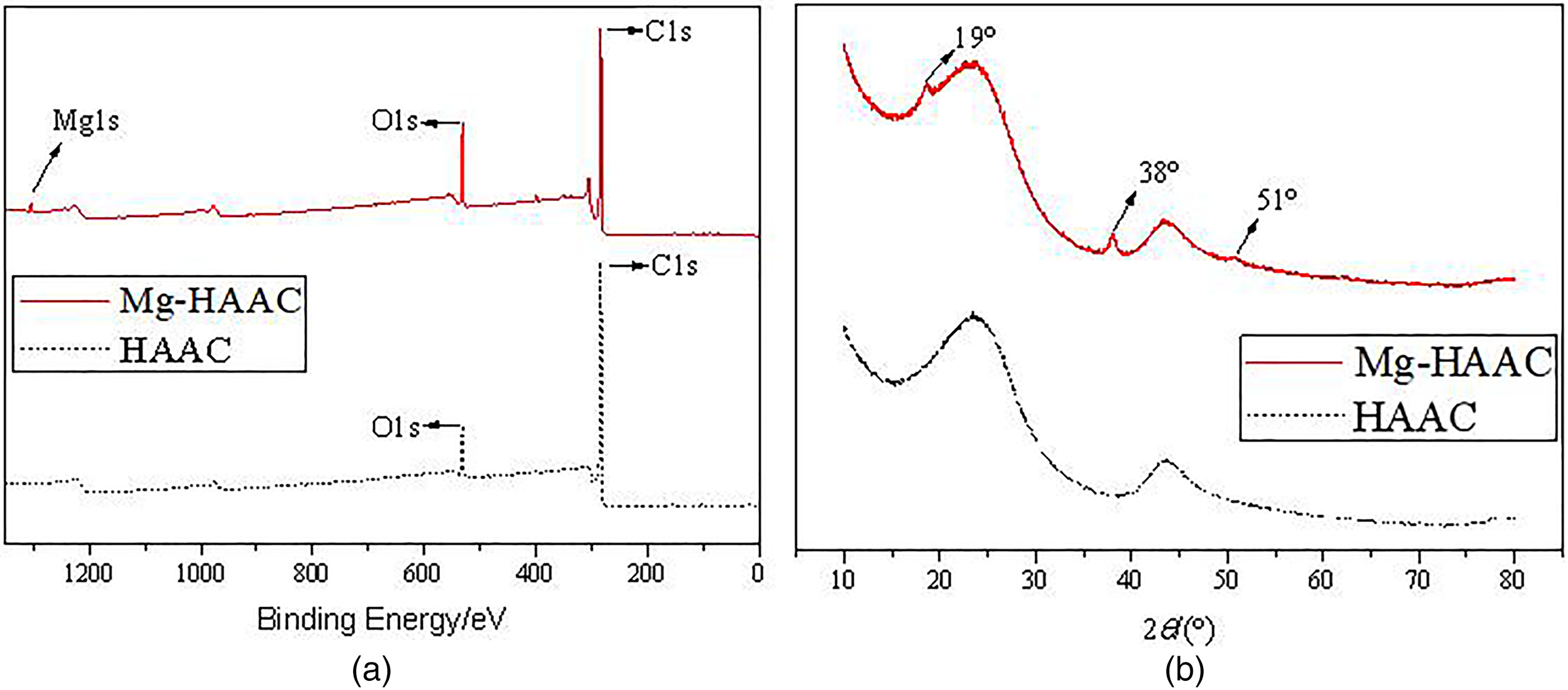
XPS spectra (a) and XRD patterns (b) of HAAC and Mg-HAAC.
