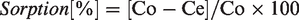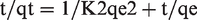Abstract
Green synthesis approach was successful used extract was successful in preparing bimetallic zero-valent Iron/Copper nanoparticles [FB-nZVFe/Cu]. Scanning Electron Microscope [SEM], Fourier Transform Infrared Spectroscopy [FTIR], and Dispersive X-ray Spectroscopy [EDX] showing the synthesizing of FB-nZVFe/Cu. The removal efficiency of Caffeine [5 mg L−1] reached 86% under the conditions [0.2 g L−1, 45 min, and pH 5]. The adsorption data are more appropriate by the Langmuir model [R2 = 0.9987] with qmax = 34.34 mg g −1. Kinetic results showed that Caffeine uptake is following pseudo-second-order. Langmuir and pseudo-second-order are more appropriate in linear and non-linear models. Overall, FB-Fe/Cu is a committed green substance for removal Caffeine from aqueous solutions. Functional parameters affect investigated using the Linear regression analysis, we found them to account for over 98% of the variables affecting the removal procedure.
Introduction
Caffeine [CAF, C8H10N4O2], an alkaloid belonging to the methylxanthine family (Al-Qaim et al., 2015). It is the most used pharmaceutical material worldwide which has a psychological effect (Elhalil et al., 2018; Ganzenko et al., 2015). Caffeine is founds in many products such as coffee, tea, energy drinks, and drugs, and acts as a psychomotor stimulant (Buerge et al., 2003; Yamal-Turbay et al., 2012). Caffeine is a primary compound in the manufacture of many medicines. It is one component of the medication for a migraine, fatigue, breathing problems, drowsiness, and pain relievers (Bueno et al., 2011; Indermuhle et al., 2013). Caffeine is safe in moderate doses, but can increase alertness, and cause nervousness, insomnia, a headache, and dizziness (Aly et al., 2013; Guo et al., 2011; Habibi et al., 2012). Extensive use of Caffeine lead to mutation effects such as DNA inhibition, irritation, tremors and anxiety (Zhang et al., 2011). It mobilizes calcium from cells which leads to bone mass loss and risk factor for cardiovascular mobilizes (Ali et al., 2012; Torres et al., 2014).
Caffeine is resistant to natural degradation. It detected with an exorbitant amount in wastewater treatment plants and water bodies worldwide (Ghosh et al., 2019). The concentration of CAF in several surface streams and WWTP effluents is as high as 230 μg/L (Rosal et al., 2010). The presence of Caffeine in the water will produce negative effects on human life and aquatic organisms (Elhalil et al., 2018). Wastewater processes can remove CAF include Fenton process (Elhalil et al., 2016), biologic treatments (Bonakdarpour et al., 2011), coagulation/flocculation (Freitas et al., 2015), and membrane process (Taheran et al., 2016), but these techniques have limits in removing CAF contaminant from water effluents (Bolong et al., 2009). Therefore, an alternative environmentally friendly and efficient method must implement for the removal of CAF from wastewater.
Researchers have focused on developing new techniques using Nano-technology to overcome these restrictions (Abdel-Aziz, 2020; Abdel-Gawad et al., 2016; Farag et al., 2018). Since the 1990s, Fe nanoscale particles [Fe-NPS] used for water treatment because of its advantages e.g. good surface area, wide dispersion of reactive sites, and unique adsorption (Abdel-Gawad et al., 2016; El-Shafei et al., 2016; Farag et al., 2018; Mostafa et al., 2017).Copper nanoparticles [Cu-NPS] have considered [chemical–physical] properties, high surface area, and less cost (Fathima et al., 2018; Khani et al., 2018). Cu-NPS are the most stable in comparison with these zero-valent metals (Mahmoud et al., 2019; Zhang et al., 2017). Nanoparticles play a serious role in contaminants removal from the aquatic surroundings (Tatarchuk et al., 2019, 2020a, 2020b). In current years, nanoscale particles prepared by NaBH4 (Mahmoud, 2017; Mahmoud et al., 2018a; SaryEl-deen et al., 2017), but they cause major contamination. It is critical to find alternate, low-cost, ecologically friendly techniques (Zhu et al., 2018). Traditional methods adopted for the synthesis of Fe-NPS or Cu-NPS by sonochemical synthesis, vacuum sputtering, physical method [attrition], and thermal decomposition. These methods have many restrictions viz. high temperature and pressure or energy requirement, and so they relative cost (Danish et al., 2017).
Green chemistry approaches become the most significant ecology friendly and low-cost alternative (Farag and Peters, 2018; Mahmoud et al., 2019a). Green leaves contain an extensive amount of reducing and capping agents via. polyphenols, flavonoids, and other reducing substances, which can reduce iron and copper salts to zero-valent and protecting them from agglomeration (Abdel-Aziz et al., 2020).