Both HAAC and Mg-HAAC showed a smooth curve with no apparent impurity peaks in XRD pattern (Figure 1(b)). But Mg-HAAC exhibited a strong diffraction peaks at 2θ of 19°, 38°, and 51°comparing with that of HAAC. This confirmed the magnesium hydroxide was successfully loaded onto the HAAC forming Mg-HAAC once again according to the standard XRD pattern information of Mg(OH)2 released by the International Centre for Diffraction Data (ICDD) in PDF2-2003 No.84-2164. Similar findings could be seen in the report of Li et al. (2019), where the unique XRD absorption peak of magnesium hydroxide was detected (Li et al., 2019).
SEM characterization
The SEM images of HAAC (a, c) and Mg-HAAC (b, d) were shown in Figure 2. Both HAAC and Mg-HAAC have uniformly distributed and dense pore structures. HAAC surface contains a small amount of material debris, while Mg-HAAC not only contains a small amount of material debris, but also has precipitates of magnesium hydroxide, which can be more visually compared in images c and d in Figure 2. Combining the results of XPS and XRD analysis, it can be determined that magnesium hydroxide has been successfully loaded onto the activated carbon.
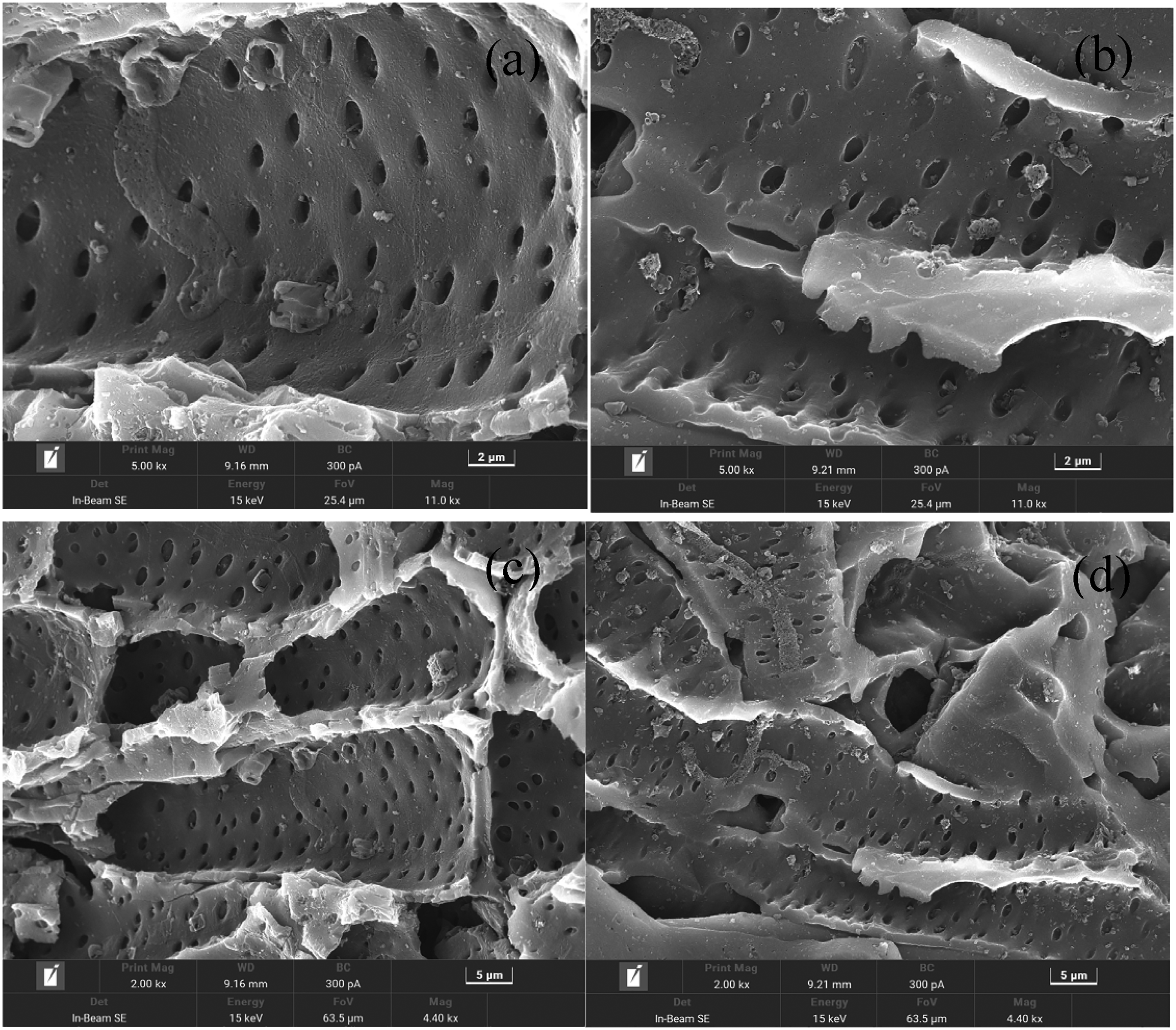
SEM image of HAAC (a, c) and Mg-HAAC (b, d).