Bimetallic Iron/Copper is more efficient than mono-metallic NPS to remove several pollutants (Danish et al., 2017). Green nanoparticles prevent fungi, bacteria, and microbes from growing (, 2017b). Plants abundance, cost, efficiency, and non-toxic nature, all these advantages are important in the water treatment process (Abdel-Aziz et al., 2019; Shanehsaz et al., 2015). Ficus Benjamina-nZVFe/Cu is a novel idea used for the removal of Caffeine.
Therefore, the present study is to assess the ability of FB-nZVFe/Cu and applied to contaminated water of Caffeine removal.
Experimental
Chemicals and reagents
All chemicals used were of the analytical reagent grade and the highest purity. We adjusted the change of pH using 0.1 M NaOH and 0.1 M HCl solutions.
Methods
Preparation of Ficus Benjamina extract
Ficus Benjamina leaves were washed with Tap water to remove dust, then with distilled water, then were dried in the oven at 50 °C. The leaves were grinded into small pieces and sieved using a 2.5 mm sieve. In an Erlenmeyer flask, 20 g of leaves were added to 100 ml distilled water, and the previous solution was boiled for 5 min at 60 °C followed by filtrating using Whatman no. 1 filter paper, and the filtrate was stored at 4 °C until used as capping and reducing agent.
Synthesis of FB-nZVFe/Cu
About 0.18 g CuSO4.5H2O, 0.94 g FeSO4.2H2O were dissolved with distilled water in a 100 ml volumetric flask. Ficus extract [50 ml] was added drop by drop to Fe/Cu solution, followed stirred for 20 min. Changing of the solution color from yellowish to brown then to black was indicating the synthesizing of zero-valent nanoparticles (Asghar et al., 2018; Katata-Seru et al., 2018). FB-ZVFe/Cu was separated using centrifugation for 10 min, then washed with anhydrous alcohol. Ficus-ZVFe/Cu was placed at 65° C for 3 hr, then was stored at 4 °C until used (Abdel-Aziz et al., 2019).
Characterization of FB-nZVFe/Cu
The prepared FB-nZVFe/Cu sample was examined using SEM with EDX [QUANTA FEG250], and FTIR spectroscopy [Shimadzu IRAffinity-1]. Before field emission, SEM FB-ZVFe/Cu nanoparticles were layered by gold which rises electrons scatter to give a notable contrast. EDX was used for defining the composition of the sample. FTIR spectroscopy to prove the formation of nanoparticles.
Batch adsorption studies
About 100 mg of CAF was dissolved in 1000 ml water [Stock 100 mg L−1]. From the stock with volumetric pipettes we took various mills to volumetric flasks to get different concentrations [5, 10, 15, and 20 mg L−1]. The removal was studied by mixing 0.2 g L−1 of FB-nZVFe/Cu particles to the CAF solutions. The removal was calculated using the following equation:
Where Co is the initial CAF concentration [mg L−1] and Ce is the equilibrium CAF concentration [mg L−1]. The amount of CAF removed by Ficus-nZVFe/Cu particles was calculated using the following equation:
Effect of different operating parameters
pH
About 0.2 g of FB-nZVFe/Cu was added to 1000 ml of CAF [5 mg L1], at different pH values [3, 5, 7, and 9], different times [15, 30, and 45] minutes, respectively, and stirring rate was fixed at 100 rpm at room temp.
Contact time
About 0.2 g of FB-nZVFe/Cu was added to 1000 ml of CAF [5 mg L−1] at different contact times; [15, 30, 45, 60, 90, and 120 min], at pH 5, and stirring rate was fixed at 100 rpm.
Adsorbent dose
Different weights of FB-nZVFe/Cu were varied between [0.1 and 0.3 g L−1] were added to 1000 ml of CAF [5 mg L−1], and other operational influences via. pH 5, and time 45 min, and stirring rate was fixed at 100 rpm at room temp.
Stirring rate
About 0.2 g of FB-nZVFe/Cu was added to 1000 ml of CAF [5 mg L−1] at contact time 45 min, room temp, at pH 5, at a different stirring rate [100, 150, 200, and 250] rpm.
Initial concentration
About 0.2 g of FB-nZVFe/Cu was added to 1000 ml of different concentrations of CAF [5, 10, 15, and 20 mg L−1], at pH 5, time 45 min, and stirring rate was fixed at 100 rpm at room temp.
Adsorption study
Freundlich and Langmuir isotherm models are the most two public isotherms applications (Abdel-Gawad and Abd El-Aziz, 2019).