Specific surface area and pore size distribution analysis
As the results shown in Figure 3, the isotherms of HAAC and Mg-HAAC presented a sharp increase at relatively low pressures. This indicated that the isotherms of two samples were both Type I and there were microporous structures in the pore size of HAAC and Mg-HAAC according to the classification by the International Union of Pure and Applied Chemistry (IUPAC). However, the adsorption isotherms of the two samples also exhibited a clear hysteresis loop as the relative pressure increases to 0.42, which indicated the adsorption isotherms of HAAC and Mg-HAAC belong to a mixed type of I and IV(Bag et al., 2020). In addition, the pore size distribution of the two samples was mainly distributed below 10 nm. This mean that they contained both microporous and mesoporous structures, which was also conducive to the adsorption of small-sized metal ions.

Nitrogen adsorption–desorption isotherm and Barrett–Joyner–Halenda (BJH) adsorption pore-size distribution plot of HAAC and Mg-HAAC.
The results of specific surface area and other pore size parameters listed in Table 2 indicate that the specific surface area (1617.5 m2/g) and pore volume (0.982 cm3/g) of Mg-HAAC were both higher than that of HAAC (1551.3 m2/g) and (0.861 cm3/g). This was consistent with the previous research that an appropriate loading amount of magnesium hydroxide could effectively increase the specific surface area and pore volume of activated carbon (Tolkou et al., 2023; Wang et al., 2016). This was mainly because an appropriate amount of magnesium hydroxide load would turn large pores in activated carbon into more micropores and medium pores, thereby increasing its specific surface area and pore volume, magnesium hydroxide will block some of the micropores, leading to an increase in mesopore quantity, thereby increasing the average pore size (Wang et al., 2016).
Surface area, pore volume parameters of HAAC and Mg-HAAC.

Effect of pH on the Copper (II) adsorption capacity
The Copper (II) adsorption capacity of activated carbon was closely related to the pH of the adsorbed solution. As the results shown in Figure 4, the Copper (II) adsorption capacity of HAAC and Mg-HAAC gradually increased as the increases of pH of the adsorbent, and reaching its maximum value of 36.24 and 53.87 mg/g at pH 7, respectively. This might be because the large amount of H+ present in the adsorption solutions with a low pH could enhance the protonation degree of functional groups on the surface of the adsorbent and competing with Copper(II) to adsorb active sites. At the same time, like charges would also inhibit Copper (II) from approaching the active sites of the activated carbon, thereby reducing the adsorption of Copper(II) by the activated carbon. As the pH increases, H+ would dissociate from the functional groups on the activated carbon surface, exposing more active sites for the adsorption of Copper (II), thereby increasing the adsorption capacity of activated carbon for Copper (II). When magnesium hydroxide was loaded onto the activated carbon surface, magnesium hydroxide itself could adsorb metal ions and also had a buffering effect on the pH of acidic solutions. Therefore, the adsorption capacity of Mg-HAAC for Copper (II) was significantly increased compared to HAAC (p<0.05) (Alcaraz et al., 2020; Massadeh and Massadeh, 2019). When the pH of the adsorbent was 3, some magnesium hydroxide would dissolve and lose its adsorption capacity, resulting the reduce of the adsorption capacity of activated carbon on Copper (II) because of the competitive effect for the adsorption sites between magnesium ions and Copper (II) (Abu-Daabes et al., 2023). This was similar to the findings of other activated carbon adsorption studies on chromium and cadmium ions (Liu et al., 2023; Tolkou et al., 2022; Wang et al., 2016). Therefore, pH 7.0 was chosen as the experimental condition for the subsequent analysis of the adsorption mechanisms of HAAC and Mg-HAAC.