Freundlich adsorption isotherm
Freundlich is an empirical calculation used for labeling dissimilar adsorption surface and is given by:
Where n [dimensionless] and Kf [[mg g−1] [mg L−1]−1/n] are Freundlich constant related to the adsorption intensity and adsorption capacity, respectively, [Kf] and [n] evaluated by plotting Ln qe and Ln Ce.
Langmuir adsorption isotherm
Langmuir assumes mono-layer coverage of CAF over a similar surface of FB-nZVFe/Cu. The Langmuir is given by the equation:
Where qe [mg g−1] is, the mass of CAF adsorbed per mass of adsorbent used, Ce [mg L−1] is the equilibrium concentration of Caffeine, qmax [mg g−1] is the maximum monolayer capability of adsorption, and KL [L/mg] is the Langmuir constant correlated to binding sites affinity and adsorption energy. The plot of Ce/qe versus Ce used to generate the values of qmax and KL.
Kinetic study
Factors from two kinetic models, including pseudo-first-order and pseudo-second-order, have been used for describe adsorption kinetics in solid-liquid systems.
Pseudo-first-order kinetic model (PFO)
The adsorption rate can be labeled using the PFO and is expressed as follows:
Where qe and qt [mg/g] represent adsorption value of CAF in aqueous mediums at equilibrium and at time [min], respectively, and k1 is the first-order equilibrium constant which calculated the plot of ln [qe-qt] against t.
Pseudo-second-order kinetic model (PSO)
PSO equation is the most simplified and frequently used kinetic equation. The pseudo-second-order kinetic model is usually described as thus:
Where qe and qt are the adsorption capability [mg/g], at equilibrium, and at time [min], respectively, and k2 [g/[mg. min] represents the rate constant of the adsorption. We can calculate values of k2 from the plot of t/qt against t.
Reusability of ficus-ZVI/Cu NPS
Reusability of the adsorbent is a significant factor for estimating the cost-effective applicability, and to satisfy the ecology and economic thresholds. Caffeine removal by FB-nZVFe/Cu was carried out at [5 mg L−1]. To further examine the reusability, the experiments were reiterated [up to 5 times] by exposing a reacted FB-nZVFe/Cu to a fresh CAF solution. Each time following the reaction, the FB-nZVFe/Cu was collected immediately from the solution by centrifugation [10 min] and was washed with ethanol, then was dried in an oven at 45 °C before being used for the next adsorption recycle.
Results and discussion
Characterization and analysis of FB-nZVFe/Cu
SEM and EDX
Figure 1 displays the SEM image of a semi-spherical shape FB-ZVFe/Cu nanoparticles, and the size of these particles range around 19–63 nm. Many pores allow the CAF to be transported better and the spread of pollution mass to the inner FB-nZVFe/Cu.

SEM of FB-nZVFe/Cu nanoparticles.
Figure 2 shows the EDX analysis of FB-ZVFe/Cu nanoparticles. Fe and Cu peaks indicate the occurrence of bimetallic. Other beaks via C, O, Si, and S of Ficus-leaves extract (Abdel-Aziz et al., 2019).

EDX of prepared FB-nZVFe/Cu sample.
FTIR measurements
Figure 3 displays that the FTIR of FB-nZVFe/Cu before the reaction between 4000 and 400 cm−1. The broad-band at 3400 to 3000 cm1 was attributed to O-H stretching vibration, indicates the occurrence of polyphenols. The strength of phenolic peaks can reduce metals and is a strong indicator for the synthesizing of the zero-valent nanoparticles. The band at 1539 cm−1 indicates the existence of the Ficus amid group; the peak at 1362 cm−1 signposts the occurrence of polyphenols Ar-ring C = C stretching vibration (Abdel-Aziz et al., 2019).

FTIR spectrum of FB-nZVFe/Cu.
Effect of operating parameters
Effect of pH
pH system plays the most role in the CAF removal efficiency process. The removal efficiency of CAF at different pH values [3, 5, 7 and 9], at a different time [15, 30, and 45] min, respectively, when the dose of Ficus-nZVFe\Cu was 0.2 g L−1, concentration 5 mg L−1, and stirring rate was fixed at 100 rpm at room temp, the CAF removal efficiency was [43, 78, 69, and 23%], [44, 81, 71, and 23%], and [45, 86, 73, and 24%], respectively, as shown in Figure 4. Ficus Benjamina has a low Zero charge point of 4.85, because of the high acidity of the solution of leaves extract (Abdel-Aziz et al., 2019). It was observed that the most appropriate pH for the removal was 5. At pH is PZC the high removal efficiency due to increasing the number of e- are generated from the zero-valent adsorbent which enhance the degradation of the CAF, and a lot of available vacant adsorption sites, which allows CAF mass to be transported better and the spread to the inner FB-nZVFe/Cu. At pH<pHpzc [pH 3], Low in the removal capacity due to the presence of an excess of H+ ions that occupy available vacant adsorption sites, and the free e- neutralize the solution, and portion of nanoparticles dissolve in the medium (Mahmoud et al., 2018c). At pH> PZC [pH 9], because of the repulsion between OH- ions and negatively surface of FB-nZVFe/Cu is negative (Misra et al., 2018; Shan et al., 2016); and maybe the low availability of nanoparticles which precipitates in the solution (Mahmoud et al., 2018b), thus leading to low removal efficiency. Based on the above pHpzc is the optimal removal of Caffeine.

Effect of pH on caffeine removal (dose 0.2 g L−1, stirring100 rpm, initial concentration 5 mg L−1).
Effect of contact time
Different time effect at [15, 30, 45, 60, 90 and 120 min], was studied on the Caffeine removal [5 mg L−1], using 0.2 g L−1 of Ficus-Fe/Cu at pH 5, and the stirring rate was fixed at 100 rpm; and the removal percent was [78, 81, 86, 88, 89 and 89%], as shown in Figure 5. The increase in connection time gradually, lead to increase the interfere of the mass contaminant in the unfilled sites of the nano-particles, and increasing the generation of the amount of degradation free e-, all this has led to increasing the removal.

Effect of contact time on Caffeine removal (pH 5, dose 0.2 g L−1, stirring100 rpm, initial concentration 5 mg L−1).
Effect of adsorbent dose
Caffeine removal efficiency was studied as a function of the adsorbents dose effects, which was varied between [0.1 and 0.3 g L−1], and other operational influences were pH 5, and time 45 min. Caffeine concentration was [5 mg L−1], and the removal ratios were [48, 86, and 100%], as shown in Figure 6. The optimal adsorbent dose for the removal of CAF was found to be 0.2 g L−1, as shown in Figure 7. As expected, increasing the FB-nZVFe/Cu dose leads to an increase in the number of both free e- and unoccupied sites; so, the removal increased.

Effect of adsorbent dose on Caffeine removal (pH 5, time 45 min, stirring100 rpm, initial concentration 5 mg L−1).
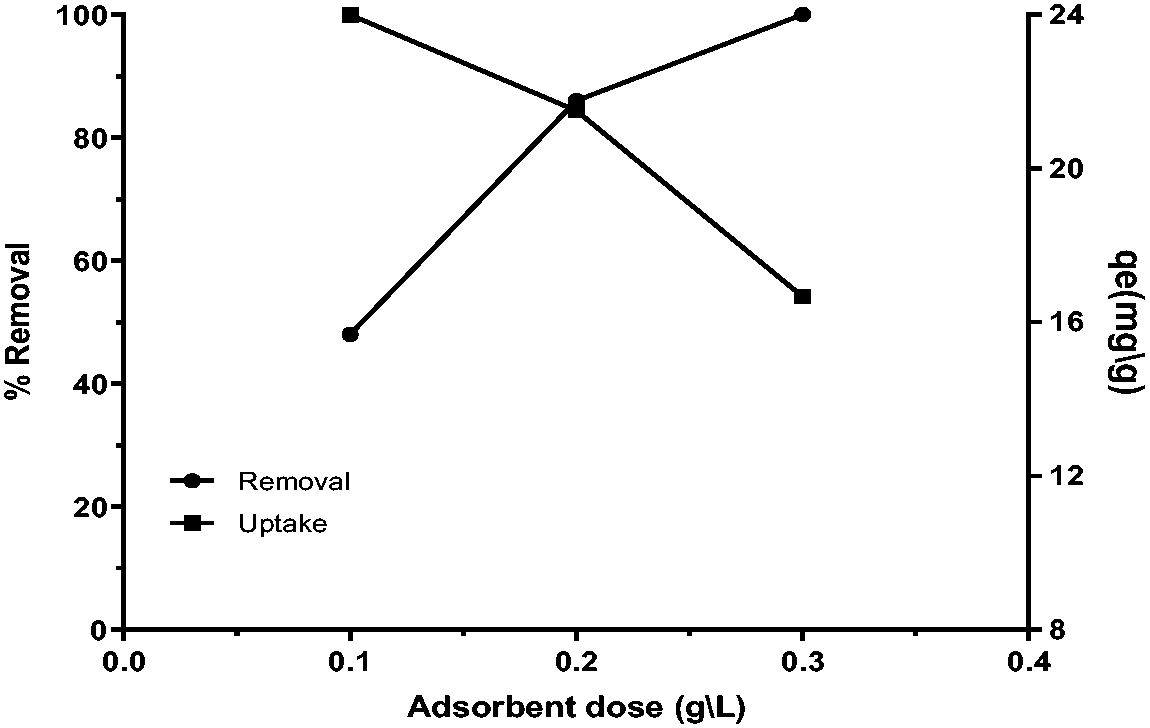
The optimum effective dose for caffeine removal.