The relationship between adsorption capacity of Copper (II) and pH of solution.
Isothermal adsorption model of HAAC and Mg-HAAC for Copper (II)
In the lower concentration range (≤ 20 mg/L), the Copper (II) adsorption capacity of HAAC and Mg-HAAC increased sharply with the increase of initial concentration of the Copper (II) solution (Figures 5 and 6). The efficient adsorption might be caused by the larger concentration difference between the solid and liquid phase which could promote the movement of the adsorbate into the adsorbent. When the concentration of Copper (II) exceeded 20 mg/L and continued to increase, the adsorption capacity of HAAC and Mg-HAAC gradually reaches its maximum, and almost all active sites bind to Copper(II) at this time (Li et al., 2021).

Sorption isotherm of Copper (II) onto HAAC (a) and Mg-HAAC (b) under different temperatures.
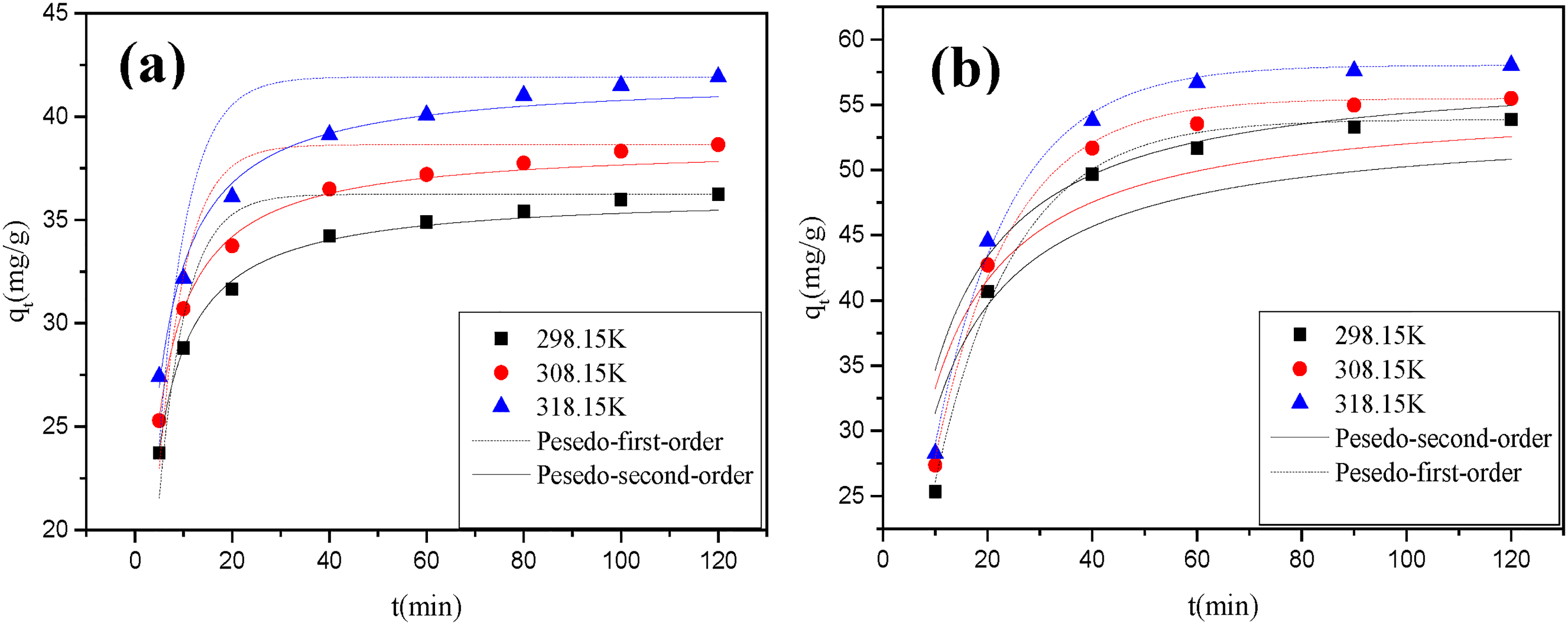
The plots of pseudo-first-order and pseudo-second-order model for the kinetic data of HAAC (a) and MG-HAAC (b) under different temperatures.