Effect of stirring rate
Figure 8 displays Caffeine removal by Ficus-nZVFe/Cu as a function of stirring rate. The stirring rate was varied between 100 and 250 rpm, and other operational influences were pH 5, and time 45 min. Caffeine concentration was [5 mg L−1] and the removal ratios were [86, 87, 88, and 88%]. The optimal stirring rate for CAF removal was found to be 100 rpm. The increase in the stirring rate from [100 to 250 rpm] has not a significant effect on the removal efficiency.
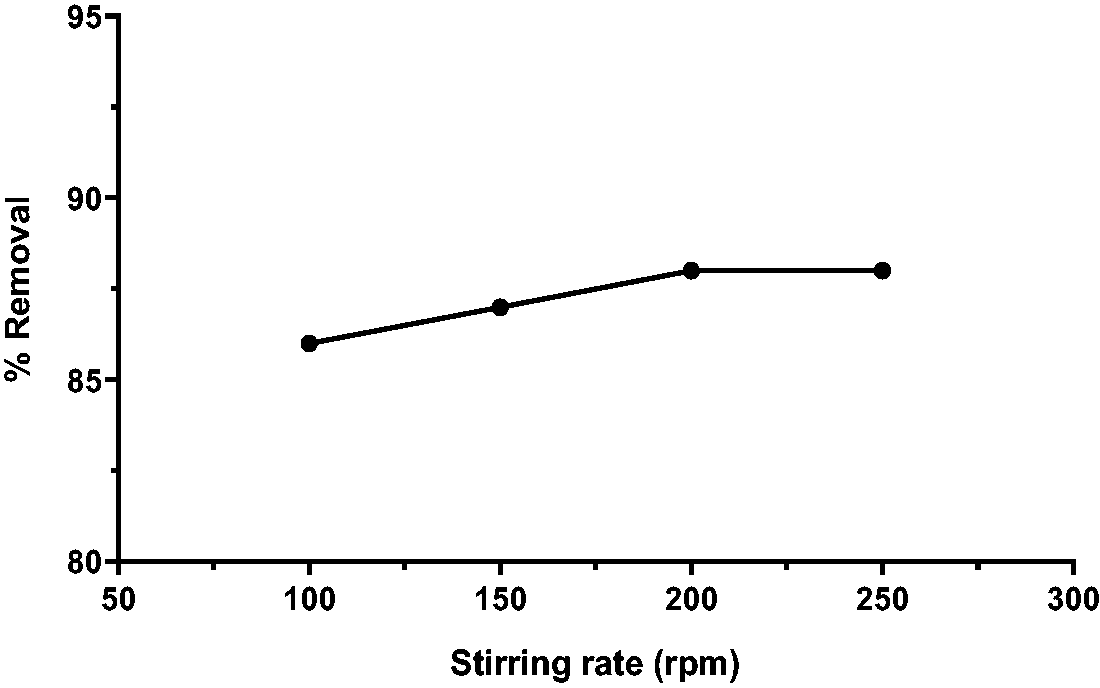
Effect of stirring rate on caffeine removal (pH 5, dose 0.2 g L−1, time 45 min, initial concentration 5 mg L−1).
Effect of the concentration
The removal experiments using Ficus-ZVFe/Cu particles were carried out on Caffeine solution having several concentrations of [5, 10, 15 and 20 mg L−1], at pH 5, time 45 min, and adsorbents dose was 0.2 g L−1, and the removal percentage was [86, 60, 45, and 33%], as shown in Figure 9. At the beginning of the study process at low concentration, the removal ratio was high, which compared to the high concentration of the pollutant.

Effect of concentration on caffeine removal (pH 5, dose 0.2 g L−1, stirring100 rpm, time 45 min).
Adsorption isotherm studies for caffeine removal
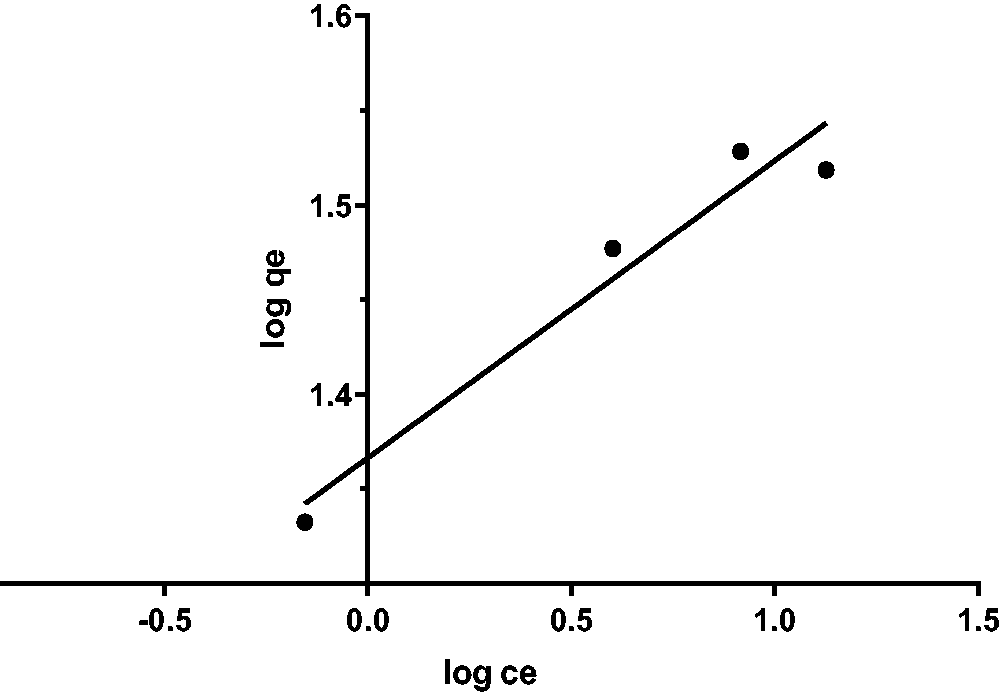
The adsorption capability of the Ficus-nZVFe/Cu predicted and estimated by Freundlich and Langmuir adsorption isotherms models, as shown in Figures 10to 13, which are the two most common isotherms applications (Abdel-Gawad, and Abd El-Aziz, 2018; Soha et al., 2017). Langmuir model was fitted well with isotherm in both linear and non-linear by higher determination coefficients R2 and low Error sum compared to Freundlich, and maximum adsorption capability of 34.34 mg g−1, as shown in Table 1.

Effect of equilibrium concentration on caffeine removal.
Freundlich for caffeine contributing component.

Langmuir for caffeine contributing component.

Isotherm studies for nonlinear models.
Isotherm models for the adsorption of caffeine.

Kinetics studies
Table 2 realized that the PSO model is a better fit for the data in both linear and non-linear than the PFO, as shown in Figures 14to 16. The value qe [cal] = 46.26 is approximately same to qe [exp] 44.5 for Caffeine. Results show that CAF uptake on FB-nZVFe\Cu is following the PSO.
Kinetic models for the adsorption of caffeine.

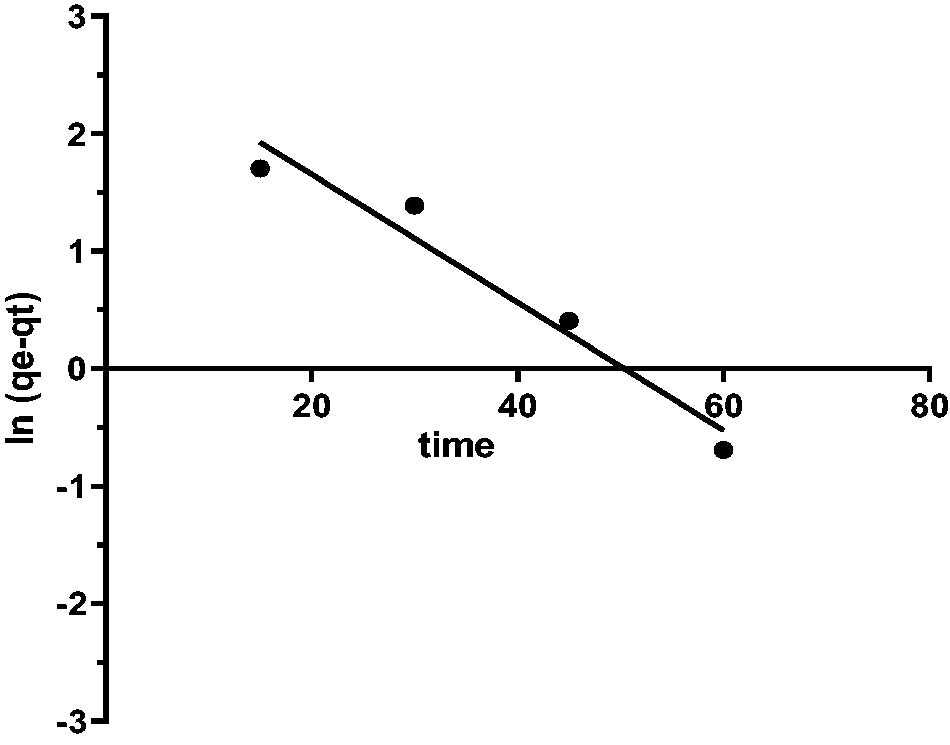
The PFO kinetics model d.