Similarly, the Copper (II) adsorption capacity of the two activated carbons also rapidly increases to equilibrium as the temperature increases. Temperature usually has four main effects on the adsorption process of activated carbon. Firstly, the increase in initial concentration of the adsorbate leads to an increase in the viscosity of the solution, which affects the diffusion rate of the adsorbate in the pores. However, increasing temperature can decrease the solution viscosity. Secondly, the increase of temperature can cause the break of chemical bonds at the edges of the active sites on the adsorbent surface and the formation of new adsorption sites, which will enhance the adsorption capacity of the adsorbent. Thirdly, adsorption is generally an endothermic process, and higher temperatures can increase its adsorption performance. Fourthly, the expansion of the porous structure in activated carbon and magnesium hydroxide after heating also contributes to the adsorption of Copper (II) by activated carbon (Ark et al., 2022; Nowruzi et al., 2020).
Therefore, increasing the initial concentration and temperature of the adsorbate could increase the diffusion rate between the solid–liquid interface and the number of adsorption sites, respectively, thereby enhancing the adsorption capacity of HAAC and Mg-HAAC. The maximum adsorption capacities of HAAC and Mg-HAAC at a temperature of 318.15 K, were 41.92.18 and 58.03 mg/g, respectively, which were higher than its adsorption capacities at other two temperatures. The adsorption performance of Mg-HAAC was significantly better than that of HAAC as well.
Table 3 showed the fitting results of the isotherm adsorption parameters for the two types of activated carbon. The correlation coefficients (R2) of Copper(II) in the Langmuir model were all higher than 0.981, indicating that the Langmuir model was more suitable for simulating the isotherm adsorption experiments of the two types of activated carbon. It also indicated that the adsorption of Copper (II) by the two types of activated carbon was a monolayer adsorption. Furthermore, the predicted values (qm(cal)) of the Langmuir model at different temperatures were very close to the experimental values (qe(exp)), and all RL values are positive and less than 1, which indicated there was a effective adsorption of activated carbon for Copper(II). The equilibrium constant KL increases with temperature, which indicated that the adsorption capacity of activated carbon for Copper(II) also increases. This is consistent with the kinetic behavior of most activated carbons in adsorbing metal ions (Bazana et al., 2019; Gao et al., 2019; Hafizuddin et al., 2021).
The parameters for Langmuir and Freundlich isotherm models of Copper (II) sorption onto HAAC and Mg-HAAC under different temperatures.