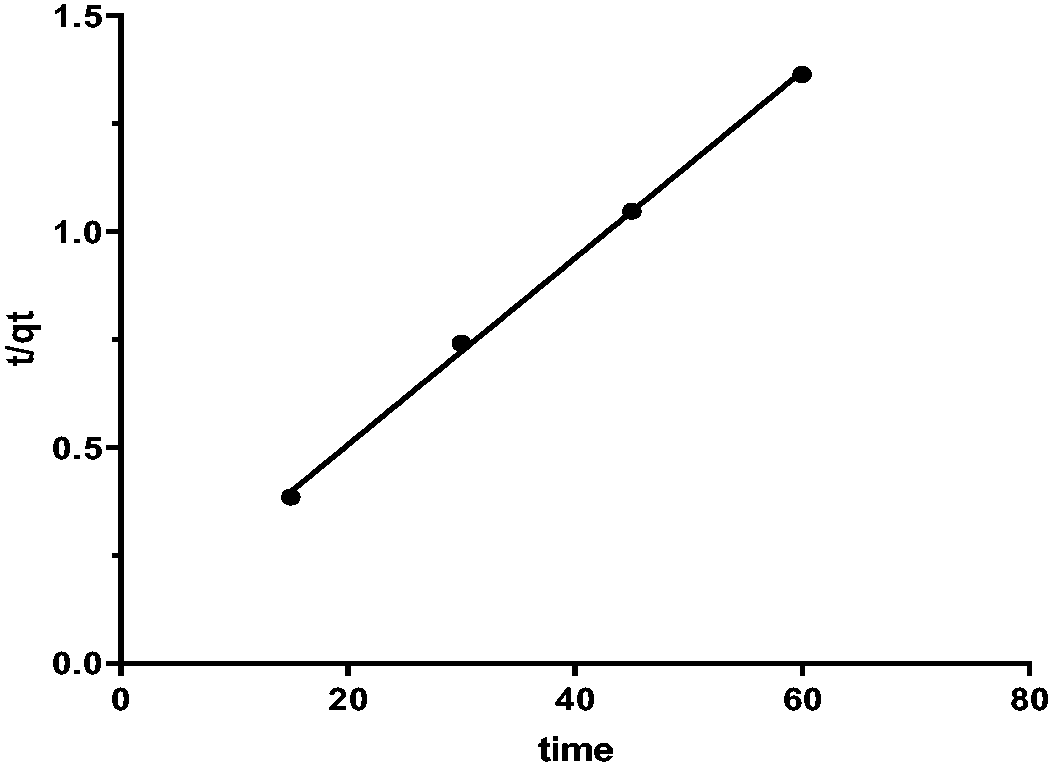
The PSO kinetics model data for caffeine.
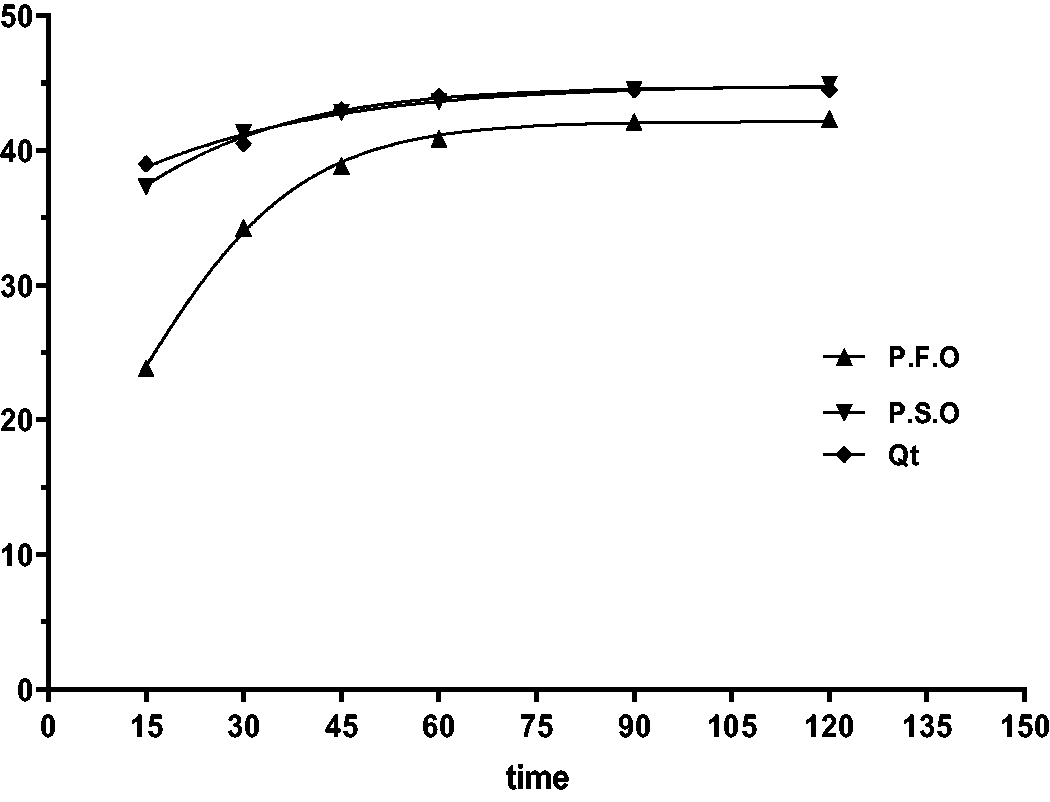
Kinetic studies for nonlinear models.
Statistical analysis
The effect of the variables on the removal process [e.g. pH, time, dose, concentration, and stirring rate] have been studied using the entered technique, where it was found that R2 = 0.953. This means that the studied variables profane conquer over 95% of the total of the variables affecting the removal procedure as that the standard error of the estimate is 4.03260, so the percentage of error in this study is low.
Response surface methodologies (RSM)
The effect of various parameters was examined using Linear regression analysis [IBM-SPSS Statistics] where results support the practical results. By applying the B values shown in Table 3, the removal equation can be deduced whereas:
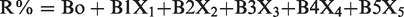
Coefficients.a

Where R is the removal percent, B is constant, X1 is the effect of pH, X2 is the effect of contact time, X3 is the effect of adsorbent dose, X4 is the effect of stirring rate, and X5 is the effect of concentration.
Reusability of FB-ZVI/Cu NPS
Figure 17 represents the removal efficiencies decreased with each reuse cycle. Thus, the removal efficiency was [82, 78, 83, 70, and 69%] after being used in the 1st, 2nd, 3rd, 4th, and 5th recycles, respectively. Yet the removal efficiency of Caffeine was still high even in the fifth recycle. The slight decrease in the removal efficiency was mainly happened in the second cycle, this may be caused by the loss of nanoparticles or by an irreversible occupation of partial-adsorption sites. The regenerated adsorbent still kept good adsorption efficiency after the five rounds of adsorption recycles. This showed that the synthesized FB-ZVFe/Cu NPs had good stability and durability and possesses excellent reusability for the removal of Caffeine from aqueous mediums. These results reveal that FB-nZVFe/Cu offered a high potential to be used repeatedly in removing CAF without a considerable decrease in its removal efficiency. One of the advantages of FB-ZVFe/Cu NPs is their easy to separate from the reaction mixture.

Reuse the performance of the synthesized FB-nZVFe/Cu.
Conclusions
In this study, the green nanoscale zero-valent Fe/Cu [FB-nZVFe/Cu] is accomplished of Caffeine removal under various operational factors. Maximum Caffeine removal was observed at pH 5. Caffeine removal efficiency between [86 and 33%] was achieved after using different CAF concentrations [5, 10, 15, and 20 mg L−1] with 0.2 g L−1 of FB-nZVFe/Cu, stirring rate 100 rpm, pH 5, and contact time 45 min, and when the dose increased from [0.1 to 0.3 mg L−1], the removal of CAF increased by 52% [C0 = 5 mg L−1]. The adsorption data were fitted well with the Langmuir isotherm study with the highest [R2 = 0.9987]. PSO kinetic model is a better fit to the experimental data than PFO kinetic model. According to the results in this study, there is no difference between linear and non-linear models. The removal efficiency of CAF was still over 68% after reusing the material for five times. FB-nZVFe/Cu being an eco-friendly alternative procedure and can lead to success in wastewater treatment and produce high-quality treated effluent.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.