Analysis Copper (II) adsorption kinetics of HAAC and Mg-HAAC
The effect of adsorption time on the Copper (II) adsorption capacity of HAAC and Mg-HAAC solution at different temperatures when the initial concentration of the adsorbent was 160 mg/L was investigated and the adsorption kinetics curve was showed in Figure 6. With the prolongation of adsorption time, the Copper (II) adsorption of the two activated carbons showed an overall trend from fast to slow, and ultimately decreased to 0. This was mainly because the initial Copper (II) concentration in the adsorbent is the highest, the activated carbon has the most binding active sites, which is conducive to the rapid occurrence of adsorption; while the concentration difference between the Copper (II) in the solution and the surface of the activated carbon decreases, and the repulsive force of Copper (II) between the surface of HAAC and Mg-HAAC and adsorbent solution gradually increases when the adsorption reaches a certain level, the adsorption sites on the surface of the activated carbon also continuously decrease, so the adsorption rate begins to decrease and gradually reaches equilibrium(Ahya et al., 2022; Nowruzi et al., 2020; Wong et al., 2021). In addition, the Copper (II) adsorption ability of Mg-HAAC was higher than HAAC under the same adsorption time and temperature. For example, after 20 min of adsorption at 318.15 K, Mg-HAAC and HAAC could adsorb 67% and 75% of Copper(II)s in the solution, respectively, indicated that Mg-HAAC has superior adsorption efficiency.
The Copper (II) adsorption kinetics of HAAC and Mg-HAAC were analyzed using pseudo-first-order and pseudo-second-order models, and the kinetic parameters were shown in Table 4. The pseudo-first-order kinetic equation fitted well for Mg-HAAC (R2 > 0.994), while the pseudo-second-order kinetic equation was relative better for HAAC (R2 > 0.985). It was worth noting that, although Mg-HAAC was more in line with the Pseudo-first-order kinetic model, this did not directly indicate that the adsorption rate of Copper (II) by Mg-HAAC was decided by physical adsorption (Ren et al., 2021). Because some of the magnesium hydroxide loaded onto the activated carbon would chemically react with Copper(II) to form copper hydroxide precipitate. This process would reduce the rate of Copper(II) in the solution entering into the micropores and mesopore due to the partial blockage of the mesopore and mesopore, resulting in lower a adsorption rate of Mg-HAAC than that of HACC. However, the adsorption capacity of Copper(II) was higher than that of HAAC. Therefore, the adsorption of Copper(II) by Mg-HAAC was still predominantly governed by chemical adsorption. The experiments involving other metal ions adsorbed by magnesium hydroxide differed from the present study in that magnesium hydroxide in the micropores and mesopores of activated carbon reacted with Copper(II), which was not observed in those experiments (Chen et al., 2023).
The kinetic model parameters for HAAC and MG-HAAC under different temperatures.

The adsorption thermodynamic analysis of HAAC and Mg-HAAC
The adsorption thermodynamic fitting parameters of both activated carbons samples were listed on Table 5. The R2 of HAAC and Mg-HAAC were both higher than 0.983, and ΔG were less than 0, indicating that the adsorption process of Copper (II) by both samples was spontaneous. In addition, ΔG also gradually decreased with the temperature increases, which mean that increasing the temperature could enhance the adsorption efficiency of activated carbon towards Copper (II). Of course, ΔH of HAAC and Mg-HAAC were all positive value indicated that the Copper(II) adsorption process of the two activated carbon samples was endothermic. And this was consistent with the effect of temperature on adsorption. Meanwhile, the positive value ofΔS represented an increase in disorder at the solid-liquid interface during adsorption, which indicated an increase in randomness of the contact between the two types of activated carbons and Copper (II) at the solid–liquid interface. The thermodynamic behavior of the adsorption of metal ions by HAAC and Mg-HAAC was consistent with that of most activated carbons(Tolkou et al., 2022; Xu et al., 2020).
Thermodynamic parameters for the Copper (II) adsorption of HAAC and Mg-HAAC at different temperatures.

Conclusion
Two kinds of activated carbon including the raw activated carbon (HAAC) prepared by phosphoric acid activation method and its modified activated carbon (Mg-HAAC) by the magnesium hydroxide modification method was obtained from
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Tibet Autonomous Region Science and Technology Major Project (grant number XZ201901NA04).
